Abstract
Background:
Premature ejaculation (PE) is a common sexual disorder characterized by a lack of voluntary control over ejaculation. Current treatments often fail to produce consistently satisfactory outcomes. Peripheral electrical stimulation (PES) is an emerging neuromodulation technique that applies electrical currents to targeted body areas and has shown promise across various conditions.
Objectives:
Although the use of PES for PE is relatively underexplored, this study aimed to synthesize existing research to better understand its potential as a treatment option.
Design:
Systematic review.
Data sources and methods:
A systematic search was conducted in PubMed, Embase, Scopus, Web of Science, and Google Scholar using relevant keywords. Studies were included if they investigated PES as a treatment for PE and reported outcomes such as intravaginal ejaculatory latency time (IELT) or other clinical measures.
Results:
Ten studies met the eligibility criteria. Due to the limited number and heterogeneity of studies, a meta-analysis was not feasible, and a narrative synthesis was performed. Stimulation was applied transcutaneously in various ways, including at acupuncture points, the dorsal penile nerve, and the posterior tibial nerve. Protocols varied considerably, though commonly reported parameters included a pulse frequency of 20 Hz and a pulse width of 200 µs, typically administered in 30-min sessions. Some studies lacked detailed descriptions of stimulation settings. Overall, the studies demonstrated a positive trend in favor of PES for prolonging IELT, and no consistent or significant adverse events were reported.
Conclusion:
The findings suggest that PES may be a promising adjunctive therapy for men with PE by prolonging IELT. Further high-quality research using validated patient-reported outcomes is needed to clarify the impact of PES on perceived ejaculatory control and sexual satisfaction.
Trial registration:
The study protocol was prospectively registered in PROSPERO (ID: CRD42024551360).
Keywords
Introduction
Premature ejaculation (PE) is a common sexual disorder characterized by the inability to regulate ejaculation. When left untreated, PE can lead to distress and frustration for both partners. 1 The prevalence of PE, whether acquired or lifelong, does not exceed 8%–10%. However, broader estimates suggest that 20%–30% of men report occasional premature ejaculation, which may not meet strict diagnostic criteria. 2
Despite numerous advances in the management of PE, current treatment options—including selective serotonin reuptake inhibitors (SSRIs), topical anesthetics, and behavioral techniques—do not consistently provide satisfactory results for all individuals. 3 Treatment responses vary, and some patients may not experience significant improvement in ejaculatory control or intravaginal ejaculation latency. Consequently, there is increasing interest in exploring novel therapeutic strategies for PE management.
Peripheral electrical stimulation (PES), which involves applying electrical impulses to specific body areas, is one such emerging neuromodulation technique. 4 It is believed to modulate neuronal activity and influence the central nervous system’s response to sexual stimuli. PES has been used in various clinical contexts, including pain management,5,6 lower urinary tract dysfunction,7,8 bowel dysfunction,9,10 and motor function. 11
According to a hypothesis proposed by Gruenwald et al. 12 neuromuscular transcutaneous electrical stimulation could potentially benefit individuals with PE by inhibiting contractions of the bulbospongiosus muscle, potentially reducing ejaculatory reflexes. The use of PES, particularly for PE, has only been explored in a fragmented body of studies so far. Therefore, a systematic review is warranted to synthesize the currently available evidence and critically assess the existing body of research.
Method
Study design
To ensure a rigorous and transparent methodology, this systematic review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (Supplemental Appendix 1). The study protocol was prospectively registered in PROSPERO, the International Prospective Register of Systematic Reviews (ID: CRD42024551360).
Search strategy
A comprehensive literature search was conducted across major electronic databases, including PubMed, Embase, Scopus, Web of Science, and Google Scholar. The search strategy was developed using appropriate keywords and Medical Subject Headings (MeSH) terms, such as “premature ejaculation,” “electrical stimulation,” and related synonyms. The search was conducted in English and covered all records from inception to May 2024. Additionally, the reference lists of relevant articles were manually screened to identify any additional studies. The detailed search syntax is provided in Supplemental Appendix 2.
Eligibility criteria
Two independent reviewers screened the retrieved articles based on predefined inclusion and exclusion criteria. Inclusion criteria were as follows:
(a) Studies investigating the use of PES, including transcutaneous or percutaneous techniques, for the management of PE.
(b) Studies reporting outcomes such as ejaculatory control, intravaginal ejaculation latency time (IELT), masturbation ejaculation latency time (MELT), or other relevant clinical measures.
(c) Original research articles, including randomized controlled trials (RCTs), quasi-experimental studies, observational studies, and case reports or case series.
Exclusion criteria were as follows:
(a) Studies not focusing on the use of PES for PE.
(b) Studies with inadequate data or insufficient information for analysis.
(c) Non-original publications, such as reviews, letters, commentaries, and editorials.
Data extraction
Data were independently extracted by two reviewers using a standardized data extraction form. The following information was collected:
(a) Study characteristics: author, year of publication, study design, sample size, and country.
(b) Participant characteristics: age, clinical condition, and number of participants.
(c) Intervention details: type of peripheral electrical stimulation, stimulation parameters (e.g., frequency, intensity), duration, and frequency of application.
(d) Outcome measures: primary and secondary outcomes assessed, measurement tools used, and corresponding results.
(e) Safety profile: any reported adverse events or complications related to PES.
(f) Main findings.
Data synthesis
Due to several factors—including the limited number of studies, heterogeneity in interventions and study designs, and a restricted study population—conducting a meta-analysis was not a suitable option for generating reliable results. The small number of included studies did not offer a sufficient sample size for robust quantitative analysis. Moreover, the studies demonstrated considerable variation in intervention protocols, participant characteristics, and methodological designs, making data pooling for meta-analysis impractical. Therefore, a narrative synthesis was adopted to allow for a comprehensive and descriptive analysis of the available evidence. This qualitative approach facilitated the identification of similarities, differences, and emerging trends across the studies.
Risk of bias and quality assessment
Quality assessment was conducted independently by two reviewers, and any discrepancies were resolved through discussion with a third reviewer. The JBI Critical Appraisal Guidelines were followed, and the appropriate checklist was selected based on the study design (randomized controlled trial, clinical trial, or case report, as detailed in Supplemental Appendix 3). The quality of the studies was categorized as high, moderate, low, or very low based on the number of “no” or “unclear” responses: 0–1 (high), 2–3 (moderate), 4–5 (low), and more than 5 (very low). The checklists used for quality appraisal are available in the Supplemental Materials.
Certainty of the evidence
The certainty of the evidence was assessed using the GRADE approach (Grading of Recommendations Assessment, Development and Evaluation). 13 Given the heterogeneity of study designs and interventions, and the narrative synthesis of results, we rated the certainty of evidence for the main outcomes narratively.
Results
Selection process
The systematic search yielded a total of 446 citations, of which 224 were duplicates and were removed. Screening the titles and abstracts resulted in the exclusion of 206 citations due to irrelevance. The full texts of the remaining 16 papers were assessed, and, ultimately, 10 studies met the eligibility criteria.14–23 The PRISMA flow diagram illustrating the selection process is presented in Figure 1. Also, Supplemental Appendix 4 provides a comprehensive list of excluded studies12,24–28 along with reasons for each decision.

PRISMA 2020 flow diagram for new systematic reviews which included searches of databases, registers, and other sources.
General characteristics of the included studies
Most of the included studies were clinical trials; four were RCTs,17,19,20,22 one was a cross-over trial, 20 and only one was a case report. 18 The study populations predominantly consisted of patients with primary PE, although one study included individuals with PE associated with chronic prostatitis/chronic pelvic pain syndrome (CP/CPPS). 15 The main tool for assessing PE across the included studies was IELT. In one study, 20 MELT was used for measuring PE-related outcomes. Treatment durations varied from 2 to 24 weeks across the studies. Further details are provided in Table 1.
General characteristic of the included studies.

CP/CPPS, Chronic prostatitis/chronic pelvic pain syndrome; EMLA, Eutectic mixture of local anesthetics; EPAS, Electrical pulse acupoint stimulation; IELT, Intravaginal ejaculatory latency time; NR, Not reported; PE, Premature ejaculation; RCT, Randomized controlled trial; TENS, Transcutaneous electrical nerve stimulation; TES, Transcutaneous electrical simulation; TNES, Transcutaneous neuromuscular electrical stimulation; TPTNS, Transcutaneous posterior tibial nerve stimulation.
In addition, as shown in Table 2, the PES protocols varied considerably among the studies that provided such information. The most commonly reported parameters were a pulse frequency of 20 Hz and a pulse width of 200 µs. Each intervention session, conducted one to three times per week, lasted approximately 30 min. Some trials did not provide detailed information regarding the stimulation protocols.15,17,22,23
The characteristics of electrical stimulation protocol reported by the included studies.

The majority of studies reported delivering transcutaneous stimulation14–21,23 using various methods, including transcutaneous electrical acupoint stimulation (TEAS), 23 transcutaneous dorsal penile nerve stimulation (TDPNS), 18 transcutaneous neuromuscular electrical stimulation (TNES), and transcutaneous posterior tibial nerve stimulation (TPTNS).14,21
Characteristics of each individual studies
In the earliest study by Lin et al. 16 conducted in 2007, 14 individuals suffering from CPPS or chronic prostatitis underwent 6 weekly sessions of PES. The results of this study indicated that half of the patients showed improvement, while the other half remained unchanged after treatment.
In the study by Mallat et al. 17 90 patients with PE were randomly assigned to three groups receiving stimulation PES, EMLA cream, or a placebo. The PES group showed significantly better results in terms of IELT, intercourse satisfaction, and adverse events compared to both the EMLA and placebo groups.
In the phase II clinical trial by Uribe et al., 21 participants received 3 weekly sessions of TPTNS for 12 consecutive weeks and were followed for 9 months after treatment completion. Eleven patients completed the trial, and 54.5% showed a tshreefold increase in baseline IELT scores at week 12 (p = 0.037). At weeks 12, 24, and 48 of follow-up, IELT increased by 4.8-fold, 6.8-fold, and 5.4-fold, respectively, in the study population. Therefore, TPTNS therapy appeared to delay ejaculation in patients with lifelong PE, with no serious adverse effects.
Moreover, Aydos et al. 14 recruited 60 patients with PE and divided them equally into two groups: the treatment group, which received TPTNS for 12 weeks, and the control group. The results of the study showed an increase in ejaculation duration in the TPTNS group.
Shechter et al. 20 administered transcutaneous electrical stimulation to 23 patients with PE, 20 of whom completed the trial. Seventeen patients experienced an improvement in MELT compared to the sham treatment. Mean MELT values increased significantly by 3.5-fold.
In another study of Sechter et al., 19 the efficacy and safety of the vPatch, a miniaturized transcutaneous electrical stimulation device that delivers electrical impulses to the perineal region for treating PE, were assessed. Following vPatch treatment, the mean IELT significantly increased from 67 to 123 s in the active group, compared to an insignificant increase in the sham group. Additionally, the increase in IELT was significantly higher in the active group, with IELT increasing by 3.1 times in the active group versus the sham group, with no serious adverse events reported.
Further, Zhang et al.’s study 23 reported a statistically significant improvement in the Premature Ejaculation Diagnostic Tool (PEDT) score in patients with erectile dysfunction who received TNES.
In another study by Xu et al. 22 the efficacy of dapoxetine, with and without TNES, was investigated in the treatment of primary PE. Their results showed that dapoxetine combined with TNES led to statistically significant improvements in IELT, sympathetic skin response latency in the penis, and a reduction in PEDT score, compared to dapoxetine alone.
In the study by Li et al., 15 69 patients with PE were randomly assigned to receive either oral paroxetine 20 mg/day, mid-frequency electrical pulse acupoint stimulation (EPAS), or a combination of oral paroxetine 10 mg/day and mid-frequency EPAS (P + EPAS) for an 8-week period. The results suggested that P + EPAS had better safety and efficacy than either paroxetine or EPAS alone in treating PE.
Lastly, a case report by Moussa et al. 18 involved the treatment of a 28-year-old man with lifelong PE using TDPNS applied three times per week for 24 weeks. IELT increased significantly from 0.6 min at baseline to 3.9, 4.0, 4.8, and 4.9 min at weeks 24, 36, 48, and 60, respectively.
Safety profile
No major or consistent adverse events were associated with the application of PES in the studies. In one study, 20 three patients reported unpleasant but tolerable sensations during the stimulation, and one patient experienced a transient burning sensation during urination after the treatment. In another study, 21 transient constipation, and a sensation of heat in the leg during one therapy session were associated with the treatment.
Quality of the included studies
Only one study was rated as high quality, which was a case report. 18 Two RCTs were rated as very low quality,17,22 two had low qualities,15,23 and others had moderate qualities.14,16,19–21 The results of the quality assessment are presented in Table 3.
The quality of the included studies, based on the JBI checklist.

N, No; NA, Not applicable; Q, Question; RCT, Randomized controlled trial; U, Unclear; Y, Yes.
Certainty of evidence
The certainty of the evidence was assessed using the GRADE approach for the outcome of increased IELT or MELT, the certainty of the evidence was rated as low due to concerns about risk of bias, as some studies were non-randomized or had unclear blinding, and concerns about inconsistency, due to heterogeneity in stimulation protocols and outcome measurements. Symptom improvement, including changes in PEDT scores and CPPS-related symptoms, was also supported by low-certainty evidence, reflecting similar concerns regarding study design limitations and indirectness resulting from variations in symptom assessment methods. The certainty of the evidence regarding the safety of PES was rated as moderate. Although no serious adverse events were reported, minor and transient side effects, such as discomfort during stimulation and transient constipation, led to a slight downgrade due to risk of bias. A detailed summary of the GRADE assessment is provided in Table 4.
Summary of findings and certainty of evidence (GRADE assessment).
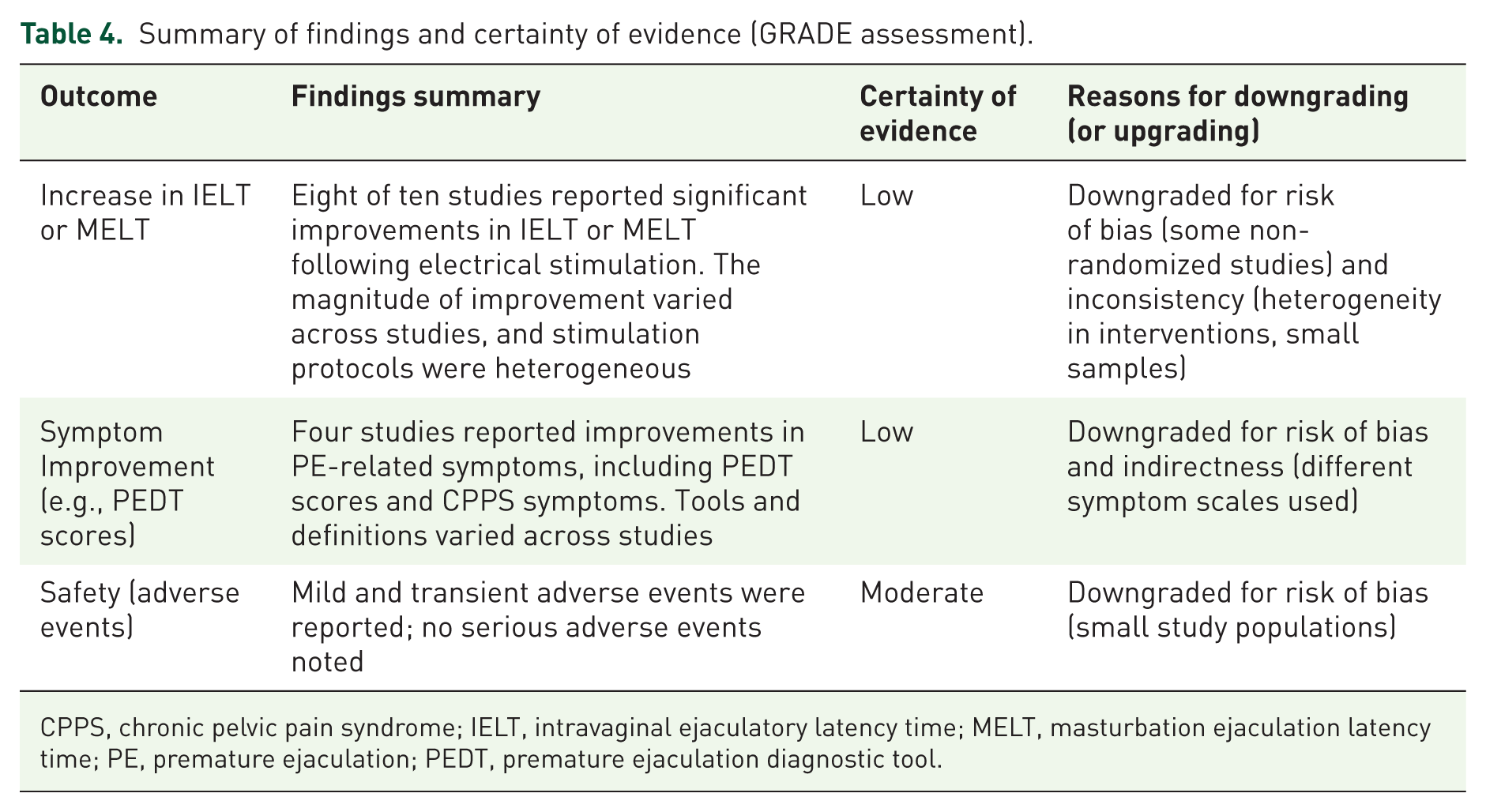
CPPS, chronic pelvic pain syndrome; IELT, intravaginal ejaculatory latency time; MELT, masturbation ejaculation latency time; PE, premature ejaculation; PEDT, premature ejaculation diagnostic tool.
Discussion
The current study aimed to evaluate the efficacy and safety of PES as a potential therapeutic approach for managing clinical outcomes in individuals with PE. The pooled analysis of the included studies demonstrated an improvement in IELT following PES interventions compared to control groups or baseline measures.
The mechanisms through which PES can enhance the clinical outcomes of PE are not yet fully understood and require further investigation. However, several potential mechanisms have been proposed based on the physiological effects of PES. PES may also affect the neuromuscular pathways involved in ejaculatory reflexes. According to the concept proposed by Gruenwald et al., 12 administrating transcutaneous electrical stimulation to the neuromuscular junctions in bulbospongiosus and ischiocavernosus muscles prior to sexual intercourse can potentially inhibit rapid stereotyped rhythmic contractions and may have a beneficial effect on the treatment of PE.
This theory is further supported by the findings of Cizmeci et al., 29 who demonstrated in a rat model that low-frequency (2 Hz) neuromuscular stimulation applied to the bulbospongiosus muscle significantly prolonged ejaculation latency compared to both high-frequency stimulation and control groups. The authors proposed that this effect may be due to the induction of subtetanic continuous contractions that interfere with the rhythmic activity required for ejaculation. These findings highlight the importance of stimulation frequency and mode in shaping therapeutic outcomes and suggest that on-demand application of low-frequency PES may be more effective than high-frequency or continuous regimens.
Also, PES may influence sensory thresholds by altering the transmission of sensory signals from the genital region to the central nervous system. PES can potentially desensitize or modulate sensory nerve fibers, leading to a reduction in the intensity of sexual sensations and delaying the ejaculatory response. PES has the potential to influence central neural circuits involved in ejaculatory control. It is hypothesized that electrical stimulation may activate or inhibit specific neural pathways in the central nervous system, thereby modulating the excitability and coordination of neuronal activity associated with ejaculation. 30 By altering the central neural circuitry, PES could potentially enhance the coordination and synchronization of the ejaculatory process, resulting in improved ejaculatory control. PES has been shown to induce neuroplastic changes in the nervous system.31,32 Prolonged or repeated PES may trigger adaptive changes in neural connectivity and synaptic plasticity. These neuroplastic effects could potentially lead to long-lasting improvements in ejaculatory control by rewiring neural networks involved in the regulation of ejaculation.
The mechanism by which PES delays ejaculation likely involves both peripheral and central pathways. Peripheral low-frequency stimulation may prevent the typical patterned contractions of pelvic floor muscles by inducing subtetanic sustained contractions, disrupting the ejaculatory reflex arc. Central mechanisms may also be involved; by modulating afferent sensory input and engaging descending inhibitory pathways, PES might recalibrate excitability thresholds within spinal or supraspinal centers responsible for ejaculatory timing.
Given this dual possibility, future studies should aim to map the site of action—whether peripheral neuromuscular, spinal, or cortical—through neurophysiological assessments, functional imaging, or targeted interventions. Additionally, comparative trials assessing different stimulation frequencies, durations (acute vs chronic), and delivery modes (e.g., on-demand vs maintenance regimens) are necessary to optimize the stimulation protocols and define patient-specific indications.
Furthermore, future studies may benefit from incorporating physiological measures such as the index of twitch times to better quantify neuromuscular response. Although this index was not reported in any of the studies included in our review, its use could provide valuable insight into how electrical impulses interact with pelvic floor musculature and contribute to a more nuanced understanding of its therapeutic effects.
Despite the overall positive findings, several limitations affected the conduction and the results of this study. First, it is worth noting that the heterogeneity among the included studies was rather high, especially in terms of the stimulation parameters, such as frequency, intensity, and duration. Second, several included studies were published in languages other than English, making it impossible to retrieve methodological details, preventing thorough assessment of their methodological quality. Also, the long-term effects and durability of the observed improvements remain unclear, as most studies had relatively short follow-up periods. This highlights the need for high-quality, English RCTs with robust methodologies to further investigate the efficacy of PES for treatment of PE.
In conclusion, PES may represent a promising adjunctive therapy for the management of PE, with current evidence suggesting that it can prolong IELT. However, it is important to clarify that IELT should not be conflated with ejaculatory control, which refers to a patient’s subjective perception of their ability to delay ejaculation—a construct not directly measured in the included studies. As such, while PES appears effective in extending IELT, further research is needed to determine its effect on perceived ejaculatory control and overall sexual satisfaction. Future studies should include validated patient-reported outcomes and longer-term follow-up to comprehensively assess both physiological and subjective dimensions of treatment response.
Conclusion
While PES may hold potential as either a primary or adjunctive therapy for the management of PE, the evidence supporting its efficacy remains limited due to the low quality of studies and the significant heterogeneity observed among the available data. Although the findings suggest that PES interventions may improve IELT, further well-designed studies are needed to strengthen the evidence base, address the existing limitations, and more clearly define its clinical utility in individuals with PE.
Supplemental Material
sj-docx-1-tau-10.1177_17562872251382317 – Supplemental material for Peripheral electrical stimulation for premature ejaculation: a systematic review of clinical studies
Supplemental material, sj-docx-1-tau-10.1177_17562872251382317 for Peripheral electrical stimulation for premature ejaculation: a systematic review of clinical studies by Fateme Tahmasbi, Alireza Rahimi-Mamaghani, Farzin Soleimanzadeh, Omid Sedigh, Sarvin Sanaie and Mohsen Mohammad-Rahimi in Therapeutic Advances in Urology
Supplemental Material
sj-docx-2-tau-10.1177_17562872251382317 – Supplemental material for Peripheral electrical stimulation for premature ejaculation: a systematic review of clinical studies
Supplemental material, sj-docx-2-tau-10.1177_17562872251382317 for Peripheral electrical stimulation for premature ejaculation: a systematic review of clinical studies by Fateme Tahmasbi, Alireza Rahimi-Mamaghani, Farzin Soleimanzadeh, Omid Sedigh, Sarvin Sanaie and Mohsen Mohammad-Rahimi in Therapeutic Advances in Urology
Supplemental Material
sj-docx-3-tau-10.1177_17562872251382317 – Supplemental material for Peripheral electrical stimulation for premature ejaculation: a systematic review of clinical studies
Supplemental material, sj-docx-3-tau-10.1177_17562872251382317 for Peripheral electrical stimulation for premature ejaculation: a systematic review of clinical studies by Fateme Tahmasbi, Alireza Rahimi-Mamaghani, Farzin Soleimanzadeh, Omid Sedigh, Sarvin Sanaie and Mohsen Mohammad-Rahimi in Therapeutic Advances in Urology
Supplemental Material
sj-docx-4-tau-10.1177_17562872251382317 – Supplemental material for Peripheral electrical stimulation for premature ejaculation: a systematic review of clinical studies
Supplemental material, sj-docx-4-tau-10.1177_17562872251382317 for Peripheral electrical stimulation for premature ejaculation: a systematic review of clinical studies by Fateme Tahmasbi, Alireza Rahimi-Mamaghani, Farzin Soleimanzadeh, Omid Sedigh, Sarvin Sanaie and Mohsen Mohammad-Rahimi in Therapeutic Advances in Urology
Supplemental Material
sj-docx-5-tau-10.1177_17562872251382317 – Supplemental material for Peripheral electrical stimulation for premature ejaculation: a systematic review of clinical studies
Supplemental material, sj-docx-5-tau-10.1177_17562872251382317 for Peripheral electrical stimulation for premature ejaculation: a systematic review of clinical studies by Fateme Tahmasbi, Alireza Rahimi-Mamaghani, Farzin Soleimanzadeh, Omid Sedigh, Sarvin Sanaie and Mohsen Mohammad-Rahimi in Therapeutic Advances in Urology
Supplemental Material
sj-docx-6-tau-10.1177_17562872251382317 – Supplemental material for Peripheral electrical stimulation for premature ejaculation: a systematic review of clinical studies
Supplemental material, sj-docx-6-tau-10.1177_17562872251382317 for Peripheral electrical stimulation for premature ejaculation: a systematic review of clinical studies by Fateme Tahmasbi, Alireza Rahimi-Mamaghani, Farzin Soleimanzadeh, Omid Sedigh, Sarvin Sanaie and Mohsen Mohammad-Rahimi in Therapeutic Advances in Urology
Footnotes
Appendix
Acknowledgements
We would like to thank the Student Research Committee of Tabriz University of Medical Sciences and the Clinical Research Development Unit of Tabriz Valiasr Hospital, Tabriz University of Medical Sciences, Tabriz, Iran, for their support of this research.
Authors Note
Fateme Tahmasbi is now affiliated to Clinical Research Development Unit of Tabriz Valiasr Hospital, University of Medical Sciences, Tabriz, Iran.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
