Abstract
Introduction:
In recent years, several preliminary reports have suggested that the robot-assisted approach may decrease the surgical morbidity of artificial urinary sphincter (AUS) implantation in female patients with stress urinary incontinence (SUI). However, for now, only short-term outcomes have been reported. The present study aimed to report the 5-year outcomes of robot-assisted AUS implantation in female patients.
Patients and methods:
All female patients who underwent a robot-assisted AUS implantation between January 2014 and September 2019 at a single academic center were included in a retrospective study. All robot-assisted female AUS implantations performed after September 2019 were excluded to ensure a 5-year minimum follow-up duration. The indication for AUS implantation was SUI due to intrinsic sphincter deficiency. The primary endpoint was the explantation-free survival and revision-free survival.
Results:
Forty-two patients were included. The median age was 66 years (28–84), and 83.8% of the patients had a history of previous anti-incontinence procedure. After a median follow-up of 64 months (16–110), 8 patients were lost to follow-up before the 5-year time point. The 5-year estimated revision-free survival was 89.2 and the 5-year estimated explantation-free survival was 88%. Five AUS explantations were needed (11.9%), and six revisions were required (14.3%). The median time to explantation was 14 months. Four explantations (80%) occurred within the first 18 months, and all of them within the first 27 months. Thirty patients (71.42%) had a complete or improved continence with a complete continence rate of 59.52% and an improved continence rate of 11.9%. There were 10 intraoperative complications (23.8%): 5 bladder injuries and 5 vaginal injuries. Thirteen patients had postoperative complications (30.9%), but only two were Clavien grade ⩾3.
Conclusion:
The 5-year outcomes of robot-assisted AUS implantation seem to confirm the promising short-term outcomes that have been reported so far, although revision rates increased with time, which warrants further investigation.
Plain language summary
Keywords
Introduction
Stress urinary incontinence (SUI) is a frequent and debilitating symptom that can severely affect women’s quality of life. 1 Among available treatments, the artificial urinary sphincter (AUS) is an emerging therapeutic option in the treatment of SUI. 2 It is offered, in some parts of the world, to female patients with SUI due to intrinsic sphincter deficiency (ISD) which definition remains not consensual but generally encompasses a positive cough stress test with a fixed/poorly mobile urethra on physical examination, and a low maximum urethral closure pressure or Valsalva leak point pressure.3,4 One of the main drawbacks of female AUS is the lack of high-quality data to support its use.5,6 Despite female AUS has been used for over 40 years, very limited long-term data have been published so far. The few series that have studied the long-term outcomes of open abdominal female AUS implantation reported a variable continence rate ranging from 65.1% to 85.6%.7–9 A meta-analysis by Shokri et al. 10 suggested a lower rate of erosion with the laparoscopic approach. In addition, a recent retrospective study comparing the open and laparoscopic implantation techniques reported lower rates of revision and explantations, along with slightly better functional outcomes. 11 Several studies have demonstrated promising functional results with the robotic approach; however, the available data remain limited by short follow-up durations.12–14 To confirm and strengthen these findings, the present study aims to report the 5-year outcomes of robot-assisted AUS implantation in female patients.
Methods
Study design
After institutional board review (Commission Nationale de l’Informatique et des Libertés number: 2235713v0), the data of all female patients who underwent a robotic AUS implantation at a single academic center between January 2014 and September 2019 were collected prospectively in a dedicated computerized dataset.
Female AUS was offered as a surgical option for women with severe SUI due to ISD, defined by a positive cough stress test, a poorly mobile urethra on physical examination, and low maximum urethral closure pressure on urodynamic studies. Alternative treatment options, including fascial sling, bulking agent injections (Bulkamid®), and/or periurethral balloons (Adjustable Continence Therapy, ACT®), were proposed to all patients.
In accordance with current guidelines, preoperative evaluation of SUI included a clinical interview, physical examination with a cough stress test, free uroflowmetry, post-void residual (PVR) assessment by ultrasound, urodynamic studies, and validated questionnaires. 3 Regarding urodynamic parameters, Valsalva leak point pressure and cough leak point pressure were considered positive if any urinary leakage occurred during the respective tests. To evaluate the long-term functional outcomes with a minimum of 5-year follow-up duration for each patient, we excluded all robot-assisted female AUS implantations performed after September 2019. Robotic was the only surgical approach used for female AUS implantation over the study period. The database was analyzed retrospectively for the present study.
Data collection
The study was approved by the local ethics committee and was conducted following the principles of the Declaration of Helsinki. Due to the retrospective nature of the study, and regarding the national research guidelines, written consent was waived; however, patients were notified about the use of their data for research purposes and had the option to request its withdrawal. The study was not supported by the industry.
The following baseline characteristics were recorded in a dedicated computerized dataset for all patients: age at the time of AUS implantation, body mass index (BMI), ASA score (Physical status score of American Society of Anesthesiologists), etiology of incontinence (neurogenic vs non-neurogenic), history of radiotherapy, history of previous anti-continence surgery, number of pads per day, 24 h pad weight test, presence of urgency, maximum free urinary flow (Qmax), PVR volume, mean operative time, estimated blood loss, cuff size, and length of hospital stay.
If no data were available with a minimum of a 5-year follow-up, patients were contacted by phone to update the device status, the continence status, the overall satisfaction, the presence of post-operative urgency or frequency, and for any subsequent treatments for incontinence or overactive bladder (OAB). If the patient could not be contacted, she was considered lost to follow-up, as we do not have a minimum of a 5-year follow-up.
Perioperative management, surgical technique, and follow-up
All AUS were implanted using an anterior transperitoneal approach with robotic assistance, according to the technique previously described. 15
Briefly, after a 14 Fr bladder catheter has been inserted, the patient is placed in a 23° Trendelenburg position, 5 ports are placed according to the usual port placement for Intuitive® Da-Vinci robotic pelvic surgery, and a transperitoneal access is created (an extraperitoneal approach was used only in a handful of very selected cases). The Retzius space is dissected, dropping the bladder down from the abdominal wall to the endopelvic fascia on both sides of the bladder neck. The assistant then places a finger in one of the anterior vaginal fornix and the endopelvic fascia is opened laterally on the tip of the assistant finger and freed sufficiently to enter into the vesicovaginal space which is developed medially behind the posterior aspect of the bladder neck under direct vision. The same step is repeated on the contralateral side until the two spaces created are joined to place a measuring tape around the bladder neck, which is sized properly. The cuff is then inserted through the 12 mm port and placed around the bladder neck. The 61–70 cmH2O pressure regulating balloon (PRB) is inserted in the abdomen through a 3 cm subinguinal incision, which is also used to insert the pump in the labia majora and to make the connections.
The urethral catheter was removed in the operating room in case of day surgery or on day 1 postoperatively, except in case of bladder injury, where it was kept for 10–14 days. The AUS was activated at 6 weeks during a 6- to 10-h hospitalization. The patients were taught how to manipulate the pump and had to perform multiple voids under the close supervision of a specialized nurse.
In the case of bladder neck or vaginal injuries, procedure is not interrupted and the wound is repaired layer by layer and a peritoneal flap is interposed between the wound and the cuff. However, in the case of concomitant vaginal and bladder injuries, implantation is canceled.
At 3 months, the PVR and pump manipulation were systematically reevaluated. Mechanical failure was screened at each visit using suprapubic ultrasound of the PRB diameter. Subsequently, all female patients underwent a yearly follow-up visit in clinics with the same protocol.
Outcomes of interest
The primary endpoints of the present study were the explantation-free and revision-free survivals. Explantation was defined as any surgical intervention requiring complete removal of the AUS without immediate reimplantation (infection, erosion, decreased bladder capacity/compliance). Revision was defined as any surgical intervention consisting of replacing one or more components of the device (pump, balloon, tube, cuff, or connectors). A revision of the device was performed for mechanical or non-mechanical failure. On rare occasions, it was used to relocate the pump after difficulties manipulating it or because of pain in a seated position. Reoperation was defined as either revision or explantation.
The secondary endpoints were (1) the continence status at last follow-up as self-reported by the patient on clinical interview, categorized as: complete resolution of SUI (0 pad), improved SUI (1 pad), or unchanged SUI (>1 pad). (2) Continence status at 3 months postoperatively. (3) Comparison between patients’ reported outcomes. (4) Postoperative complications. (5) Major postoperative complications defined as a Clavien-Dindo Grade of 3 or higher. (6) Device status at last follow-up and any event of revision or explantation.
Postoperative complications were recorded and graded according to the Clavien-Dindo classification and were assessed during the 3-month postoperative period. 16
The last follow-up was defined as the most recent date on which information was gathered, either during an outpatient clinic visit or by phone call to update the patient’s data.
Statistical analysis
Means and standard deviations were reported for continuous variables, medians and ranges for categorical variables, and proportions for nominal variables. Comparisons between groups were performed using the χ2 test or Fisher’s exact test for discrete variables, and the Mann–Whitney test for continuous variables as appropriate. Change of continuous variables over time was assessed using the paired Student’s t-test.
The probability of revision-free and explantation-free survivals was estimated using the Kaplan–Meier method. Patients without any event (revision or explantation) during the study period were censored at the date of the last follow-up available as per the principle of the Kaplan–Meier analysis. For the functional outcomes, their continence status at last follow-up was reported at the 5-year timepoint. Statistical analyses were performed using JMP v.18.0 software (SAS Institute Inc., Cary, NC, USA).
All tests were two-sided with p < 0.05 as a threshold to define statistical significance.
Results
Patients’ characteristics
Forty-two patients were included in the present analysis. A total of eight patients were lost to follow-up before the 5-year time point. The patients’ characteristics are summarized in Table 1. The median age was 66 years (range: 28–84). The mean body mass index was 27.3 kg/m2 (±4.6). There was no patient with a history of pelvic radiotherapy, and only three patients had neurogenic SUI. Most of the patients had a history of previous anti-incontinence surgery (83.3%), with 71.4% having a history of mid-urethral slings and 26.2% having a history of Burch colposuspension. The median number of pads per day was 3 (1–20), and the median 24-h pad weight test of 375 g (range: 40–1000). Regarding urodynamic parameters, there were seven patients with detrusor overactivity (17%), and the mean maximum cystometric capacity was 412.6 ml (±131.7).
Patients’ characteristics.

Perioperative and postoperative outcomes
The perioperative outcomes are presented in Table 2. Six patients underwent concomitant mesh sacrocolpopexy (14.3%). The mean operative time was 187.4 min (±54), the median AUS cuff size was 75 mm (range: 50–90), and the mean estimated blood loss was 84.7 ml (±134.3). There were 10 intraoperative complications (23.8%) with 5 bladder injuries and 5 vaginal injuries. Of the 13 postoperative complications, only 2 were ⩾3 were Clavien-Dindo grade: 1 vaginal erosion and 1 urethrovaginal fistula.
Perioperative outcomes.
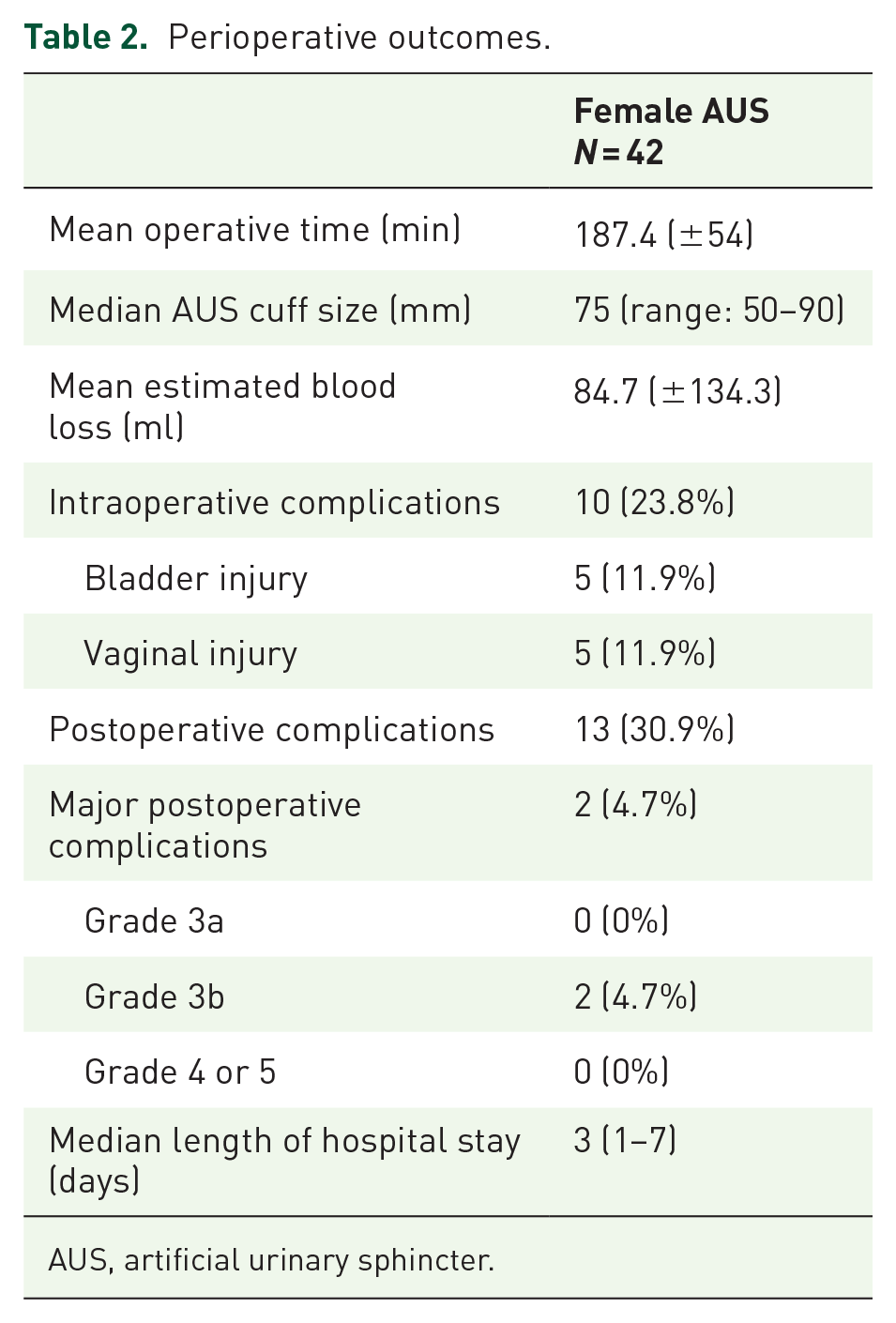
AUS, artificial urinary sphincter.
Device outcomes
The median follow-up was 64 months (range: 16–110). There were eight patients lost to follow-up before the 5-year time point (19%), including two who had been permanently explanted. There were five explantations over the study period (11.9%): three for vaginal exposure of the cuff (60%), one for urethral extrusion of a midurethral sling next to the cuff, and one for a labial exposure of the pump, likely due to a subclinical infection of the device. The median time to explantation was 14 months. Four explantations (80%) occurred within the first 18 months with one of them in the first 3 months (vaginal erosion) and all of them within the first 27 months. The 5-year estimated explantation-free survival was 88% (Figure 1).

Explantation-free survival.
There were six revisions (14.3%): four for mechanical failures (66.7%), one pump relocation, and one non-mechanical failure. Only one revision occurred during the first 3 months (pump relocation). The median time to revision was 38 months, and the last revision occurred at 99 months. The 5-year estimated revision-free survival was 89.2% (Figure 2). The 5-year estimated reoperation-free survival was 79.9% (Figure 3). At the last follow-up available, 31 patients still had their device in situ and functioning (73.8%), two had a new AUS reimplanted after explantation, three had their AUS permanently explanted, and six had their initial AUS in situ but non-functioning and permanently deactivated (14.3%) because of difficulties to manipulate the pump, two of which occurred upfront and four occurring over the course of the follow-up.

Revision-free survival.

Reoperation-free survival.
Functional outcomes
At 3 months postoperatively, 78.6% of patients were fully continent, and 4.76% of the patients described an improved continence. Four patients (9.52%) had an unchanged continence. At last follow-up, the continent rates were lower: 30 patients (71.42%) had a complete or improved continence with a complete continence rate of 59.52% and an improved continence rate of 11.9%. Twelve patients had an unchanged continence. Overall, the functional outcomes remained roughly stable over time (Table 3), with 83.3% of patients improved at 3 months (PGII 1–3) versus 78.7% at the last follow-up available. Only the USP OAB subscore increased significantly between the 3 months and the last follow-up timepoint (1.3 vs 4.3; p = 0.02). Eighteen patients required treatment for OAB at some point during follow-up (42.9%), and five required a third-line therapy (sacral neuromodulation or botulinum toxin injection) for OAB postoperatively (11.9%).
Changes in patient-reported outcomes between baseline, 3-month, and the last follow-up available.

p < 0.05 compared to baseline. **p < 0.05 compared to the 3 months post-op results.
ICIQ-SF, International Consultation on Incontinence Questionnaire – Short Form; OAB, overactive bladder; PGII, Patient Global Impression of Improvement; USP score, Urinary Symptoms Profile score.
Discussion
This study presents long-term outcomes of robot-assisted AUS implantation in women, considering that this approach may reduce surgical morbidity and that long-term data on robotic implantation techniques remain scarce. We found a satisfactory continence rate at the 5-year follow-up, but this must be weighed against the observed complications: the rate of late complications in this series was significant, especially when it came to revision. When comparing early postoperative continence rates with those at the last follow-up, we observed a lower continence rate in the long-term group as expected. This raises important questions about potential factors contributing to late failure and complications.
Many factors could explain both early and late device failures. First, difficulties in manipulating the pump were quite common, resulting in permanent deactivation in six patients (14.3%). Patients who are unable to operate the pump due to a lack of dexterity, cognitive dysfunction, age-related conditions, or high BMI can have incomplete bladder emptying, urinary tract infections, or persistent urinary leakage. One may postulate that improvement in patients’ selection with increasing experience and the upcoming surge of electromechanical sphincters may improve the long-term failures due to difficulty in manipulating the pump. 17
Another possible explanation for functional failure is bladder neck atrophy, though it seems to be much rarer in the female population than in males. 10 The mechanism of atrophy in female patients remains poorly understood and is likely under-evaluated. In the cohort by Chartier-Kastler et al., 18 the lower median cuff size in the revision group is in favor of a potential existence of atrophy over time.
Mixed incontinence and de novo OAB are more challenging causes of failure, as their mechanisms remain unclear, and the recurrence of urinary leakage can be a difficult experience for patients after AUS implantation. In concordance with Vayleux et al., 9 a high proportion of patients in our cohort required treatment for OAB postoperatively (42.9%). Some of the first-line and second-line treatments required have been evaluated in this specific population and have shown good results.19,20
There are several potential explanations for the development of OAB. First, many patients have a history of suburethral sling procedures or Burch colposuspension. Since OAB symptoms can be a consequence of bladder outlet obstruction, 21 it is reasonable to assume that prior surgeries may contribute to some degree of chronic bladder outlet obstruction, which could explain the subsequent development of OAB unmasked after the AUS implantation. Second, while the AUS is generally considered to allow for physiological voiding with low urethral resistance when the cuff is opened, this remains a hypothesis, and the AUS could potentially cause anatomical bladder outlet obstruction. Finally, the bladder neck dissection, which occurs close to the trigone where many afferent nerve fibers are located, may damage these fibers, triggering bladder overactivity.
In line with Bazinet et al., 22 the rate of revision was relatively high (14.3%), with a vast majority of mechanical failure. This finding may hypothetically be explained by the intraoperative manipulation of the AUS tubing with the robotic tools, which lack tactile feedback. One may postulate that the tubing might be weakened by the robotic manipulation. They may also be compromised during the extraction and insertion through the subinguinal incision. The long-term revision rate after robotic female AUS implantation should therefore remain under scrutiny in the next few years.
Most explantations occurred within the first 18 months, and all occurred within the first 27 months. Hence, any series reporting outcomes of female AUS with a median follow-up of over 30 months may provide a good overview of the risk of erosion/explantation. Our explantation rate is in line with or slightly lower than open approaches series reported in the literature.8,9 While urethral erosion is the main issue, it does not seem to be the case in female patients, which is likely due to the bladder neck location of the cuff, the bladder neck wall being much thicker and stronger than the bulbar urethra in male patients. In fact, vaginal exposure was the main cause of explantation. This finding is of interest as the reasons for explantation were rarely detailed in the previous studies on female AUS. This may be due to the vaginal atrophy and more fragile tissues commonly seen in women during the peri-menopausal period. The potential benefit of preoperative or long-term local estrogen therapy to improve tissue health and reduce the risk of erosion warrants consideration. Identifying risk factors for erosion in the future could also support the use of a concomitant flap during implantation to mitigate this risk.
Another important factor that may contribute to treatment failure is the high rate of previous incontinence surgeries. While our data do not allow us to draw definitive conclusions, we believe that patients with a history of multiple incontinence procedures, urethral or bladder neck mesh exposure, or bladder neck exstrophy represent complex cases that should be avoided during the initial phase of the learning curve. The number of previous incontinence procedures, the presence of associated neurogenic disease, and simultaneous augmentation procedures have been identified as factors associated with device survival by Costa et al. 8
Our study has several limitations that should be noted. First, it is a retrospective analysis conducted at a single center, which introduces inherent biases. The series includes our initial implantations, which may have led to poorer outcomes due to the early stage of development of this technique. We were unable to include open AUS implantations, as only a limited number have been performed at our center. Providing a comparison of different approaches with longer follow-up would be valuable to further support the use of the robotic approach. Our secondary endpoint remains limited as there is no standardized definition of continence, and it does not properly reflect patients’ overall quality of life, and we believe that patients with incomplete continence can still benefit from this treatment. We did not standardize the evaluation of de novo OAB; therefore, the question of whether these high rates of OAB are due to unmasked overactivity of the bladder reservoir versus de novo OAB due to poor manipulation of the pump or other factors has not yet been determined. Similarly, we did not define clear criteria for evaluating bladder neck atrophy, potentially leading to its underestimation. Further studies with larger populations, a multicenter design, and extended follow-up are needed to generate robust data that could support broader adoption of this approach and potentially influence guidelines. Such studies may also help identify risk factors for failure and erosion, thereby improving patient selection for female AUS implantation in the long term. This would contribute to reducing complication rates and assist surgeons, particularly those at the beginning of their learning curve, in selecting appropriate candidates more effectively. Additional research on de novo OAB with urodynamic parameters would be of interest to address these unresolved questions and reduce failure rates.
Conclusion
The 5-year outcomes of robot-assisted AUS implantation seem to confirm the promising short-term outcomes that have been reported so far, although revision and erosions rates increased with time. These results highlighted the challenges of mechanical failures, vaginal exposure of the cuff, and de novo OAB which warrants further investigation.
Footnotes
Acknowledgements
None.
