Abstract
Renal cell carcinoma (RCC) represents 2.2% of all malignancies worldwide; however, its mortality rate is not negligible. Surgery is the primary treatment for most nonadvanced cases, with its indications and techniques evolving over the years. To provide an update on RCC management in Brazil, focusing on surgery. The Latin American Cooperative Oncology Group-Genitourinary Section and the Latin American Renal Cancer Group gathered a panel of Brazilian urologists and clinical oncologists to vote on and discuss the best management of surgically resectable RCC. The experts compared the results with the literature and graded them according to the level of evidence. For small renal masses (SRMs; less than 4 cm), biopsy is indicated for specific/select cases, and when intervention is needed, partial nephrectomy should be prioritized. Radical nephrectomy and ablative techniques are exceptions for managing SRMs. Patients with small tumors (less than 3 cm), slow tumor growth, or a risk for surgery may benefit from active surveillance. Localized carcinoma up to 7 cm in diameter should be treated preferably with partial nephrectomy. Lymphadenectomy and adrenalectomy should be performed in locally advanced cases if involvement is suspected by imaging exams. Patients with venous tumor thrombi usually require surgical intervention depending on the extent of the thrombus. Neoadjuvant therapy should be considered for unresectable cases. Even in the era of targeted therapy, cytoreductive nephrectomy still has a role in metastatic disease. Metastasectomy is indicated for most patients with resectable disease. This consensus presents recommendations for surgical treatment of RCC based on expert opinions and evidence from the medical literature. Surgery remains the best curative option for nonadvanced cases, and it still has a role for select patients with metastatic disease.
Keywords
Introduction
Kidney cancer represents 2.2% of all malignancies worldwide, with approximately 431,288 new cases in 2020 1 and 200,000 deaths in 2022. 2 Brazil registered approximately 10,000 new cases of kidney cancer and 5000 deaths in 2020, with an estimated 5-year prevalence of 31,000, including both sexes. 3
Clear cell renal cell carcinoma (RCC) is the most common type of kidney cancer, representing up to 70% of all RCC cases. 4 The leading cause of RCC remains unknown, but older age, a history of chronic renal disease, arterial hypertension, diabetes, obesity, smoking, and some hereditary conditions increase the risk. 2
Over time, the mortality of patients with RCC has decreased, 5 probably due to the increase in early detection and treatment methods. Treatment of RCC depends on the stage, and surgery is the main treatment option for patients without advanced disease. However, indications and techniques concerning surgery have changed over the past few years, and new evidence has become available, though other topics remain undefined. This paper provides an update on the Brazilian consensus on management of RCC, focusing on surgery, which was first published in 2019. 6
Methods
The Latin American Cooperative Oncology Group-Genitourinary Section (LACOG-GU) and the Latin American Renal Cancer Group (LARCG) selected 72 Brazilian clinical oncologists and urologists to compose a panel of experts based on their expertise. All of these professionals are board certified by the Brazilian Societies of Clinical Oncology and Urology and are members or collaborators of the LACOG and/or LARCG. The vast majority of them develop academic activities or have participations in clinical trials. Many of them already participated in the previous consensus publication.6,7 This paper is an update of the RCC management consensus in Brazil, which were last published in 2019 and 2020.6,7 The outcomes of these deliberations have been documented in two separate papers. This paper focuses on surgery and was mainly assembled by urologists, except for advanced/metastatic disease and metastasectomy topics, in which both specialists participated.
The questionnaire included 162 multiple-choice questions and was developed by two authors (S.C.Z. and A.S.), based on an extensive literature review over the past 3 years, and reviewed by two other authors (M.L.W. and W.H.C.). Each question presented an “abstain” option for those who were uncertain or unable to answer.
The panelists answered the questionnaire between February and July 2023 through an online platform. An independent facilitator compiled the responses, considering answers chosen by at least 75% of the participants as a consensus. A virtual meeting was convened to address questions for which a consensus was not reached, followed by a second round of voting. Once again, responses selected by at least 75% of the participants were considered a consensus; the most frequently chosen answer was deemed a recommendation endorsed by most of the panel when consensus was not reached. The complete questionnaire, along with the detailed results, can be found in the Supplemental Material.
The selected answers were cross-referenced with findings from the medical literature that was carried out using the Pubmed, Google Scholar, and Cochrane Library databases. Inclusion criteria were meta-analysis of randomized and nonrandomized clinical trials or observational studies, randomized or nonrandomized, double-blind or open clinical trials, and observational studies on surgical treatment of RCC. Nonsystematic review articles, case reports, preclinical studies, and conference abstracts were excluded. The quality of the studies was assessed using a modified version of the Oxford Levels of Evidence classification 8 (Tables 1 and 2).
Classification of the level of evidence, as adapted from the Oxford classification 2009. 8

The grade of recommendation was adapted from the Oxford classification 2009. 8

Results
A summary of the main findings as a result of this consensus update is presented in Tables 3–6.
Summary of small renal mass management.
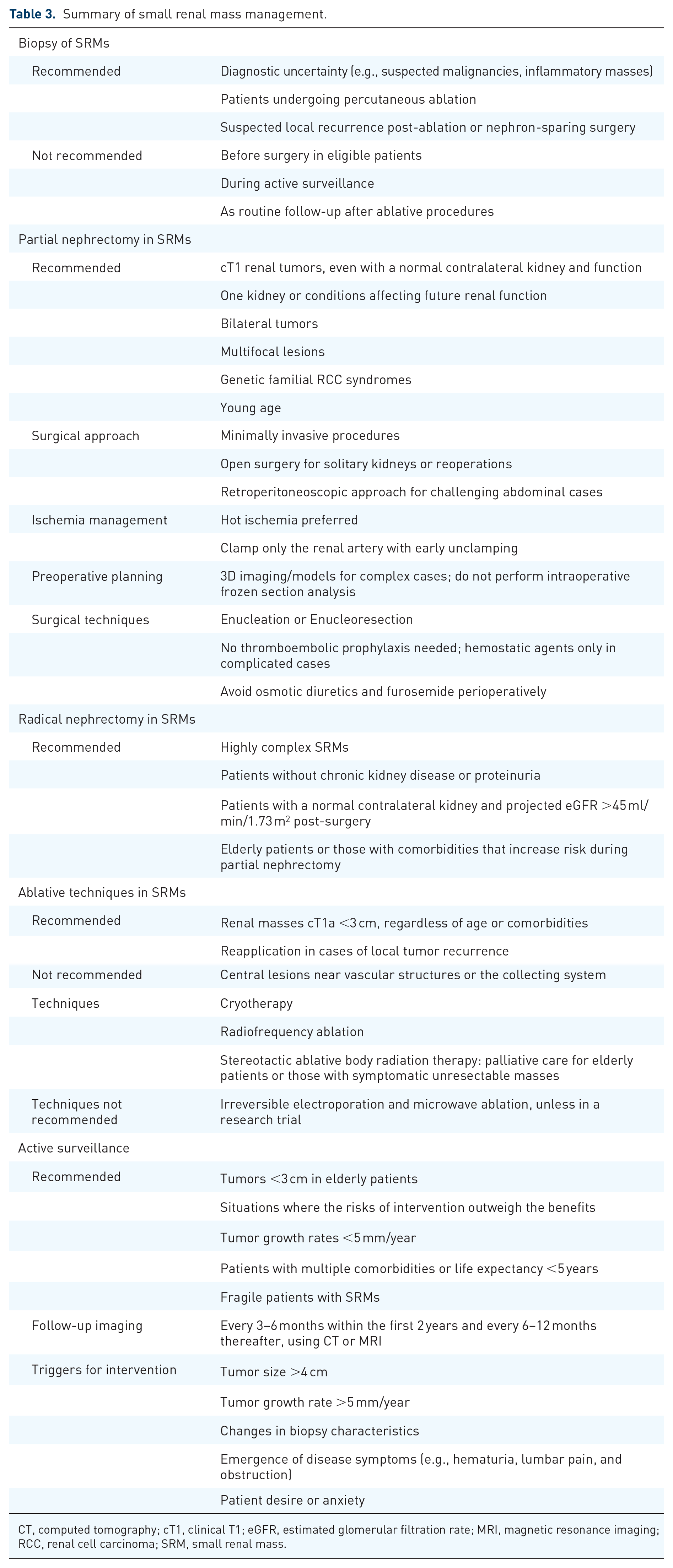
CT, computed tomography; cT1, clinical T1; eGFR, estimated glomerular filtration rate; MRI, magnetic resonance imaging; RCC, renal cell carcinoma; SRM, small renal mass.
Summary of localized renal cell carcinoma management.

RCC, renal cell carcinoma.
Summary of locally advanced carcinoma management.

CT, computed tomography; MRI, magnetic resonance imaging.
Summary of advanced/metastatic disease management.

CN, cytoreductive nephrectomy; IMDC, International Metastatic renal cell carcinoma Database Consortium; LND, lymphadenectomy; MDACC, MD Anderson Cancer Center.
Management of small renal masses
Biopsy of small renal masses
Small renal masses (SRMs) are defined as a tumor with up to 4 cm as the largest diameter and present a lower risk of malignancy than larger tumors. 9 The panel recommends biopsy for SRMs when there is diagnostic uncertainty, as seen in suspected renal infiltration due to lymphoproliferative disease, metastasis (another primary tumor), or suspicion of an inflammatory mass (consensus; level of evidence (LE): 5, Grade (GR): D). Biopsy is also required for patients receiving percutaneous ablation modalities (consensus; LE: 2b, GR: B).10,11 Suspicion of local recurrence after percutaneous ablation or nephron-sparing surgery should also lead to biopsy (consensus; LE: 5, GR: D).
The panel does not recommend biopsy before the procedure (good performance status and no comorbidities) in patients who are candidates for surgery (consensus; LE: 5, GR: D) or in patients who are candidates for active surveillance (consensus; LE: 2b, GR: B).12,13 Biopsy should not be routinely carried out in follow-up after ablative procedures (consensus; LE: 5, GR: D), during active surveillance (change of tumor characteristics; trigger for intervention; consensus; LE: 1a, GR: A), 12 or for complex renal cysts with a solid area <2.0 cm (consensus; LE: 5, GR: D).
Renal mass biopsy should be carried out under computed tomography (CT) and/or ultrasound guidance, according to availability (consensus; LE: 2b, GR: B).10,11 The panel recommends conscious sedation and/or local anesthesia (consensus; LE: 2b, GR: B).10,14 A minimum of two fragments should be collected (consensus; GR: 2b)15–17 from both the central and peripheral regions of the mass (consensus; LE: 2b, GR: B). 18
Partial nephrectomy in SRMs
The panel recommends performing partial nephrectomy (PN) for clinical T1 (cT1) renal tumor management when the intervention is indicated, even in patients with a normal contralateral kidney and normal renal function (consensus; LE: 2b, GR: B).19,20 PN is highly effective for treating T1aN0M0 RCC, with cancer-specific mortality comparable to that of radical nephrectomy (RN) and a lower risk of developing chronic kidney disease and its associated comorbidities.19,20
Considering these findings, nephron-sparing procedures should be prioritized for patients with:
only one kidney, one functional kidney (consensus; LE: 2a, GR: B),21,22 chronic kidney disease, proteinuria, imminent renal insufficiency, or any other conditions that might affect renal function in the future (consensus, LE: 2a, GR: B) 23 ;
bilateral tumors (consensus; LE: 2a, GR: B) 24 ;
multifocal tumors (consensus; LE: 5, GR D);
genetic familial RCC syndrome (consensus; LE: 2b, GR: B)25,26;
young age (consensus; LE: 2b, GR: B). 27
For patients with multiple multifocal tumors, where the removal of all tumors could leave nonfunctional residual kidney tissue, RN may be preferred, particularly if the patient has a normally functioning contralateral kidney.
In older or frail patients with limited life expectancy, such as those with extensive atherosclerosis in the renal arteries, nephron-sparing surgery may be more complex and carry a higher risk. In these cases, RN is often a better option than partial surgery.
For patients with hereditary RCC syndromes, nephron-sparing surgery should be deferred until the tumor diameter exceeds 3.0 cm. The panel recommends considering nephron-sparing procedures in patients with familial RCC syndromes and masses larger than 3 cm (consensus; LE: 2b, GR: B),28,29 except for aggressive RCC syndromes, such as hereditary leiomyomatosis and RCC syndrome and fumarate hydratase-deficient RCC, in which the surgical approach must be indicated immediately, independent of the tumor size (consensus; LE: 5, GR: D).
Regarding the surgical approach, minimally invasive procedures should be considered standard (consensus; LE: 2b, GR: B).30–32 The panel recommends considering nephron-sparing procedures even for patients with highly anatomically complex tumors (consensus; LE: 2b, GR: B),33,34 and there are advantages of robotic-assisted surgery in these cases (consensus; LE: 2b, GR: B).33–35 Robotic-assisted PN has a lower incidence of complications than open surgery, such as infections and bleeding (consensus; LE: 2a, GR: B), 36 though the operative time is usually longer (recommendation, LE: 2a, GR: B). 36 Open surgery is no longer the standard for PN (consensus; LE: 2b, GR: B),30–32 but it is still appropriate, especially for patients with a solitary kidney and patients who undergo reoperation (consensus; LE: 5, GR: D).
In the case of a “hostile peritoneal cavity,” with multiple previous abdominal surgeries and scars or patients with unfavorable body habitus or with posterior tumors, the panel recommends a retroperitoneoscopic approach (consensus; LE: 2a, GR: B). 37
Ischemia during nephrectomy is a topic that remains unclear and debated. During PN, the panel recommends hot ischemia for most cases (consensus; LE; 5, GR: D), with an ideal total ischemia time of up to 25–30 min (consensus; LE: 5, GR: D). However, in specific situations, such as in patients with high nephrometric scores and expected ischemia times >25–30 min, the use of cold ischemia could be considered (consensus; LE: 2a, GR: B). 38
During nephrectomy, only the renal artery should be clamped (consensus; LE: 2b, GR: B), resulting in more preserved renal function and a lower complication rate.39,40 Clamping both vessels, that is, the renal artery and vein, is indicated only in challenging cases, such as central tumors or tumors with high nephrometric scores and the risk of intrarenal venous bleeding (consensus; LE: 5, GR: D). The panel recommends early artery unclamping (after medullary suture; consensus; LE: 2b, GR: B) 41 and off-clamp or zero ischemia (clampless surgery) only for select cases (consensus; LE: 5, GR: D).
Use of 3D images/3D-printed models should be considered for preoperative surgical planning in challenging cases, such as in patients with centrally located tumors, high morphometric scores, nearness of secondary blood vessels or excretory systems, a solitary kidney, or multiple tumors in the same organ, or who underwent previous surgeries or ablative procedures (consensus; LE: 2a, GR: B). 42
The panel does not recommend performing intraoperative frozen section analysis of the margins or surgical bed of PNs (consensus; LE: 5, GR: D).
The panel recommends enucleation and resection (enucleo-ressection) versus wedge resection (consensus; LE: 5, GR: D).
Thromboembolic event prophylaxis is not indicated for patients undergoing PN (consensus; LE: 5, GR: D). Hemostatic agents should be used intraoperatively only for complicated cases with difficult intraoperative hemostasis (consensus; LE: 5, GR: D). The panel does not recommend use of osmotic diuretics, mannitol (consensus; LE: 2b, GR: B), 43 or furosemide (consensus; LE: 5, GR: D) perioperatively.
RN: what is its role in SRMs?
RN can be an option for surgical management of highly complex SRMs (consensus; LE: 2a, GR: B).44,45 In these cases, the lower incidence of surgical complications outweighs the significance of preserving parenchymal tissue. 45 RN can be performed in patients with no previous chronic kidney disease or preexisting proteinuria (consensus; LE: 5, GR: D) and in patients with a functional normal contralateral kidney and a projected estimated glomerular filtration rate greater than 45 ml/min/1.73 m2 after surgery (consensus; LE: 5, GR: D).
RN is an option for elderly patients and patients with comorbidities that contraindicate renal artery clamping or who are at high risk of complications after PN (consensus; LE: 5, GR: D).
Ablative techniques: when should they be considered in SRMs?
Ablative techniques, such as cryo- and radiofrequency ablation, are gaining relevance in treatment of cT1 RCCs; however, cryoablation seems to have advantages when comparing the two ablation techniques,15,46,47 despite its higher costs.
Cryoablation results in oncological outcomes comparable to those of PN, with a 5% complication rate. 15 Both cryotherapy and radiofrequency ablation are viable options (consensus; LE: 2b, GR: B),46,47 with percutaneous access as the preferred approach (consensus; LE: 2a, GR: B). 48
The panel recommends ablative techniques for renal masses with cT1a smaller than 3 cm (consensus; LE: 2b, GR: B),46,49 regardless of age (recommendation; LE: 5, GR: D) or comorbidity status (consensus, LE: 2a, GR: B). 50
In 4%–8% of cases, reapplication of ablative techniques is necessary. 51 In the case of local tumor recurrence, ablation techniques can be reapplied (consensus; LE: 2a, GR: B).15,51 However, ablative techniques should not be applied in central lesions near vascular structures or the collecting system (recommendation; LE: 5, GR: D).
Irreversible electroporation and microwave ablative modalities are not recommended in clinical practice, unless they are part of a research protocol/trial (consensus; LE: 2b, GR: B). 52
Stereotactic ablative body radiation therapy can be implemented in a palliative setting, in elderly or frail patients, for intracranial metastasis, and for poor surgical candidates with symptomatic unresectable large renal masses (consensus; LE: 2a, GR: B).53,54
Active surveillance: for whom is it indicated?
In localized renal lesions, especially SRMs, active surveillance—defined as the initial observation of the tumor by serial imaging, potentially resulting in no or a delayed intervention—is a safe and well-established option for a carefully selected and well-informed group of patients. 55 Nevertheless, structured protocols for patient selection, intervention triggers, and long-term safety studies are still lacking; thus, active surveillance remains underutilized.55,56
Active surveillance is indicated, especially for suspicious SRMs <3 cm (consensus; LE: 2a, GR: B), 57 and can be considered for tumors less than 2 cm in size, regardless of patient age. 58
The panel recommends active surveillance as primary disease management for suspected renal cancer masses, particularly those under 3 cm (consensus; LE: 5, GR: D). However, active surveillance is not appropriate for all small kidney tumors. A minority of SMRs (10%–12%) can harbor high-risk or high-grade tumors, making prior biopsy advisable. 59
Active surveillance is recommended for tumors smaller than 3 cm in elderly patients (consensus; LE: 2a, GR: B). 57 Expectant management is indicated when the risks of any procedure or intervention and the risk of death override the benefits of oncologic treatment (consensus; LE: 5, GR: D), when the tumor growth rate is <5 mm/year (consensus; LE: 2b, GR: B), 20 for patients with multiple comorbidities (consensus; LE: 2a, GR: B), 57 for those with a life expectancy <5 years (consensus; LE: 2b, GR: B), 12 and for fragile patients with SRMs (consensus; LE: 2b, GR: B). 58
For younger patients without comorbidities, upfront surgery is often preferred over active surveillance. Surgical treatment of SRMs can help avoid the numerous risks and inconveniences associated with long-term active surveillance protocols, such as anxiety, radiation exposure, iodinated contrast infusions, and allergies. For healthy patients, surgery typically results in few significant postoperative complications or morbidities. Some authors suggest that there is insufficient evidence to support active surveillance for SRMs in young patients, indicating a need for more studies in this area. 60
Regardless of the size of the mass, active surveillance should not be regarded as the standard treatment when solely considering factors such as marginal renal function (recommendation; LE: 5, GR: D) or low-grade tumors (consensus; LE: 2b, GR: B). 20
As mentioned above, follow-up protocols for active surveillance are still lacking, and the best interval for optimizing the risk–benefit relationship has yet to be defined. 56 Based on currently available data, the panel recommends performing follow-up imaging every 3–6 months within the first 2 years (consensus; LE: 2b, GR: B) 61 and every 6–12 months thereafter (consensus; LE: 2b, GR: B) 61 with CT or magnetic resonance imaging (MRI; consensus; LE: 2a, GR: B). 62 Ultrasound is not recommended (recommendation; LE: 2a, GR: B), 55 despite being utilized in some centers (Jonh Hopkins Active Surveillance Program).
The triggers for delayed intervention during active surveillance have also not been clearly defined, and further research is needed. The panel recommends considering the following events as triggers for intervention:
tumor size >4 cm (consensus; LE: 2a, GR: B) 62 ;
tumor growth rate > 5 mm/year (consensus; LE: 2a, GR: B) 62 ;
changes in biopsy characteristics (consensus; LE: 5, GR: D);
emergence of disease symptoms (hematuria, lumbar pain, obstruction; consensus; LE: 2a, GR: B) 62 ;
patient desire or anxiety (consensus; LE: 2a, GR: B). 62
Localized RCC
PN for localized tumors
PN is an option for
RN for localized tumors
The panel recommends RN for localized RCC greater than 7 cm in size (recommendation; LE: 2a, GR: B) 45 and for tumors with high anatomic complexity (consensus; LE: 5, GR: D). RN should be carried out in cases in which lymph node involvement is suspected (recommendation; LE: 2b, GR: B), 66 for masses with venous tumor thrombus (consensus; LE: 2b, GR: B), 67 and in cases with direct extension to the ipsilateral adrenal gland (consensus; LE: 2b, GR: B). 68 The panel also recommends RN for localized tumors within a functionally excluded kidney affected by a tumor (consensus; LE: 5, GR: D) or at the surgeon’s discretion (consensus; LE: 5, GR: D).
RN is not the standard of care for tumors ⩽7 cm (consensus; LE: 2b, GR: B). 69
Locally advanced carcinoma
Lymphadenectomy for locally advanced tumors
The role of lymphadenectomy (LND) during RN seems to be beneficial only for a select group of patients, and performing it routinely is not recommended, as it does not improve oncological outcomes.70,71
Current data consistently agree that LND allows for better pathological staging but does not yield any therapeutic benefit (consensus; LE: 2b, GR: B).70,71
Based on available data, the panel does not recommend LND in the following cases:
localized tumors (cT1/cT2; consensus; LE: 2b, GR: B) 72 ;
locally advanced disease (cT3/cT4), with no suspicion of lymph node involvement on abdominal CT or MRI (recommendation; LE: 2b, GR: B) 66 ;
Mayo Clinic: grade III/grade IV/necrosis/T3–4/sarcomatoid/rhabdoid (recommendation; LE: 2b, GR: B), 73 as most of this information is available only after surgery and it is difficult to make intraoperative decisions based on frozen section analysis for these pathological features.
LND should be performed for patients with clinically suspected involved nodes (cN+) identified by imaging methods (consensus; LE: 2b, GR: B). 74 The panel recommends using nomograms for preoperative decision-making (recommendation; LE: 2b, GR: B) 75 and intraoperative inspection and detection of lymphadenomegaly (consensus; LE: 2b, GR: B).76,77
If LND is performed, it should encompass the perihilar template (recommendation; LE: 5, GR: D) following the following anatomical limits:
lateral and anterior: large ipsilateral vessels (consensus; LE: 2b, GR: D)76,77;
cephalic: upper mesenteric artery (consensus; LE: 5, GR: D);
distal: lower mesenteric artery (recommendation; LE: 2b, GR: B). 77
Adrenalectomy in locally advanced tumors
The indications and outcomes of concomitant adrenalectomy during RN are relatively underexplored in the literature. 78 Adrenal involvement is becoming more infrequent with earlier diagnosis of RCC. 79 Nevertheless, concomitant ipsilateral adrenalectomy remains overused. 80 Thus, the reasons for removing the adrenal gland during nephrectomy have yet to be clarified. 81
According to available data, adrenalectomy is only indicated in the following situations:
if a solitary adrenal metastasis or tumor invasion is identified by imaging (CT or MRI; consensus; LE: 2b, GR: B) 82 ;
intraoperative findings suggest adrenal invasion (consensus; LE: 2b, GR: B). 82
Considering the low level of evidence on this subject, the panel does not recommend routinely performing adrenalectomy during nephrectomy even if the lesion is in the upper pole and > 7 cm (recommendation; LE: 2b, GR: B),83,84 in tumors not confined to the upper pole (T3 or greater; recommendation; LE: 5, GR: D), in tumors with perirenal fat invasion (consensus; LE: 5, GR: D), or in cases with intraoperative lymph node invasion (recommendation; LE: 5, GR: D).
The most reliable diagnostic method for identifying adrenal invasion and deciding on concomitant adrenalectomy remains CT or MRI, as they have the highest positive predictive value for this diagnosis (consensus; LE: 2b, GR: B). 85
Special situations in locally advanced RCC
Venous thrombus
RCC extending into the venous system—the renal vein, inferior vena cava (IVC), or even the right atrium—is a rare but aggressive scenario with poor prognosis.86–88 Invasion of the venous wall or the presence of adverse pathological factors such as advanced stage ⩾pT3, high grade, necrosis, lymph node involvement, and sarcomatoid features compromise the prognosis of patients with RCC with venous tumor thrombus (consensus; LE: 2b, GR: B).88,89
Available data corroborate the panel’s recommendation of performing thrombectomy simultaneously at the time of RN for patients with evidence of a thrombus involving the IVC or the right atrium (i.e., cavoatrial involvement; consensus; LE: 2b, GR: B).90,91 A thrombus invading the renal veins also necessitates surgical intervention (consensus; LE: 2b, GR: B).92,93
The appropriate surgical technique depends on the individual case and surgeon’s experience but mainly on the extent of the thrombus. For thrombi above level II, the panel recommends applying hepatic mobilization or the Pringle maneuver as a practical surgical approach to help expose the retrohepatic IVC and improve safety (consensus; LE: 2b, GR: B). 94 Additionally, for a thrombus that extends beyond the major hepatic veins, cardiopulmonary bypass with or without hypothermic circulatory arrest is necessary to achieve complete resection of the venous tumor thrombus (consensus; LE: 5, GR: D).
Ligation of the IVC can be performed, rarely, during the nephrectomy of right kidney tumors if the renal and gonadal veins or left adrenal veins are present (consensus; LE: 5, GR: D).
Thrombi in the renal vein and/or infrahepatic vena cava, below level III, of a favorable conformation can be approached with a minimally invasive technique by qualified surgeons (consensus; LE: 2b, GR: B). 95
Neoadjuvant therapy should not be routinely prescribed for a large venous tumor thrombus because it may have a limited effect on the thrombus and may be related to increased mortality (consensus; LE: 2b, GR: B).58,96
Unresectable cases
Concerning primarily unresectable tumors, the possibility of neoadjuvant therapy is emerging after discussion among a multidisciplinary team, with the aim of enabling surgical intervention in these patients, as it is the standard treatment for locally advanced RCC. Nevertheless, research and data are still scarce.
Therefore, the panel’s recommendation is to discuss neoadjuvant therapy as an exception in initially unresectable cases (consensus; LE: 2b, GR: B),97,98 using tyrosine kinase inhibitors (TKIs) + immuno-oncology (recommendation; LE: 5, GR: D).
Advanced/metastatic disease
Nephrectomy in advanced/metastatic disease
As targeted therapy for kidney cancer has improved, the role of surgery in metastatic RCC has become more controversial. 99 However, cytoreductive nephrectomy (CN) remains an essential element in multimodal therapy for metastatic RCCs; patients must be carefully selected to achieve benefits, as CN seems to lead to greater morbidity and mortality than standard nephrectomy. 100 Thus, the panel recommends CN for a minority of patients with advanced/metastatic disease (recommendation; LE: 2b, GR: B), 99 including those with a venous thrombus (recommendation; LE: 2b, GR: B).101,102 Nevertheless, a multidisciplinary discussion for patient selection for CN is necessary (consensus; LE: 2b, GR: B). 103
The panel suggests using the International Metastatic Database Consortium (IMDC) risk model as a tool in the decision-making process (consensus; LE: 2b, GR: B),104,105 as follows:
IMDC favorable risk patients benefit from CN (consensus; LE: 2b, GR: B) 106 .
IMDC intermediate-risk patients with only one risk factor can be selected for CN (consensus; LE: 2b, GR: B)106,107.
IMDC intermediate-risk patients with ⩾2 factors must be carefully selected for CN (consensus; LE: 2b, GR: B)106,108.
IMDC poor-risk patients should not undergo CN (consensus; LE: 3a, GR: C).109,110
The panel does not recommend using MD Anderson Cancer Center (MDACC) risk criteria as a decision tool (recommendation; LE: 5, GR: D). However, if this tool is chosen, the panel recommends CN only for patients with <4 MDACC risk factors (recommendation; LE: 5, GR: D); it should be avoided in patients with ⩾4 MDACC risk factors (recommendation; LE: 5, GR: D).
The panel also recommends considering the following factors when selecting patients for CN:
primary tumor burden versus systemic tumor burden (consensus; GR: 2b)111,112;
number of metastatic sites (consensus; LE: 2b, GR: B).99,106,113,114
Cytoreductive PN (in metastatic patients) is an option for favorable tumors (consensus; LE: 2a, GR: B).115,116
Most patients with one metastatic site benefit from CN (consensus; LE: 2b, GR: B),106,114 whereas only a minority of patients with ⩾2 metastatic sites have an improved outcome after CN (recommendation; LE: 2b, GR: B). 106
As different sites of metastases seem to be associated with different prognoses and overall survival (OS) even after CN, taking into consideration the location of the metastasis can be helpful when selecting patients for CN (consensus; LE: 2b, GR: B).106,114 In the following scenarios, the panel recommends careful consideration and proceeding with CN only for the minority of patients with liver (consensus; LE: 2b, GR: B), 114 brain (recommendation; LE: 2a, GR: B), 109 bone (recommendation; LE: 2b, GR: B),111,114 or lymph node (recommendation; LE: 2b, GR: B)103,114 metastasis. On the other hand, the panel recommends CN for most patients with lung metastasis (recommendation; LE: 2b, GR: B). 106
LND should not be carried out routinely in metastatic disease during CN (consensus; LE: 2b, GR: B).117,118
The panel recommends CN for the minority of patients with nonclear cell histology (recommendation; GR: 2b) because, depending on the histology, the prognosis is usually poorer; the benefit appears to be related to independent factors such as risk criteria, age, and tumor staging.119–122
As multimodal therapy for metastatic RCC (mRCC) is common in clinical practice, therapeutic sequencing is also important. 116 In patients who did not receive upfront CN, the panel recommends deferred CN for patients who respond well to systemic therapy (consensus; LE: 1c, GR: A).123,124
In the presence of tumor complications such as bleeding, urinary obstruction, refractory pain, or hypertensive crisis, the panel recommends palliative nephrectomy (consensus; LE: 2b, GR: B). 125
Metastasectomy in advanced/metastatic disease
Current data suggest that local surgical therapy for mRCC metastases still has a significant role in improving OS and delaying disease progression. 126
As a single metastasis in metastatic RCC has a more favorable prognosis than multiple metastases, 127 the panel recommends performing metastasectomy for most patients with a single metastasis (consensus; LE: 2a, GR: B)128,129 and a select minority of patients with multiple metastases (recommendation; LE: 2a, GR: B). 130 A select group of patients with lymph node metastasis can undergo metastasectomy (recommendation; LE: 2b, GR: B), as the benefit is related to the number of positive lymph nodes. 117 The panel only recommends metastasectomy for a select group of patients with bone metastasis (consensus; LE: 2b, GR: B), especially those with solitary lesions.130,131
Along with the site of metastases, certain factors are also relevant for the prognosis of patients receiving local metastasis treatment. The panel recommends metastasectomy only for a minority of patients with a disease-free interval <1 year (recommendation; LE: 2a, GR: B) because they typically have poor prognosis.128,129 However, most patients with a disease-free interval >1 year should undergo metastasectomy (consensus; LE: 2a, GR: B).128,129
The panel recommends metastasectomy only for a minority of patients with synchronous metastasis (recommendation; LE: 2a, GR: B) because they have poor prognosis.128,129
Metastasectomy is indicated for most patients with favorable IMDC risk (consensus; LE: 2b, GR: B) 132 and for a minority of patients with intermediate risk (recommendation; LE: 2b, GR: B) 132 but is not indicated for those with poor risk (consensus; LE: 2b, GR: B). 132
The panel recommends metastasectomy in patients with local recurrence for most patients (consensus; LE: 2b, GR: B), as it improves OS. 133
Conclusion
This consensus provides evidence-based recommendations for surgical management of RCC in Brazil. Surgery, mainly PN, remains the best curative option for patients with localized RCC, and it still has a role in select patients with metastatic RCC.
Supplemental Material
sj-docx-1-tau-10.1177_17562872241312581 – Supplemental material for Renal cell cancer treatment: the Latin American Cooperative Oncology Group (LACOG) and the Latin American Renal Cancer Group (LARCG) surgery-focused consensus update
Supplemental material, sj-docx-1-tau-10.1177_17562872241312581 for Renal cell cancer treatment: the Latin American Cooperative Oncology Group (LACOG) and the Latin American Renal Cancer Group (LARCG) surgery-focused consensus update by Stênio de Cássio Zequi, Anderson de Oliveira Galvão, André Costa Matos, Gilberto Laurino Almeida, Marcelo Esteves Chaves Campos, Marcelo Langer Wroclawski, Thiago Camelo Mourão, Wagner Eduardo Matheus, Arie Carneiro, Augusto Modesto de Sousa Neto, Aurus Meneses, Breno Dauster, Daher Cezar Chade, Deusdedit Cortez Vieira da Silva Neto, Éder Silveira Brazão, Eduardo Café Cardoso Pinto, Eliney Faria, Felipe de Almeida e Paula, Felipe Lott, Fernando Korkes, Fernando Meyer, Francisco Hidelbrando Alves Mota Filho, Frederico Mascarenhas, Giuliano Betoni Guglielmetti, Guilherme Antônio Veloso Coaracy, Gustavo Cardoso Guimarães, Gustavo Franco Carvalhal, Jonatas Luiz Pereira, Leandro Koifman, Lucas Fornazieri, Lucas Nogueira, Lucas Teixeira Batista, Luciano Alves Favorito, Luiz Henrique Araújo, Marcos Lima de Oliveira Leal, Marcos Tobias-Machado, Mauricio Cordeiro, Mauricio Murce Rocha, Nilo Jorge Carvalho Leão Filho, Rafael Ribeiro Meduna, Renato Beluco Corradi, Ricardo de Lima Favaretto, Roberto Machado, Rodolfo Borges dos Reis, Roni de Carvalho Fernandes, Victor Espinheira Santos, Vladmir Pinheiro De Oliveira, Walter Henriques da Costa, Wilson F. S. Busato and Andrey Soares in Therapeutic Advances in Urology
Footnotes
Acknowledgements
The authors acknowledge writing assistance from Dr. Mariana Matos, MD, a medical writer, on behalf of Springer Healthcare. This manuscript was prepared according to the International Society for Medical Publication Professionals—Good Publication Practice for Communicating Company-Sponsored Medical Research: the GPP3 Guidelines.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
