Abstract
Purpose
To analyze the recurrence in patients with clinic stage T1 renal cell carcinoma (RCC) who were upstaged to stage T3a after partial nephrectomy (PN) using a new sub-classification criterion.
Methods
A retrospective study of pathological characteristics was performed in patients who were upstaged to pT3a on the basis of fat invasion (FI).
Results
After analyzing the pathological findings, we proposed the following new sub-classification criteria for pT3a RCC with FI: (1) renal tumor invades the pseudo-capsule and contacts the perinephric adipose tissue directly or the tumor protrudes into the perinephric adipose tissue like a tongue (Type A); and (2) tumor nodules are distributed in perinephric adipose tissues (Type B). A significant difference was observed in the recurrence rate between the two subtypes A and B. For Type B, the recurrence rate after radical nephrectomy (RN) and PN was 15.79% and 63.64%, respectively. The recurrence rates for Types A and B after PN were 11.11% and 63.64%, respectively.
Conclusions
T3a RCC with tumor nodules in perinephric adipose and/or an irregular tumor protruding into the adipose tissues lead to a higher recurrence rate. We recommend that T3a RCC be carefully analyzed and patients be treated on an individual basis.
Keywords
Background
Renal cell carcinoma (RCC) is the most frequently diagnosed malignancy among renal tumors; 1 it accounts for 2% to 3% of all malignancies and its incidence has increased globally by about 2% annually over the past 20 years. 2 The incidence of RCC continues to steadily increase in most countries.3–5
Advancements in modern radiologic imaging tools, such as computed tomography (CT) and ultrasound (US), have contributed to an overall improvement in the incidental detection of localized RCC, among which most are classified as clinical stage T1. 6 T1 RCC is commonly associated with favorable treatment outcomes, so the main guidelines from both the European Association of Urology (EAU) and the American Urologic Association (AUA) recommend nephron-sparing techniques, such as partial nephrectomy (PN), as the primary treatment for patients with clinical stage T1 RCC.7,8 However, some patients who undergo PN for T1 RCC may be upstaged to pT3a on the basis of the final pathological examination results, and this becomes a challenging obstacle for surgeons. 9
To date, it is uncertain which surgical method (PN or radical nephrectomy [RN]) should be used when patients who undergo PN for T1 RCC are upstaged to pT3a. Some studies have reported similar survival outcomes between the two groups (PN vs. RN), while others showed contradictory findings.10–12 The cause underlying the controversy needs to be characterized in further studies, so we recommend a new sub-classification criterion for pT3a RCC with fat invasion (FI) to resolve this controversy.
Materials and methods
The study was approved by the Institutional Review Board (The First People’s Hospital of Lianyungang Ethics Committee, Lianyungang, October 2018; approval number 20180103), and written informed consent was obtained from all included patients. The reporting of this study conforms to the STROBE statement. 13 A retrospective analysis of our nephrectomy database was performed, which identified T1 RCC patients who consecutively underwent PN or RN between 2010 and 2018. The PN technique was performed as previously reported. 14 Cold ischemia was not performed for any of the patients. The criteria for performing a RN were as follows: 1) insufficient volume of remaining parenchyma to maintain proper organ function; 2) unfavorable tumor location; and 3) adherence to the renal vessels. Except for the abovementioned criteria, all of the criteria related to performing a PN. The inclusion criteria were as follows: patients diagnosed with T1 RCC on the basis of preoperative images, and postoperative pathological findings confirmed that they were upstaged to pT3a. The exclusion criteria were as follows: 1) benign lesions; 2) non-T3a RCC; or 3) missing follow-up data. Following surgery, all the specimens were evaluated by two specialized genitourinary pathologists. The RCC specimens were stained at the edge using ink in accordance with routine procedures and cut into sections that were approximately 3 mm thick. The sections were then stained with hematoxylin–eosin. The pathological findings were classified in accordance with the 2017 TNM classification standard. 15
Postoperative evaluations were performed using kidney and chest CT imaging 6 months after surgery and then annually thereafter until 5 years after surgery. If necessary, magnetic resonance imaging or bone scans were also performed when clinically indicated. Disease relapse was determined upon local recurrence that was identified by radiologic or pathologic evidence, which includes lymph node and/or distant metastasis. Surgical resection of locally recurrent disease was offered when a complete resection was possible, and targeted therapies were used for metastatic RCC. Additionally, the R.E.N.A.L. nephrometry score was used to evaluate the tumors. The R.E.N.A.L. score comprises the following parameters: (R)adius, which corresponds to the maximum tumor diameter; (E)xophytic/endophytic properties of the tumor; (N)earness of tumor’s deepest portion to the collecting system or sinus; (A)nterior (a)/posterior (p) descriptor; and the (L)ocation relative to the polar line.
Statistical analysis
All statistical analyses were performed using GraphPad Prism7 software (GraphPad Software, La Jolla, CA, USA). Pearson’s Chi-square test and Fisher’s exact test were conducted to compare the categorical variables. Recurrence-free survival was analyzed using the Kaplan–Meier method and the log–rank test. Two-sided P-values <0.05 were considered to represent statistically significant differences.
Results
This study included 2927 patients on the basis of the final pathological examination results, among whom 1705 patients had pathological T1 tumors and 99 patients were upstaged to pathological T3a tumors, which is a rate of 5.81%. Among the 99 patients whose tumors were upstaged, 38 of them underwent PN and 61 underwent RN. No differences were observed in gender, age, tumor size, laterality, tumor histologic type, R.E.N.A.L. score, or tumor grade between the two groups (Table 1).
Patient characteristics (n = 99).

RN, radical nephrectomy; PN, partial nephrectomy; IQR, interquartile range; R.E.N.A.L., Radius, Exophytic/endophytic properties, Nearness, Anterior (a)/posterior (p) descriptor, and Location.
After analysis of the sample sections, we proposed the following set of new classification criteria for pT3a RCC with FI: Type A and Type B. Type A includes direct renal tumor contact with the perinephric adipose tissue (Figure 1) or tumor that protrudes into the perinephric adipose tissues (Figure 2). Type B includes tumor nodules that are distributed in perinephric adipose tissue (Figure 3).

Direct renal tumor contact with the perinephric adipose tissue. Blue arrow: tumor; Red arrow: adipose tissue (200×). Scale bar = 100 μm.

The tumor protrudes into the perinephric adipose tissue like a tongue. Blue arrow: tumor; Red arrow: adipose tissue (100×). Scale bar = 50 μm.

Tumor nodules are distributed in the perinephric adipose tissue. Blue arrow: tumor; Red arrow: adipose tissue (200×). Scale bar = 100 μm.
Sixteen patients had tumor recurrence during the follow-up (average duration: 56 months; range: 15–60 months). On the basis of the pathological sub-classification of pT3a diseases, there were three and three cases of recurrence in the Type A category and seven and three cases of recurrence in the Type B category when patients underwent PN or RN, respectively (P = 0.005) (Table 2). For Type A, the recurrence rates of RN and PN were 7.14% and 11.11%, respectively, which was not significantly different (Figure 4a). For Type B, the recurrence rates of RN and PN were 15.79% and 63.64%, respectively, which was a significant difference (P = 0.014) (Figure 4b). The recurrence rate for patients who were classified as Type A or B and who underwent RN was 7.14% or 15.79%, respectively, which was not significantly different (Figure 4c). Moreover, the recurrence rate for patients who were classified as Type A or B and who underwent PN was 11.11% or 63.64%, respectively, and this difference was significant (P = 0.002) (Figure 4d). A Kaplan–Meier analysis showed that overall recurrence-free survival was significantly better in patients with Type A tumors compared with those with Type B tumors (P = 0.002) (Figure 5a). Patients who underwent RN treatment had a better prognosis compared with those who underwent PN (P = 0.027) (Figure 5b). In addition, Types A and B after PN showed significant differences in the recurrence-free survival, with better survival shown in patients with Type A compared with those with Type B tumors (P = 0.0002) (Figure 5c). Recurrence-free survival for patients with Type A or B tumors after RN was not significantly different (Figure 5d).
Postoperative recurrence data (n = 99).

PN, partial nephrectomy; RN, radical nephrectomy.
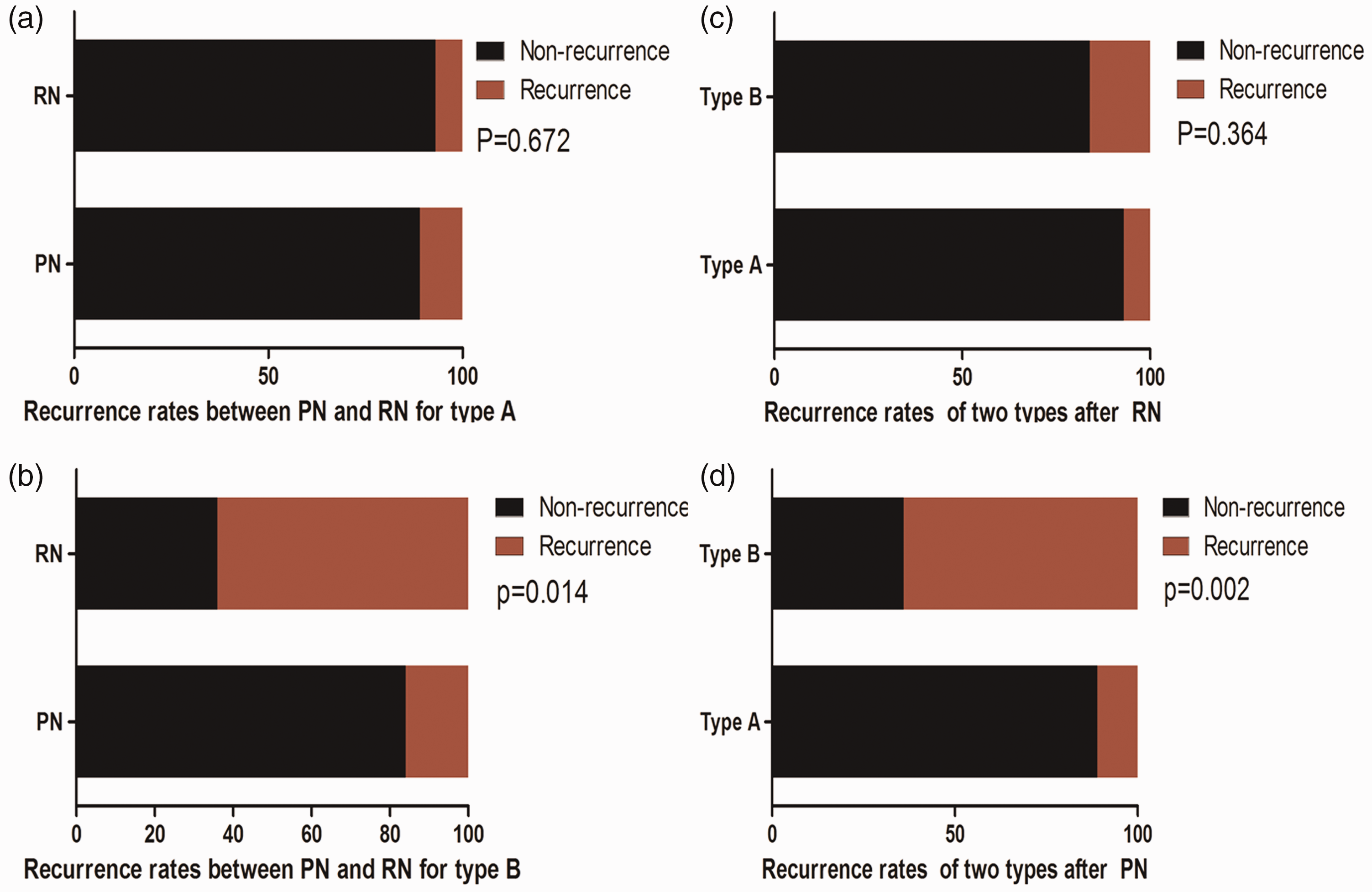
Recurrence rates. a) Recurrence rate for PN and RN for Type A was not significantly different; b) Recurrence rate was higher for PN compared with RN for Type B (P=0.014); c) Recurrence rate for Types A and B after RN was not significantly different; d) Recurrence rate was higher for Type A compared with Type B after PN (P=0.002). P-value determined by Pearson’s Chi-square and Fisher’s exact test.

Kaplan–Meier survival analyses of recurrence-free survival in RCC patients. a) Overall recurrence-free survival was better in patients classified as Type A compared with those classified as Type B (P = 0.002); b) RN was associated with a better prognosis for patients with a pT3a renal tumor with fat invasion compared with PN (P = 0.027); c) Recurrence-free survival for Type A and B after PN. Patients with Type A showed better survival compared with Type B (P = 0.0002); d) Recurrence-free survival for Types A and B after RN was not significantly different.
Discussion
With the improvement of surgical skills and the development of new technologies in recent years, PN has become widely used in patients with complex RCC, and this has also led to an increased amount of tumor upstaging after surgery. 16 In the present study, approximately 5.81% of the patients with cT1 RCC were upstaged to pT3a after surgery, which is consistent with the findings of previous studies in this field. Srivastava et al. collected data from 28,854 T1 RCC patients who underwent surgery, and this is the largest study of this type that has been conducted to date. 17 They found that the tumor was upstaged to pT3 in 4.2% of these patients. 17 The latest study on this topic showed that the proportion of patients who were upstaged from cT1 to pT3 was 19%, and the logistic regression analysis in this study indicated that age, radius, and touching the main vessels were significantly associated with pathologic upstaging. 18 Pathological upstaging of RCC after surgery is common, and this situation should be investigated by urologists.
PN is not currently recommended as the preferred treatment for T3a RCC by the EAU or AUA guidelines on RCC.19–21 Recent studies suggest that RN should be used to treat patients with T3 RCC, and open surgery, laparoscopic surgery, and even robotic surgery are all feasible. 22 It is a challenging and non-ideal situation when cT1 RCC is upstaged to pT3a after surgery. Therefore, some scholars have different perspectives because they had already identified this phenomenon.
Lee et al. retrospectively analyzed data from 1324 T1a RCC patients and 43 T1a RCC patients who were upstaged to T3a, and they concluded that that pT3a small RCC after PN had similar oncological outcomes as those of pT1a RCC. The 5- and 10-year recurrence-free survival in these patients was 98.0% and 95.2% for T1a patients and 94.4% and 95.2% for T1a patients who were upstaged to T3, respectively, which was not significantly different. 23 Therefore, PN is suitable to treat patients with T3a RCC. Oh et al. reviewed 3567 patients and showed similar findings that PN has a similar recurrence-free survival outcome compared with RN in patients with cT1a that was upstaged to pT3a RCC. 24 A similar finding was also reported in a study in 2018. They found that in patients with unexpected pT3a RCC at final pathology, PN does not appear to negatively affect cancer control with regard to metastatic progression and cancer-specific mortality. 25
Different findings indicate that further in-depth investigations are required. We found that T3a RCC with FI can also be subdivided into the following two types on the basis of pathological microscopic examination of the sections: Type A and Type B. Type A includes renal tumors that are in direct contact with the perinephric adipose tissue or that protrude into the perinephric adipose tissues. Type B includes tumor nodules that are distributed in perinephric adipose tissue. There have been no previously published studies to clarify this finding. Our finding may contribute to the current controversy that is mentioned above.
Among the 99 upstaged individuals in this study, 69 were classified as Type A, and 27 of them underwent RN, while 42 of them underwent PN. During a median 56-month follow-up period, only three patients who underwent RN and three patients who underwent PN relapsed, which was not significantly different. For Type A, complete removal of the tumor and adipose tissue covering the tumor surface is not difficult using the PN method, and the relapse rate after surgery is low. The tumor protrudes into the fat like a tongue, and if the adipose tissue on the tumor surface is improperly removed during surgery, it can lead to the occurrence of positive margins due to the residual tumor tissue that has protruded into the adipose tissue. This may be a risk factor for local tumor relapse. Positive margins were reported to result in tumor metastasis and recurrence. 26 Therefore, the adipose tissue on the surface of the tumor may not be able to be completely removed during surgery, especially with irregularly shaped tumors.
Only 30 patients had tumors that were classified as Type B in our study, among whom 11 patients were identified after PN and 19 were identified after RN. During the follow-up, there were seven patients who underwent PN and three patients who underwent RN who experienced a relapse. The difference between the two groups was statistically significant (P = 0.014). Because the tumor nodules cannot be easily recognized using US or CT before surgery or by the naked eye during surgery, it is difficult to completely remove the scattered nodules during PN. Thus, radical surgery should be used for this type of tumor (Type B). We compared the recurrence rates of patients with Types A or B after RN and PN, and the results showed 7.14% and 15.79% recurrence after RN for Type A, which was not significant, and 11.11% and 63.64% recurrence after PN for Type B, which was significantly different (P = 0.002). However, tumor nodules are difficult to recognize both preoperatively and intraoperatively, and they are commonly found when examining the slices pathologically. Therefore, close follow-up is required for patients with type B tumors who have undergone PN treatment, and radical surgery should be performed as soon as possible for those who show early local recurrence.
This study has some limitations including that it was a single-center retrospective study with a relatively small sample size. Despite these limitations, it provides new evidence to explain the current controversy about patients with T1 RCC who are upstaged to pT3a after surgery.
Conclusion
The treatment outcomes of patients with T1 RCC that is upstaged to pT3a after PN remains controversial. The key findings of this current study indicate that a higher recurrence rate was identified in T3a RCC with tumor nodules in perinephric adipose or/and with an irregular tumor shape that protrudes into adipose tissue. Additionally, we recommend that patients with T3a RCC be carefully assessed and treated on an individual basis.
Footnotes
Authors’ contributions
DSZ and XFX conceived and directed the project. DSZ contributed to writing the manuscript. DSZ analyzed the data. DSZ and XFX collected the samples. All authors read and approved the final manuscript.
Acknowledgment
We acknowledge and appreciate our colleagues for their valuable efforts and comments on this paper.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
