Abstract
Background:
Stress urinary incontinence (SUI) presents as unintentional urine leakage associated with activities. It significantly affects quality of life (QoL) and is the most common type of incontinence in women. Current treatment options, particularly non-surgical therapies, are lacking.
Objective:
To assess the efficacy of mechanotherapy provided by the Flyte® intra-vaginal device during pelvic floor muscle training (PFMT).
Design:
This was a randomized, controlled, double-blinded trial.
Materials and methods:
Flyte is a repeat use device for conditioning and strengthening the pelvic floor muscles (PFMs). It provides two-part mechanotherapy. Part 1 is the stretching and preloading of the PFM from the internal wand. Part 2 integrates mechanical pulses which elicit muscle cellular and tissue level responses that trigger cellular regeneration, improve neuromuscular facilitation and motor learning. Subjects used the device for 5 min/day for 12 weeks. Subjects (144) were randomized and evaluated at 6 and 12 weeks. Arm A (72) received both Part 1 and Part 2 mechanotherapy for 12 weeks, whereas Arm B (72) received Part 1 therapy for 6 weeks, then crossed over to full therapy. Mean age was 50, 49, respectively, prior pelvic/abdominal surgery 26%, 46%, and previous incontinence treatments 13%, 22%. The primary endpoint was 24-h pad weight (24-HR PW) at 6 weeks. Secondary endpoints were 24-HR PW at 12 weeks and QoL [International Consultation on Incontinence Questionnaire (ICIQ), Urinary Incontinence Quality of Life (IQOL)].
Results:
Part 1 therapy had a greater than anticipated therapeutic effect. Thus, the study was underpowered to identify differences between study arms. Therefore, data were pooled to assess the effects of mechanotherapy. Twenty four-HR PW was significantly reduced at 6 weeks (p = <0.0001), with further reduction from 6 to 12 weeks (p = <0.0001). Data were stratified based on 24-HR PW severity. Significant reductions were noted in all severity groups (mild p = <0.0001, moderate p = <0.0001, severe p = <0.01). QoL was similarly improved at 6 weeks (ICIQ p = <0.0001, IQOL p = <0.0001), and 12 weeks (ICIQ p = <0.0001, IQOL p = <0.0001). Compliance was >80% at 6 weeks and 70% at 12 weeks.
Conclusion:
Two-part mechanotherapy significantly improved 24-HR PW and QoL across all severities of SUI. Improvements were noted in as little as 2 weeks and appeared to be sustained through 2-year follow up.
Trial registration:
Registered on ClinTrials.gov (NCT02954042).
Plain language summary
Stress urinary incontinence is the involuntary loss of urine during activities such as sneezing, coughing, lifting and exercise. It negatively affects quality of life, and is the most common type of incontinence in women. Current treatment options, particularly non-surgical therapies, are lacking. Pelvic floor muscle training, sometimes referred to as Kegel exercise, is the usual initial therapy to treat stress urinary incontinence. This study assessed the effectiveness and safety of the two types of mechanical stimulation therapy provided by the Flyte® intra-vaginal wand during pelvic floor muscle training. The first type stretches the muscles to optimize the effect of the pelvic floor muscle exercises. The second type is the delivery of specially tuned mechanical pulses that trigger muscle cell and tissue responses that accelerate muscle cell healing, increase muscle awareness and improve coordination. Due to an inadequate number of study subjects in the two study arms, the study data were combined into one analysis group to better assess the benefits of Flyte therapy in the participants. The volume of urine loss was very significantly reduced at 6-weeks, with further reduction from 6-12 weeks. Data were then divided into subgroups based on the severity of urine loss at the beginning of the study. Very significant reductions were noted in women with Mild, Moderate and Severe incontinence. Quality of life was similarly improved at 6 and 12 weeks, as measured by two quality of life questionnaires. These improvements were noted in as little as 2 weeks. More than 80% of participants performed the therapy at 6 weeks and 70% at 12 weeks. Improved quality of life was sustained through two years after the study ended in women who voluntarily continued participation in the study.
Keywords
Background
Urinary incontinence (UI) is very common in adult women. World-wide, more than 300 million women have UI, with studies reporting incidence rates of 11–70%, approximately half of which are stress urinary incontinence (SUI).1–3 Clinically, SUI presents as brief urine leakage provoked by physical activity. Common risk factors include increasing age, obesity, parity, smoking, alcohol consumption, and hysterectomy. 4 SUI is related to urethral sphincter failure associated with increased intra-abdominal pressure. In mild SUI, this pressure may be the result of sudden or forceful activities such as exercise, sneezing, laughing, or coughing. In more severe forms, urine leakage occurs with less forceful activities like standing up, walking, or bending forward. The condition causes significant reductions in patients’ quality of life (QoL), proportional to its severity. It is more common in middle-aged and older women. 5
Pelvic floor muscle training (PFMT) is recommended by numerous professional medical associations as the first-line treatment for patients diagnosed with SUI. 6 Other non-surgical options include lifestyle changes, pads, vaginal weights, pessaries, biofeedback, pelvic floor physical therapy (PT), and electrical stimulation.7,8 Despite the long history of these interventions, significant need exists for more effective therapies, especially for women not interested in surgical interventions. Recent research has explored the effect of mechanical forces as a therapeutic modality for treating muscle injuries and weakness in patients with UI.9–11 Application of mechanotherapy presents a potentially new and beneficial approach for treatment of SUI in women.
This randomized clinical trial examined the clinical benefits of the Pelvital Flyte® device, which provides mechanical therapy imposed on the pelvic floor muscles (PFMs) as they voluntarily contract and relax (Kegels). Biofeedback via lights on the device representing movement of the PFM builds coordination and motor control as the mechanotherapy is applied. The Flyte device improved PFM function as measured both objectively (24-hour pad weight, 24-HR PW) and subjectively through SUI-related symptoms and QoL questionnaires. The study was sponsored and funded by Pelvital USA, Inc., and registered on ClinTrials.gov (NCT02954042).
Materials and methods
The reporting of this study conforms to the Consolidated Standards of Reporting Trials statement. 12
Physiologic basis of Flyte therapy
Flyte provides two-part mechanotherapy. Part 1 is the effect of the carefully designed internal wand itself, which provides stretching and preloading of the pelvic muscles, as well as a resistive surface against which to contract the muscles during PFMT. Part 2 is the addition of precisely tuned mechanical pulses which enhance therapeutic benefit by intentionally disrupting the natural state of the cell. The mechanical signals elicit a response from the muscle resulting in functional changes at the cellular and tissue levels. These, in turn, promote growth and survival, influence metabolic responses, and re-establish tissue architecture, all of which contribute to a strong and healthy muscle system. This well-described process is called mechanotransduction, defined as the mechanism by which cells convert mechanical stimuli into cellular responses to a variety of mechanical loads.10,11,13
In the Flyte device these mechanical forces originate at the interface of the wand and the muscle tissues. They are then transmitted to the nuclei of the muscle cell via the cytoskeleton, from the cytoskeleton to the extracellular matrix (connective tissues) and finally to the pelvic bones. This process functions because skeletal muscle systems are designed by nature to have a natural state of tension, called tensional integrity or ‘tensegrity’, which assures that the muscle systems, inclusive of muscle units and connective tissues, will respond immediately when exposed to mechanical loads.14,15 In addition, mechanotherapy modulates nerve signaling via the stimulation of mechanoreceptors in muscles that activate the proprioceptive sensory system. This enhances afferent and efferent neural communication with the reflexive spinal sacral plexus pathways, as well as the higher conscious control centers of the brain.
Study device
After initiation of the Pelvital Stress Urinary Incontinence Training device (P-SUIT) research trial, the Pelvital wand used as the study device was branded as Flyte®; the device will be referred to as Flyte in the remainder of the paper. Flyte is a repeat use, non-sterile, intra-vaginal device intended to strengthen, re-train, and coordinate the PFM system (Pelvital USA, Inc., Eagan, MN, USA). It is designed for repeated, in-home use by adult women to enhance PFM training.
The product (Figure 1) consists of an internal wand that is inserted into the vagina and a handheld Control Unit that provides a biofeedback function to inform the user when the correct muscles are being contracted and relaxed during each Kegel contraction, further enhancing its efficacy. The Control Unit also contains data storage and a rechargeable battery. The stored data can be transferred from the Control Unit when connected by a cable into a computer for data display and clinical assessment. (The next generation Flyte device is anticipated to provide cloud connectivity and data collection/storage.) The device’s battery is recharged via a charging cable via a standard wall outlet. Furthermore, the internal components of the wand are measuring the difference between resting PFM tone and contraction muscle tone during the mechanical pulses. This determines a dampening coefficient, or the amount of pressure change on the wand. This information is then visually provided as biofeedback to the user, thereby enhancing their conscious control of the PFM. The mechanotherapy and biofeedback are provided simultaneously as part of one therapy. While mechanotherapy is the primary mechanism of action, biofeedback is provided simultaneously to enhance user understanding and conscious control.

Flyte study device.
Study design
This was a prospective, randomized, controlled, double-blinded trial. Subjects were randomly assigned to device treatment with mechanical pulses (Arm A, Parts 1 and 2 therapy) for 12 weeks or treatment without mechanical pulses (Arm B, Part 1 therapy only) for the first 6 weeks, after which Arm B participants crossed over to Arm A to receive two part therapy for the second 6 weeks. The study hypothesis was that use of the Flyte mechanotherapy device with or without mechanical pulses would improve outcomes as compared to baseline, and that therapy with mechanical pulses in conjunction with PFMT would quantitatively improve UI as compared to PFMT without device pulses. The primary endpoint was the change in the severity of involuntary urine loss, determined by an improvement in 24-HR PW from baseline to week 6. Secondary endpoints included 24-HR PW at 12 weeks, 24-HR PW for the full cohort from baseline to 6 and 12 weeks, and changes in QoL from baseline to 6 and 12 weeks using validated, disease-specific instruments [International Consultation on Incontinence Questionnaire – Urinary Incontinence – Short Form (ICIQ-UI-SF) and Urinary Incontinence Quality of Life (IQOL)]. Subject adherence to treatment was also assessed at 6 and 12 weeks. Voluntary follow-up was continued up to 24 months.
Subjects who met all inclusion/exclusion criteria and provided informed consent were enrolled in the trial and randomized using a random number generator. Participants were allocated to either the treatment or control arm. Subject follow-up visits occurred at weeks 2, 4, 6, 8, 10, and 12, with the final visit being at the completion of the study. The routine visits at weeks 2, 4, 8, and 10 were for verification of patient compliance, data download, and the review, as well as documentation of the voiding diary and pad weight. The visits at 6 and 12 weeks consisted of data collection and comparison with the baseline assessment at the initiation of the study. These included the 24-HR PW test and QoL questionnaires.
Potential study subjects were screened for study enrollment using the following inclusion and exclusion criteria:
Female gender
Ages 18–75
Clinical diagnosis of SUI
Ability to contract the PFMs
Able to document incontinence and voiding in a diary
Provision of written informed consent form
Minimum 10-g pad weight increase at initial 24-HR PW test.
Exclusion criteria included:
Diagnosed mixed or urge UI
Inability to contract the PFMs
Impaired cognitive function or neurologic conditions
Physical limitations that impede the patient’s ability to participate (e.g. ability to stand)
Acute infection or hematuria
Pregnant or actively trying to conceive
History of pelvic irradiation
Concurrent medications with α-adrenergic antagonists or diuretics
Pelvic organ prolapse stage III or IV
Severe urethral sphincter weakness and/or defect
Suspected urethral and/or vesical fistula.
The trial was performed at three sites in the U.S. Investigational Review Board approval was obtained by each site prior to initiation of subject recruitment. Further determination of eligibility for inclusion into this study was made using the current standard of care at each site for diagnosis of SUI, in conjunction with the above listed inclusion/exclusion criteria. Urodynamic testing and formal muscle strength testing were not performed in potential study subjects, as strength was not a study endpoint. To be accepted into the study, subjects had to have clinical SUI diagnosed by a physician, demonstrate at least 10 mg of urine leakage on the initial 24-HR Pad Test, and have the ability to contract the PFM.
Upon verification that the patient met these criteria, they were led through the informed consent process and written informed consent was obtained. Site monitoring was performed to determine protocol compliance; deviations were recorded. Once subjects were randomized, investigators, subjects, and trial sponsor were blinded to treatment allocation assignment. After informed consent was obtained, potential participants were provided supplies to collect 24-HR PW.
At the ‘Baseline Assessment Visit’ it was determined whether the patient met the inclusion criteria of ‘minimum 10 g initial 24-HR PW test’ as well as other inclusion/exclusion criteria, including ability to contract the PFMs, that is, ability to perform a Kegel contraction via subjective digital assessment. Urodynamic and formal PFM muscle testing were not performed during the study, as PFM strength was not an endpoint. Subjects who met the inclusion/exclusion criteria were then enrolled into the trial and randomized. Patients who later met all the inclusion/exclusion criteria were eligible for enrollment.
Statistical analysis
Based on the findings from a pilot study, a sample size of 118 persons (1:1 randomized) was determined to give 90% statistical power to detect a threshold of 20% difference assuming a 40% relative difference in the actual data of 24-HR PW testing. 16 Assumptions for the power calculation were based on the results from a pilot study. The exact effect size was not known a priori but was be assessed during interim analyses. All analyses were intent-to-treat, with a two-sided alpha level of 0.05 with comparison of intervention versus control arm, using an independent t test. Randomization allotment was provided by the trial statistician using a computer-generated randomization list generated by Power Analysis and Sample Size Software (2016); NCSS, LLC., Kaysville, UT, USA.
Patient population
Two hundred fifteen subjects were screened for the trial and 144 were randomized and assigned a treatment (72 Arm A, 72 Arm B). Twenty-four participants were screened for the study and provided a baseline 24-HR PW but did not complete follow-up activities after randomization (n = 15 Arm A, n = 9 Arm B). These 24 participants were censored for all endpoints except the adverse event (AE) reporting. The majority of withdrawals occurred early due to inability to use the device (e.g. the device could not be inserted). One hundred twenty continued participation through at least 6 weeks. The trial was stopped when enrollment was complete.
One subject improperly performed the 24-HR PW collection and was censored. This left 119 subjects (57 Arm A, 62 Arm B) with data for the primary endpoint analysis. The study was stopped after achieving target enrollment. Voluntary long-term follow-up of up to 24 months was undertaken and completed in December of 2021. Figure 2 provides the Consolidated Standards of Reporting Trials (CONSORT) flow diagram of the progress through the phases of the study.

CONSORT study flow diagram.
Results
Baseline characteristics
Table 1 summarizes the participants’ characteristics at baseline. Baseline severity was defined as follows: mild 10–20 g, moderate 21–70 g, severe >70 g. In the study cohort, there were 43 participants with mild, 57 with moderate, and 18 with severe incontinence. A large number of women in both arms were White.
Baseline characteristics.

24-H pad weight
Overall, a statistically significant reduction in 24-HR PW occurred among all participants from baseline to 6-weeks (p < 0.001) and to 12-weeks (p < 0.001). Of participants, 57% (68/119) had a >50% reduction in 24-HR PW in the first 6 weeks, and 71% (81/114) achieved a clinically meaningful reduction (>50%) in pad weight by 12 weeks of therapy. The median reduction in 24-HR PW was 68% by 12 weeks. Overall, 61% of participants achieved continence with <10 g.
No significant difference occurred in the primary endpoint: 24-HR PW at 6 weeks in subjects in Arm A declined from 36.8 to 11.7 g and Arm B from 27.0 to 13.9 g (p = 0.40). Both arms improved over baseline. It is believed this was due to the greater than expected therapeutic effect of Part 1 mechanotherapy (muscle stretching/preloading from the wand alone). A post hoc analysis found that an additional 50 patients (25 each study arm) were needed to achieve statistical significance between study arms. As a result, analyses focused on the treatment effects for the entire study cohort, as therapy provided in both study arms exhibited significant improvement over baseline.
Full study cohort 24-HR PW improved significantly compared to baseline at 6 weeks, with further improvement at 12 weeks; incremental improvement from 6 to 12 weeks was also significant [Figure 3(a)]. The 24-HR PW results were further stratified by baseline severity, as illustrated in panel B. Baseline severity was defined as follows: mild 10–20 g, moderate 21–70 g, severe >70 g. In the study cohort, there were 43 subjects with mild, 58 with moderate, and 18 with severe SUI. Significant differences were noted in all severity levels from baseline, with incremental improvement noted from week 6 to week 12. While not statistically significant, trends favored the full treatment arm (part 1 + part 2 mechanotherapy) compared to baseline between 6 and 12 weeks.

24-Hour pad weight: Full cohort (a) and stratified by baseline severity (b).
Quality of life
QoL results as assessed by the ICIQ were found to be statistically significant from baseline to 6 and 12 weeks, with incremental improvement noted from 6 to 12 weeks [Figure 4(a)]. (Note that better QoL for the ICIQ is represented by a lower score.) As presented in panel B, when stratified by baseline 24-HR PW severity, statistically significant improvements were noted from baseline to 6 weeks and baseline to 12 weeks in all groups. While not statistically significant, additional improvements were noted between 6 and 12 weeks.

ICIQ. Full cohort (a) and stratified by baseline 24-h pad weight severity (b) (lower score denotes better QoL).
As presented in Figure 5, QoL in the full cohort as assessed by the IQOL instrument revealed similar improvements from baseline to 6 and 12 weeks, with incremental improvement noted from 6 to 12 weeks [Figure 5(a)]. As presented in panel B, when stratified by baseline 24-HR PW severity, statistically significant improvements were noted from baseline to 6 weeks and baseline to 12 weeks in all severity groups. Better QoL for the IQOL is represented by a higher score.

IQOL. Full cohort (a) and stratified by baseline 24-h pad weight severity (b) (higher score denotes better QoL).
Long-term follow-up
Long-term follow-up data were collected at 6, 12, 18, and 24 months on a voluntary basis. Fifty-four subjects performed voluntary follow-up at 6 months; this decreased to 31 at 24 months. The significant improvements in QoL noted at 12 weeks were maintained for both QoL instruments over 2 years, with IQOL scores being slightly improved over this 2 year time frame, as represented in Table 2 (pooled data). These trends were noted at all levels of baseline severity (Figure 6) and device usage (Figure 7).
Quality of life during long-term follow-up.

ICIQ, International Consultation on Incontinence Questionnaire; IQOL, Urinary Incontinence Quality of Life.

Quality of life findings during long-term follow-up stratified by baseline 24-h pad weight severity.
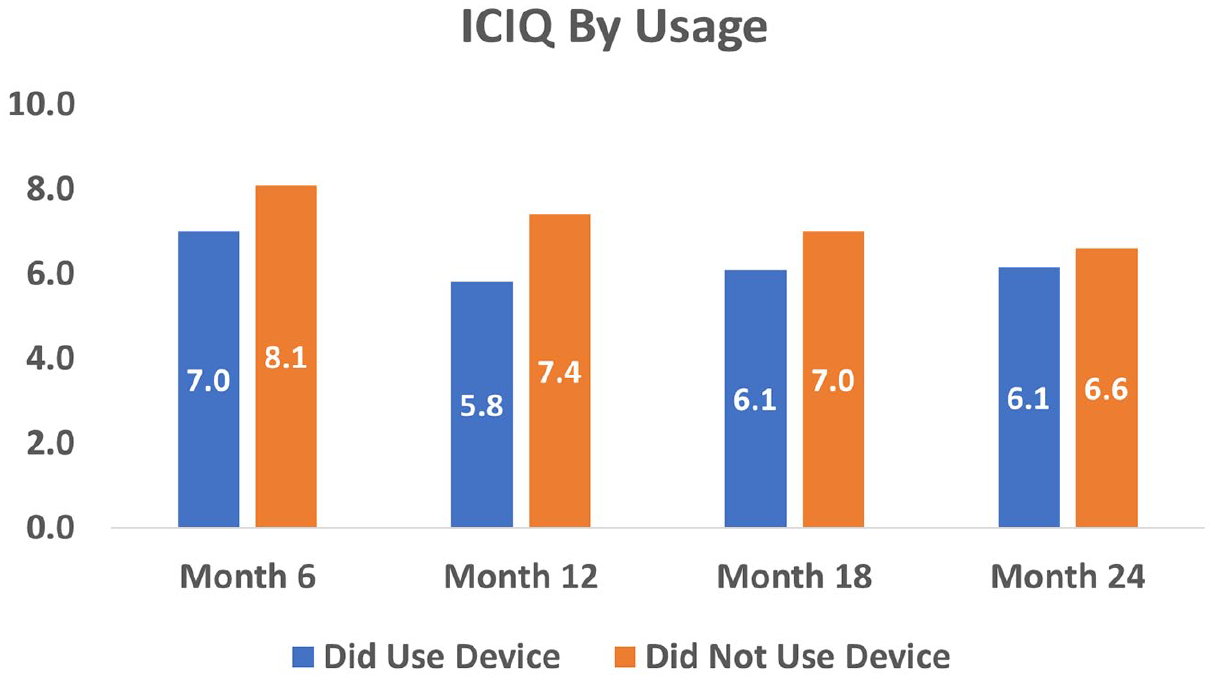
Quality of life by Flyte device usage.
While continued daily therapy was not specified, further analyses of device usage after study completion showed that those women who continued Flyte therapy at any frequency fared slightly better than those who did not in all severity categories. This is illustrated in Figure 6 for ICIQ scores; similar results were noted in the IQOL scores.
Compliance
Compliance data were obtained from information that was downloaded from the device (input from each muscle contraction). A software change instituted for the last 25% of subjects resulted in data loss. Data from these first 75% of subjects indicated that overall median compliance rate was high in both study arms. Specifically, compliance in Arm A was 80% at 6 weeks and 72% at 12 weeks; Arm B compliance was 84% at 6 weeks and 67% at 12 weeks. This is summarized along with the IQRs in Table 3. It was noted that there was no correlation between net change in 24-HR PW and the percent daily compliance at either 6 or 12 weeks.
Therapy compliance by study arm.

IQR, interquartile range.
Adverse events
All AEs were adjudicated by a Data Safety and Monitoring Board. Overall, 84 AEs occurred among 51 consented participants during the 12 week intervention period. All AEs were collected among all subjects randomized, including those who withdrew from the trial. The incidence of AEs did not differ greatly between arms. There were 37 AEs among 25 participants in Arm A, and 47 AEs among 26 participants in Arm B. Overall 65 AEs were of mild severity (grade 1), and 18 of moderate severity with limitations of daily activity (grade 2), and there was 1 grade 3 AE which was a serious AE due to hospitalization for pancreatitis. This SAE was deemed unrelated to the study device. Device-related AEs included: 27 definitely related to the device (e.g. pelvic discomfort or pain), 9 probably related, 24 possibly related, and 24 not related. Of the definite device-related AEs, the majority were vaginal/pelvic discomfort, soreness, or pain with using the device, which was expected, similar to muscle soreness after any exercise. Of the 18 grade 2 AEs, only 1 was determined to be device-related, 2 were probably related, 7 possibly related, and 8 not related.
Discussion
As presented earlier, the trial was found to be underpowered to distinguish statistically significant improvements between the two study arms, likely due to the greater than anticipated clinical benefit from part 1 mechanotherapy alone. Pooled results for the entire cohort demonstrated that participants achieved a clinically and statistically significant reduction in SUI with 61% achieving functional urinary continence by 12 weeks (<10 g with 24-HR PW), and 71% achieving >50% reduction.
Professional clinical guidelines recommend a non-surgical approach to SUI as first line therapy. This may include lifestyle changes (e.g. fluid restriction, bladder training, caffeine avoidance) and PFMT.17,18 A large amount of published literature has assessed the effectiveness of PFM strengthening, which is currently the non-surgical standard of care, as identified in numerous international clinical guidelines.7,8,19 Many well-designed systematic reviews and meta-analyses have been done evaluating the efficacy of PFM strengthening.20,21 Furthermore, numerous studies have been done assessing various adjunctive therapies in addition to PFMT. A systematic review by Ayeleke et al., 22 identified 13 trials involving 585 women with UI using PFMT along with another active treatment compared to women receiving the same active treatment alone. Overall, there was not enough evidence to determine if the addition of PFMT to another active treatment would result in more reports of cure or improvement in urine leakage and better QoL, compared to the same active treatment alone. Neuman and colleagues performed a systematic review that included 24 studies assessing PFMT, most of which included adjunctive therapies (biofeedback, electrical stimulation, abdominal strengthening). The authors found no benefit in adding biofeedback, electrical stimulation, or abdominal muscle training to a PFMT protocol. 23
In aggregate, the results from the current study are superior to findings reported in the PFMT literature, including those that utilized additional adjunctive therapies. In particular, it was the short time between therapy initiation and SUI improvement from device treatment of only 5 min/day.
Mechanotherapy in UI
Mechanotherapy is a foundational component of PT when treating musculoskeletal disorders. PT for incontinence may include manual intra-vaginal digital mechanical techniques of the pelvic floor structures, as well as other integrative approaches. 10 In addition, other forms of mechanical therapy have recently been investigated in women with UI. Sønksen et al. evaluated transcutaneous mechanical nerve stimulation using perineal vibration in 33 women with SUI. They found that after 6 weeks of treatment in subjects with SUI, there was a significant reduction in daily incontinence episodes and pad use. At 6 weeks 73% had no incontinence episodes, with durability through 3 months in combination with a standard PFMT program, but without additional vibratory stimulation. 24
Whole body vibration (WBV) for the treatment of UI was evaluated in numerous studies and summarized in several systematic reviews.25,26 In synopsis, these studies report that WBV alone or in combination with physiotherapy:
Improves PFM strength and QoL in individuals with UI
Does not cause PFM fatigue in nulliparous continent women
Leads to higher PFM activation in participants with weakened PFM, and achieves higher pelvic floor activation than maximum voluntary contraction alone
Has a significant effect on the electro-myographic response and the rating of perceived exertion significantly increased with increased frequency of the mechanical vibration
Leads to reactive activation of weakened PFMs
Improves pelvic floor dysfunction and myometric parameters, including the muscle logarithmic decrement, muscle frequency, and muscle stiffness, as well as reduction in perceived disability.
A systematic review by a Mazur-Bialy and colleagues explored the possibilities of using physiotherapeutic techniques (including vibration training) in the treatment of UI in women with attention to the techniques of PFM activation. They found that intra-vaginal vibratory stimulation significantly increased the effectiveness of PFMT, while being more effective in improving PFM strength than transvaginal electro-stimulation when applied in women with SUI. They state that intra-vaginal vibratory therapy can be considered supportive of the therapeutic process in patients with PFM or UI dysfunction. 9
Flyte mechanotherapy builds on the methods reported in the above studies by developing an optimally shaped intra-vaginal probe that delivers specifically tuned mechanical pulses in close proximity to the target muscle groups in the pelvic floor. Furthermore, benefits were achieved with only 5 min of daily use.
Limitations of the study
There are several limitations in this study. First, it was found that the sample size was underpowered to identify superiority of mechanical pulses (Part 2 mechanotherapy) added to the muscle stretch and preloading provided by the wand itself (Part 1 mechanotherapy). Thus, the study design was not adequate due to the larger than expected positive effect of the wand alone. Post hoc calculation indicated that 50 additional subjects (25 in each arm) were needed to demonstrate superiority of the added mechanical vibratory effect. The practical consequence of this distinction between study arms is uncertain, as the commercial device provides both components of the mechanotherapy. When the study data is pooled for both groups, the benefit of this therapy over baseline was highly significant for both objective and subjective endpoints. In addition, some women experienced discomfort using the wand. While this was anticipated, as with muscle soreness noted after any muscle exercise, a smaller wand configuration is now the standard product available. Finally, the study did not assess variations in therapy delivery, for example, duration of muscle contraction against the device, length of each therapy session, and length of therapy beyond 12 weeks.
Finally, further investigation is needed into the effectiveness of Flyte therapy in other patient populations who might benefit from PFM strengthening, such as post-partum urinary stress incontinence, mixed incontinence, urge incontinence, and fecal incontinence.
Conclusion
To our knowledge, Flyte is the first UI therapeutic device that utilizes the scientific principles of mechanotransduction and tensegrity. Two-part mechanotherapy provided by this intra-vaginal mechanotherapy device significantly improved objective endpoints assessed by 24-HR PW, as well as subjective measures evaluated by two disease-specific QoL instruments with only 5 min of daily use. These improvements were noted in as little as 2 weeks and appear to be sustained. Future studies are needed to determine optimal therapy protocols, and to assess the efficacy of this promising therapy in other pelvic floor disorders, or to skeletal muscle abnormalities more broadly.
Supplemental Material
sj-docx-1-tau-10.1177_17562872241228023 – Supplemental material for Randomized trial of mechanotherapy for the treatment of stress urinary incontinence in women
Supplemental material, sj-docx-1-tau-10.1177_17562872241228023 for Randomized trial of mechanotherapy for the treatment of stress urinary incontinence in women by Nissrine Nakib, Suzette Sutherland, Kevin Hallman, Marcus Mianulli and David R Boulware in Therapeutic Advances in Urology
Supplemental Material
sj-docx-2-tau-10.1177_17562872241228023 – Supplemental material for Randomized trial of mechanotherapy for the treatment of stress urinary incontinence in women
Supplemental material, sj-docx-2-tau-10.1177_17562872241228023 for Randomized trial of mechanotherapy for the treatment of stress urinary incontinence in women by Nissrine Nakib, Suzette Sutherland, Kevin Hallman, Marcus Mianulli and David R Boulware in Therapeutic Advances in Urology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
