Abstract
Objective
This study aimed to evaluate the clinical outcomes and safety of combined duloxetine and tolterodine therapy in patients with mixed urinary incontinence.
Methods
A retrospective analysis was conducted on patients with mixed urinary incontinence treated between January 2021 and December 2023. Patients received tolterodine (4 mg/day) and duloxetine (40 mg twice daily). Clinical outcomes were assessed at weeks 4 and 12 using the Overactive Bladder Symptom Score, International Consultation on Incontinence Questionnaire–Short Form, pad tests, and bladder diaries. Patient satisfaction was evaluated using the Clinical Global Impression scale.
Results
The study included 115 patients (mean age: 46.6 ± 13.4 years). Significant improvements were observed in Overactive Bladder Symptom Score, International Consultation on Incontinence Questionnaire–Short Form scores, and pad weights at weeks 4 and 12 compared to baseline (p < 0.001). Daily micturition, nocturia, urgency, and incontinence episodes significantly decreased, whereas mean voided volume increased. According to Clinical Global Impression scores, 73.9% of patients reported partial or complete improvement. Treatment-related adverse events were observed in 12.1% of patients.
Conclusion
The combination therapy of duloxetine and tolterodine appears to be safe and potentially beneficial, reducing symptom scores and incontinence episodes. However, these findings need to be supported by controlled studies.
Keywords
Introduction
Urinary incontinence (UI) is the involuntary leakage of urine. 1 Three types of UI are commonly observed. The first is urge urinary incontinence (UUI), which refers to involuntary urine leakage following a sudden urge to urinate. The second is stress urinary incontinence (SUI), which occurs with increased intra-abdominal pressure, such as coughing, sneezing, or laughing. The combination of these two types of UI is called mixed urinary incontinence (MUI).1,2
The prevalence of these types of UI, which are commonly observed in women, ranges between 25% and 45%. 3 Several factors may contribute to the etiology of UI. Although UUI is typically idiopathic in origin, it may also occur in various neurogenic disorders, including spina bifida, multiple sclerosis, and Parkinson’s disease. 4 SUI is caused by defects in the pelvic floor due to conditions such as childbirth, pelvic surgery, and weight gain. These two types of UI often occur together in most patients. 5
UI affects quality of life, interactions with the physical environment, and psychological well-being. 2 SUI is most commonly observed in middle-aged women, whereas UUI and MUI are observed in older individuals. A detailed medical history, bladder diary, basic physical examination, and urine analysis are recommended before treating the disease. 6
MUI may be more resistant to treatment than the other two types. Typically, treatment should be directed toward the predominant component of UI.7,8 Some behavioral therapies, including weight loss, avoidance of excessive fluid intake, abstinence from alcohol and caffeine, prevention of constipation, and pelvic floor muscle exercises, are considered first-line treatment methods.9,10 Anticholinergic agents and/or beta-3 agonists are used for the medical treatment of UUI, 11 and duloxetine, a serotonin–norepinephrine reuptake inhibitor (SNRI), may be preferred for SUI. 12 Onabotulinum toxin A injection or sacral neuromodulation can be considered in cases resistant to medical treatment for UUI, and mid-urethral sling surgery, autologous sling procedures, or urethral bulking agents can be used for the surgical treatment of SUI. 13
The only drug indicated for MUI treatment so far is duloxetine. 12 However, its efficacy alone may be insufficient, particularly in MUI with predominant urge symptoms. In the literature, the combination of duloxetine and mirabegron has been reported to be potentially effective in the treatment of urge-dominant MUI. 14 However, the long-term effects of beta-3 agonists are not well known because their therapeutic effects occur late.
This study aimed to evaluate the clinical outcomes and safety of duloxetine and tolterodine treatment in patients with urge-dominant MUI. Tolterodine was selected based on its proven efficacy and tolerability in reducing UI episodes, as demonstrated in previous studies. 15
Materials and methods
Data from patients with MUI aged >18 years who presented to the urology outpatient clinic between January 2021 and December 2023 were analyzed retrospectively after obtaining approval from the Ethics Committee (2023/483). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines, 16 and the study was conducted in accordance with the Declaration of Helsinki (1975, as revised in 2024).
Patients with urge-predominant MUI were included in the study. This condition was defined as having predominant UUI symptoms with at least one stress incontinence episode and at least three urge incontinence episodes on a 3-day voiding diary. Patients with active urinary tract infection, urinary tract calculi, a history of urinary tract malignancy, previous surgical treatment for UI, grade 3–4 cystocele, or prolapse were excluded. Patients presenting with isolated SUI or isolated UUI were also excluded from the study. Post-micturition residual volume (PMR) was calculated using the ellipsoid formula (length × width × height × 0.52). A PMR >150 mL was established as an exclusion criterion, as increased residual urine volume may indicate voiding dysfunction or bladder outlet obstruction, thereby complicating MUI management.
Prior to treatment, patients were evaluated based on urine analysis, urine culture, ultrasonography (to determine PMR after urination), and clinical examination (abdominal, pelvic, and perineal).
SUI was demonstrated by a provocative stress (cough) test in both lithotomy and standing positions. The type of incontinence was determined through UI-related questioning, and the predominant symptoms were identified based on the patient’s history. The Overactive Bladder Symptom Score (OABSS) and the International Consultation on Incontinence Questionnaire–Short Form (ICIQ–SF) were completed both before and after treatment to evaluate symptom severity.
OABSS is a validated patient-reported questionnaire evaluating daytime frequency, nocturia, urgency, and UUI. 17 A total score between 0 and 15 points can be obtained from the questionnaire. The urgency question is considered fundamental for diagnosis, and a score of at least 2 points on this item is interpreted as favoring overactive bladder. Higher questionnaire scores are associated with greater severity of UUI. The language validation was performed by Culha et al. 18
The ICIQ–SF is a validated patient-reported measure evaluating the frequency, severity, and impact of urinary leakage using three questions. A score between 0 and 21 can be obtained from the questionnaire. Higher scores indicate greater symptom severity and poorer quality of life. The language validation was performed by Çetinel et al. 19
A 24-h pad test was performed. The test began with an empty bladder, and the patients were asked to perform normal daily activities. To avoid inaccurate results, the pads were recommended to be worn with waterproof underwear. At the end of the 24-h period, the pads were weighed, and the difference between the weights before and after the test was calculated.
Patients received 4 mg tolterodine daily (Detrusitol, Viatris; USA) and 40 mg duloxetine twice daily (total 80 mg/day) (Nexetin 40 mg, Nobel İlaç; Turkey). Treatment responses were evaluated after weeks 4 and 12. Adverse events were assessed according to the Common Terminology Criteria for Adverse Events (CTCAE). 20 Patient satisfaction with the treatment was assessed using the Clinical Global Impression (CGI) scale. A bladder diary was completed at baseline and at weeks 4 and 12.
Statistical analysis
Data analysis was performed using Statistical Package for Social Sciences (SPSS) 25.0 (IBM; Armonk, USA) software. The distribution of the data was evaluated using the Kolmogorov–Smirnov test. A paired-samples t-test and Fisher’s exact test were applied for pre- and post-treatment comparisons. A p-value <0.05 was considered statistically significant.
Results
After applying the exclusion criteria to a total of 137 patients, data from 115 patients were analyzed (Figure 1). The mean age of the patients was 46.6 ± 13.4 years, and the mean body mass index (BMI) was 30.6 ±8.2 kg/m2. The duration of complaints was 41.9 ± 43 months. Smokers and nulliparous patients constituted 20.9% and 15.7% of the study population, respectively. The mean number of births was 2.2 ± 1.7. A total of 20.9% of the patients were smokers. Demographic characteristics of the patients who participated in the study are presented in Table 1.

CONSORT flow diagram.
Demographic characteristics.
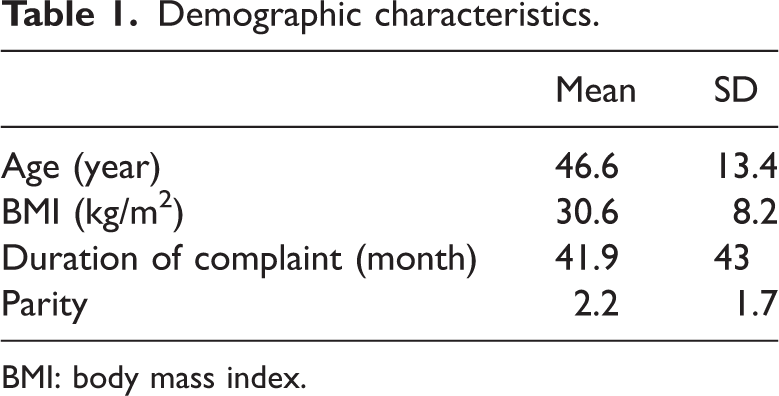
BMI: body mass index.
Prior to treatment, the mean OABSS was 7.7 ± 2.1, mean ICIQ–SF was 9.7 ± 2.4, and mean 24-h pad weight was 29.1 ± 11.5 g. A combination treatment of duloxetine and tolterodine was administered for 12 weeks. After treatment, significant improvements were observed in OABSS, ICIQ–SF scores, and pad test results beginning from the week 4 and continuing up to week 12 (p < 0.001; Table 2).
Symptom scores and pad test comparison between pretreatment and post-treatment.

ICIQ–SF: International Consultation of Incontinence Questionnaire–Short Form; OABSS: Overactive Bladder Symptom Score.
Table 3 presents a comparison of bladder diary entries before and after treatment. Analysis of the bladder diary data showed a statistically significant decrease in the number of daily voids and urgency episodes and a statistically significant increase in the mean micturition volume (p < 0.001). Prior to treatment, the mean numbers of nocturia episodes and incontinence episodes were 2.7 ± 0.8 and 3.3 ± 2.2, respectively. After treatment, these values changed to 1.1 ±1.1 and 1.8 ± 2.1 at week 4 and 1 ± 1.4 and 1.9 ± 2.4 at week 12, respectively. Significant reductions in both nocturia and incontinence episodes were observed at weeks 4 and 12 (p < 0.001).
Bladder diary comparison between pretreatment and post-treatment.

When patient satisfaction was assessed according to the CGI scale, 85 (73.9%) patients reported partial or complete improvement. During the 12-week period, no patient was excluded from the study. Treatment-related adverse events were observed in 14 (12.1%) patients. The most common side effects were constipation in 8 (6.9%) patients (7 patients experienced intermittent (CTCAE grade 1) symptoms and 1 patient had persistent (CTCAE grade 2) symptoms), dizziness and nausea in 4 (3.4%) patients (mild symptoms in all patients (CTCAE grade 1)), and dry mouth in 2 (1.7%) patients (mild symptoms in all patients (CTCAE grade 1)). However, none of the patients discontinued treatment due to side effects.
Discussion
The combination treatment of duloxetine and tolterodine for MUI was associated with improvements in symptom scores and pad test results. Patients reported satisfaction with the treatment, and no discontinuations due to adverse effects were observed.
UI is a major contributor to medical expenditure for patients and healthcare systems worldwide. Early diagnosis and treatment of the disease can play an important role in patient care and cost reduction. 21 Patients should be evaluated using a detailed medical history, a bladder diary, and validated questionnaires to assess the severity of UI. When planning treatment, underlying preventable causes should first be eliminated. Subsequently, conservative treatments, such as pelvic floor muscle exercises, are recommended as the first step according to the type of incontinence. 22
MUI is slightly more resistant to treatment than isolated UUI or isolated SUI. Particularly in the treatment of stress-dominant MUI, surgical treatment may be considered as a primary option. Although mid-urethral sling surgeries have high success rates, mesh removal procedures have come to the forefront in recent years because of mesh-related complications. Therefore, it is recommended that treatment decisions for SUI be made jointly with the patient after thoroughly informing them of the advantages and disadvantages of these treatment options. 23
In addition, procedures such as transobturator tape (TOT) and tension-free vaginal tape (TVT), which are used in the treatment of urge-dominant MUI, may increase urgency symptoms. 24 In the treatment of MUI, tolterodine, an anticholinergic agent, and duloxetine, a SNRI, have been used. 12 Guidelines recommend duloxetine with a high level of recommendation for the treatment of MUI. However, duloxetine treatment alone may be insufficient to prevent UUI, particularly in patients with predominant urgency symptoms.
Ariman et al. 14 evaluated the efficacy of duloxetine in combination with mirabegron, a beta-3 agonist. The authors reported a patient satisfaction rate of 62.5%. There was also a decrease in symptom scores and the number of pads used per day. In the group receiving this combination therapy, mirabegron-induced hypertensive attacks and jaw spasms associated with the duloxetine combination were observed. The desired therapeutic effect was achieved later, likely due to delayed receptor saturation with mirabegron. In our study, duloxetine was combined with tolterodine, a fast-acting anticholinergic. The findings demonstrated that this combination therapy provided significant improvement in OABSS and ICIQ–SF scores within the first month, and this effect persisted until the end of week 12. However, both studies are single-arm retrospective studies, and the lack of a control group prevents comparative evaluation of the efficacy of combination therapies, allowing only the expression of potential benefits.
When side effects were analyzed, duloxetine-related central nervous system and gastrointestinal adverse effects were frequently observed in previous studies. In the study by Ariman et al., 14 hypertensive attacks were also reported in addition to these side effects. In our study, adverse effects were observed in a total of 14 patients, and the most common adverse effect was constipation, which was believed to be related to tolterodine. None of the patients experienced side effects that led to discontinuation of treatment.
The study has several limitations. The most important is the small number of patients. The relatively short follow-up period can also be considered another limitation, as it is a disadvantage when evaluating the durability of the effect and long-term treatment-related adverse effects of anticholinergics. Furthermore, pre- and post-treatment evaluations of the patients were conducted through questionnaires, and no invasive tests were used. The use of invasive tests may provide a more objective evaluation. Another important limitation of the study is its single-arm retrospective design. The absence of a control group prevents evaluation of the placebo effect, the effectiveness of concomitant behavioral therapies, or changes due to the natural course of the disease, thereby preventing definitive determination of whether the observed clinical outcomes are solely attributable to the efficacy of the drug combination. Therefore, our data need to be supported by controlled prospective studies.
Conclusion
The combination therapy of duloxetine and tolterodine appears to be safe and potentially beneficial, reducing symptom scores and incontinence episodes. However, these findings need to be supported by controlled prospective studies with longer follow-up periods.
Footnotes
Acknowledgments
None.
Author contribution
Hakan Polat: Methodology, Writing-Original draft preparation, Data Curation, and Supervision; Mithat Ekşi: Conceptualization, Writing-Reviewing and Editing; Alican Çatık: Data Curation; Azad Akdağ: Data Curation; Yusuf Arıkan: Visualization and Investigation; Ubeyd Sungur: Software and Formal analysis; Taner Kargı: Visualization and Investigation; Serdar Karadağ: Writing-Original draft preparation and Resources; Alper Bitkin: Supervision and Project administration.
All authors read and approved the final version of the manuscript.
Declaration of conflicting interests
No conflict of interest was declared by the authors.
Ethics committee approval
Ethics committee approval was received for this study from the ethics committee of Health Science University, Bakırköy Dr. Sadi Konuk Training and Research Hospital.
Experimental-informed consent
Written and verbal informed consent was obtained from the patients included in the study.
Financial disclosure
The authors declare that this study received no financial support.
