Abstract
Several mobile healthcare (mHealth) apps are available in various marketplaces, but there is still concern about their accuracy, data safety, and regulation. The goal of this review was to critically analyze the mobile apps created for education, diagnosis, and medical and surgical treatment of patients with kidney stone disease (KSD), as well as to assess the level of data security, the contribution of physicians in their development and adherence to the Food and Drug Administration (FDA) and Medical Device Regulation (MDR) guidance. A comprehensive literature search was performed using PubMed (September 2022), in the Apple App Store and Google Play store using relevant keywords and inclusion criteria. Information was extracted for the name of the app, primary and additional functionalities, release and last update, number of downloads, number of marks and average rating, Android/iOS compatibility, initial and in-app payments, data safety statement, physician involvement statement, and FDA/MDR guidance. A total of 986 apps and 222 articles were reviewed, of which based on the inclusion, 83 apps were finally analyzed. The apps were allocated to six categories about their primary purpose: education (n = 8), fluid trackers (n = 54), food content description and calculators (n = 11), diagnosis (n = 3), pre- and intra-operative application (n = 4), and stent trackers (n = 2). Of these apps, the number of apps supported for Android, iOS, and both of them were 36, 23, and 23, respectively. Despite a wide range of apps available for KSD, the participation of doctors in their development, data security, and functionality remains insufficient. Further development of mHealth should be carried out properly under the supervision of urological associations involving patient support groups, and these apps must be regularly updated for their content and data security.
Introduction
Urolithiasis, commonly known as kidney stone disease (KSD), is a widespread and growing health concern worldwide. 1 The incidence of this chronic disorder ranges from 1% to 13% depending on geographic region, age, and sex.1,2 The management of KSD involves multiple steps, including patient education, diagnosis, and medical or surgical treatment. Mobile health technologies, or mHealth, have emerged as promising tools to help manage KSD and improve patient outcomes. 2 The use of smartphones and related software has made it easier for patients to access medical resources and manage their disease, which can ultimately improve their well-being and disease prevention. 2 As of 2018, there were over 318,000 mHealth apps available on various marketplaces, which are nearly twice as many as in the 3 years prior.2,3 There are concerns about the accuracy of app content, data safety, and a lack of centralized regulation, however. 4
Despite the availability of several reviews on endourology apps, most of them focus on a limited number of categories. This leaves a gap in the literature on the role of mHealth in the education, diagnosis, and treatment of patients with KSD. 5 Therefore, the goal of this review is to critically analyze the mobile apps created for the education, diagnosis, and treatment of patients with KSD. In addition, we will evaluate the level of data security, the involvement of physicians in the app development process, and adherence to regulatory guidance from the Food and Drug Administration (FDA) and Medical Device Regulation (MDR).
In this review, we conducted a comprehensive literature search using PubMed, as well as searching the Apple App Store and Google Play store using relevant keywords and inclusion criteria. We extracted information on the name of the app, primary and additional functionalities, release and last update, number of downloads, number of marks and average rating, Android/iOS compatibility, initial and in-app payments, data safety statement, physician involvement statement, and FDA/MDR guidance. Despite the wide range of apps available for KSD, we found that the participation of physicians in their development, data security, and functionality remains insufficient. The further development of mHealth for KSD should be carried out under the supervision of urological associations involving patient support groups. In addition, these apps must be regularly updated for their content and data security.
In conclusion, this review highlights the potential benefits of mHealth in the management of KSD. There are concerns about the accuracy of app content, data safety, and a lack of centralized regulation, however. Further research is needed to better understand the role of mHealth in the management of KSD and to ensure its safety and effectiveness.
Materials and methods
The search was conducted in September 2022 in the Apple App Store and Google Play store with the following queries: ‘urolithiasis’, ‘kidney stone disease’, ‘KSD’, ‘water OR drink tracker’, ‘oxalate’, ‘purine’, ‘ureteral stent’, ‘stone’, ‘kidney stones’, ‘kidney calculi’, ‘ureteral stones’, ‘Calcium’, ‘Phosphate’, ‘Citrate’, and ‘diet’. The inclusion criteria included at least 1000 downloads OR at least 100 reviews OR an availability on both marketplaces OR statement on the physician involvement OR the presence of the statement that the app in question was not a medical device OR that the presented app could be used in the treatment of the patients suffering from KSD OR apps withdrawal from markets but assessed in studies. Applications designed for meetings and congresses, designed specifically for physician education, and those not available in English were excluded. Applications were considered suitable for further selection if they met at least one of the following criteria: (1) inform users about KSD, (2) calculate the daily water volume, (3) describe food content, (4) may be implemented in the diagnosis of the patients (according to the description), (5) support preoperative planning or intraoperative performance, and (6) facilitate postoperative follow-up. These criteria served as the basis for further clustering of the finally included apps.
Published literature on various smartphone and tablet-based apps for the treatment of KSD was also extracted in PubMed without restrictions on the year of publication with the following advanced query: (((((((((mobile) OR (app)) OR (mheath)) OR (android)) OR (iOS)) OR (smartphone)) OR (tablet)) OR (water tracker)) OR (stent tracker)) AND (urolithiasis). Moreover, the name of included apps was used to find studies with their clinical use. In the presence of several papers dedicated to the same software, the most informative one regarding app parameters was selected. Reviews as well as editorial comments were excluded. The selected articles were analyzed and classified according to the above-mentioned groups. Figure 1 shows the process of selecting the studies from the available literature for inclusion in the study.

Inclusion criteria for studies from the literature.
Information on all apps meeting the inclusion criteria was extracted from the description on the marketplaces or from the manuscript to disclose the following items: the name of the app, publisher/authors, primary and additional functionalities, release and last update years, number of downloads (available only for Android devices), number of marks and average rating, Android/iOS compatibility, initial and in-app payments, data safety statement, physician involvement statement, FDA/MDR guidance statement, and physician consultation statement. Data safety statement means its presence in the app description or within the app’s privacy policies provided by the developer, indicating that user-related data cannot be achieved by third parties or the statement that the app was developed in compliance with the Health Insurance Portability and Accountability Act (HIPAA). According to FDA and MDR guidance6,7 apps developed for education, water tracking, and food content calculation are not considered a medical devices. So, apps from other groups were checked for FDA/MDR guidance statements. A physician consultation statement means advice for users with KSD to seek specialist visits to interpret all app-provided data. Furthermore, all accessible apps were downloaded to Samsung Galaxy S21 Ultra (Android OS, version 11.0) or iPhone 12 (iOS, version 15.5) to recheck the above-mentioned features and statements. Moreover, the readability of the context within educational apps was calculated by entering all text into a dedicated website (https://readabilityformulas.com/free-readability-formula-tests.php) to reveal the average readability score among the next validated algorithms: the Flesch Reading Ease score, the Gunning Fog, Flesch–Kincaid Grade Level, The Coleman–Liau Index, The Simple Measure of Gobbledygook (SMOG) Index, the Automated Readability Index, and the Linsear Write Formula. The number of apps meeting the American Medical Association (AMA) recommendation and the National Institutes of Health (NIH) recommendation was calculated. Figure 2 shows the app selection process with the details of the process of selecting individual apps for inclusion in the study.

Inclusion criteria for selecting individual apps.
Features of all apps found only in the literature were extracted from articles. When analyzing the drawbacks in the ‘Discussion’ section, we took into account those available for usage (can be downloaded via Google Play, App Store, or dedicated websites) to reliably calculate proportions within each criterion, whereas the ‘Results’ section was formulated based on all included apps according to the study goal. Descriptive statistics were performed using SPSS, version 22.0 (SPSS, Inc., Chicago, IL, USA).
Results
A total of 986 apps and 222 articles were reviewed, of which based on the inclusion, 83 apps were permissible for the final analysis. A total of 78 apps were presented in Google Play and App Store and two of them were described in studies; one app is described in the literature and available via a dedicated website; the description of four apps was presented in the literature only. The apps were allocated to six categories in relation to their primary purpose: education (n = 8), fluid trackers (n = 54), food content description and calculators (n = 11), diagnosis (n = 3), pre- and intra-operative application (n = 4), and stent trackers (n = 3). Of these apps, the number of apps supported for Android, iOS, and both platforms were 36, 23, and 23, respectively. One app was withdrawn from marketplaces [Ureteral Stent Tracker (UST)], so extracting most of the above-mentioned features was not possible, but the app was included as it was clinically investigated in studies.
Education
Eight apps directed to the education of patients with KSD were detected (Table 1). Seven apps are present in Google Play and do not require initial and in-app payments. One app was described in the dedicated study. 8 The data security statement was specified in the description of the two apps. None of those presented in Google Play are created with physician participation and only three developers added the statement that their software is not a medical device and therefore should not replace professional advice. Notably, the apps’ content is at the secondary school level (6–12th grade level), with only one of them meeting the AMA recommendation for the sixth-grade reading level and five meeting the NIH recommendation for the eighth-grade level.
Education-related apps.

FAQ, frequently asked question; n/a, not applicable.
Water trackers
Fluid balance apps provide a huge potential to favorably affect self-management strategies in KSD. Summarily, 54 out of 83 software were dedicated to the calculation and tracking of daily consumed water (Table 2), and all of them are available on marketplaces. Two apps required initial payment (both for the IOS). The in-app payment was offered in 52 apps for the expanded functionality without any other ads. Of these apps, the number of apps supported for Android, iOS, and both platforms was 22, 18, and 14, respectively. Data safety was confirmed in the description of 33 apps. Specialist involvement in app development statements was missing for all apps. Seven apps advised users not to consider them as medical devices and obtained final recommendations from the physicians.
Water trackers and related apps.

Various parameters were used to calculate users’ daily water intake: weight (n = 51), sex (n = 47), daily physical activity (n = 22), wake-up and bedtime (n = 19), age (n = 10), climate (n = 9), the purpose of water balance (n = 5), type of fluid consumed (n = 5), pregnancy/breastfeeding status (n = 4), height (n = 4), comorbidity (n = 1), and toileting habits (n = 1). Interestingly, only one application indicated the source of the formula used for the daily water estimation.
Food content and calculators
A total of 11 apps were specifically developed to provide food content information and their calculation for patients with KSD (Table 3). Of these apps, the number of apps supported for Android, iOS, and both platforms them were 4, 4, and 3, respectively. The physician involvement statement was presented in only one app, whereas data safety was provided in six apps. The initial and in-app payments were required in three and four apps, respectively. Two developers indicated that such software was not the only resource for dietary information for KSD and should not replace physician consultation. Description and calculation on almost all food components essential for the patients with KSD were provided: oxalate (n = 7), uric acid (n = 3), calcium (n = 2), sodium (n = 1), phosphate (n = 1), and citrate (n = 1).
Apps related to food content and calculators.

Natural remedies were described in two apps. Notably, both were created without physician participation and did not recommend users seek professional help.
Diagnosis
In accordance with the inclusion criteria, three apps could be implemented in the diagnosis of KSD according to their description, enabling to record, and sharing the urinalysis (one app), 24-h urine with the risk calculation (one app available on a dedicated website), 9 and albumin-to-creatinine ratio (one app); the latter could be used only in the United Kingdom with the designated test kit (Table 4). Data safety statements were presented in all three apps, whereas only two apps were created with physician participation. Initial and in-app payments were required in one and two apps, respectively. Finally, only an app for the urinalysis recorder was accomplished with the statement on seeking a doctor’s help to interpret and correct urinary parameters. None of the apps from this group indicated accordance with FDA/MDR.
Apps related to diagnosis.

FDA, Food and Drug Administration; MDR, Medical Device Regulation; n/a, not applicable.
Pre- and intra-operative application
Despite active attempts to reduce the incidence and recurrence of KSD, there remains a huge percentage of patients who need surgical intervention, which makes it important to introduce mHealth in this area as well. Only two apps were available, which are free without any in-app payments and created with physician involvement (Table 5). Notably, ‘Stone Pass – Kidney Stone Pain: Passing vs surgery’ (KnowStone LLC) aims to predict the chance of spontaneous stone passage and warn patients not to use the calculation results without consulting a urologist, while app with the calculation of percutaneous nephrolithotomy (PCNL) STONE and GUY’S scores recommend urologists to check results according to hospital protocols. None of these are provided with FDA/MDR or data safety statements.
Apps related to pre- and intra-operative applications.
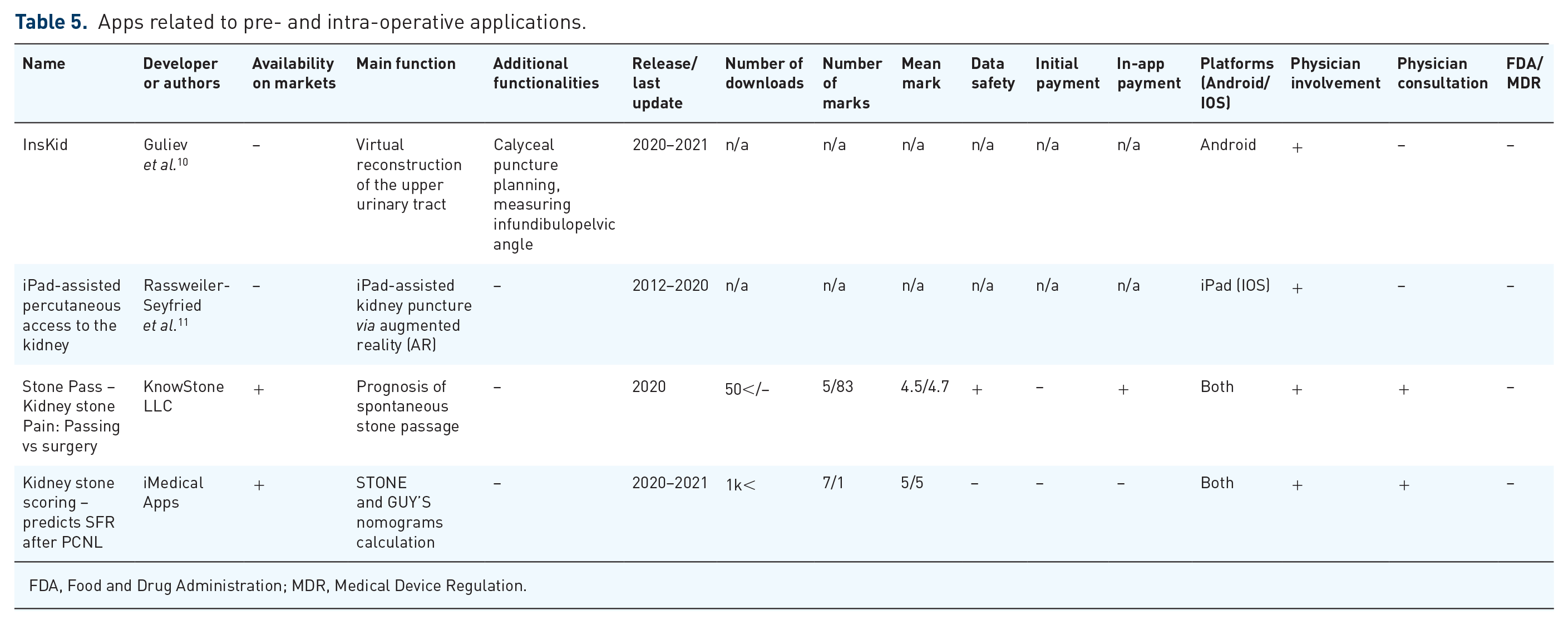
FDA, Food and Drug Administration; MDR, Medical Device Regulation.
Another two methods using smartphone and tablet apps were revealed from the literature, one to measure pelvicalyceal anatomy and the other to help with PCNL puncture. InsKid, described by Guliev et al., is an app for smartphones based on Android OS for mobile virtual endoscopy. 10 With the help of a smartphone and three-dimensional (3D) reconstruction of the pelvicalyceal system (PCS), it is possible to better advise patients, measure the infundibulopelvic angle (IPA) as well as determine the orientation of small calyces to plan percutaneous access trajectory preoperatively.
Rassweiler-Seyfried et al. 11 formulated the results of intraoperative iPad-assisted percutaneous access to the kidney via augmented reality (AR) technology compared with standard PCNL. The iPad was used as a camera, and a screen was to send the data via wireless local-area network (WLAN) to the server and display various visualization models. The computer used was a laptop for real-time calculation and playback. No intraoperative complications occurred using this. The radiation exposure was higher in the iPad-assisted group, which could be explained by the dependency of the puncture on the stability and precision of the program.
Stent trackers
The placement of ureteral stents is an essential element of stone procedures. Forgotten ureteral stents (FUSs) are associated with encrustation and upper urinary tract obstruction.
Two apps purposefully developed to track ureteral stents are available (Table 6). Moreover, in one app (StoneMD), stent tracking includes an additional function including the calculating risk of stone recurrence. 12 Urostentz (RAYZ CHARTERED Inc.) is free of charge app for android-based gadgets; the iOS version is submitted and the same shall be available soon on the App Store. Moreover, Urostentz makes it possible to track stent-related symptoms and be in touch with patients in a two-way discussion. In the study by Hameed et al., 13 among 92 patients who downloaded the Urostentz application, there was no case of FUS, which emphasizes the need to keep in touch with patients and remind them to remove the stent.
Stent tracking apps.

FDA, Food and Drug Administration; MDR, Medical Device Regulation; n/a, not applicable.
Stent tracker (One Advertising and Communication Services Ltd.) is a freely available app for both platforms with the function of tracking stent indwelling time. There is no statement on data safety or physician involvement, however. In the literature, there are several papers describing UST™ usage, developed by Visible Health Inc. in partnership with Boston Scientific Corporation; 14 however, it was recalled from iOS and Android markets.
Discussion
Role of digital technologies
Digital technologies are rapidly gaining popularity around KSD and are mostly presented as Social Media (SoMe), telehealth, and mhealth. SoMe include different public use platforms such as Facebook, Twitter, 15 and YouTube. 16 Concerns, however, exist about the use of social media in the clinical context. Not all authors follow guidelines specific to the relevant use of social media in decision-making processes which is detrimental to the information quality. 17 According to Jamnadass et al. 18 SoMe and search engines provide valuable information to patients with KSD. While the information provided regarding dietary aspects and fluid management was good, however, it was not comprehensive enough to include advice on other aspects of KSD prevention. Besides, it compromises patient confidentiality, especially when public platforms like Twitter are utilized. 19
Telehealth and mHealth
Telehealth encompasses distant patient monitoring, and other methodologies allowing remote care and may be introduced across different modalities such as telephone, email, audiovisual recording devices, videoconferencing, and soft and wearable devices. Telehealth, however, is now being reservedly popularized in urology. Hindrances to its embodiment contain technological competence, refund uncertainties, and resistance to change in working processes. 20
The World Health Organization (WHO) characterizes mHealth as medical and public health practice supported by mobile devices such as smartphones, tablets, and wearable devices. 21 ‘Mobile’ is the key word to understand the difference between telehealth and mHealth. The former refers to all examples of healthcare via contemporary technology, while mHealth indicates the concept of mobile self-care and customer technologies such as smartphone and tablet apps. Although they can surely be helpful for patients and attending physicians, the central issue concerns the regulation of their use.
FDA and MDR regulations
According to FDA regulation, apps developed for education, water tracking, and food content calculation are not considered a medical devices, and so may be popularized without any associated statements. Those used precisely for diagnosis, treatment planning, intraoperative assistance, and post-treatment follow-up could be potentially harmful if do not work correctly and should correspond to FDA regulations. 6 Among the apps investigated, 10 referred to these groups; 6 were presented in Google Play and App Store and 1 was available via a website. None of these are provided with the such statement in their description, however.
The same is stated by the MDR [EU] 2017/745. 7 Apps created for education and lifestyle correction (water and food consumption) refer to class I (low-risk) and should not be developed within regulation statements. Mobile apps and software that are created for the diagnosis, monitoring, prognosis, and treatment or alleviation of disease are considered medical devices and correspond to the class IIa (low-to-medium risk) or higher and should be accomplished with Quality Management System (QMS) certificate at least. Again, investigated apps corresponding to these purposes were not provided with an MDR compliance statement. From this point of view, it becomes obvious that developers have a huge responsibility in the interpretation of the app’s designation and must follow current policies or advise to seek a doctor at least.
Physician guidance with mHealth apps
In our review, among the 79 apps available for usage (both placed on the markets or dedicated website), only 15 (19%) accomplished a notice that users should not interpret app-provided data themselves and seek physician consultation. In theory, users of the remaining 64 applications could perceive obtained recommendations as guidelines and may not seek a physician consultation to help them. For example, according to the systematic review performed by Gamage et al., 22 fluids low in calcium seem to reduce the risk of KSD. Healthy users may not have any risks when correcting the fluid balance, while this needs to be tailored to individual patients with other diseases such as KSD or heart failure. For this reason, all applications in their description should advise patients with any diseases to consult a doctor.
Although some studies confirm the benefits of such applications for patients with KSD,8–14 recruited patients were under physician supervision, which prevented patients from falsely interpreting the apps’ recommendations and making decisions about their treatment themselves. According to our review, only 5/79 (6.3%) apps available for usage were developed with the participation of doctors and were from diagnosis, pre-, and intra-operative use, and stent tracker groups. Of the other app groups, only one had physician involvement.
Limitations in mHealth app
There are limitations to mHealth apps, especially around the updates available. Indirectly, the year of the last update of the application/date of publication may also correspond to the quality of information available to users. In our review, only 35/79 apps available to download (43.3%) were updated in 2022, and the remaining 44 contained data from 2021 and earlier, which is a clinical problem due to the constant updating of KSD guidelines and the origin of a huge number of publications regarding the treatment of patients with urolithiasis. Another issue lies in the guarantee of the safety of user data. Ideally, medical apps should be compliant with the HIPAA and provide a special statement in the description. Among 79 apps accessible for download by nonmedics, only 46 provided statements on data safety.
Future of mHealth
mHealth is a steadily developing field and has the potential to play an essential role in urological care and education. 23 In a recent survey, Mantica et al. 24 identified an almost eightfold increase in these apps in the field of urology between 2012 and 2020. The same trend was observed in our review for the fluid apps, from 18 to 54, 5 although their inclusion criteria were different. The lack of regulatory supervision and healthcare professionals involved in the development of apps raises concerns regarding their accuracy and safety.
All applications aimed at treating kidney stone formers therefore should be developed only with the participation of specialists and leading organizations in the field of urolithiasis. Recommendations should be personalized because what is useful for one is harmful to the other. The application interface should include items listed in the guidelines. If the software is created as a medical device, developers must follow special regulatory norms. To ensure patient privacy, developers should satisfy dedicated acts, such as HIPAA. Finally, it is necessary to constantly monitor the effects of such apps on the treatment of patients with KSD globally with the participation of a larger number of patients to easily detect application flaws and to update them regularly.
This is the first study to critically analyze apps targeted toward all aspects of the management of patients with KSD. In the absence of a unified policy for evaluating mHealth apps, we used a set of search items and grouping in accordance with the apps’ function. Authorship disclosure within the description in the markets and apps themselves can be difficult to recognize; hence, the extent of physician involvement may be undervalued. Moreover, without any structured assessment of educational apps, we were not able to formally assess them. Our searches were limited to the largest stores (Google Play and the Apple App Store), not reflecting the state of the less popular marketplaces.
Conclusion
In conclusion, the prevalence of urolithiasis has been increasing globally, and the use of mobile health technologies (mHealth) has become a popular method for managing this condition. The availability of mHealth apps has significantly increased over the past years, with over 318,000 apps present on various marketplaces. There are concerns regarding the accuracy of their content, data safety, and centralized regulation, however. This review aimed to critically analyze the mobile apps developed for the education, diagnosis, and treatment of patients with KSD and to assess the level of data security, physician involvement, and adherence to FDA and MDR guidance. Our findings revealed that there are a wide range of apps available for KSD, but the participation of doctors in their development, data security, and functionality remains insufficient. The majority of the apps were fluid trackers, and only a few were designed for diagnosis, pre- and intra-operative use, stent tracking, and food content description and calculators. Furthermore, most of the apps lacked data security statements and only a few indicated physician involvement in their development. Adherence to FDA and MDR guidance was also inadequate. Therefore, it is recommended that the development of mHealth apps for KSD should involve the supervision of urological associations and patient support groups. Furthermore, apps should be regularly updated for content and data security, and adherence to regulatory guidance should be ensured. This will help to improve the accuracy and safety of the apps and enhance their usefulness for patients with KSD.
