Abstract
Introduction:
Aims of this study were to evaluate the functional outcomes of a vaginal wall sling technique in patients with stress urinary incontinence at 20 years after surgery and to evaluate the patient’s satisfaction after the surgical procedure.
Material and Methods:
This was a prospective single-center study on patients with stress urinary incontinence who underwent in situ vaginal sling surgery. Presurgery evaluation included history, pelvic examination, and urodynamic test. All patients completed Urogenital Distress Inventory–6 (UDI-6) questionnaire. They underwent checkups at 1, 3, 6, and 12 months postoperatively and then annually. The sling was created by making a rectangle (15–20 × 25 mm) on the anterior vaginal wall and it was reinforced by one roll of Marlex mesh on each side of the sling. The sutures were passed through the vagina at the suprapubic level after suprapubic incision, above the rectus fascia and tied without excessive tension.
Results:
From May 1996 to May 2002, 40 women underwent vaginal wall sling surgery for stress urinary incontinence. Last visit was performed on 20 women between March 2020 and April 2020. Median follow-up was 251.3 months (20.9 years) (range = 204.3–285.4 months). The success rate after 5 years of surgical procedure was 80%; over 5 years, the objective cure rate was 45%. Considering only the group of 13 patients with pure stress urinary incontinence, the objective cure rate decreased to 38%, in particular 7 years after surgery. Women who did not resolve their urinary incontinence needed to undergo a new treatment. At over 5 years after surgery, there was an increase in urgency (p = 0.001) and voiding symptoms (p = 0.008) and urgency urinary incontinence (UUI) (p = 0.04). Ninety-five percent were very much worse or much worse according to the Patient Global Impression of Improvement (PGI-I) scale.
Conclusion:
The in situ vaginal wall sling does not guarantee good long-term functional outcomes in women with stress urinary incontinence.
Introduction
For years, autologous sling surgery has been the gold standard in the treatment for stress urinary incontinence (SUI). 1 Different muscles have been used over time as support for the urethra, such as the pyramidalis, 2 levator ani, 3 gracilis, 4 and bulbocavernosus muscles. 5 Hohenfelner reported that the muscle graft was first used by Giordano 1907. 2 It was hypothesized that the use of muscle as a support material could preserve the contractile capacity of the urethra and act as a neosphincter, creating urethral compression and partial obstruction. These procedures, however, came with complications, such as recurrent cystitis, fistulas, and urethral sloughing.
The studies of McGuire and Lytton 6 later introduced procedures that utilize the abdominal-vaginal approach. In these suspension procedures, slings of varying lengths (from 7–10 cm to 2–4 cm) were used and suspended by needle. The materials used varied from autografts (rectus fascia, fascia lata, dermis, vaginal skin), to cadaveric allografts (fascia, dermis, dura mater), to xenografts (porcine dermis, small intestinal submucosa, pericardium, bovine dermis).
The use of autologous slings has decreased with the introduction of less invasive surgical techniques such as the transvaginal tape (TVT) in 1995, the transobturator tape (TOT) in 2001, and the synthetic sling (nylon, polyethylene, polytetrafluoroethylene, and polypropylene). 7 By the year 2010, 250,000 surgical procedures by synthetic sling were performed using a mid-urethral sling (MUS). 8
The choices of materials and surgical techniques have changed since the US Food and Drug Administration (FDA) issued its first warning in 2008 on the use of synthetic material in pelvic surgery for both SUI and urogenital prolapse (POP). This was due to reports of complications such as vaginal pain (especially during sexual intercourse), vaginal exposure or extrusion, and urinary infections. 9 Complications due to vascular injury or bladder perforation also occurred less frequently. 9 Treatment of these complications occasionally requires hospitalization and new surgeries.
With the FDA update in 2011, complications related to the use of mesh in the vaginal correction of urogenital prolapse were reported. 10 More than 650 postmarket surveillance investigations were initiated for both POP and SUI mesh surgeries. 11 In 2016, POP meshes were classified as class III or high-risk devices by the FDA. 12 Although SUI slings were not included in the high-risk classification, numerous insurance claims and the spread of negative news in mass media created fear of the sling regardless of their use for POP or SUI. This fear affected both patients and surgeons, such that the use of MUS decreased over time. 13
Following these developments, procedures and materials that had been abandoned, 13 such as pubovaginal slings, were reintroduced. Our hypothesis is that the reintroduction of autologous sling procedures is not necessarily the right choice due to the lack of long-term results compared with MUS and the lack of an effective ban by the FDA on the use of synthetic material for urinary incontinence.
The primary objective of our study was to evaluate the long-term (20.9 years) outcomes of autologous sling procedures performed in our clinic in 1996. We utilized a modified in situ vaginal wall sling, a material first used by Raz in 1989. 18 The secondary objective was to evaluate long-term patient’s satisfaction with this procedure.
Material and methods
This was a prospective single-center study on patients with SUI and mixed urinary incontinence with predominant stress form who underwent in situ vaginal sling surgery. 14 From May 1996 to May 2002, 40 consecutive women with SUI underwent in situ vaginal sling surgery. 14 In a previous study, 14 we reported the results 5 years after the surgical procedure. Follow-up continued and the last visit was performed between March 2020 and April 2020.
We included women with severe SUI, Valsalva leak point pressure (VLPP) < 60 cm H2O, and/or maximum urethral closure pressure (MUCP) < 30 cm H20. 14 Twenty-seven patients had hypermobility. We excluded patients who have already undergone anti-incontinence surgery. Presurgery evaluation included history, pelvic examination using the Halfway system, 15 urodynamic test (including urethral pressure Profile), micturitional diary, a 1-h pad test, and transrectal dynamic ultrasound. 14 Urinary symptoms were evaluated using the standardized questionnaire Urogenital Distress Inventory–6 (UDI-6). 16 All patients underwent check-ups at 1, 3, 6, and 12 months postoperatively and then annually, with the preoperative protocol except for urodynamic test. They performed uroflowmetry, and at last visit, they completed the Patient Global Impression of Improvement (PGI-I) scale questionnaire. 17
Surgical technique
All surgical procedures were performed by one senior surgeon (EC) according to a previously described surgical technique. 14 The sling was created by making a rectangle with two horizontal and two vertical incisions on the anterior vaginal wall. The upper horizontal incision was at the level of the bladder neck, while the lower on was about 1 cm posterior to the urethral meatus. The rectangle was about 15–20 × 25 mm. After preparing the sling, the proximal anterior vaginal wall edge was prepared to cover the vaginal island; the dissection was continued along the lateral edges of the sling and the endopelvic fascia was opened. One roll of Marlex mesh (Cousin Biotech, Wervicq-Sud, France) was positioned within a helicoidal suture (in 0-nonreabsorbable monofilament) on each side of the sling to ensure reinforcement. The sutures were passed through the vagina at the suprapubic level after suprapubic incision (1.5 cm) by ligature carrier. After adjusting the tension of sling aligning the posterior semicircle of the bladder neck with the anterior, the two suprapubic sutures were tied above the rectus fascia. The proximal vaginal wall flap was sutured with interrupted 2.0 Dexon sutures to cover the island of the vaginal wall. The suprapubic incision was closed. The roll of Marlex mesh was inserted in 14 cases; 24 patients had urethral hypermobility; in these cases, sutures were positioned on the urethropelvic ligaments. Patients have not undergone further simultaneous surgical procedure. The study was approved by the local ethics committee (CEAS Umbria) in 1996 14 As the present study is a continuation of this work, it was not necessary to ask for further approval. The results reported are those of the regular follow-up expected. Informed consent was obtained from all study participants.
Statistical analysis
McNemar for paired data and Fisher’s exact tests were used to compare categorical variables. The probability of SUI was calculated using Kaplan–Meier survival analysis. Patients were censored if they reached the end of follow-up without SUI. All calculations were performed using IBM-SPSS® version 22.0 (IBM Corp., Armonk, NY, USA, 2013). A two-sided p value < 0.05 was considered significant.
Results
From May 1996 to May 2002, 40 consecutive women underwent vaginal sling surgery for SUI. Functional outcomes 58.5 months after surgery were reported in our previous study in 2004. 14 Last visit was performed between March 2020 and April 2020. Median follow-up was 251.3 months (20.9 years) (range = 204.3–285.4 months).
Six patients were lost to follow-up and 12 had passed away. In this last report, we have analyzed 20 patients. Of 20 patients, 14 underwent the implantation of a roll of Marlex mesh. In five women, we used only the vaginal wall sling without other supports. Table 1 showed the baseline demographic informations and clinical characteristics of the study population. None of the women evaluated at the last visit had undergone previous anti-urinary incontinence surgical procedures.
Baseline characteristics of the study population.
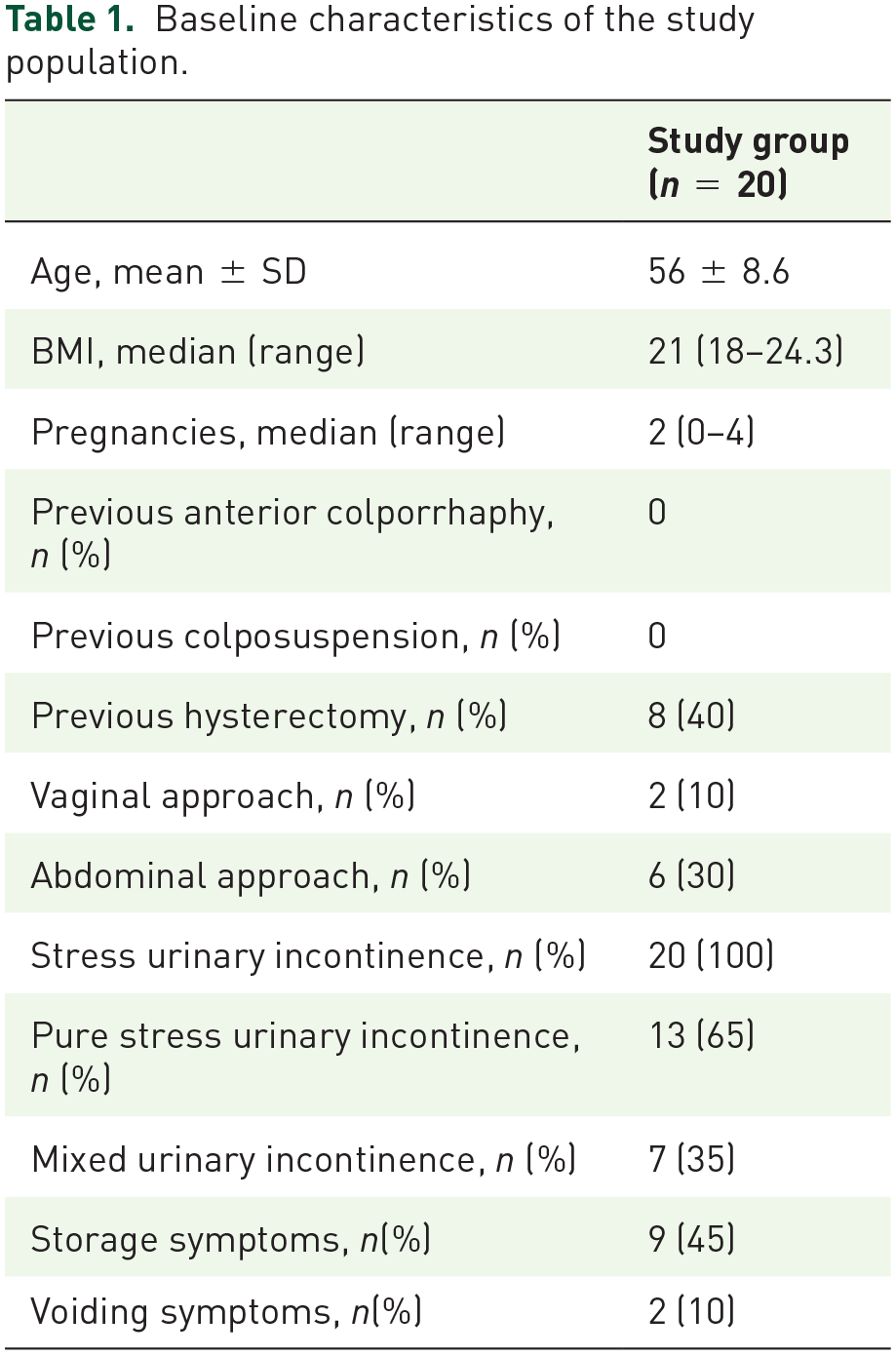
Table 2 showed the functional outcomes at 5 years and over 5 years after surgery. The data were the results obtained only with the vaginal wall sling, without further treatment to correct persistent or de novo urinary incontinence.
Functional data at baseline, 5 years after surgery, and over 5 years after surgery with only the implantation of vaginal wall sling.
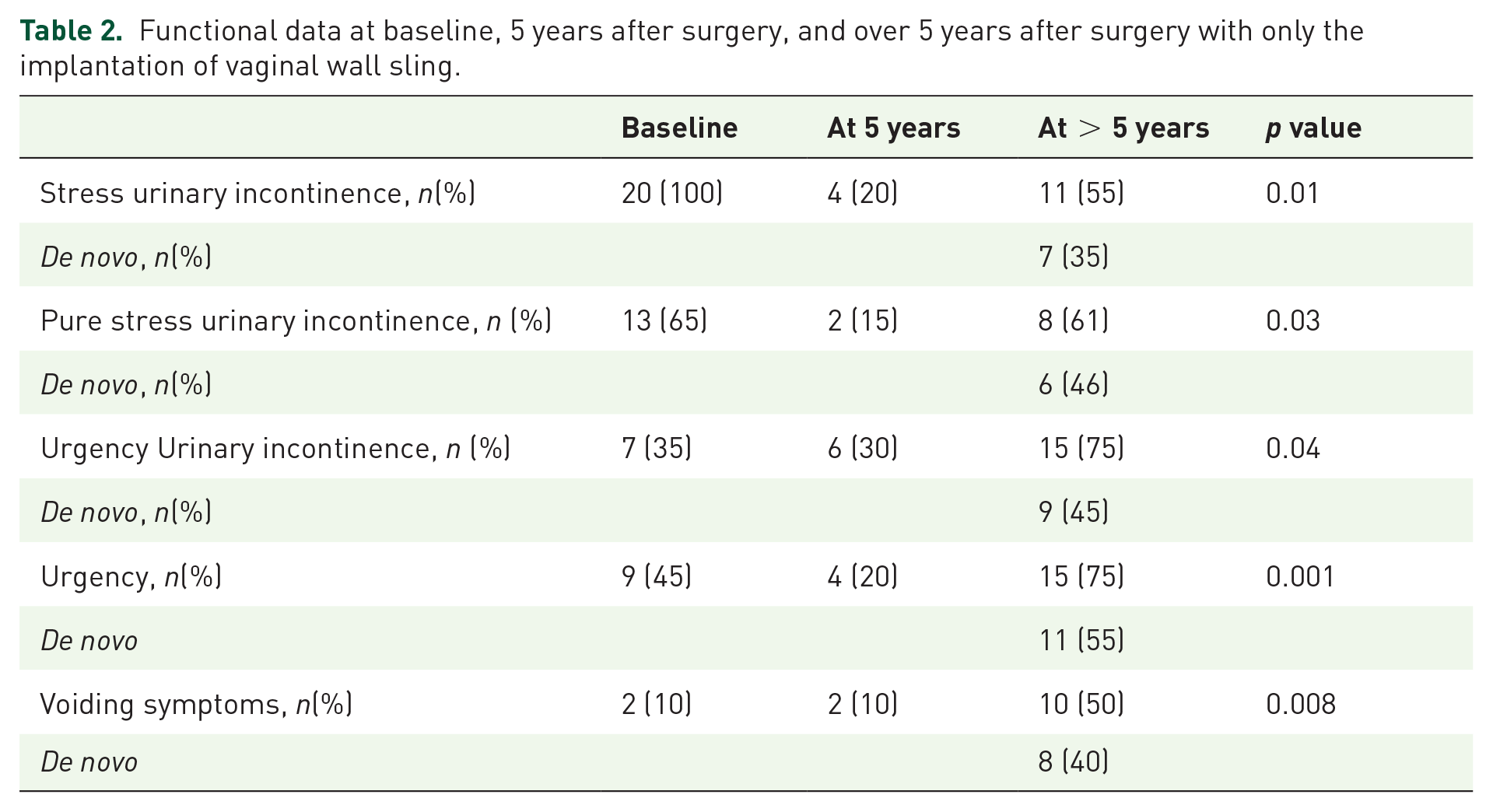
Urinary incontinence success rate
At over 5 years after surgery, we had a statistically significant increase in urgency from 20% to 75%, p = 0.001. Urgency appeared de novo in 55% of the entire sample (11 patients). Of the nine patients with preoperative urgency, seven also had urgency urinary incontinence. The urgency urinary incontinence resolved in 5% (1/20 patients) and persisted in 30% (6/20 patients). It appeared de novo in 45% of the entire sample (9/20 patients). All patients with overactive bladder (OAB) symptoms underwent treatment with functional electrostimulation or anticholinergic drugs. It persisted in 60% (9/15 patients).
The success rate 5 years after of surgical procedure was 80%; 14 over 5 years if we consider the whole sample of subjects with SUI, the objective cure rate was 45% (9/20 patients) and a subjective cure rate was 35% (7/20 patients). Four women underwent only vaginal wall sling without Marlex were incontinent at last visit. Incontinence appeared in the first 7 years after surgery.
Considering only the group of 13 patients with pure SUI (70% of the entire sample), we observed only with the implantation of vaginal wall sling an objective cure rate of 85% (11/13 patients) 5 years after surgery; over 5 years, the objective cure rate decreased to 38% (5/13 patients) and a subjective cure rate was 23% (3/13 patients). Considering only the group of seven patients with mixed urinary incontinence, the objective cure rate was 85.7% (6/7 patients) 5 years after surgery; over 5 years, the objective cure rate decreased to 42.8% (3/7 patients) and a subjective cure rate was 28.5% (2/7 patients).
The Kaplan–Meier curve (Figure 1) showed that over time there has been a progressive worsening of the cure rate of SUI after an initial success. In particular, 7 years after surgery, SUI increased noticeably. At the last visit, women reported the absence of stress incontinence because in the meantime (already 10 years after the surgical procedure) they underwent to other types of treatment. They are to be considered failures for the vaginal sling surgery.

Kaplan–Meier stress urinary incontinence after implantation with vaginal wall sling.
Women who did not resolve their urinary incontinence needed to undergo a new treatment. Of the 11 failed women, six women had not the rolls of Marlex mesh, of which four were incontinent within the first 5 years after surgery, 14 and they subsequently underwent anti-incontinence surgery by synthetic sling; the other seven patients became incontinent over 5 years. After an initial improvement with autologous sling, they developed incontinence again and were retreated by MUS.
Storage and voiding symptoms
In women with pure SUI, de novo urgency appeared in 92% of cases (12/13 patients) and de novo UUI appeared in 7.6% (1/13 patients), while in women with mixed urinary incontinence, de novo urgency appeared in 71.4% (5/7 patients) and de novo UUI appeared in 14.2% (1/7 patients).
Voiding symptoms increased from 10% (two patients) to 50% (10 patients) (p = 0.008). In particular, voiding dysfunctions persisted in 10% (2/20 patients) and they appeared de novo in eight women. In no patients did we observe a PVR >50 ml.
Not all patients annually completed the UDI-6 questionnaire so it is not analyzed in this study.
Complications
In the long term, they did not develop further complications beyond those already reported previously. 14 No complications related to the sling and the material used have been recorded. Ninety-five percent were very much worse or much worse according to the PGI-I scale.
Discussion
Our study showed that functional outcomes tend to decline after 20 years post-SUI correction using the in situ vaginal wall sling. Our previous study showed that after a mean follow-up period of 58.5 months (range = 12–92 months), the success rate of the procedure was 83.4% with a persistence rate of 16.6%. In this update, over the years the success rate has dropped dramatically. The patients were then referred to other types of treatment. There was also an increase in the number of patients with storage symptoms and voiding symptoms. Our results can hardly be compared with the data in the literature as there have been numerous variants of Raz’s technique 18 and recorded success rates have varied between 61% and 100%. 19
Our technique included a Marlex mesh reinforcement, 14 as we hypothesized that its absence could result to a weaker sling. Our results differ from those of Goldman et al. 20 and Kilicarslan et al., 21 possibly because their studies made use of a different technique and shorter follow-up period. These studies evaluated preoperative predictive factors of postoperative failure. Goldman et al. 20 found that 16 months after vaginal wall sling implantation, the success rate of SUI correction was 40% in patients with Valsalva leak point pressure (VLPP) < 50 cm H2O and 93% in patients with VLPP >50 cm H2O.
Kilicarslan et al. 21 evaluated maximum urethral pressure (MUP) as a predictive factor in addition to VLPP. The success rate was 65.4% in patients with VLPP < 50 cm H2O and MUP < 30 cm H2O; meanwhile, the success rate was 90.6% in patients with VLPP > 50 cm H2O and MUP > 30 cm H2O. Raz also stated that this technique was not indicated in women with severe sphincter deficit. 18 If we wish to use VLPP as a predictor of postoperative failure, our mean VLPP at preoperative evaluation was 39 cm H2O; this could explain our failures, but this factor and the preoperative urodynamic parameters are not sufficient to explain the long-term functional results. On the contrary, we recorded an immediate postsurgery success rate of 83.4% despite the mean VLPP being 39 cm H2O; this could signify that there are other contributing factors to the decrease in continence rate over time, probably related to the technique and materials used.
Our results instead agree with those of Rodrigues et al. 22 and Metin et al. 23 In both studies, the techniques utilized were different from our own, but a decrease in the long-term success rate was observed; the failure rate was 16% at 3 years postprocedure, 26% after 4 years, and 30% after 5 years.
The technical reasons for the decrease of vaginal wall slinging effectiveness over time are not known or explainable. One of the most probable reasons is that the pathophysiological mechanism of stress incontinence is based on the presence of an intrinsic sphincter deficit (ISD); this may be associated with urethral hypermobility. 24 Raz et al., 18 however, stated that vaginal wall slinging is not indicated in ISD and this could explain the decrease in success rate after an initial period of therapeutic success. In spite of numerous variations, vaginal wall sling implantation follows biophysical rules that, in our opinion, do not allow adequate support in the presence of increased abdominal pressure. Over the time, the fascia may become looser and the sutures anchoring the vaginal wall sling to the fascia become weaker. We used reinforcing Marlex mesh to avoid pull-through and to reinforce the patch. Marlex is biomechanically different from the modern polypropylene, monofilament macroporous mesh materials. If we were to perform these surgeries today, we would not have a Marlex mesh; probably the use of the different heterologous material would not have changed the results, as the failure is mostly caused by suspension sutures break or tear through the origin or insertion sites. Another fundamental role is played by the material. Probably the different material could influence the outcome if it were the part affixed under the bladder neck. No studies have compared the variations of material characteristics (autologous and heterologous) during the increase of abdominal pressure. It is probable that heterologous slings are more appropriate for the aim. Few studies have compared vaginal wall sling with other procedures, particularly with autologous rectus fascial sling. were compared in one RCT and two nonrandomized retrospective studies. In a nonrandomized study, 71 months after the success rate was higher in women who underwent autologous rectus fascial sling compared anterior vaginal wall sling (93.7% versus 79.8%). Unfortunately, there are no randomized studies in literature that compare the long-term results of vaginal wall slings and synthetic slings. Based on our results, however, the success rate of heterologous sling was higher.25 –27
In our previous study, 28 we have shown that after 10 years from TOT implantation, the objective cure rate is 80%; this is certainly higher than those recorded after vaginal wall sling implantation.
As a comparison, the autologous sling has often been used as a pubovaginal sling. Fusco et al.’s 8 review and meta-analysis of the literature on the functional outcomes and complications of synthetic and autologous slings (such as pubovaginal slings) showed that the objective and subjective cure rate are comparable; however, only one randomized controlled trial (RCT) has a follow-up period of more than 5 years. Conversely, the use of devices for MUS exposes the patient to an increased risk for complications such as bladder perforation and sling exposure or extrusion. In addition, the use of pubovaginal slings results in increased storage symptoms and risk of reintervention. 29 Also in our study there was an increase of urinary symptoms. The increase in storage symptoms may be attributed in part to aging and the utilized technique; a slight increase was actually recorded after the first year postimplantation. However, the complications related to autologous sling are possibly unavoidable as they seem to persist despite numerous technical variations.18 –20 Nonetheless, the complications related to synthetic slings could be potentially avoided by adequately training physicians and allowing only centers with a large volume of cases and adequate expertise to perform the implantation. 30 In 2015, the Scientific Committee on Emerging and Newly Identified Health Risks (SCENIHR) report 31 reassured urogynecologists by stating that synthetic slings are adequately studied, with more than 200 published clinical trials that establish its long-term efficacy and good safety profile when used in experienced hands. On the contrary, numerous devices that are now available on the market make heterologous slinging an increasingly less invasive technique that comes with shorter operating times and hospital stays. Choosing to implant an autologous sling is not necessarily the right choice both for the long-term results and for the actual immediate advantages perceived by the patient (operating time, hospital stay). The satisfaction rate after 10 years from the implant of synthetic sling is 80%. 25
The strengths of our study lie in its prospective design and long-term follow-up period. There are no known studies on the use of the autologous sling that was done with a follow-up period as long as ours. The use of the vaginal wall sling, which is an autologous sling, is also less frequently studied than the pubovaginal sling.
One limitation of our study is the small sample size; however, it may be justified by the long follow-up period. Our study also lacks a control group that would have been subjected to synthetic sling implantation. We do not offer these services at our center, making an RCT difficult to perform. UDI-6 scores were not analyzed because not all patients completed it throughout the follow-up. There are missing data on the duration of treatment for OAB.
Conclusion
Our results demonstrated that the use of vaginal wall sling does not guarantee a good objective and subjective long-term cure rate in women with SUI.
Footnotes
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
