Abstract
Introduction:
Erectile dysfunction (ED) following prostate cancer treatment is not uncommon and penile rehabilitation is considered the standard of care in prostate cancer survivorship (PCS), where both patient and his partner desire to maintain and/or recover pre-treatment erectile function (EF). There is a clinical interest in the role of regenerative therapy to restore EF, since existing ED treatments do not always achieve adequate results.
Aim:
To review regenerative therapies for the treatment of ED in the context of PCS.
Materials and Methods:
A review of the existing PubMed literature on low-intensity extracorporeal shockwave therapy (LIESWT), stem cell therapy (SCT), platelet-rich plasma (PRP), gene therapy, and nerve graft/neurorrhaphy in the treatment of ED and penile rehabilitation, was undertaken.
Results:
IESWT promotes neovascularization and neuroprotection in men with ED. While several systematic reviews and meta-analyses showed positive benefits, there is limited published clinical data in men following radical prostatectomy. Cellular-based technology such as SCT and PRP promotes cellular proliferation and the secretion of various growth factors to repair damaged tissues, especially in preclinical studies. However, longer-term clinical outcomes and concerns regarding bioethical and regulatory frameworks need to be addressed. Data on gene therapy in post-prostatectomy ED men are lacking; further clinical studies are required to investigate the optimal use of growth factors and the safest vector delivery system. Conceptually interpositional cavernous nerve grafting and penile re-innervation technique using a somatic-to-autonomic neurorrhaphy are attractive, but issues relating to surgical technique and potential for neural ‘regeneration’ are questionable.
Conclusion:
In contrast to the existing treatment regime, regenerative ED technology aspires to promote endothelial revascularization and neuro-regeneration. Nevertheless, there remain considerable issues related to these regenerative technologies and techniques, with limited data on longer-term efficacy and safety records. Further research is necessary to define the role of these alternative therapies in the treatment of ED in the context of penile rehabilitation and PCS.
Keywords
Introduction
Scientific advances in prostate cancer (PC) treatment have resulted in a better survival rate in men with clinically localized PC. 1 However, this has led to greater demands for better patient health-related quality-of-life domains under the concept of prostate cancer survivorship (PCS), where both the patient and his partner desire to maintain and/or recover pre-treatment physical functioning, including sexual function. 2 Contemporary literature shows that erectile dysfunction (ED) remain as high as 60% following robotic radical prostatectomy. 3 In those who receive radiation therapy, ED tends to occur later and is generally more severe in the external beam radiation group compared with those who received brachytherapy. 4 Men who receive androgen deprivation therapy and those who have undergone salvage therapies are more likely to experience significant ED. 2 Furthermore, the recovery of sexual function can vary from one individual to another, including the response to various sexual therapies.
Penile rehabilitation is accepted as the standard of care in men who received surgery or radiation therapy for PC. While oral phosphodiesterase type 5 inhibitor (PDE5i) remains the first-line therapy and current standard in penile rehabilitation, the evidence supporting its clinical efficacy in restoring natural spontaneous erection is far from convincing.5–7 Intracavernosal therapy is more likely to be effective, especially in the setting of neuropraxia, while penile prosthesis implantation provides a permanent mechanical solution.8,9
Over the past decade there has been considerable interest in the role of regenerative therapy to restore erectile function (EF) without the need for dependency on medical therapy. Although there is substantial preliminary research undertaken on the role of various regenerative strategies for ED, there is a paucity of high-quality human data to support their use as a standard of care in clinical practice; particularly in the setting of penile rehabilitation in PCS. The following article explores our evolving understanding of the topic, with an overview of the basic science on regenerative technology and its role on EF recovery and penile rehabilitation in the setting of PC treatment and PCS (Table 1).
Published studies on regenerative technology to restore EF in men following prostate cancer therapy.
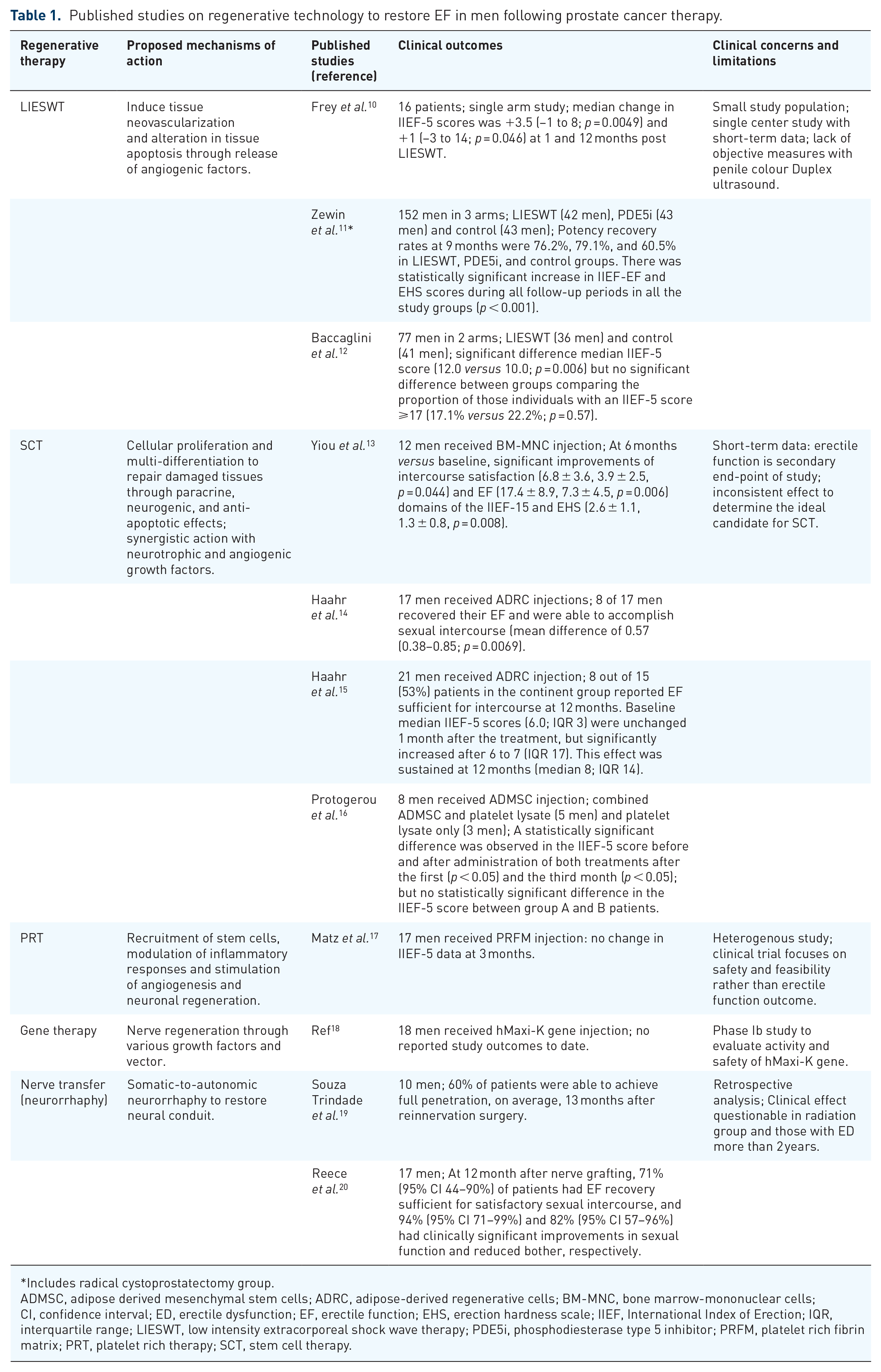
Includes radical cystoprostatectomy group.
ADMSC, adipose derived mesenchymal stem cells; ADRC, adipose-derived regenerative cells; BM-MNC, bone marrow-mononuclear cells; CI, confidence interval; ED, erectile dysfunction; EF, erectile function; EHS, erection hardness scale; IIEF, International Index of Erection; IQR, interquartile range; LIESWT, low intensity extracorporeal shock wave therapy; PDE5i, phosphodiesterase type 5 inhibitor; PRFM, platelet rich fibrin matrix; PRT, platelet rich therapy; SCT, stem cell therapy.
Materials and methods
This article was formulated based on a review of the PubMed database for the English language original articles, narrative reviews, and evidence-based clinical guidelines published up to September 2020. The keyword search included regenerative therapy, low-intensity shockwave therapy (LIESWT), stem cell therapy (SCT), platelet-rich plasma (PRP), gene therapy, nerve graft/neurorrhaphy, erectile dysfunction, and penile rehabilitation. This article is not meant to provide a full systematic review and meta-analysis on this topic; furthermore, published animal studies were excluded. Discussion relating to regulatory pathways or commercial registration of these regenerative technologies is also not included within the scope of this paper.
Results
Low-intensity extracorporeal shockwave therapy (LIESWT)
There is great interest among clinicians and patients regarding the use of LIESWT for ED, since this technology has been shown to improve EF through the release of various angiogenic factors such as vascular endothelial growth factor (VEGF) and endothelial nitric oxide synthase (eNOS), which are responsible for tissue neovascularization and alteration in cellular apoptosis.21,22
At the moment, there are many LIESWT machines on the commercial market offering different types of shockwave energy (electrohydraulic, electromagnetic, or piezoelectric) with varying degrees of energy flux densities, penetration depth, and treatment template. The later is often derived from earlier published studies and based on manufacturer guidelines.22,23 While there is no direct comparative clinical trial amongst the various LIESWT machines and with recognition of the fact that each machine offers its own protocol, 23 published studies including systematic reviews and meta-analyses have revealed a statistically significant improvement in EF following LIESWT in men with ED.24–27 Furthermore, several clinical guidelines advocate the use of LIESWT as an effective and safe treatment; in particular, in men with mild to moderate vascular ED.22,23
While preclinical studies based on cavernous nerve injury in an animal model of ED have shown neuroprotection and nerve regeneration, 28 as well as improvement in cavernosal blood flow 29 following LIESWT, there is limited published clinical data in men following radical prostatectomy (Table 1).30,31
In a pilot study of 16 patients who underwent bilateral nerve-sparing surgery, Frey et al. 10 reported minimal improvement in International Index of Erectile Function (IIEF)-5 scores following LIESWT. In contrast, Zewin et al. 11 found that LIESWT resulted in higher recovery of potency compared with the control group (76.2% versus 60.5%; p < 0.001); however, the positive effects of LIESWT are lower than PDE5i use (79.1%). A recent open-label randomized clinical trial with 2 parallel arms and an allocation ratio of 1:1 between tadalafil versus tadalafil and LIESWT 13 reported that LIESWT was associated with an improvement in IIEF-5 score but this was not clinically significant (17.1% versus 22.2%, p = 0.57). In addition, studies in men with mixed etiologies of ED showed that men with vasculogenic ED responded better to LIESWT than men who developed ED following radical prostatectomy.32,33
Corporal hypoxia, secondary to ED, often resulted in higher expression of pro-fibrotic factors; the subsequent development of corporal fibrosis can cause irreversible ED. 2 As a result, it is not uncommon for these men following radical prostatectomy to complain of penile length loss and the development of Peyronie’s disease.8,9 These penile changes can limit cavernosal neovascularization and the neuro-regenerative effects of LIESWT. At present, there are numerous variables in LIESWT machines in terms of device specifications and treatment protocols, 23 in addition to existing limitations in the literature such as lack of objective measures (e.g. penile blood flow) and relatively short-term data. 22 The result is aa limited general acceptance of LIESWT as a standard of care for ED, let alone as a proper tool for penile rehabilitation in the PCS setting. 24 Current clinical guidelines and position statements by various sexual medicine organizations advocate a cautious approach to the adoption LIESWT. Further studies should be conducted to better define strategies to optimize patient selection and shock wave energy delivery.21,22
Cellular-based therapy
Stem cell therapy (SCT)
SCT has emerged as a promising regenerative approach due to its ability to drive cellular proliferation and multi-differentiation to repair damaged tissues. 34 Over the past decade, there has been considerable interest in SCT in the treatment of ED. The most popular method of SC delivery in ED treatment is intracavernosal injection, 35 given its ease of administration and proven success rate in both preclinical and clinical trials. 36 Various studies with animal ED models have demonstrated the paracrine, neurogenic, and anti-apoptotic roles of stem cells and various synergistic approaches, while certain growth factors (e.g. brain-derived neurotrophic factor, [BDNF]) have been utilized to improve clinical effects.34–36 Recent studies have coupled more novel technologies such as recombinant DNA (lenti-rBDNF), 37 nanoparticles, 38 exosomes, 39 and shockwaves 40 to further optimize these therapeutic effects.
There have been a limited number of clinical trials investigating the role of SCT for ED indications such as post-prostatectomy, Peyronie’s disease, diabetics, and vascular compromised men (Table 1).13–15,16,41,42 In a study with escalating doses of mesenchymal stem cells in a phase I trial involving 12 patients, Yiou et al. 13 reported significant EF improvements in 9 out of 12 patients based on IIEF domains on intercourse satisfaction (6.8 versus 3.9, p = 0.044), EF (17.4 versus 7.3, p = 0.006), and EHS (2.6 versus 1.3, p = 0.008), and without serious adverse events. The use of a single dose of autologous adipose-derived stem cell has been shown to improve IIEF scores at 6 months of follow-up (53% of patients were able to achieve penetrative sexual intercourse without the use of oral medications), 14 and this positive effect was found to be sustained even after 12 months at subsequent follow-up study. 15 More recently, a single-center pilot study showed that a combination of autologous adipose-derived stem cells and PRP significantly improved EF based on IIEF scores. 16
While the risk of tissue rejection is likely minimal and short-term safety data including cancer risk has been reassuring, the use of stem cells remains a concern from both a bioethical and regulatory perspective. There exists a need to evaluate longer-term clinical outcomes and standardize SCT protocols for ED. In addition, the stability of allogenic stem cells remains a considerable challenge for clinical applications on a larger scale.
Platelet-rich plasma (PRP)
The PRP is an autologous product obtained from whole blood with more than four times normal human physiological serum platelet concentration. PRP contains many growth factors (e.g. VEGF, platelet-derived growth factor [PDGF], and fibroblast growth factor [FGF]) responsible for regenerative functions, including the recruitment of stem cells, the modulation of inflammatory responses, and the stimulation of angiogenesis and neuronal regeneration.43–45 The PRP is prepared by centrifugation of the patients’ blood to remove red blood cells, with the addition of a common platelet activator or agonist immediately before tissue application. This activates the clotting cascade and releases these growth factors in order to promote angiogenesis and tissue healing. 43 There are several commercial PRP separation systems with different applications for use in a wide variety of conditions. 46
The use of PRP to improve EF has been reported in both preclinical and clinical studies (Table 1).43,47 In an animal model of cavernous nerve injury, the administration of PRP growth factors resulted in a reduction in cellular apoptotic markers, reduced tissue fibrosis, neuronal regeneration, and an improvement in EF.44,48–50 Several phase I–II human PRP clinical trials have shown promising data with good short-term safety.17,50 In a mixed cohort of patients and different etiologies and reasons for PRP injections, Matz et al. 17 reported that the IIEF scores improved by an average of 4.14 points in 5 patients who received PRP therapy with no serious adverse event.
The evidence to support PRP use in the treatment of ED is not strong, especially in the setting of PC men. Furthermore, medical hype and the commercialization of this technology within a limited regulatory framework has deterred high-quality research data. None of these studies is designed specifically as part of a penile rehabilitation protocol; in addition, the clinical application and specification of PRP in terms of EF recovery needs to be optimized and standardized.
Gene therapy
In the era of personalized medicine, gene therapy is very attractive since it offers several clinical advantages. These include single-dose therapy to restore erectile function for long-term, an ability to be combined with other therapies to optimize dose requirements and minimize side effects, and also the opportunity to develop patient-specific treatment approaches. 51 Current gene therapies in ED could be categorized into activators of the nitrergic-neural system, endothelial growth factors promoters, and modulators of ion channels in smooth muscle cells. These act on various molecular targets including eNOS, neurotrophic and angiogenic factors, potassium channels, prostacyclin I2 synthase, and peptides.51,52
Most gene therapy research in ED remains preclinical at this stage 52 with various growth factors such as brain-derived neurotrophic factor (BDNF) and neurturin having been explored as gene therapy options for ED after cavernous nerve injury. 53 Vector-vehicles such as adeno-associated and herpes simplex 54 have also been tested. To date, the only ongoing clinical trials using gene therapy for the treatment of ED are in diabetic patients.55–57 While there is currently a phase IIA registered clinical trial evaluating the potential activity and safety of the hMaxi-K gene for ED, it is not specifically designed in the context of treating post-prostatectomy ED men. 18 Further clinical studies are required to investigate the optimal use of growth factors and the safety aspects of vector delivery system in terms of immunogenicity, cytotoxicity, inflammatory reaction towards viral vectors, and insertional mutagenesis of gene delivering viral vectors.
Interposition nerve graft and nerve transfer (neurorrhaphy)
From the identification of the cavernous neurovascular bundles to subsequent refinements in nerve-sparing radical prostatectomy, there have been considerable advances made to preserve EF in men undergoing radical prostatectomy. 58 The use of an intraoperative tool to facilitate the identification of cavernous nerves, such as the CaverMap® surgical aid (UroMed, Boston, MA) has permitted various interpositional nerve grafts to be placed onto the surgical bed to repair these critical nerves. The goal is to restore the neural conduit for the preservation of EF postoperatively. In a rat model of cavernous nerve injury it has been shown that the autologous vein can also serve as a guide for cavernous nerve regeneration, with its effect further enhanced when the vein is filled with PDGFs. 59 While numerous single center studies have shown some positive benefits in interpositional nerve grafts (Table 1),60–62 the largest randomized study with unilateral sural nerve grafting following nerve-sparing radical prostatectomy was terminated early as a result of an increased potency rate at 2 years while not reaching the threshold significance level of at least a 20% (absolute) improvement. 63
While conceptually interpositional cavernous nerve grafting is logical, criticisms of this surgical technique include a lack of clearly-defined proximal and distal stumps of the neurovascular bundles within the surgical bed (since these neurovascular bundles form a complex intricate prostatic plexus along the posterior aspect of the prostate, pelvic floor, and the urethral sphincter) and the ability to accurately repair a neural gap within a convoluted network of transected nerves. 58 In addition, there are questions about whether a true nerve-sparing surgery is possible, given that potential neuropraxia and postoperative inflammatory changes may affect neural ‘regeneration’.9,64 In addition, the situated grafts may be damaged if adjuvant or salvage radiotherapy is necessary. 8 Many questions remain about whether bilateral nerve-sparing is superior to unilateral nerve-sparing, and the exact role of penile rehabilitation in this setting can be largely unpredictable. The National Comprehensive Care Network (NCCN) prostate cancer guidelines advocate that the replacement of resected nerves with a nerve graft is not beneficial for the recovery of EF after surgery. 65
There is also renewed interest in the role of penile re-innervation techniques with an end-to-side nerve graft using a somatic-to-autonomic neurorrhaphy.64,65 Souza Trindade et al. 19 reported this novel technique in 10 patients following radical prostatectomy at least 2 years previously, and that 60% of patients were able to achieve full penetration, on average, 13 months after reinnervation surgery. Those that had radiotherapy experienced a slower return of EF. Reece et al. 20 found that 71% (95% confidence interval [CI] 44–90%) of patients had EF recovery sufficient for satisfactory sexual intercourse. This side-to-end neurorrhaphy allows for the use of the femoral nerve as the donor to provide budding or sprouting of the donor nerve axons. This neural (brain) plasticity is thought to be central to the establishment of functional communications between genital nerve receptor centres and the central nerve centres, mediated by femoral nerve sensory fibers (mixed nerve). 66 The reported high success rate has not been replicated by other centres. It is important to note that the neurotransmitter acetylcholine has different effects in the somatic and autonomic nervous systems; implanting a somatic nerve end into the corporal tissue (instead of directly onto the cavernous nerve) may not restore the neural conduit. In addition, it is questionable whether EF recovery is possible in some men after 2 years post-prostatectomy with ensuing irreversible corporal fibrosis. This novel surgical technique requires further basic scientific research and confirmation from larger multi-center trials.
Conclusions
The potential of regenerative medicine in restoring EF following radical prostatectomy is exciting and highly innovative. In contrast to the existing treatment regime, regenerative ED technology aspires to promote endothelial revascularization and neuro-regeneration. The use of various novel agents, micro-energy applications, and novel neural ‘reconstruction’ will continue to push boundaries and perhaps promote symbiotic therapy with existing penile rehabilitation protocols. Despite this enthusiasm, there remain considerable issues related to these regenerative technologies and techniques; due in part to the limited data on longer-term efficacy and safety records.
Defining true success using regenerative therapy to restore EF is likely to be dependent on many factors, including sexual desire, penile size and shape, the ability to successfully have penetrative intercourse, and sexual practice, as well as ejaculatory and orgasmic dysfunctions. In addition, there are many interplaying factors such as changes in psychosexual, body image and relationship dynamics, and the presence of coexisting urinary or bowel disturbances may exacerbate the sense of loss of masculinity and sexual distress. 2 The need for adjuvant or salvage radiation or hormonal therapy will adversely effect EF recovery and worsen ED. These factors underscore the limitations in current and future treatment strategies to restore EF, as well as highlight the need towards a more holistic approach to male sexuality beyond achieving just an erect penis.
Footnotes
Acknowledgements
Professor Eric Chung is the Chairperson for the Prostate Cancer Survivorship committee on sexual health, function, and rehabilitation at the International Consultation on Sexual Medicine.
Author’s note
Eric Chung is also affiliated with AndroUrology Centre, Brisbane QLD and Sydney NSW, Australia.
Author contributions
Eric Chung is responsible for conception and design, data acquisition, analysis and interpretation, manuscript drafting, revision and the final approval of the completed article.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
