Abstract
Objective
To investigate the therapeutic effect of implanting a three-piece inflatable penile prosthesis (IPP) combined with the phosphodiesterase-5 inhibitor sildenafil in severe erectile dysfunction (ED) patients.
Methods
This randomized controlled study included 123 ED patients. Sixty-two patients received the IPP implantation and 61 patients received the IPP implantation and the phosphodiesterase-5 inhibitor sildenafil. Erectile function and sexual life quality were evaluated using the five-item International Index of Erectile Function (IIEF) and modified Sexual Life Quality Questionnaire–Quality of Life domain (mSLQQ-QoL), respectively. Serum interleukin (IL)-6, IL-8, tumor necrosis factor (TNF)-α, vascular cell adhesion molecule (VCAM)-1, and intercellular adhesion molecule (ICAM)-1 levels were assessed. Kaplan–Meier curves were used to assess the overall IPP survival.
Results
Implantation of the three-piece IPP with sildenafil improved erectile function and sexual life quality, alleviated the inflammatory response, reduced the complication rate, and improved overall IPP survival.
Conclusion
Implantation of the three-piece IPP combined with a phosphodiesterase-5 inhibitor significantly improved clinical outcomes and the prognosis in ED patients.
Keywords
Introduction
Erectile dysfunction (ED) is defined as a persistent inability to obtain or maintain an erection for satisfying sexual intercourse. 1 Worldwide, up to 50% of men aged 40 to 70 years have ED. 2 An increasing amount of evidence shows that ED is caused by the interaction of multiple physiologic systems, such as endocrine, neural, and vascular systems. 3 Although ED is considered to be an age-related disease, it might have begun to occur in adolescence, especially for those who have risk factors, including cardiovascular diseases, metabolic syndrome, or diabetes. 4
Currently, various methods are used to treat ED including seeking assistance from a mental health professional, lifestyle modifications, oral phosphodiesterase type 5 inhibitor (PDE5I), testosterone therapy, intracavernosal injection therapy, a vacuum erection device, and penile prosthesis implantation. 5 PDE5Is including avanafil, lodenafil, mirodenafil, and sildenafil have been used clinically as first-line pharmaceuticals to treat ED. 6 As previously reported, PDE5Is have a therapeutic effect on ED in patients with spinal cord trauma. 7 Additionally, PDE5I administration was shown to be well-tolerated for ED in diabetic men, and it also showed excellent effectiveness. 8 The anti-inflammatory effect of PDE5I was also reported in several studies. Kosutova et al. 9 revealed that sildenafil treatment suppressed pro-inflammatory mediator release and attenuated oxidative damage in acute lung injury. Therefore, we hypothesized that PDE5I might play an anti-inflammatory role in patients with ED.
Implantation of an inflatable penile prosthesis (IPP) is an effective and safe treatment option for men who do not respond to conventional medical therapy. 10 Although IPP implantation was reported to result in better sexual function, and the patients had better feeling as well as higher satisfaction than with other treatment options, it also might result in mechanical failure that requires prosthesis repair, explant, or replacement. 11 Moreover, infections commonly result from bacterial implantation on the surface of the device during IPP implantation. 12 The three-piece IPP was reported to be a safe and effective approach that has a high satisfaction rate, and its minimally invasive approach could decrease common postoperative complications. 13 Therefore, more attention needs to be paid to decreasing the complication rate and alleviating inflammation.
In this study, we performed a randomized controlled trial to investigate the effect implanting a three-piece IPP combined with an PDE5I in patients with severe ED.
Materials and methods
Patients
The present randomized controlled study enrolled 123 patients with severe ED who were admitted to our hospital from March 2011 to February 2015. All of the patients underwent implantation of a three-piece IPP for the first time. A diagnosis of ED was made on the basis of the patient’s medical history and physical examination and laboratory test results, including the free testosterone and lipid profile. Penile duplex sonography and neurologic examination were used to detect the causes of ED. The diagnostic criteria details were as follows: 1) over 50% failure on sexual intercourse (minimum of four attempts); and 2) a score of less than 11 for the five-item of International Index of Erectile Function (IIEF-5) for more than 6 months. 14 Inclusion criteria were as follows: 1) ED patients with no response to conventional oral pharmacotherapy or intracavernosal injection; 2) ED patients with severe penile curvature, shortening, and impaired penile rigidity; and 3) patients with no intention of maintaining nonsurgical options to treat ED. Patients with a penile curvature greater than 90° were excluded. 15 The present study was approved by the ethics committee at the Second People’s Hospital of Nantong City, and written informed consent was obtained from all of the parents who participated in the study.
Treatments and implantation procedure
All ED patients were randomly divided into the following two groups using a SPSS software (v.18.0, SPSS Inc., Chicago, IL, USA)-generated number list: 1) Control group: IPP implantation; or 2) Observation group: IPP implantation combined with the PDE5I sildenafil at a dose of 50 mg/day for 1 month.
A minimally invasive technique was performed for the implant. First, the patients’ inguinal and penile areas were shaved, and prophylactic antibiotics (1 g vancomycin and 160,000 U gentamicin via intravenous injection) were then administered under general or spinal anesthesia. The three-piece IPP was also immersed in an antibiotic solution (400 mg vancomycin, 800,000 U gentamicin, and 1000 mL 0.9% NaCl). The implantation procedure was conducted under antibiotic cover through a transverse penoscrotal incision. An artificial erection was induced to identify the dorsal nerve and any pathology that required correction, and stay sutures were placed using a lateral placement. After the skin and bilateral corporotomy incisions, the proximal and distal corpora cavernosa were evaluated and dilated. To avoid pump damage from excessive backpressure, the three-piece penile prosthesis was inflated to the maximum distension. The wound was protected by a silver sulphadiazine-impregnated wound dressing (Urgotul-SSD, Urgo Medical, Dijon, France), covered with sterile gauze, and treated with proper pressure. The drainage tube was removed 24 hours after surgery, and the catheter was removed 5 to 7 days later. Additionally, more attention should be paid to preventing urethral injury. Patients with obvious pain were treated with opioid-based analgesia. The prosthesis was activated 8 to 10 days after surgery and patients started using the prosthesis for sexual intercourse 6 weeks later.
Measurement of serum factors
Serum interleukin (IL)-6, IL-8, tumor necrosis factor (TNF)-α, vascular cell adhesion molecule (VCAM)-1, and intercellular adhesion molecule (ICAM)-1 levels were detected using an enzyme-linked immunosorbent assay (ELISA) at baseline and 5 days after the surgery. Briefly, peripheral venous blood samples were collected from all the ED patients and evaluated using commercial ELISA kits (all purchased from Abcam, Cambridge, MA, USA): human IL-6 ELISA kit (ab46027); human IL-8 ELISA kit (ab214030); human TNF alpha ELISA kit (ab100654); human VCAM1 ELISA kit (ab223591); and human ICAM1 ELISA kit (CD54) (ab174445).
Data collection
Demographic information from all ED patients was recorded, including age, body mass index (BMI), ED cause, ED duration, and intraoperative complications. The IIEF-5 and the modified Sexual Life Quality Questionnaire–Quality of Life domain (mSLQQ-QoL) were used to evaluate erectile function and sexual life quality in patients before treatment and at 2, 3, 6, and 12 months after the surgery. Postoperative complications within 2 weeks were recorded. Follow-up lasted for 5 years from admission to the last follow-up or IPP failure (mechanical or non-mechanical).
Statistical analysis
Continuous data are presented as the mean ± standard deviation (SD). Comparisons among three or more groups were performed using a one-way analysis of variance (ANOVA) followed by Tukey’s post hoc test, while comparisons between two groups were made using the Student’s
Results
Basic characteristics of all erectile dysfunction patients
One hundred twenty-three patients with severe ED were included in the present randomized controlled study. Patients in both groups received a penile prosthesis implant using the same type of three-piece IPP, but the observation group also received sildenafil at a dose of 50 mg/day for 1 month. There were 62 patients in the IPP group and 61 patients in the IPP and sildenafil group, and their average age was 55.02 ± 7.64 years and 57.74 ± 8.29 years, respectively. During the study period, no patient withdrew or was lost to follow-up. The basic characteristics of all the included patients are shown in
Baseline characteristics of the ED patients.

IPP, inflatable penile prosthesis; BMI, body mass index; ED, erectile dysfunction.
Three-piece inflatable penile prosthesis combined with a phosphodiesterase-5 inhibitor improved erectile function and sexual life quality
To investigate the effect of the different therapies on the two study groups, the erectile function and sexual life quality were evaluated using the IIEF-5 scale and the mSLQQ-QOL, respectively. As shown in

A three-piece inflatable penile prosthesis combined with a phosphodiesterase-5 inhibitor improved erectile function and sexual life quality. (a) The IIEF-5 scale was used to evaluate the erectile function at 0, 2, 3, 6, and 12 months in the two study groups. The difference was significant between at months 2 and 3. (b) The mSLQQ-QOL was used to measure the sexual life quality. ***P < 0.001
Three-piece inflatable penile prosthesis combined with a phosphodiesterase-5 inhibitor alleviated inflammation
We then determined the serum inflammatory factor levels at admission and 7 days after the treatment. As shown in
Serum level of pro-inflammatory markers/mediators before and 5 days after treatment.
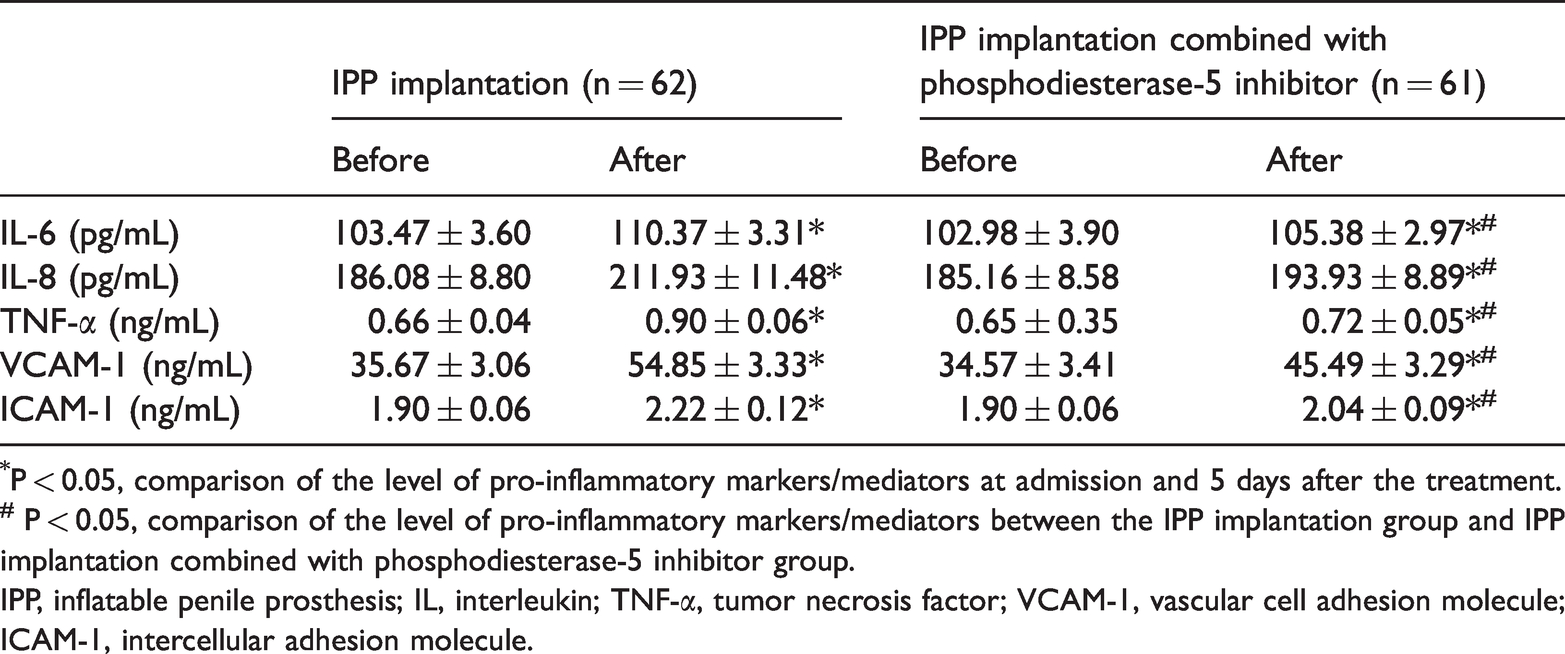
*P < 0.05, comparison of the level of pro-inflammatory markers/mediators at admission and 5 days after the treatment.
# P < 0.05, comparison of the level of pro-inflammatory markers/mediators between the IPP implantation group and IPP implantation combined with phosphodiesterase-5 inhibitor group.
IPP, inflatable penile prosthesis; IL, interleukin; TNF-α, tumor necrosis factor; VCAM-1, vascular cell adhesion molecule; ICAM-1, intercellular adhesion molecule.
Three-piece inflatable penile prosthesis combined with a phosphodiesterase-5 inhibitor decreased complications and increased the overall prosthesis survival rate
We finally analyzed the postoperative complications rate and overall IPP survival rate (mechanical survival and non-mechanical survival) over a 5-year follow-up. As shown in
The incidence of complications after surgery in the two groups after a 5-year follow-up.

IPP, inflatable penile prosthesis.

The Kaplan–Meier method was used to estimate the overall survival rate of the three-piece inflatable penile prosthesis over a 5-year follow-up in two groups.
Discussion
A variety of treatments have been successfully used to treat patients with ED, such as vacuum devices, intracavernosal injections, PDE5Is, and penile prosthesis implantation. 16 Although PDE5I use is considered to be a first-line therapy, IPP implantation has gradually become a popular treatment because of its low complication rate and high satisfaction rate. 17
The three-piece IPP is composed of corporal cylinders, a flow regulator or pump, and a reservoir. 18 The three-piece IPP has been the most frequently used type of penile prosthesis, accounting for over 75% of the currently placed IPPs because of its rigidity, girth expansion, and optimum flaccidity. 19 As reported, the IPP implantation significantly improved the quality of life in ED patients. 20 Another previous study also demonstrated that the three-piece IPP could be safely implanted in ED patients with various urinary diversions, and it had satisfactory outcomes with no increase in the risk of infection or damage to adjacent structures. 21 In this study, we found that the implantation of the three-piece IPP significantly improved the erectile function and sex life quality, which could be further improved using a PDE5I.
The inhibitory effect of sildenafil on the inflammatory response and oxidative stress have been recently illustrated in several studies.22,23 Laxmi et al. 24 found that sildenafil served as an inhibitor for inflammation in bronchial asthma in rats. In addition, as stated in a study of severe acute pancreatitis using a rat model, sildenafil administration decreased the level of inflammatory factors (IL-1β, IL-6, and TNF-α). 25 We also, for the first time, observed that a three-piece IPP combined with sildenafil remarkably downregulated IL-6, TNF-α, VCAM-1, and ICAM-1 levels, which means that sildenafil administration might alleviate the inflammatory response in ED. Because IPP implantation is an invasive treatment for ED patients, it might be associated with various complications, such as urinary retention, mechanical failure, and infection. 26 In this study, we also investigated the clinical outcome after implantation. The results showed that IPP implantation combined with sildenafil could increase the overall IPP survival rate and reduce the complication rate, swelling, and pain duration.
The present study also has some limitations. First, the number of ED patients included in this study was limited. Second, we did not enroll a healthy group as a control. Therefore, further study is required.
Conclusion
We conducted a randomized controlled study to investigate the effect of three-piece IPP implantation combined with a PDE5I on erectile function and sexual life quality, inflammation, and the complication rate in severe ED patients. We showed, for the first time, that implanting a three-piece IPP combined with PDE5I administration might significantly improve the clinical outcomes and patient prognosis.
Footnotes
Ethics approval and consent to participate
The present study was approved by the ethics committee at the Second People’s Hospital of Nantong City. Written informed consent was obtained by all participants.
Consent for publication
All authors agreed with the submission and the journal’s policies and copyright.
Availability of data and material
All of the data in this study can be obtained from the authors upon reasonable request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors were supported financially by the project “The research of phosphodiesterase type 5 siRNA carried by adipose stem cells transplanted into the human corpus cavernosum to promote erection” (Jiangsu Health Committee and Nantong Second People’s Hospital, Jiangsu, China; Z2019025).
