Abstract
Background:
Urethroplasty may influence ejaculatory function due to the potential for nerve injury during perineal dissection. It is essential to understand postoperative ejaculatory outcomes for effective patient counseling and surgical planning.
Objectives:
To compare ejaculatory function and satisfaction following excision and primary anastomosis (EPA) versus substitution urethroplasty (SU), and to identify independent predictors of these outcomes.
Design:
Retrospective cohort study.
Methods:
A total of 63 sexually active men who underwent anterior urethroplasty between July 2017 and July 2022 at two tertiary centers were evaluated. Group I (EPA, n = 33) underwent transecting end-to-end urethroplasty, while Group II (SU, n = 30) underwent substitution urethroplasty utilizing buccal mucosa grafts. Ejaculatory function and satisfaction were assessed using the Male Sexual Health Questionnaire (MSHQ).
Results:
The mean stricture length was 3.3 ± 7 cm, and the overall urethroplasty success rate was 87% after a mean follow-up of 45.6 ± 21 months. The mean score on the MSHQ Erection Scale differed significantly between groups (EPA = 10.13 ± 4.66 vs SU = 12.69 ± 3.12, p = 0.008). Conversely, scores on the Ejaculation Scale (EPA = 27.24 ± 7.63 vs SU = 27.57 ± 6.84, p = 0.443) and Satisfaction Scale (EPA = 24.17 ± 6.73 vs SU = 25.79 ± 5.64, p = 0.790) were comparable. Multivariate analysis identified age as the sole independent predictor of Ejaculation Scale scores (β = −0.219, 95% CI: −0.351 to −0.087, p = 0.001).
Conclusion:
Postoperative ejaculatory function showed no significant difference between EPA and SU urethroplasty. Younger age was associated with improved ejaculatory outcomes, whereas no surgical or stricture-related factors predicted patient satisfaction. These findings underscore the importance of considering patient age during postoperative counseling and expectation management.
Introduction
The prevalence of urethral stricture disease is approximately 2% among patients with urologic pathology and more than 50% among those with urethral disorders observed in urologic practice. 1 Urethral reconstruction primarily focused on stricture recurrence, lower urinary system symptoms, and issues related to the urethra. However, there has been a recent shift toward the treatment of sexual dysfunction after surgery. 1
The ejaculatory dysfunction (EjD) may be affected by nerve damage sustained during ventral dissection and splitting of the bulbospongiosus muscle, as this procedure can damage the neural circuits responsible for maintaining erectile function. 2 Finally, surgical procedures on the urethra can potentially cause EjD.
The primary objective of urethroplasty is to restore normal urine flow while ensuring a high standard of living with minimum adverse outcomes. Furthermore, considering the central aim of urethral reconstruction to enhance quality of life, urologists have shown an increased inclination toward subjective encounters among patients after urethroplasty. This includes treating nonobstructed voiding areas during urethroplasty, such as sexual function, discomfort, esthetic outcomes, and overall voiding quality, including the absence of post-void dribbling or urine spraying. Therefore, this psychometric comparative analysis assessed ejaculatory function and satisfaction between the excision and primary anastomosis (EPA) and substitution urethroplasty (SU) groups after urethroplasty.
Methods
This retrospective study was conducted from July 2017 to July 2022 on 63 patients who underwent urethral reconstruction at two centers: King Abdulaziz Medical City in Riyadh, Saudi Arabia, and King Khalid University Hospital in Riyadh, Saudi Arabia. The patients, with an average age of 36.6 years, underwent the procedure after obtaining approval from the institutional review board and providing their consent. The objective of this study was to conduct a psychometric comparison between EPA and SU to assess ejaculatory function and satisfaction after urethroplasty.
The study encompassed sexually active males aged 18 years or older diagnosed with anterior urethral strictures, specifically penile or bulbar regions. Patients presenting with posterior, meatal, distal strictures, or multiple stricture sites were excluded from the study. In addition, individuals with histories of radiation therapy, hypospadias, or lichen sclerosus were also excluded, owing to their higher risk of long-term recurrence. These exclusion criteria were implemented to ensure cohort homogeneity and to reduce confounding variables linked to elevated recurrence rates and potential effects on ejaculatory function associated with these conditions. Patients with lichen sclerosis, radiation, and infectious etiologies have a higher risk of long-term recurrence, 3 which may impact both the objective and subjective outcomes. The final sample consisted of 63 male volunteers diagnosed with anterior urethral stricture.
Group I underwent EPA urethroplasty employing a transecting technique, which involves the excision of the stricture segment followed by an end-to-end anastomosis. Group II received SU utilizing dorsal or ventral buccal mucosa grafts, selection based on stricture length, urethral plate condition, and degree of spongiofibrosis. The determination of graft placement (dorsal or ventral) was made intraoperatively by the surgeon. The Male Sexual Health Questionnaire (MSHQ) was administered postoperatively in an office setting, with a median time of 6 months (range: 3–12 months) from surgery to data collection. Furthermore, the type of surgical procedure was determined by examining electronic medical data. This questionnaire was chosen due to its efficiency and ease in meeting the necessary time constraints. The data were gathered via postoperative interviews conducted in an office setting, whereas preoperative data were acquired using a recall method. Questionnaires were not administered until at least 3 months after the surgery. A formal sample size calculation was not performed due to the retrospective design of the study, which made the sample size contingent upon the number of eligible patients treated within the study period (July 2017–July 2022).
Male sexual health questionnaire:
The MSHQ 4 was utilized to assess ejaculatory function and satisfaction. The MSHQ is a validated, self-administered instrument comprising 25 items that evaluate domains such as erection, ejaculation, satisfaction, sexual activity, and distress. Postoperative MSHQ data were collected through in-office interviews conducted at a minimum of 3 months following surgery (median, 6.2 months; range, 3–12 months). Preoperative MSHQ data were obtained via a recall method, wherein patients were instructed to complete the questionnaire based on their memory of preoperative sexual function during the postoperative interview. The potential for recall bias inherent in this retrospective methodology is a limitation that has been appropriately recognized in this study. The MSHQ is a straightforward and helpful psychometric tool for quantifying a three-domain sexual function scale, encompassing ED, EjD, and satisfaction. 4
The survey is a self-administered questionnaire consisting of 25 items. The assessment had three distinct scales: an erection scale composed of three questions, an Ejaculation scale consisting of seven items, a Satisfaction scale comprising six items, and an additional nine items that pertain to sexual activity, duration since the most recent sexual encounter, level and fluctuations in sexual activity, and the amount of distress associated with sexual dysfunction. Response choices include binary scales (Yes/No) and five-point and six-point Likert scales. The MSHQ was designed to evaluate several aspects of sexual function and pleasure, with a specific focus on ejaculatory function.
Permission to utilize the MSHQ was officially secured from the copyright holder (Sanofi-Aventis R&D, France) via the Mapi Research Trust (Special Terms No. 124865). The questionnaire was employed in accordance with these permissions solely for non-commercial academic research purposes (Supplemental Material).
Follow-up and outcome
Urethral openness was assessed using peri-catheter urethrography at 3 weeks post-surgery for catheter removal and semirigid cystoscopy at 9 months post-surgery. Success was defined as the absence of lower urinary tract symptoms, no need for further procedures (e.g., urethral dilation), and confirmation of urethral patency via urethrography and cystoscopy.
Statistical analysis
Data normality was evaluated employing the Shapiro–Wilk and Kolmogorov–Smirnov tests. Categorical variables (e.g., stricture location, etiology) were analyzed using Chi-square or Fisher’s exact tests. Continuous variables (e.g., MSHQ scores, age) were compared using independent t-tests for normally distributed data or Mann–Whitney U tests for data that did not conform to normal distribution. Analysis of variance (ANOVA) and Kruskal–Wallis tests were applied for multi-group comparisons. Assumptions of linear regression, including linearity, normality of residuals, and absence of multicollinearity, were validated utilizing scatter plots, residual plots, and variance inflation factors < 5. Covariates such as age, hypertension, diabetes, stricture location, etiology, and surgical history were selected a priori, based on their clinical significance in relation to sexual function and urethroplasty outcomes. Statistical significance was determined at p < 0.05, using SPSS Version 27, Armonk, New York, USA.
Results
Group I comprised patients who underwent transecting end-to-end primary anastomosis (EPA) urethroplasty, whereas Group II included patients who underwent SU utilizing dorsal buccal mucosa grafts (90%) or ventral buccal mucosa grafts (10%), with an average graft length of 4.2 cm (ranging from 2.5 to 6.0 cm). The selection of the surgical technique was determined by factors such as stricture length, condition of the urethral plate, spongiofibrosis, and previous interventions. Urethral patency was confirmed through a peri-catheter urethrogram conducted at 3 weeks post-surgery and via semirigid cystoscopy at 9 months. Success was delineated as a maximum urinary flow rate (Qmax) exceeding 15 mL/s, a post-void residual (PVR) less than 50 mL, and a patent urethra observed on urethrogram and cystoscopy, without the necessity for additional interventions such as urethral dilation.
The overall success rate of urethroplasty was 87.3% (55 out of 63 patients) following a mean follow-up period of 45.59 ± 21 months. Objective outcomes encompassed a mean Qmax of 18.2 ± 4.1 mL/s (EPA: 18.5 ± 4.0 mL/s; SU: 17.9 ± 4.2 mL/s; p = 0.612) and a mean PVR of 35.4 ± 15.2 mL (EPA: 34.8 ± 14.9 mL; SU: 36.0 ± 15.5 mL; p = 0.784). Urethral patency was verified in 87.3% of patients through urethrography at 3 weeks and cystoscopy at 9 months.
The cause of urethral stricture was idiopathic in 78.8% (26/33) of Group I and 83.3% (25/30) of Group II (p = 0.547), with an overall idiopathic rate of 80.68%, as shown in Table 1.
Patient characteristics.
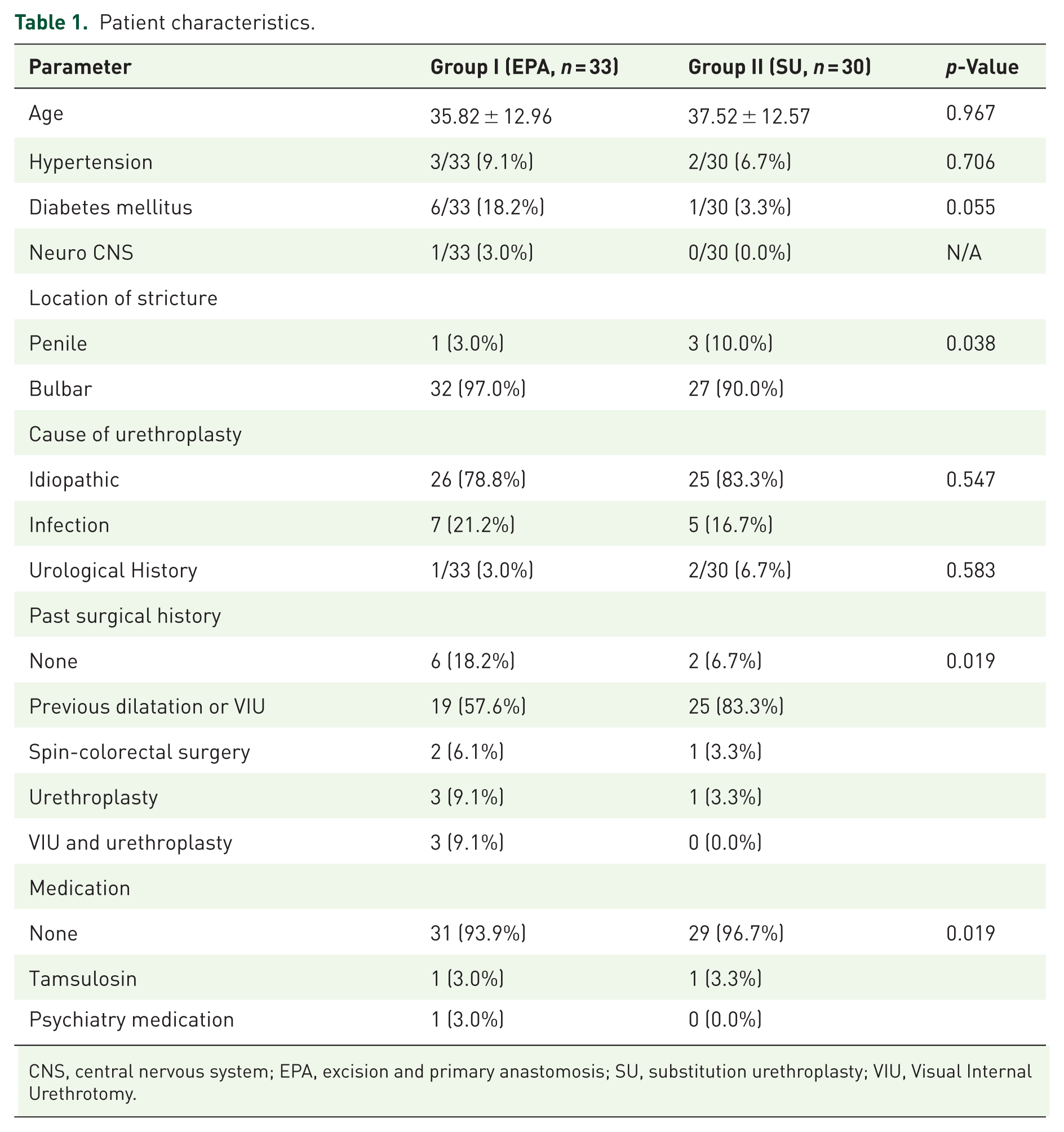
CNS, central nervous system; EPA, excision and primary anastomosis; SU, substitution urethroplasty; VIU, Visual Internal Urethrotomy.
The mean MSHQ Erection Scale score significantly differed after 3 months of EPA and SU (10.130 ± 4.6600 vs 12.690 ± 3.1195) (p = 0.008). In comparison between the cohorts, the mean scores of the Ejaculation Scale and Satisfaction Scale were comparable between the groups (p = 0.443 and p = 0.790, respectively; Table 2).
Summary of the male sexual health questionnaire in both groups.
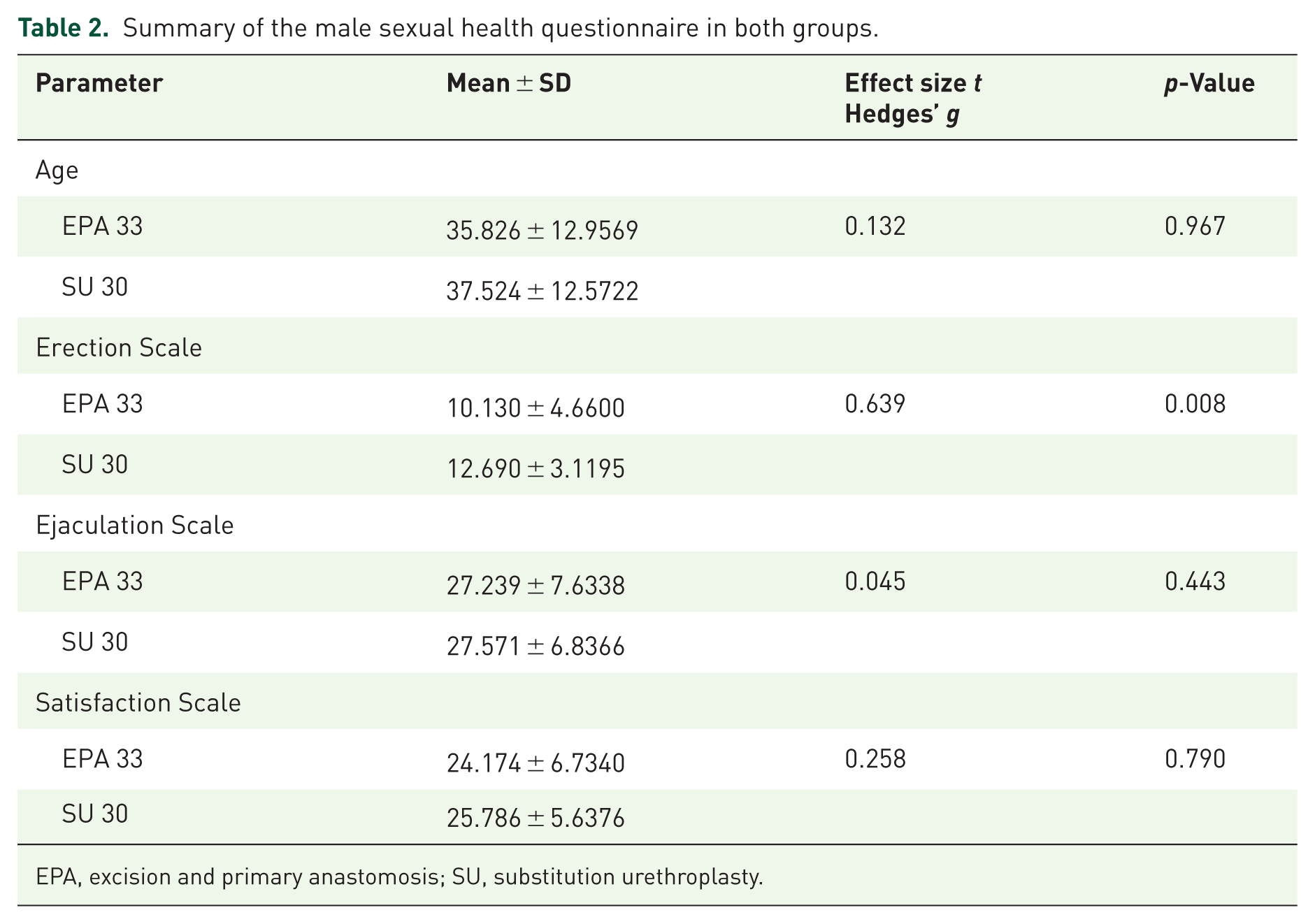
EPA, excision and primary anastomosis; SU, substitution urethroplasty.
The subgroup analysis based on age (<40 vs ⩾40 years) showed higher scores on the ejaculation scale among younger patients (28.5 ± 6.8 vs 25.2 ± 7.1, p = 0.042), indicating that age is a predictor of ejaculatory function outcomes.
The multivariate analysis revealed that patient age was the only significant independent predictor of the Ejaculation Scale (β: −0.219, 95% CI: −0.351 to −0.087, p = 0.001). Other factors, including comorbidities, stricture location, etiology, and surgical history, showed no significant correlation (p > 0.05), with wide confidence intervals indicating limited precision for these predictors (Table 3). However, other factors, such as comorbidities, cause, and type of urethral stricture, showed no significant correlation (p > 0.05) with the ejaculation scale (Table 3). However, the multivariate analysis revealed that no significant predictors were identified for the Satisfaction Scale (Table 4).
Multivariate analysis model to explore the independent predictors of the Ejaculation scale.

β, beta coefficient, CI, confidence interval; EPA, excision and primary anastomosis; SU, substitution urethroplasty.
Multivariate analysis model to explore the independent predictors of the satisfaction scale.
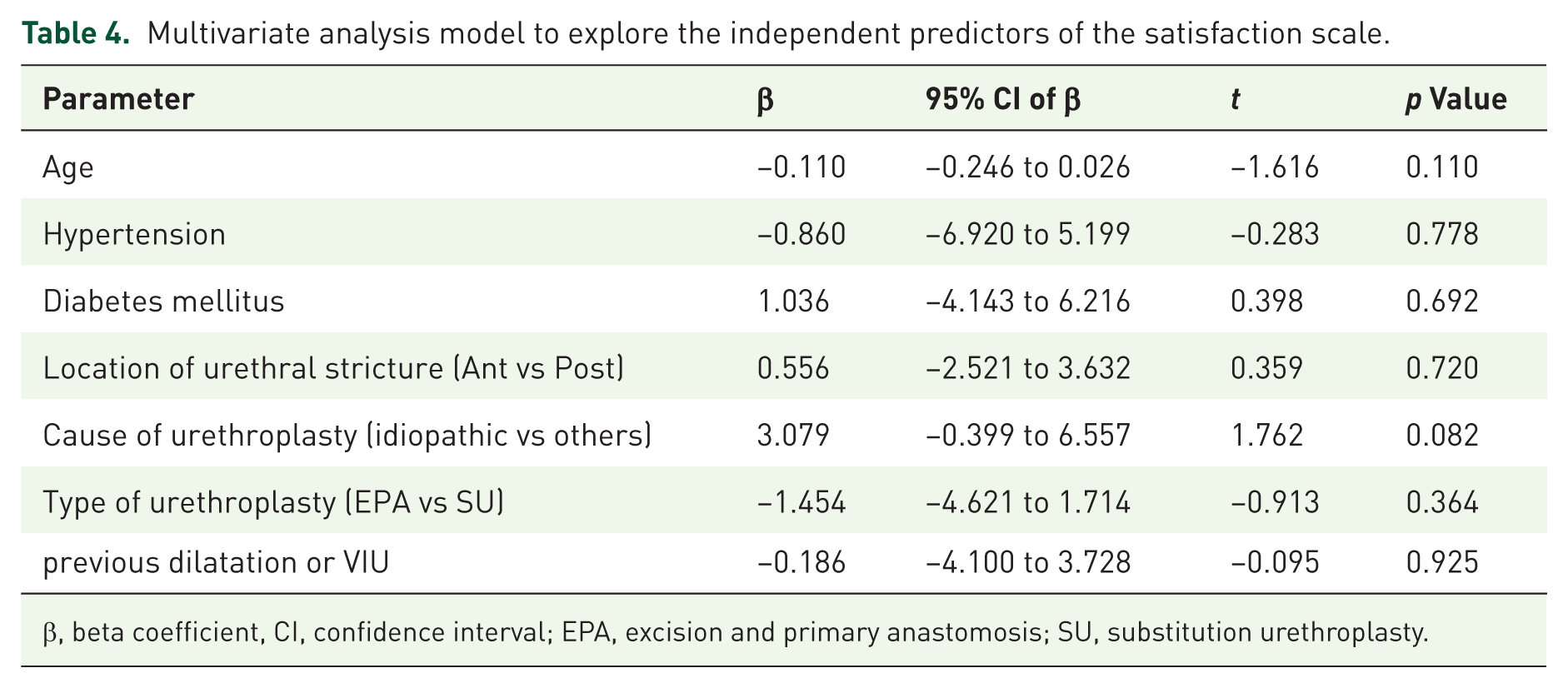
β, beta coefficient, CI, confidence interval; EPA, excision and primary anastomosis; SU, substitution urethroplasty.
A stacked bar chart showing the proportion of patients in Group I (EPA) and Group II (SU) reporting levels of bother (None, Mild, Moderate, Severe) for erectile dysfunction (ED) and EjD. Approximately 52.2% of Group I and 58.6% of Group II reported no bother for EjD, as shown in Figure 1.
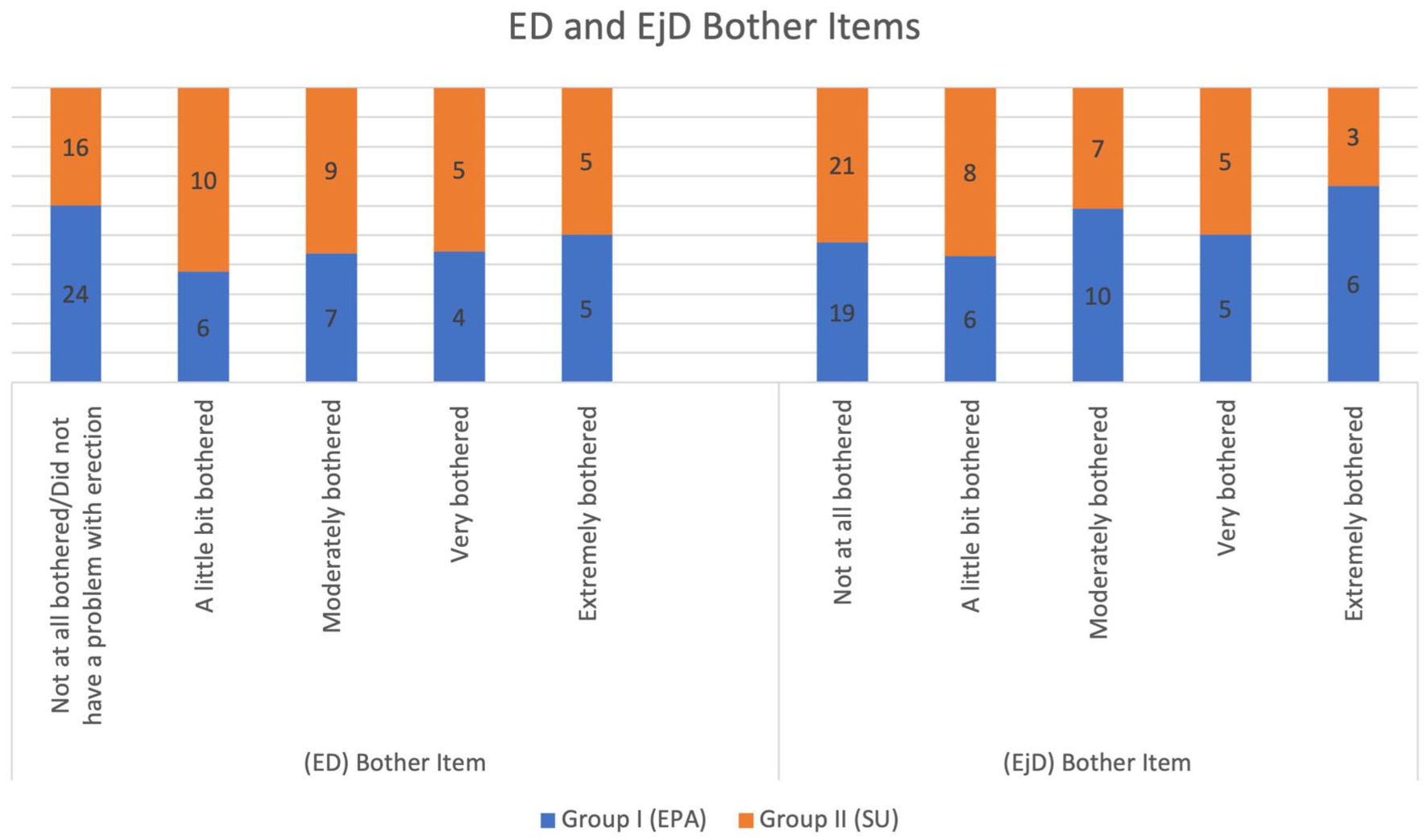
ED and EjD Bother items in both groups.
Discussion
The long-term success rates of urethroplasty have been firmly established in large cohorts, independent of stricture location, and across a diverse range of procedures. Nevertheless, a significant portion of the published findings after urethroplasty mainly consist of metrics established by the surgeon, with a few studies including patient outcome measures. 5 Evaluating EjD is challenging; many authors have used non-validated questionnaires.
Preoperatively, the reported prevalence of EjD ranges from 0% to 85%. 6 Up to 25% of men with urethral stricture develop EjD because of the obstruction. 7 Redmond et al.8 suggested this may be resolved after urethroplasty. Most studies reported improved ejaculatory function after restoration of the urethral lumen. However, sometimes, EjD persists or worsens. 6 The incidence of postoperative erectile dysfunction has been reported to range from 0% to 38%, while ejaculatory dysfunction varies from 7.7% to 67%. Changes in mean ejaculatory function scores, by the MSHQ-EjD, ranged from 0.7 to 7.0. 9 Furthermore, a mean difference of −0.87 in the IIEF-EF score and 1.77 in the Male Sexual Health Questionnaire-Ejaculatory Dysfunction (MSHQ-EjF) score was observed following anterior urethroplasty (p = 0.003). 9 These findings highlight that, although sexual function may be affected after anterior urethroplasty, such changes are generally modest, and patients should receive appropriate counseling.
The hypothesis is that division of the bulbospongiosus muscle and subsequent damage to the perineal nerves during dissection may disrupt or reduce ejaculatory force. 8 Barbagli et al. 10 introduced a muscle-sparing technique to mitigate this problem. Nevertheless, research conducted across many institutions showed no improvement when using the earlier approach. 7 Almost 82.0% of patients experienced no change in ejaculatory function, and 7.1% experienced the development of de novo or worse ejaculatory dysfunction after reconstruction. 8 This might be attributed to the accumulation and aggregation of semen at the repair site.
There has been a lack of evaluation or documentation of ejaculatory function in individuals who have undergone urethroplasty. Prior studies have used the MSHQ to assess ejaculatory function in individuals undergoing urethroplasty. Erickson et al. 7 observed that the overall effect on postoperative ejaculatory function was minimal, with limited changes noted. Most patients who reported functional decline reported decreased volume, vitality, and discomfort. 7 By contrast, Singh et al. 11 observed a notable improvement in ejaculatory function among patients. In this cohort, approximately 52.2% and 58.6% of groups I and II, respectively, did not bother at all.
Age is correlated with EjD before and after urethroplasty. Erickson et al. reported EjD Improvement in younger men aged <49 years, whereas men >60 years did not experience improvement in posture after urethroplasty.11,12 Moreover, Sharma et al. 13 compared anterior urethroplasty, both anastomotic and substitution, showing EjD improvement, but only in patients <40 years. Urkmez et al. 2 observed that patients over the age of 65 experienced statistically significant variations in postoperative IIEF and sexual satisfaction scores when compared to preoperative baselines, whereas younger patients did not demonstrate any notable changes. Our analysis indicated that patient age was the only predictive factor for the ejaculation scale.
Furthermore, diabetes has been recognized as a substantial predictor of reduced short-term erectile function outcomes, with a coefficient of −7.47 at 3 months following surgery. 2 This reinforces that patient-specific factors, rather than procedural details, drive sexual recovery after urethroplasty.
Furr et al. 14 compared EPA with dorsal BMG urethroplasty and reported no significant differences between the groups regarding post-urethroplasty IIEF5 and MSHQ-EJD ejaculatory score or bother items in the ED and EjD, respectively. Similarly, the average length of urethral strictures in our study was 3.7 cm. There were no significant differences between the ED- and EjD-bothered groups. The mean scores of the Ejaculation and Satisfaction Scales were similar among the cohorts. This finding is particularly important, given that EPA was typically reserved for shorter strictures, while BMG was used for longer strictures; yet, functional outcomes remained comparable.
There is no substantial difference in ejaculatory outcomes among these techniques. Beysens et al. 15 prospectively compared anastomotic urethroplasty with grafting techniques and observed that, although patients undergoing EPA experienced a transient, significant reduction in ejaculatory and orgasmic scores at 6 weeks, these scores reverted to baseline levels at 6 months, with no overall significant postoperative alteration. Similarly, patients undergoing EPA demonstrated no significant difference in their perception of ejaculation compared to grafting techniques when analyzed according to the surgical approach. 6 Nevertheless, patients with mild or no baseline erectile dysfunction experienced a noteworthy decline in function at 3 months post-operation; however, this impairment was restored by the 18 months. 6
The literature continues to debate the functional benefits of non-transecting approaches in bulbar urethroplasty. While some studies have reported that non-transecting urethroplasty is associated with a significantly lower risk of erectile dysfunction compared with transecting techniques, 16 others have found no statistically significant differences between the two approaches in terms of postoperative erectile function. 6 Chapman et al. 17 observed that de novo sexual dysfunction occurred more frequently following EPA than in non-transecting repairs (14.3% vs 4.3%), with urethral transection being the only independent predictor of postoperative sexual dysfunction on multivariate analysis. A recent study comparing perineal urethrostomy (PU) with buccal mucosal graft urethroplasty (BMGU) for complex anterior urethral strictures found high levels of patient satisfaction in both groups (86.7% vs 89.1%). 18 Although patients undergoing PU were significantly older than those undergoing BMGU (median age, 64 vs 57 years), functional outcomes, such as maximum urinary flow rate and post-void residual volume, were comparable. 18 These findings suggest that PU can be considered a viable first-line option for comorbid or elderly patients, offering favorable success and satisfaction rates with acceptable functional outcomes. 18 While PU avoids urethral transection and potential ejaculatory disruption, its role should not be overstated as a “last resort,” given the high satisfaction with substitution techniques such as BMGU. 19 This aligns with our data, suggesting that SU can achieve ejaculatory outcomes equivalent to EPA without compromising success.
Our investigation demonstrates that ejaculatory function and sexual satisfaction, assessed via the MSHQ, along with multivariate analysis, identified age as the sole independent predictor of Ejaculation Scale scores (β = −0.219, 95% CI: −0.351 to −0.087, p = 0.001), with younger patients exhibiting more favorable outcomes. No factors related to stricture or surgery predicted ejaculatory function or satisfaction, thereby underscoring the importance of age in patient counseling and reassuring patients that surgical technique does not compromise outcomes. In light of these findings, the results of our study showing comparable ejaculatory and satisfaction outcomes between transecting (EPA) and SU further support the notion that urethral transection may not universally indicate inferior sexual or ejaculatory results. Instead, patient-specific factors, particularly age, seem to exert a more significant influence on postoperative sexual function than the choice of surgical technique.
Study limitations
The current investigation encountered certain limitations, including its retrospective design and the inherent variability in surgical methodologies across different institutions. The retrospective design and dependence on a recall method for preoperative MSHQ data introduce potential recall bias, which may compromise the reliability of preoperative assessments of sexual function. This limitation impedes our ability to directly compare preoperative and postoperative outcomes within individual patients; consequently, our findings primarily concentrate on comparisons between the EPA and SU groups. Future prospective studies that incorporate baseline MSHQ data are necessary to substantiate these findings. The absence of data concerning hypogonadism, testosterone therapy, opioid usage, and 5-alpha-reductase inhibitor utilization, attributable to inconsistent documentation within medical records, limits the capacity to thoroughly account for confounding variables affecting sexual function. The disparity in diabetes prevalence (17.4% vs 4.8%) was addressed through multivariable analysis; however, residual confounding may still persist. These factors, along with the retrospective recall methodology, are recognized as limitations of this study. In addition, the lack of a formal sample size calculation, consequent to the retrospective design, may restrict the statistical power necessary to detect differences between the groups. This inconsistency has also resulted in deficiencies in data collection both prior to and following surgery. Future research should incorporate prospective power calculations to guarantee an adequate sample size. In addition, there is a need for standardization in follow-up intervals and processes among various institutions. Although this study provides a crucial initial insight into the clinical and patient-reported outcomes of urethroplasty in Saudi Arabia, its findings may also explore the risks, benefits, and patient expectations in surgical preparation.
Conclusion
The specific parameters associated with stricture or surgery that influence postoperative ejaculatory function have not been definitively established. The improvement in ejaculation after urethroplasty was only reported in younger patients, and the results indicate that patient age was the only objective factor that could predict the ejaculation scale. Our research suggests that no predictive factors can predict the satisfaction scale score.
