Abstract
Aims:
To assess the efficacy in lowering post-operative urinary retention, urinary tract infection and lower urinary tract symptoms and the incidence of adverse events among 12 interventions and to perform risk–benefit analysis.
Methods:
Previous randomized controlled trials were identified from MEDLINE, Scopus and CENTRAL database up to January 2020. The interventions of interest included early ambulation, fluid adjustment, neuromodulation, acupuncture, cholinergic drugs, benzodiazepine, antispasmodic agents, opioid antagonist agents, alpha-adrenergic antagonists, non-steroidal anti-inflammatory drugs (NSAIDs) and combination of any interventions. The comparators were placebo or standard care or any of these interventions. Network meta-analysis was performed. The probability of being the best intervention was estimated and ranked using rankogram and surface under the cumulative ranking curve. Risk–benefit analysis was done. Incremental risk–benefit ratio (IRBR) was calculated and risk–benefit acceptability curve was constructed.
Results:
A total of 45 randomized controlled trials with 5387 patients was included in the study. Network meta-analysis showed that early ambulation, acupuncture, alpha-blockers and NSAIDs significantly reduced the post-operative urinary retention. Regarding urinary tract infection and lower urinary tract symptoms, no statistical significance was found among interventions. Regarding the side effects, only alpha-adrenergic antagonists significantly increased the adverse events compared with acupuncture and opioid antagonist agents from the indirect comparison. According to the cluster ranking plot, acupuncture and early ambulation were considered high efficacy with low adverse events, corresponding to the IRBR.
Conclusion:
Early ambulation, acupuncture, opioid antagonist agents, alpha-adrenergic antagonists and NSAIDs significantly reduce the incidence of post-operative urinary retention with no difference in adverse events. Regarding the risk–benefit analysis of the medical treatment, alpha-adrenergic antagonists have the highest probability of net benefit at the acceptable threshold of side effect of 15%, followed by opioid antagonist agents, NSAIDs and cholinergic drugs.
Background
Urinary retention is the inability to pass urine despite persistent effort, which can be acute and chronic urinary retention. Generally, acute urinary retention is defined as a painful, palpable or percussible bladder and the patient is unable to pass the urine, which is commonly seen after surgery with long operation time. This is known as post-operative urinary retention (POUR), which results in urinary complications including urinary tract infection (UTI), patients’ discomfort and distress, prolonged hospital stay and increased cost of the treatment. The POUR incidence varied from 2.1% to 80% according to surgery types and specific study factors (e.g. age, aggressiveness of the bladder dissection, the use of opiates and underlying comorbidities).1–4
The POUR may be caused by several mechanisms, 5 including surgery (e.g. voiding reflex interruption, perioperative medication, 6 sensation of bladder fullness impairment from anesthesia, the imbalance of sympathetic and parasympathetic systems 7 ) and other factors (e.g. immobilization, voiding in supine position, or perineal and lower abdominal pain) that could inhibit the perineal relaxation that is necessary for voiding. 8 Various non-medical and medical strategies were introduced aiming to prevent POUR, for example, early ambulation (AMB), neuromodulation (NEU), anti-spasmodic agents (ASPs), non-steroidal anti-inflammatory drugs suppository (NSD), alpha-adrenergic antagonist (ALP), et cetera. Currently, no standard guideline has been approved for preventing this condition. According to previous meta-analysis, the effect of POUR prevention was pooled and demonstrated good efficacy in ALP, AMB and ASP compared with placebo. 9 However, only a few studies have pooled the adverse events (AEs) and other outcomes such as UTI, post-void residual urine (PVRU), and lower urinary tract symptoms (LUTSs). In addition, no network meta-analysis has been reported and compared among all interventions. Our study aimed to evaluate the efficacy of all interventions, both non-medical and medical, in prophylactic POUR, UTI, PVRU, LUTS and AE, along with performing risk–benefit analysis.
Methods
Protocol
The systematic review and network meta-analysis protocol was developed following the guidelines in the PRISMA extension of network meta-analysis (NMA). The review protocol was registered with PROSPERO (CRD 42019145653).
Search strategy and criteria for study inclusion
The studies were identified primarily from PubMed, Scopus and CENTRAL up to January 2020 and updated monthly. Additionally, Thai-Journal Citation Index, WHO International Clinical Trials Registry Platform (www.clinicaltrials.gov) and references of selected articles and previous systematic reviews were used to identify the eligible studies. The search terms were constructed and followed the interested population, medication name and procedures of each intervention and outcome, which were POUR, UTI, PVRU, LUTS and AE. The full search strategies are available in the Supplemental Material Table 1 online.
This study included only the randomized controlled trials (RCTs) without language restriction with the following criteria: conducted in adults aged 18 years or older who underwent any type of surgery; compared between any of the following interventions: AMB, perioperative fluid adjustment (FLU), NEU, acupuncture (ACU), cholinergic drug (CHO), benzodiazepine (BENZ), ASP, intravenous opioid antagonist related (OPI), ALP, NSD, combined interventions (COMBs), and placebo/standard care (PLA); and having at least one of the following outcomes: POUR, UTI, PVRU, LUTS and AE. The studies with insufficient data for pooling after three attempts of contacting the author every 2 weeks and the studies published in languages which reviewers could not translate were excluded.
Interventions and outcomes
There were 11 interventions with placebo included in our study. Four non-medical interventions were AMB, FLU, NEU and ACU. AMB was defined as the patients having no absolute bed rest and were mobilized to the toilet. FLU intended to restrict oral and intravenous fluid during perioperative and early postoperative periods. ACU included acupuncture procedure with or without electrical stimulation. CHO included bethanechol, neostigmine or distigmine by both oral and intravenous routes. BENZ comprised all oral or intravenous medication in this group. ASP contained drotaverine administered perioperatively. OPI consisted of any dosage of methylnaltrexone or naloxone given along with opioid. ALP included all medications and dosage of this drug group. NSD included any form and dosage of non-steroidal anti-inflammatory drug. PLA could be placebo, or no intervention or standard care.
The primary outcome was the incidence of POUR, which was diagnosed according to original studies. Briefly, most studies diagnosed by clinical symptoms and signs 8 (e.g. inability to urinate within 7 days after the surgery, after urethral catheter removal, or the feeling of strong desire to void or palpable suprapubic mass) with or without confirmation by urine volume measurement by urethral catheterization or ultrasonography. The secondary outcomes were symptomatic UTI with/without laboratory confirmation, and PVRU measured in millimeters by catheterization or ultrasonography. LUTS was reported in scores from any questionnaires and AEs.
Data extraction and study quality assessment
Data extraction was done independently by at least two of three reviewers (PS, PN, RW) using a data extraction form. Disagreement between reviewers was discussed and solved by a team. The extracted data included general characteristics of article, baseline patients’ characteristics (e.g. number of patients, mean age, type of operations, operative time, type of anesthesia), outcome measurement, interventions and data for pooling, that is, a total number of patients and events for dichotomous outcome, and mean and SD for continuous outcome.
Risk of bias assessment was done independently by at least two of three reviewers (PS, PN, RW). The assessment of risk of bias was performed using the revised Cochrane risk-of-bias tool for randomized trials (RoB 2), 10 which comprised five domains (randomization, deviations from the intended intervention, missing outcome data, measurement of the outcome, selection of the reported result) along with overall risk of bias. Each domain and overall risk of bias was rated as low, some concerns, or high.
Data synthesis and statistical analyses
Direct meta-analysis (DMA)
DMA was performed if there were at least three studies for each comparison. Risk ratio (RR) for dichotomous outcomes and mean difference (MD) for continuous outcome were estimated. These were then pooled across studies using a fixed-effect model if heterogeneity was not present, otherwise a random-effect model was applied. Heterogeneity was assessed using Cochran’s Q and Higgins’s I² statistic, 11 and considered present if p-value was less than or equal to 0.1 or I² more than 25%. Source of heterogeneity was explored by fitting each of the co-variables (e.g. age, type of operation, type of anesthesia) in a meta-regression model. If the included variable could decrease I² more than 50%, subgroup analysis and sensitivity analysis were performed accordingly. Publication bias was assessed using funnel plot and Egger test. If asymmetric funnel plot presented, contour enhanced funnel plot was then applied to distinguish the cause of asymmetry. 12
NMA
The interventions of interest were coded as 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 and 11 for PLA, AMB, FLU, NEU, ACU, CHO, BENZ, ASP, OPI, ALP, NSD and COMB, respectively. The network of all treatments was mapped. Two-stage NMA was applied 13 as follows: first, a linear regression was applied to estimate relative treatment effects (i.e. In(RR), MDs). Second, relative treatment effects were pooled across the studies using multivariate meta-analysis with random-effect model. The probability of being the best intervention in each outcome was estimated and ranked using rankogram and surface under the cumulative ranking curve (SUCRA). Inconsistency assumption was assessed by applying the design-by-treatment interaction model. Publication bias was evaluated using comparison-adjusted funnel plot.
Cluster rank and risk–benefit analysis
A cluster ranking plot was created simultaneously considering SUCRAs of benefit (i.e. lowering POUR, UTI, PVRU, LUTS) and the risk of AE. The higher SUCRA of the outcomes and AEs represented the better efficacy and fewer AEs. The plot was divided into four quadrants, in which the right upper quadrant reflected the intervention with good efficacy and fewer AEs, whereas the left lower quadrant represented the intervention with low efficacy and high AEs.
Incremental risk–benefit ratio (IRBR) of POUR prevention was calculated as a ratio of an increment in risk (ΔR) divided by an increment in benefit of POUR prevention (ΔB) estimated by NMA. Monte Carlo method was used to simulate ΔR and ΔB with 1000 replications assuming normal distribution for both. The risk–benefit plane was constructed with the Y-axis representing ΔR and the X-axis representing ΔB along with risk–benefit acceptability threshold, 14 that is, the willingness to accept additional AEs for one additional benefit. This plot was divided into four quadrants, in which the right lower and right upper quadrants implied good benefit with fewer AEs; and good benefit, but higher AEs, respectively. For the latter quadrant, the intervention would be favored if the IRBR fell under the line of the risk–benefit acceptability threshold. In addition, risk–benefit acceptability curve (RBAC) was created based on the risk–benefit plane with any value of threshold. 15 This curve indicated the probability of the intervention to give the net benefit for all thresholds.
In terms of net benefit framework, net clinical benefit (NCB) was calculated by multiplying the acceptable risk threshold with ΔB then minus with ΔR. 16 We also generated the NCB probability curve with NCB in the Y-axis and any value of acceptability threshold in the X-axis. 15 This curve represented the number of additional AEs that occured per 100 patients treated at a given threshold.
All analyses were performed using STATA® version 16.0 (StataCorp LP, College Station, TX, USA) and the simulations were done using Microsoft® Excel 2019 (Microsoft Corp., Seattle, WA, USA).
Results
Study selection and characteristics of eligible studies
A total of 5425 studies was identified, but only 45 RCTs were included with a total of 5387 patients; see Figure 1. Most studies had two arms with included interventions of three studies for AMB, four for FLU, two for NEU, three for ACU, five for CHO, one for BENZ, one for ASP, three for OPI, 22 for ALP, two for NSD and one for COMB of CHO and BENZ. Five outcomes were considered, including POUR (N = 44), PVRU (N = 10), UTI (N = 7), LUTS (N = 4) and AE (N = 23). The characteristics of included RCTs are shown in Table 1 and Supplemental Table 2.

Flow diagram showing selection of articles for review.
Characteristics of included studies.

ACU, acupuncture; AE, adverse event; ALP, alpha-adrenergic antagonist; AMB, ambulation; ASP, anti-spasmodic agent; BENZ, benzodiazepine; CHO, cholinergic drug; COMB, combined intervention; FLU, fluid adjustment; GA, general anesthesia; LUTS, lower urinary tract symptom; NEU, neuromodulation; NSD, non-steroidal anti-inflammatory drug suppository; OPI, opioid antagonist related; PLA, placebo/standard care; POUR, post-operative urinary retention; PVRU, post-void residual urine; UTI, urinary tract infection.
Risk of bias assessment
From 45 RCTs, only three (6.7%) studies were identified as low overall risk of bias (RoB), 25 studies (55.6%) were considered as some concern and 17 (37.8%) studies were high RoB in the overall module. When each module was considered individually, there were six (13.3%) studies in randomization process, 38 (84.4%) in deviations from the intended intervention, 42 (93.3%) in missing outcome data, 33 (73.3%) in measurement of the outcome and six (13.3%) in selection of the reported result that were graded as low RoB. The RoB is shown in Supplemental Figure 1.
DMA
DMA was performed according to five outcomes, that is, POUR (39 studies, n = 4,697), PVRU (five studies, n = 490), UTI (four studies, n = 326), LUTS (three studies, n = 437) and AE (19 studies, n = 1949). For POUR, 39 studies were included with 4/6 pairs of interventions17,18,20,21,23,26,28,30–32,35–56,60–63 which were significant in the incidence of POUR relative to PLA, that is, AMB (n = 398), ACU (n = 325), OPI (n = 429) and ALP (n = 5206) with the pooled RR [95% confidence interval (CI)] of 0.44 (0.32, 0.61), 0.32 (0.11, 0.94), 0.62 (0.40, 0.95) and 0.55 (0.40, 0.76), respectively (Supplemental Figure 2). The heterogeneity was varied with the I2 ranged from 63.54 to 73.34. Subgroup analysis was performed in ALP versus PLA according to clinical significance including gender and operative type. The results indicated that the prophylactic effect of ALP was statistically significant only in male patients (pooled RR 0.45, 95% CI 0.29, 0.69). Considering the type of operation, we divided the operation into neurourological procedure (i.e. spinal cord surgery, prostate surgery and anti-incontinence surgery) and non-neurourological procedure (i.e. craniofacial surgery, cataract, thoracic surgery or limb surgery). Subgroup analysis was performed according to the type of operation in each gender and found that only non-neurourological operation had significant prophylactic effect from ALP in both genders (Supplemental Figure 3).
Only ALP versus PLA was pooled for UTI (four studies, n = 326),45,48,50,56 PVRU (five studies, n = 490)42,45,46,48,54 and LUTS (three studies, n = 437),42,46,50 but their effects were not significant in lowering these outcomes (Supplemental Figures 4–6). In terms of AE, 23 studies reported AE but only 19 studies of three comparisons which had at least three studies in each comparison were included in the analysis.17,18,30–32,61,62 Among these, overall and individual AEs were reported in 10 and nine studies (Supplemental Table 2).38,40,41,44,48–52,54–56,60 For those reported individual AEs, we used the maximum incidence of individual AE as the composite outcome. ALP significantly increased the AE relative to placebo, as shown in Supplemental Figure 7 with RR of 1.72 (1.07, 2.78, I2 0).
Publication bias was assessed using the Egger’s test, which suggested a symmetrical plot in all comparisons, corresponding to the funnel plot except POUR in ALP versus PLA. Contour-enhanced funnel plots suggested that the asymmetrical plots resulted from heterogeneity, not from publication bias (Supplemental Figure 8).
NMA
POUR
Forty-four17,18,20,21,23–26,28,30–53,55,56,58–63 RCTs with 12 interventions and 5338 patients were included; see the network map in Figure 2. Although all interventions reduced POUR, only AMB, ACU, ALP and NSD were statistically significant compared with placebo, but none of the indirect comparisons was significant; see Table 2. The probability of being the best intervention in lowering the POUR using rankogram (Supplemental Figure 9) and SUCRA demonstrated that NSD was the best prophylaxis (SUCRA 81.6%). The ranking along with SUCRA is shown in Table 3.

Network maps of (a) POUR, (b) UTI, (c) PVRU, (d) LUTS and (e) AE outcomes.
Relative treatment effects for lowering POUR and AEs.

Results were reported as risk ratios (95% confidence intervals) from network meta-analysis between each pair of treatments. Comparisons are read from left to right. Bold font indicates statistical significance. Results for POUR are presented above intervention names and results for AE are below intervention names.
ACU, acupuncture; AE, adverse event; ALP, alpha-adrenergic antagonist; AMB, ambulation; ASP, antispasmodic agents; BENZ, benzodiazepine; CHO, cholinergic agent; FLU, fluid restriction; NSD, non-steroidal anti-inflammatory drug suppository; OPI, opioid antagonist agent; POUR, post-operative urinary retention.
Treatment ranking by SUCRA from NMA in lowering POUR, UTI, PVRU, LUTS and minimizing AE outcomes accordingly.
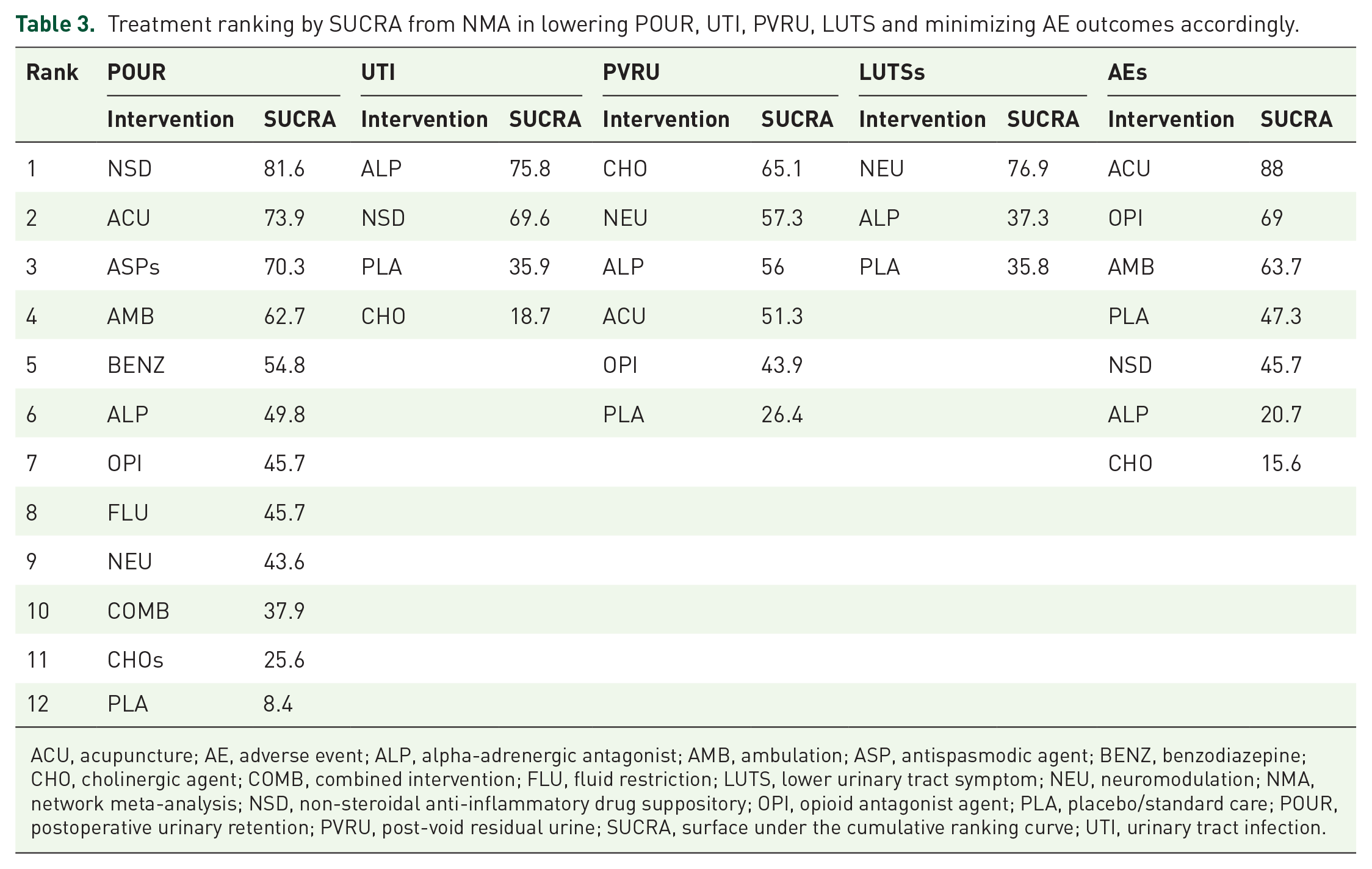
ACU, acupuncture; AE, adverse event; ALP, alpha-adrenergic antagonist; AMB, ambulation; ASP, antispasmodic agent; BENZ, benzodiazepine; CHO, cholinergic agent; COMB, combined intervention; FLU, fluid restriction; LUTS, lower urinary tract symptom; NEU, neuromodulation; NMA, network meta-analysis; NSD, non-steroidal anti-inflammatory drug suppository; OPI, opioid antagonist agent; PLA, placebo/standard care; POUR, postoperative urinary retention; PVRU, post-void residual urine; SUCRA, surface under the cumulative ranking curve; UTI, urinary tract infection.
No evidence of inconsistency was found (Chi-square = 6.71, p = 0.0816). Sensitivity analysis for the studies with variance greater than 80th percentile showed similar results. Adjusted funnel plots were performed and showed asymmetry, and heterogeneity was suspected (Supplemental Figure 10).
UTI
Data from seven RCTs with four interventions (i.e. PLA, CHO, ALP and NSD) and 652 patients were used; see Figure 2. NSD and ALP were about 0.38 (0.02, 9.15) and 0.57 (0.25, 1.27) times better in lowering UTI whereas CHO was about 1.16 (0.25, 1.27) times higher risk of UTI relative to PLA. However, none of them was statistically significant (Supplemental Table 33). The probability of being the best intervention in UTI prevention was NSD, followed by ALP, PLA and CHO (Table 3 and Supplemental Figure 11). No evidence of inconsistency was found (Chi-square = 0.36, p = 0.5498). Adjusted funnel plots were performed and showed symmetry of the plot (Supplemental Figure 12).
PVRU
A network map was constructed using data of 10 RCTs (n = 900) with six interventions, that is, PLA, NEU, ACU, CHO, OPI and ALP; see Figure 2. All interventions decreased PVRU by about 8.6–20.2 ml, but none of them was statistically significant; see Supplemental Table 3. The probability of being the best intervention in decreasing PVRU was CHO, followed by NEU, ALP, ACU, OPI and PLA (Table 3 and Supplemental Figure 13). No evidence of inconsistency was found (Chi-square = 0.99, p = 0.3189). Adjusted funnel plots were performed and showed asymmetry of the plot, suggesting possible publication bias (Supplemental Figure 14).
LUTS
Data from four RCTs with three interventions (i.e. PLA, NEU and ALP) and 523 patients were analyzed. A network map was created and no closed loop was found; see Figure 2. LUTS was reported in International Prostate Symptom Score (IPSS) or American Urological Association (AUA) symptoms index. Both NEU and ALP decreased LUTS about 2–0.03, but these were not statistically significant (Supplemental Table 3). The probability of being the best intervention in lowering LUTS was NEU, followed by ALP and PLA (Table 3 and Supplemental Figure 15). Inconsistency assumption was forced analyzed and found no inconsistency (Chi-square = 0.00, p = 0.9766). Adjusted funnel plots were performed and showed symmetry of the plot (Supplemental Figure 16).
AE
The analysis comprised data from 23 RCTs with seven interventions (i.e. PLA, AMB, ACU, CHO, OPI, ALP and NSD) and 2596 patients. A network map was constructed; see Figure 2. For direct comparison, AMB, ACU and OPI were found to decrease the incidence of AEs compared with PLA with the RRs of 0.74 (0.25, 2.17), 0.32 (0.07, 1.49) and 0.72 (0.46, 1.14), respectively. On the other hand, CHO, ALP and NSD increased the AE with RRs of 2.47 (0.57, 10.70), 1.66 (0.99, 2.78) and 1.15 (0.23, 57.40), respectively. However, no statistical significances were found. Regarding indirect comparison, statistically significant increased AE was found in ALP versus ACU and ALP versus OPI (Table 2). The probability of being the least AE occurrence was ACU, followed by OPI, AMB, PLA, NSD, ALP and CHO (Table 3 and Supplemental Figure 17). Inconsistency assumption was forced analyzed and no inconsistency was found (Chi-square = 0.00, p = 0.9446). Adjusted funnel plots were performed and showed symmetry of the plot (Supplemental Figure 18).
Cluster ranking plot and risk–benefit analysis
A cluster rank plot was created according to the SUCRA of benefit and the risks from each intervention pair; see Figure 3. ACU and AMB were in the right upper quadrant, implying that ACU was the best in high efficacy of POUR prevention and PVRU reduction with the least AE, and AMB was the second best for POUR prevention.

A cluster rank plot of benefit in lowering POUR versus risk of AE outcomes.
In terms of risk–benefit analysis, the risk–benefit plane was constructed and is shown in Supplemental Figure 19. A scatter plot of ACU fell in the right lower quadrant, implying dominant risk–benefit over the placebo, although the shape of the plot was sparse, which represented the low precision of the outcome. Supposing the risk–benefit acceptability threshold was 0.2, which meant two patients with additional AE were acceptable for the 10 additional POUR preventions, AMB, ACU and ALP were beneficial as the plots were under the threshold line. IRBRs in POUR prevention compared with PLA were 0.0118, 0.7372, −0.1291, 1.4131, −0.0608 and −0.0092 for AMB, ACU, CHO, OPI, ALP and NSD, respectively (Supplemental Table 4). RBACs of each intervention are presented in Supplemental Figure 20 and the estimations of percent chance that the IRBR would be less than risk–benefit acceptability thresholds are summarized in Supplemental Table 5. For clinical implication, RBACs among the interventions were compared in any threshold and these aided in decision making (Figure 4). Given the low acceptable threshold of 0.2, referring to only two acceptable AEs for 10 beneficial outcomes, AMB was the most favorable followed by ACU and ALP, then OPI and NSD. CHO was the least useful intervention. Another example, with the acceptable threshold moved up to 0.3, showed that AMB and ALP were the best with similar benefit, then ACU, NSD, OPI and CHO. In terms of net benefit framework, NCB acceptability curve is shown in Supplemental Figure 21, representing the probability of being the best intervention at distinct threshold.
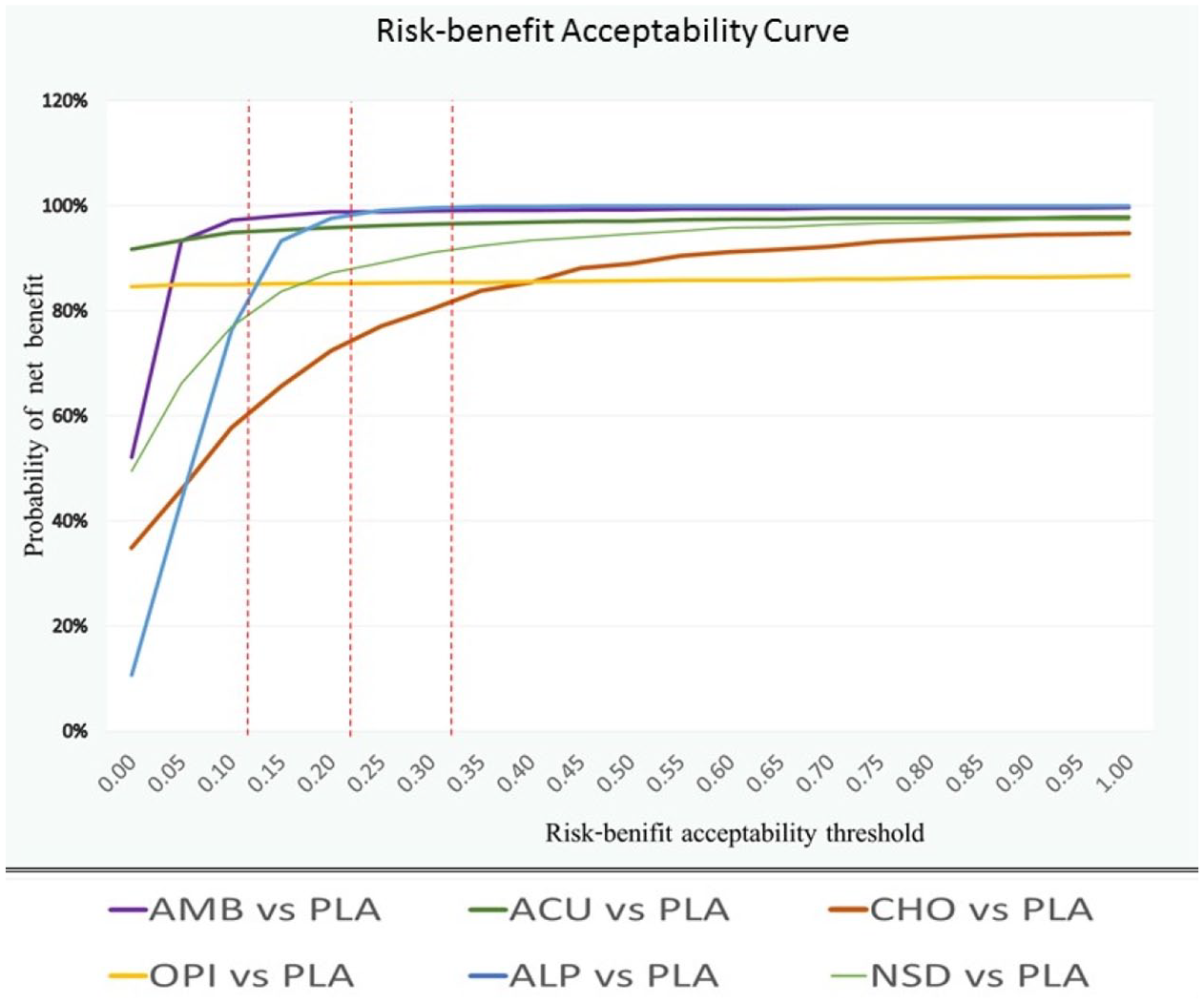
Risk–benefit acceptability curve in lowering post-operative urinary retention at each distinct threshold among six intervention pairs.
Discussion
POUR is one of the most common complications after surgeries, which results in urinary complications including UTI, patients’ discomfort and distress, prolonged hospital stay and increased cost of treatment. 8 Currently, there are many interventions reported aiming to prevent POUR. However, there is no standard protocol for preventing this condition because of the uncertainty of the efficacy and the concern about AEs. From the previous studies, there were five meta-analyses published about intervention for POUR prevention, two studies analyzed about methods and timing of catheterization.64,65 Jackson et al. 9 reported not only medical but also non-medical interventions for POUR prevention, including both RCTs and non-RCTs. Clancy et al. 66 assessed ALP from five RCTs, particularly in men who underwent inguinal hernia repair. Ghuman et al. 67 described 15 RCTs and analyzed the effect of ALP in patients who underwent various kinds of operation. The results of our study were in a similar direction compared with the previous meta-analyses, but we added a higher number of included studies and the network technique allowed for indirect comparison of all treatments available and ranking which treatment should be best in POUR prevention.
In our study, we reported the comprehensive review, first NMA and risk–benefit analysis of all available RCTs in preventing POUR. Our interventions of interest included 11 non-medical and medical interventions with five outcomes. The direct comparisons from NMA showed that AMB, ACU, OPI and ALP significantly reduced POUR. Nevertheless, due to the limited number of the studies and the missing data of covariables, further analysis including heterogeneity, meta-regression and subgroup analysis was not possible in some comparisons. The possible source of heterogeneity might be from the diversity of the studies in terms of populations, the schedule and dosage of the intervention, the definition of outcomes, time of measurement and variation in operation type.
For NMA, our results showed that the best intervention for POUR prevention was NSD followed by ACU, ASP, AMB, BENZ, ALP, OPI, FLU, NEU, COMB, CHO and PLA. However, only NSD, ACU, AMB and ALP were found statistically significant in our analysis compared with PLA. No statistical significance was observed among other interventions from both direct and indirect comparisons. For PVRU, the best intervention was CHO followed by NEU, ALP, ACU, OPI and PLA. Regarding UTI, NSD was considered the best, followed by ALP, PLA and CHO. The ranking of probability of being the best among these outcomes was sparse and not in the same direction among the outcomes. We hypothesized that the reason behind this was probably the number of involved studies in our secondary outcomes, which was quite small, and because of the heterogeneity of the studies. Concerning the AEs, the intervention with the least AEs was ACU, followed by OPI, AMB, PLA, NSD, ALP and CHO. Nevertheless, only ALP versus OPI and ALP versus ACU were statistically significant. Most AEs were reported as mild to moderate, such as nausea, vomiting, pruritus et cetera. One of the obstacles in AE analysis was the difference in reported data. Some studies reported on overall AEs while some studies described each AE individually. Therefore, we chose to analyze as the composite outcome, which counted the most common reported incidence to include into the analysis. As a result, the incidence of AE might be underestimated. Another concern, regarding RoB, was that most of the studies were considered to be high concern and some concern for RoB, so interpretation should be done with caution.
According to the cluster ranking, ACU was found to be the best in POUR and PVRU reduction, followed by AMB. However, the decision for choosing also depended on the willingness to accept any complication, which was different in each threshold. Aiming to simplify for clinical application, we constructed the RBAC (Figure 4), which compared among six interventions. This curve was valuable for intervention selection based upon acceptable threshold. Based on our findings, we recommend patients should have a routine non-medical treatment of early AMB as soon as possible after surgery. Medical intervention, such as alpha-adrenergic antagonists might be considered in male patients undergoing non-neurourological procedure, if the patients and clinicians accept that risk of AEs might occur around 10–15%.
Strengths and limitations
Our study has several strengths. To our knowledge, this is the first NMA reporting about POUR prevention. In addition, we comprehensively analyzed with a large number of population and included only RCTs which assumed the appropriate study design to evaluate the efficacy of the interventions. Finally, we reported the risk–benefit analysis with the RBAC, which is easy to understand and simple to implement and intuitively provides for the clinician to incorporate the information for decision making. There were some limitations in our study. First, the number of the studies in some comparisons were small and heterogeneous, so the analysis was limited to only some outcomes and also precluded further analysis such as subgroup or sensitivity analysis. It should be noted that the incidence of AEs might be underestimated due to the heterogeneity of outcome reporting and the analysis as the composite outcome. Another concern was regarding RoB, because most of the studies were considered high concern and some concern for RoB, so interpretation should be done with caution. Further well-designed study may be needed to reach further conclusions.
Conclusions
AMB, ACU, OPI, ALP and NSD might be useful in prevention of POUR. However, when both efficacy and AEs were considered with the acceptable threshold of side effect of 15%, ALP might be the best, followed by OPI, NSD and CHO.
Supplemental Material
sj-pdf-1-tau-10.1177_17562872211022296 – Supplemental material for Medical and non-medical interventions for post-operative urinary retention prevention: network meta-analysis and risk–benefit analysis
Supplemental material, sj-pdf-1-tau-10.1177_17562872211022296 for Medical and non-medical interventions for post-operative urinary retention prevention: network meta-analysis and risk–benefit analysis by Pokket Sirisreetreerux, Rujira Wattanayingcharoenchai, Sasivimol Rattanasiri, Oraluck Pattanaprateep, Pawin Numthavaj and Ammarin Thakkinstian in Therapeutic Advances in Urology
Footnotes
Acknowledgements
We thank Stephen Pinder, a native-English-speaking advisor to the Department of Clinical Epidemiology and Biostatistics, Faculty of Medicine Ramathibodi Hospital, Mahidol University, for proofreading our manuscript.
Author contributions
PS did the data extraction, analyzed and interpreted the data and was a major contributor in writing the manuscript. RW extracted the data. SR analyzed and helped with interpretation of data. OP analyzed and helped with interpretation of data. PN has made substantial contributions to the conception, design of the work and did the data extraction. AT has made substantial contributions to the conception and revised the manuscript. All authors read and approved the final manuscript.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
