Abstract
Objective
The efficacy of probiotics and/or prebiotics for preventing eczema in infants remains unclear. This meta-analysis evaluated published studies on pro/prebiotics for eczema prevention, investigating bacterial strain efficacy and changes to the allergy status of the children involved.
Methods
Randomized, double-blind, placebo-controlled trials were analysed, irrespective of bacterial strains used in the pro/prebiotics. Studies of pregnant women, nursing mothers and infants receiving pro/prebiotics were included. All infant participants were assessed within 2 years of birth. Incidences of eczema and systemic sensitization were measured by weighted relative risk ratios (RRR).
Results
The 14 studies on probiotics had a pooled RRR of 0.69 (95% confidence interval [CI]: 0.62, 0.78). Three studies on prebiotic consumption showed a RRR of 0.80 (95% CI: 0.54, 1.18). One study of mixed pro/prebiotic (synbiotic) strains found a RRR ratio of 0.81 (95% CI: 0.66, 0.99). No consistent sensitization changes were found. Only the combination of nonspore lactobacilli and bifidobacteria reduced the incidence of eczema.
Conclusion
This meta-analysis found that probiotics or synbiotics may reduce the incidence of eczema in infants aged <2 years. Systemic sensitization did not change following probiotic administration.
Introduction
Increased living standards are often accompanied by decreased rates of infection, due to greater attention to environmental and personal hygiene. However, near-sterile conditions do not allow the neonate to establish gastrointestinal microbial flora and immunity to pathogens, which may result in high morbidity due to allergic disease. 1 Eczema eruption presents with rashes, erythema, papules and severe paroxysmal itching, mainly involving the head and face. High rates of recurrence, bleeding, scarring and even infection (due to scratching) can seriously affect health-related quality of life. The incidence of infantile eczema has risen rapidly, especially in developed countries. 2 Unfortunately, the aetiology and pathogenesis of infantile eczema is not fully understood, and no effective treatment is available. Hence, developing a preventative strategy for atopic eczema is of great importance. Research has shown that a helper T cell-mediated immune imbalance occurs in individuals with eczema, which prevents the Th2-dominated (allergic) immune response from quickly switching back towards a Th1 immune response, to create a more balanced immune state. 3 Early establishment of gastrointestinal microflora can enhance Th1 immunity and inhibit Th2-mediated allergic inflammation. 4 At the same time, gastrointestinal microflora can stimulate the expression of immunoglobulin (Ig)A, which enhances the mucosal defence system and stimulates anti-inflammatory effects, thereby reducing the incidence of eczema. 5
Probiotics are cultures of living micro-organisms that exert health benefits beyond inherent general nutrition, when ingested in certain amounts. 6 Administration of probiotics can enhance gastrointestinal microflora colonization and balance the Th1/Th2 immune response, thereby aiding immune maturation. 7 Prebiotics are nondigestible oligosaccharides in general, and fructo-oligosaccharides in particular. They have been shown to stimulate the growth of endogenous bifidobacteria, which, after a short period of ingesting the prebiotics, become the predominant bacterial species in human faeces. 8 A clinical study found that administration of probiotics and/or prebiotics (synbiotics) early in life can help gastrointestinal microflora colonization, and has been linked to the prevention of necrotizing enterocolitis and a reduction in rates of infectious diarrhoea. 9 Since Kalliomaki et al. 10 first used probiotics to prevent eczema in 2001, many clinical trials to investigate the role of probiotics or prebiotics in eczema prevention have been conducted. However, the results of such studies have been inconsistent, with some10–17 reporting positive results and others18–27 giving opposing views.
The present report describes a meta-analysis of oral probiotics and/or prebiotics for eczema prevention, with a focus on effects on the infant immune system and any bacterial strain specific outcomes.
Materials and methods
Study selection
Randomized, double-blind, placebo-controlled trials were identified from the PubMed database using specific terms (prebiotic, probiotic or synbiotic; eczema or dermatitis prevention) and filters (human; randomized controlled trial; infant: birth to 23 months); Embase and the Cochrane Library (until March 2013) databases were also searched. Participants in the identified studies were healthy pregnant women and infants <2 years old, with or without a family history of allergic disease. Participants received oral probiotics and/or prebiotics (no strain or quantity limits) for various times (also no limits) or a placebo control. Neonates were given maltodextrin, cereals, formula or breast milk at the same time. The references of all articles retrieved were also examined. No language limitation was imposed, and unpublished studies were not included. Studies were excluded if the original data were not available. If there were multiple publications of the same study population, the most detailed study was included. Outcome measures in the infants were the incidence of eczema (primary) and systemic sensitization after probiotic consumption (secondary; measured by a positive skin prick test [SPT])18,26 and/or elevation of serum IgE (≥0.35 kIU/l).12,13,19 Certain studies also measured individual cytokine levels.18–20, 2, 4,26 All participants were followed up for ≤2 years.
Data extraction and quality of studies
Data were extracted and evaluated independently by two reviewers (D.D. and Z.J.L.). All studies were assessed according to the Cochrane Handbook for Systematic Reviews of Interventions. 28
Data synthesis and analysis
For the meta-analysis, the RevMan analytical software package (RevMan, version 5.0, Cochrane Collaboration, Oxford, UK) was used. The incidence of eczema and systemic sensitization were assessed by Mantel–Haenszel weighted relative risk ratios (RRR), with 95% confidence intervals (CI). The I2 index was calculated to assess the heterogeneity of the study results. A fixed-effects model was presented when the studies had little variance; a random-effects model was used when heterogeneity was present. Analysis was restricted to subgroups of studies defined by clinical characteristics. The statistical program Stata®, version 12.0 (StataCorp LP, College Station, TX, USA) was used for the sensitivity analysis. A P-value of <0.01 was considered statistically significant.
Results
Characteristics of studies included in a meta-analysis of the effects of pro- and prebiotics on the incidence of eczema in children aged <2 years.

Fulfill, n participants who completed treatment; loss, n lost to follow-up. ST11, Lactobacillus paracasei; LPR, Lactobacillus rhamnosus LPR; BL999, Bifidobacterium longum BL999; LGG, L. rhamnosus GG; Bb-12, Bifidobacterium animalis subsp. lactis Bb-12; La-5, Lactobaccillus acidophilus La-5; BGN4, Bifidobacterium bifidum BGN4; AD011, B. lactis AD011; AD031, L. acidophilus AD031; pectin-derived acidic oligosaccharides (pAOS); short chain oligosaccharides (scGOS); long chain fructo-oligosaccharides (lcFOS); HN001, Lactobacillus rhamnosus HN001; HN019: B. animalis subsp lactis strain HN019; ATCC, Lactobacillus reuteri ATCC; LAVRI-A1, L. acidophilus LAVRI-A1; L. rhamnosus GG(ATCC 53103); L. rhamnosus LC705 (DSM 7061); Bifidobacterium breve Bb99 (DSM 13692); Propionibacterium freudenreichii ssp. shermanii JS (DSM 7076); F19: L. paracasei ssp. paracasei strain LF19; L. rhamnosus strain GG (ATCC 53103); Bifidobacterium lactis Bb12; B. bifidum W23; B. lactis W52; Lc. lactis W58; PDX, polydextrose; GOS, galacto-oligosaccharides; LOS, lactulose; Y, yes; N, no; U, unclear (data unavailable from original article).
Fourteen studies10–13,16,18–26 were randomized, double-blind, placebo-controlled trials of probiotics for the prevention of eczema. With a total of 2 550 participants, the heterogeneity I2 index was 57% (P = 0.004). Compared with the placebo group, the pooled RRR for infants developing eczema while they/their mothers were taking probiotics was 0.69 (95% CI: 0.62, 0.78; P = 0.00001) (Figure 1). Sensitivity analysis showed that this outcome was reliable (data not shown).
Incidence of eczema in infants <2 years old after probiotic and/or prebiotic administration to the infants themselves and/or to their mothers.
The three studies14,15,27 that evaluated consumption of prebiotics had a heterogeneity I2 index of 48%. There were no significant effects of prebiotic consumption (by infants or mothers) on development of eczema in the infants (RRR = 0.80 (95% CI: 0.54, 1.18)). A large-scale study conducted by Kukkonen et al.
17
of probiotics mixed with prebiotics showed a RRR of 0.81 (95% CI: 0.66, 0.99; Figure 1). A funnel plot of the studies of eczema prevention included in the meta-analysis is shown in Figure 2.
Funnel plot of studies included in a meta-analysis of pro- or prebiotics (and a combination of both: synbiotics) in the prevention of infant eczema, suggesting there is unlikely to be a relationship between effect and study size.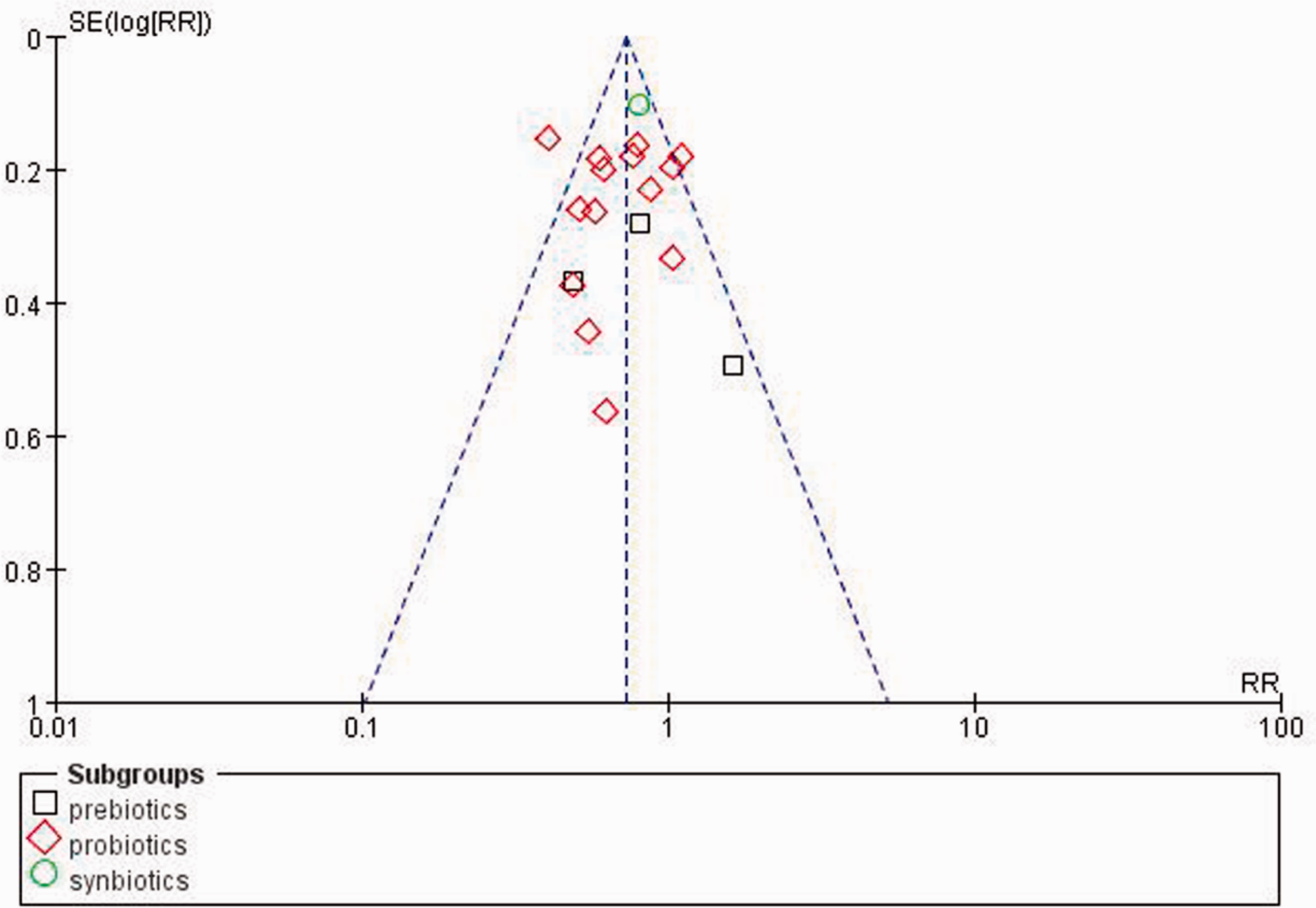
The probiotic intervention trials were classified into three groups by the strain of bacteria used as the probiotic: nonspore lactobacilli;10,11,16,18,19,23–25 bifidobacteria;16,20 mixed strains12,13,21,22,26 (containing both nonspore lactobacilli and bifidobacteria). The pooled RRR were 0.78 (95% CI: 0.60, 1.01), 0.82 (95% CI: 0.63, 1.07) and 0.58 (95% CI: 0.44, 0.76), respectively (Figure 3). Significant effects were observed only when mixed strains were used.
The efficacy of different probiotic strains for the prevention of infant eczema in the studies included in a meta-analysis.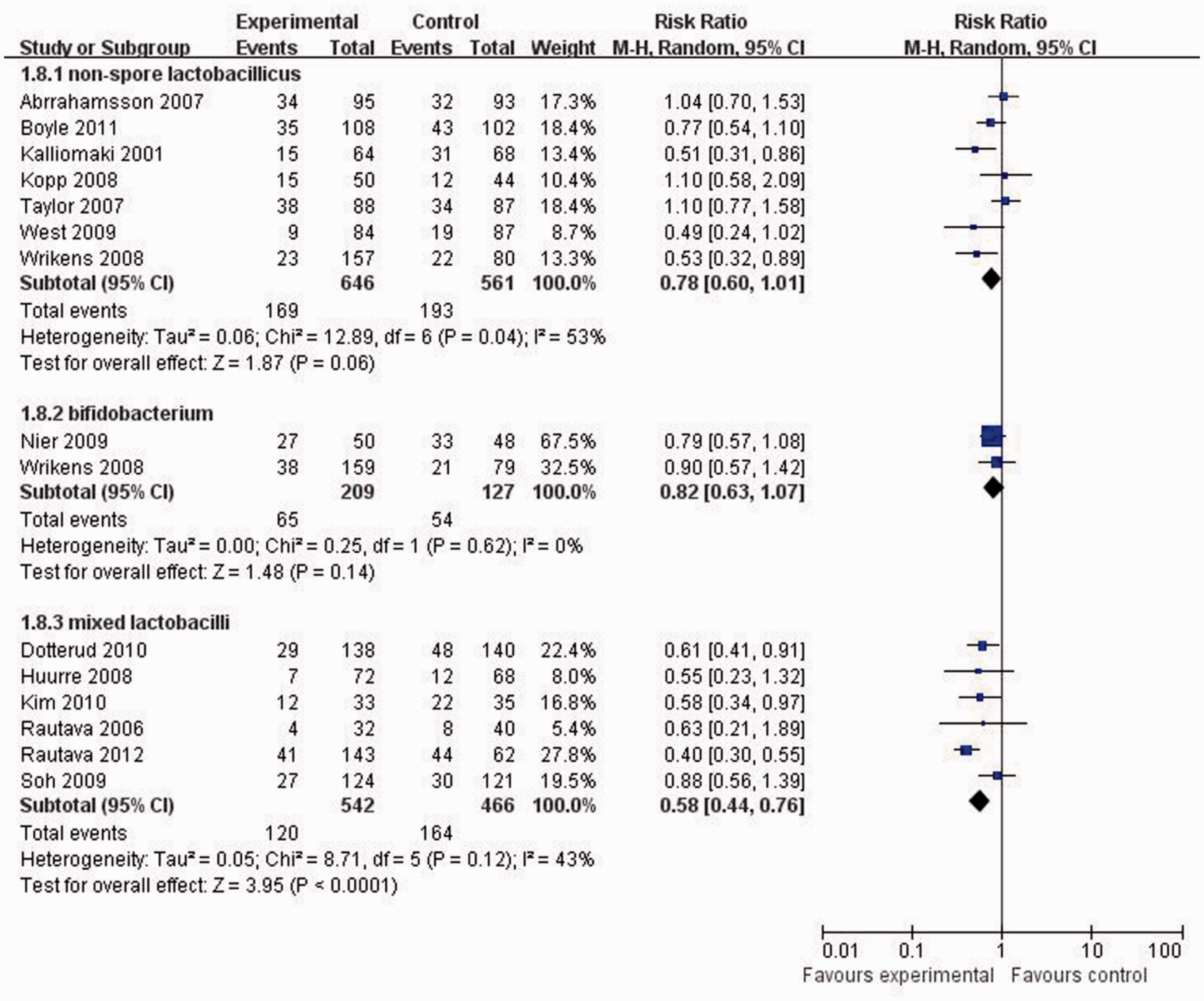
Studies were classified into three groups according to their clinical characteristics: SPT + IgE, SPT and IgE. The RRR for each group was 0.97 (95% CI: 0.69, 1.37), 0.87 (95% CI: 0.72, 1.06) and 1.04 (95% CI: 0.69, 1.56), respectively. None of these effects was statistically significant (Figure 4).
Systemic sensitization changes identified in a meta-analysis of studies of eczema in infants aged <2 years after pre- or probiotic intervention to infants and/or their mothers (during pregnancy or lactation).
Discussion
The present meta-analysis included randomized, double-blind, placebo-controlled trials of oral probiotic and/or prebiotics consumed by pregnant and/or nursing mothers and/or their offspring, for the prevention of eczema. The incidence of eczema was reduced by ∼31% with the consumption of probiotics, but was not significantly changed with the consumption of prebiotics. Furthermore, combining nonspore lactobacilli and bifidobacteria resulted in a 42% reduction in the incidence of eczema. Thus, the use of mixed probiotics may be a useful strategy for reducing infantile eczema. Probiotics and synbiotics generally cause few side-effects and are easily accessible. 3 Therefore, such cultures (especially mixed strains of probiotics), should be recommended for infants or pregnant women, due to their moderate ability to prevent infant eczema.
Asthma, rhinitis and eczema are the most common allergic diseases, among which atopic eczema has the highest incidence in infants and is the best predictor for subsequent allergic asthma and rhinitis in school-age children. 47 A positive SPT and high IgE levels (≥0.35 kU/l) have correlated with atopic and allergic disease, and systematic sensitization within 2–3 years after birth has been associated with allergic disease morbidity. 48 Probiotic interventions can reduce the incidence of eczema to a certain extent, but they cannot change the host allergic status. In the studies that measured cytokines,18–20,22,24,26 levels of certain cytokines were found to have changed at the end of the study, indicating changes in the allergic conditions of the infants. However, different cytokines were measured in different studies,18–20,22,24,26 and the numbers assessed were too small to add to the meta-analysis.
The observed effects of pro/prebiotic administration depended on many factors. Differences in bacterial strains (quantity and quality), time and duration of interventions, diagnostic criteria for eczema, genetic makeup and living environments of the infants all create clinical heterogeneities. In addition, the long duration of the studies resulted in a high rate of loss to follow-up. To strengthen the conclusion and reduce the heterogeneity, further randomized clinical trials are required.
Through conducting this meta-analysis, several issues became apparent that will require further study. In particular, more repeat studies of prebiotics or synbiotics are needed. Certain baseline demographics of the infants (including family risk of eczema, genetics and lifestyle factors) were not unified across the studies and this may have affected the results. Thus, repeated interventions are critical for confirming outcomes. Additionally, durations of completed studies varied between 4 weeks and 2 years, making it difficult to choose the appropriate duration for administering probiotics to maximize eczema prevention. Compliance also remains a problem to be explored. Most of the studies that yielded negative results were assessed within the first year of life, possibly interfering with the cumulative incidence and influencing the results.
In conclusion, based on this meta-analysis of related published studies, the administration of probiotics and synbiotics may reduce the incidence of infant eczema, whereas prebiotic administration alone has no effect. With probiotics, systemic sensitization does not change following probiotic administration, and only probiotics containing mixed bacterial strains may achieve significant results. More studies are needed to determine how synbiotics should best be administered.
Footnotes
Declaration of conflicting interest
The authors had no conflicts of interest to declare in relation to this article.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
