Abstract
Background:
A novel formulation of oral testosterone undecanoate (TU) was studied in a long- and short-term phase III trial to evaluate safety and efficacy.
Methods:
Hypogonadal men (age 18–65 years; two morning serum testosterone (T) <300 ng/dl with signs/symptoms) were recruited into a 365 day (trial I) or 105 day (trial II), randomized, multicenter trial. Patients were randomized 1:1 to oral TU (
Results:
Oral TU efficacy (% of patients with eugonadal T
Conclusion:
A new oral TU formulation was safe and effective and represents a significant therapeutic advance for the treatment of appropriate hypogonadal men.
Introduction
Testosterone (T)-replacement therapy (TRT) has evolved over time to provide healthcare providers and their hypogonadal patients with numerous treatment options. Beginning with early use of implanted T-pellets to injectable T-esters to oral methyltestosterone to a first-generation oral T-undecanoate (TU) product to scrotal and non-scrotal T patches and then to topical T-gels, the number of TRT choices continues to evolve. 1 More recent additions to the TRT armamentarium include a buccal patch, a long-acting T-undecanoate injection (intramuscular) product, a short-acting T-enanthate injection (e.g. 7 days; subcutaneous) and a nasal T-gel. Each of these delivery routes are associated with well-known drawbacks, including pain of injection, dermal irritation, T transference and potentially serious liver toxicity (e.g. oral methyltestosterone). In addition, dose adjustment to individualize patient response to these TRT methods can be challenging. Missing from the healthcare professional’s stable of TRT products is an oral T formulation that meets the current rigorous regulatory standards for safety and efficacy.
Historically, efforts to administer oral T have taken two primary paths: alkylation of T at the C-17 position to create T analogs that are resistant to first-pass hepatic metabolism (exemplified by methyltestosterone);
2
or fatty-acid esterification of T to create a T-ester (exemplified by TU) that is absorbed
To address the absence of an oral TRT product that meets current-day regulatory requirements for efficacy and safety, TU was formulated in a unique self-emulsifying drug delivery system (SEDDS) that was initially evaluated in short-term clinical studies.
9
SEDDS formulations combine hydrophilic and lipophilic excipients that enable the solubilization of highly lipophilic molecules like TU in the gut so that they may be absorbed after oral ingestion with a typical meal (no high-fat content required).
10
As depicted in Figure 1, absorption of oral TU occurs almost exclusively (>97%)
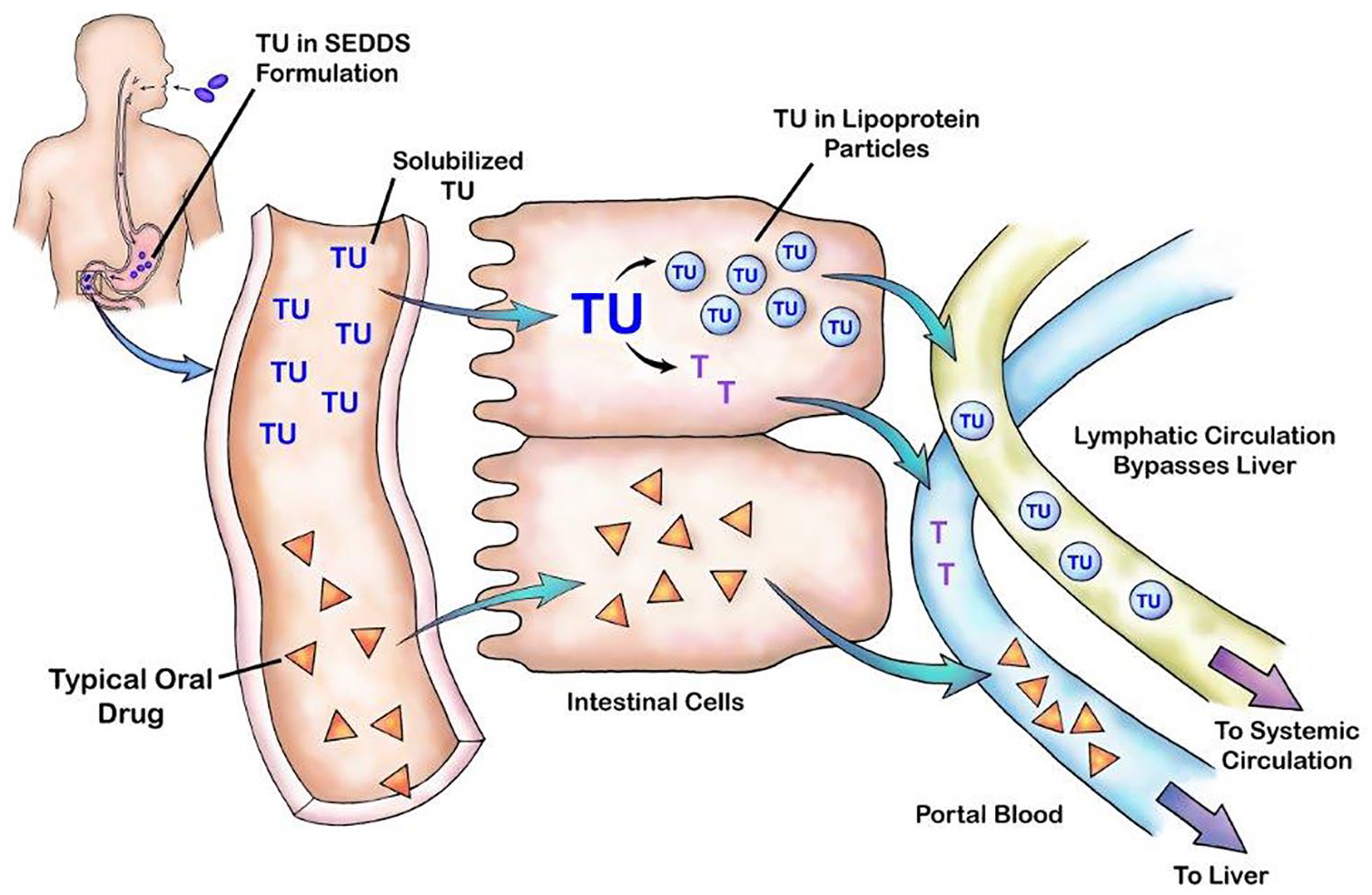
Pictorial representation of TU lymphatic absorption after oral delivery in SEDDS formulation.
This was important for two reasons. First, real-world clinical monitoring of T is based on serum T measurements. Second, it was necessary to adapt the dose-titration algorithm utilized in the pivotal trial of JATENZO (where T was assayed in NaF-EDTA plasma) for use with a single serum sample derived from blood collected into a standard plain collection tube (i.e. without added chemicals).
The present article summarizes two phase III clinical trials conducted to demonstrate long- and short-term safety and efficacy of a new oral TU formulation (JATENZO®) studies CLAR-09007, trial I [ClinicalTrials.gov identifier: NCT01403116] and CLAR-15012, trial II [ClinicalTrials.gov identifier: NCT00272278]. The primary difference between these studies was the starting oral TU dose and subsequent dose-titration algorithm employed to maximize the percentage of patients who achieved T
Materials and methods
The phase III clinical trials detailed herein were approved by a central or site-specific institutional review boards before study initiation at each clinical site and were conducted in accordance with the Declaration of Helsinki and/or all relevant federal regulations, including good clinical practice guidelines. Written informed consent was obtained from trial participants before any study-related procedures were conducted.
Patient populations
Eligible patients were men aged 18–65 years, body mass index <38 kg/m2, with hypogonadism as defined by verified low morning serum total T <300 ng/dl (blood samples collected between 0600 h and 1000 h on 2 separate days approximately 7 days apart) and a history of signs and/or symptoms consistent with hypogonadism. Patients were naïve to androgen-replacement therapy or had an adequate washout of previous androgen-replacement therapies. Patients were excluded if they had significant uncontrolled intercurrent disease of any type, hematocrit (Hct) <35% or >48%, history of polycythemia, untreated, severe obstructive sleep apnea, abnormal digital rectal exam, prostate-specific antigen (PSA) >4.0 ng/ml, International Prostate Symptom Score >19 or history of prostate cancer. Prohibited medications included those that could affect T levels, T metabolism, or levels of T metabolites (e.g. antiandrogens, 5-alpha-reductase inhibitors, estrogens, long-acting opioid analgesics, or human growth hormone), as well as nutritional supplements that could possibly increase serum T (e.g. androstenedione or dehydroepiandrosterone).
Primary and secondary efficacy parameters
The primary efficacy variable for each study was the proportion of oral TU-treated patients who achieved a 24-h T
Study design features
Both studies were open-label, randomized trials of an oral SEDDS formulation of TU [JATENZO®; TU capsules]. Key design features are summarized in Table 1. A topical T control group [i.e. T-gel (AndroGel® 1%) or T-solution (Axiron®)] served as a positive control arm for comparative safety assessments in trials I and II, respectively. However, oral TU efficacy was evaluated independent of that observed with topical T. In each study, participants in both treatment groups were provided up to two opportunities for adjustment of their T dose based on individual responses to oral TU (per protocol) or topical T (per product labeling instructions). In general, a patient’s serum/plasma T
Key design features of phase III studies.

Percentage of subjects with testosterone
Pivotal trial on which efficacy was based for FDA approval.
Meals contained typical fat content and were not required to be ‘high fat’ meals.
Blood sample taken 4–6 h after morning oral TU dose.
T
The PDQ 14 was used to assess sexual function and mood changes. Patients were asked to complete the questionnaire every day for 7 consecutive days before day 1 and the last study day (end of study). Each domain of the PDQ (sexual desire, enjoyment and performance, mood, and sexual activity score) was evaluated.
a.m., morning; BID, twice daily; BMD, bone mineral density; DEXA, dual-emission X-ray absorptiometry;
Safety measures included physical examination, vital signs, fasting clinical laboratory analysis (hematology, chemistry, urinalysis), cardiovascular (CV) biomarker monitoring [high-sensitivity C-reactive protein (hs-CRP),15,16 lipoprotein-associated phospholipase A2 (Lp-PLA2),
17
lipoprotein a (Lp(a)),
18
and apolipoprotein A1 (Apo-A1)
19
in trial I only; hs-CRP is a strong independent predictor of CV disease risk and its reduction, for example, by rosuvastatin in the JUPITER trial showed a reduction in subjects with normal LDL (see Ridker
Oral TU dose-adjustment paradigms
About 4 and 8 weeks after oral TU therapy was initiated in each trial, patients could have their TU dose adjusted based on T
Statistical methods
The proportion of oral TU-treated subjects whose 24 h total testosterone
Changes from baseline for the PDQ were summarized by treatment group, and overall for each subscale score. A 95% CI for the change from baseline within each treatment group was computed. Change from baseline for other clinical parameters of interest [body composition and bone mineral density (BMD)] and the difference between treatment groups for these endpoints was compared using an analysis of covariance (ANCOVA).
Results
Patient characteristics and disposition
Table 2 summarizes key patient characteristics and disposition parameters. Notable is that in both studies, at least 90% of enrolled subjects were included in the primary efficacy analyses. In addition, few patients discontinued due to adverse events and these typically were not a result of T therapy. Mean compliance on oral TU therapy in both studies was ⩾95%.
Summary of key oral TU patient demographic and disposition parameters.

Efficacy population comprised all randomized patients who had sufficient data at day 90 PK visit to calculate serum T
Efficacy population defined as all patients who had evaluable PK profile to calculate plasma T
Day 365 of oral TU dosing.
BMI, body mass index;
Primary and secondary efficacy and testosterone time–concentration profile in response to oral TU
Primary efficacy
The primary efficacy objective of both studies was to demonstrate that a mean average T concentration could be achieved in at least 75% of oral TU patients after one or two dose adjustments. As shown in Table 3, 83.6% (trial I) and 87.3% (trial II) of oral TU patients achieved circulating concentrations of T in the mid-eugonadal range and efficacy was sustained to the 12-month timepoint in the long-term study. Efficacy in the topical T arms of each study were similar in magnitude [79.0% (trial I) and 87.3% (trial II)] and not statistically different from oral TU. The
Efficacy results for oral TU patients in trials I and II.

On final PK day after up to two dose-adjustment opportunities.
Minimum lower bound of 95% confidence interval must be ⩾65% to satisfy FDA standard.
a.m., morning;
Testosterone time–concentration profiles on the efficacy determination timepoints for trials I and II are shown in Figure 2. Both profiles are consistent and indicate peak T levels are achieved about 4 h after oral TU was administered. Mean peak T concentrations were within the eugonadal range only in the pivotal trial (trial II).

Mean concentration–time profiles for total serum T in patients treated with oral TU at final PK visit in trial I, and for NaF-EDTA plasma total T in patients treated with oral TU at final PK visit in trial II.
Single-sample dose-adjustment paradigm for oral TU
PK modeling and simulation results for circulating T confirmed that dose-titration decisions based on a single blood sample taken 3–5 h or at 4 h after the morning oral TU dose was an effective means to guide dose adjusting to achieve/maintain T concentrations in the eugonadal range. As shown in Table 4, regardless of the three measures used to determine the need to adjust the oral TU dose (i.e.
PK simulation results evaluating mean T concentration (
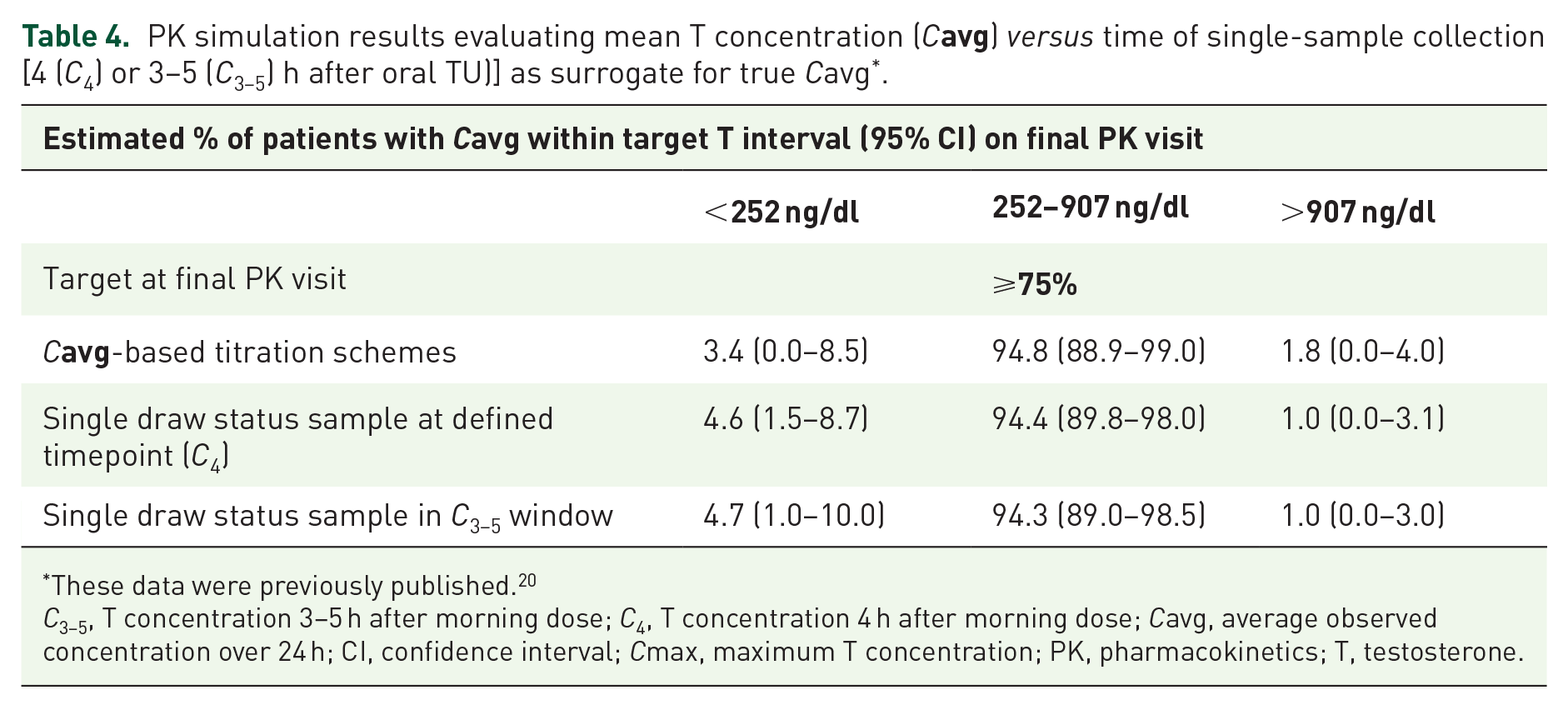
These data were previously published. 20
Secondary efficacy
In trial I, three secondary efficacy parameters were assessed: PDQ (a measure of psychosexual function), body composition (i.e. lean and fat mass) and BMD of hip and spine. Although there was no statistically significant difference in PDQ responses between the oral TU groups, oral TU was associated with a statistically significant improvement from baseline (

Effect of oral TU on change from baseline in psychosexual function (PDQ) responses over the 4-month treatment period in trial I.

Effect of 6- and 12-month oral TU therapy on mean (±) changes from baseline in lean body mass and fat mass in trial I.
Consistent with the findings observed in the long-term study, patients in study trial II also exhibited statistically significant improvements from baseline (
Safety: long-term study (trial I)
No deaths occurred in response to daily oral TU exposure for 365 days. Two subjects in the oral TU group experienced an acute myocardial infarction; one in the setting of hospitalization for pneumonia. Neither event was considered to be causally related to oral TU in light of their respective cardiovascular (CV) disease histories. Treatment-emergent adverse events (TEAEs) considered by study investigators to be likely related to oral TU and T-gel exposure are summarized in Table 5. Not surprisingly, there was a higher incidence of gastrointestinal (GI) side effects associated with oral TU than topical T-gel. However, these GI effects were mild in nature and did not result in discontinuation of oral TU. Other adverse events are typical of TRT and to the extent the incidence was higher in the oral TU
Incidence of treatment-emergent adverse events related to long-term oral TU and T-gel therapy in study trial I.

PSA, prostate-specific antigen; TU, testosterone undecanoate.
The mean baseline hematocrit for oral TU patients was 44.1 ± 2.5%, and absolute changes after 4 and 12 months of treatment with oral TU were +2.1 ± 3.4% and +2.9 ± 3.9%, respectively, with both of these being statistically greater (
Prostate safety
We assessed prostate volume, PSA, and change in AUA/I-PSS category classification over the course of the study. Mean baseline prostate volume for oral TU was 29.3 ± 14.20 cc which was similar to the transdermal T-gel group mean baseline prostate volume of 30.7 ± 25.52 cc. Mean increase from baseline on day 365 was similar between oral TU and T-gel (2.97 ± 9.83 cc and 1.81 ± 26.40 cc, respectively) and not significantly different (
Cardiovascular biomarkers: hs-CRP, Lp-PLA2, Lp(a) and Apo-A1
The impact of oral TU therapy on CV biomarkers is summarized in Table 6. For simplicity, only baseline and day 365 data are included in this table although all biomarkers were also measured on days 90 and 180. Statistical analyses compared the absolute change in baseline over time between treatment groups using a repeated-measures analysis of variance. No statistically significant difference was observed for hs-CRP [with or without exclusion of values >10 mg/l (indicative of acute infection)] or Lp-PLA2. There was a statistically significant difference in Lp(a) response in favor or oral TU [i.e. reduced mean Lp(a)
Effects of long-term oral TU therapy on CV biomarkers
1

1 For simplicity, only baseline and day 365 data shown. Biomarkers also assessed on days 90 and 180.
2
Analysis after excluding all values >10 mg/l. Note: analysis with all values did not affect outcome (
3 Comparison of absolute change from baseline between treatment groups based on repeated (over all assay times) measures ANOVA model.
ANOVA, analysis of variance; CV, cardiovascular; hs-CRP, high-sensitivity C-reactive protein; Lp-PLA2, lipoprotein-associated phospholipase A2; TU, testosterone undecanoate.
Finally, mean systolic BP increased slightly over the course of the study in both treatment groups, such that by day 365, the mean increase from baseline was about 5 mmHg and 3 mmHg in patients who received oral TU or T-gel, respectively. Heart rates in both groups increased about one beat per minute over the study.
Safety: trial II
No deaths occurred during the study, and there were no drug-related serious adverse events. The overall incidence of TEAEs considered related to study drug occurred in 18.7% of patients in the oral TU group and in 14.5% of the topical T group (Table 7). The proportion of patients who prematurely discontinued from the study due to adverse events was 1.8% in each treatment group.
Treatment-emergent adverse events (TEAEs) considered related to T therapy in trial II.

T, testosterone; TU, testosterone undecanoate.
The TEAEs which occurred more frequently in oral TU patients than in the topical T group were increased hematocrit, hypertension, and decreased HDLc, reported in between 3% and 5% of patients. Each of these events was reported as mild or moderate in intensity, and none resulted in premature discontinuation from the study. Decreased HDLc events occurred at the higher oral TU doses (316 mg and 396 mg BID), whereas events of increased hematocrit and hypertension were not related to TU dose nor to T
As expected, based on the pharmacological actions of T, mean increases from baseline in hematocrit were observed in both treatment groups at each study visit but remained within the normal range in most men (97% oral TU; 100% topical T). Shifts from normal hematocrit values at baseline to above the normal range were observed in 3% of oral TU patients at the final visit, compared with none of the topical T patients.
No clinically significant changes in the liver function tests were observed in either treatment group. Two patients on oral TU experienced an inexplicable and transient elevation in ALT and AST to levels more than twice the and upper normal limit (UNL). Bilirubin levels remained normal in these patients. A third patient experienced a transient increase of AST (also more than twice UNL) during the study. Changes in lipid profiles were more pronounced among oral TU patients compared with topical T patients. Shifts from normal baseline to below the normal range for HDLc were observed in 28.9% of oral TU patients compared with 14.8% of topical T patients at the final visit; small and clinically insignificant reductions in LDLc were observed in both oral TU and topical T patients; there was no statistically significant difference in LDLc response between groups.
Clinic systolic BP increased from baseline to the end of the study (final visit) in both treatment groups [mean ± standard deviation (SD): oral TU, 2.8 ± 11.8 mm Hg; topical T, 1.8 ± 10.8 mm Hg], whereas diastolic blood pressure was essentially unchanged at the final visit for both groups. Measurement of BP with ABPM yielded greater mean increases from baseline to end of study (approximately 2 days prior to final PK visit) in average daytime (
Discussion
The new oral TU formulation evaluated in this study becomes the first oral T-ester pro-drug approved by US regulatory authorities, and only the second oral androgen approved for TRT use in the US, the last being methyltestosterone over 60 years ago. While both trials described herein achieved primary efficacy, only trial II achieved both efficacy endpoints relative to average and peak T response. This reflects a more refined dose-titration algorithm in the pivotal
The overall safety profile of oral TU was similar in both studies and reflected the well-recognized adverse-effect profile of T therapy as a class (e.g. decreased HDLc, increased hematocrit). A minor exception to this was the occurrence of a greater number of GI-associated side effects in oral TU patients (e.g. nausea, diarrhea, burping) compared with topical T, but these were transient, minor in severity and did not result in patients discontinuing oral TU. Of particular importance is the fact that oral TU was not associated with liver toxicity in either the long- or short-term study, a sharp contrast to methyltestosterone that has been historically associated with potentially serious hepatoxicity.
Oral TU was associated with a small but statistically significant increase in systolic BP
In contrast to the potential CV risk associated with elevated BP in patients treated with oral TU, we did not observe changes in other well-recognized CV risk biomarkers, namely, hs-CRP, Lp- PLA2, and Lp(a). And while we did observe a reduction in HDLc, this is a well-known effect of androgen therapy regardless of route of administration and thus oral TU therapy is not unique in producing this effect. Moreover, HDLc biology is complicated and data now suggest that its protective role in CV disease may be less important than the CV risk posed by increases in other lipid fractions, namely, LDLc and triglycerides.25,26 Thus, of greater clinical importance than reduced HDLc concentrations in oral TU patients was the lack of clinically meaningful changes in LDLc and triglyceride levels, since significant elevations in these lipid fractions (particularly LDLc) are unequivocal risk factors for CV disease.27,28
In conclusion, the new oral TU formulation described herein is a safe and effective means to treat hypogonadal men and has an overall profile consistent with the class of available TRT products. As such, this product represents a significant therapeutic advance for the treatment of appropriate hypogonadal men, particularly those in the US where, until now, an oral T treatment option was essentially unavailable. Oral TU administration is convenient, and twice-daily dosing with food (i.e. with breakfast and dinner and without the need for a high fat content) is a simple regimen that should promote better patient adherence over transdermal and injectable T products that dominate use among hypogonadal men but are associated with pain of administration (injected T-esters) or with transfer of T to women and children.
Footnotes
Acknowledgements
The authors gratefully acknowledge the investigators who participated in the conduct of trials I and II, most notably: Christina Wang, MD (Torrance, CA), John Amory, MD (Seattle, WA), Laurence Belkoff, DO (Cynwyd, PA); Gregg Flippo, MD (Birmingham, AL), Joe Blumenau, MD (Dallas, TX), Stephen Kulback, MD (Birmingham, AL), Marc Gittelman, MD (Aventura, FL), Jed Kaminetsky, MD (New York, NY), Stanon Honig, MD (New Haven, CT), Michael Zitzmann, MD (Munster, Germany), and Herman Behre, MD (Halle, Germany). We also acknowledge James Longstreth, PhD (Longstreth & Associates) and Nastya Kassir, PhD (Pharsight Consulting Services, Division of Cetara) for their PK modeling and simulation expertise; Janet Wittes, PhD (Statistics Collaborative, Inc) and Nestor Rohowsky, MS (IDCS, Inc.) for statistical analyses; Sharon Kraus (Kraus Studios) and Esther See for assistance with figures; and Theo Danoff, MD, PhD and Sandy Faulkner, RN (both Clarus Therapeutics, Inc.) for their oversight, data analyses and report preparation for trials I and II.
Conflict of interest statement
RSS served as principal investigator for studies CLAR-09007 (trial I) and CLAR-15012 (trial II) that were funded by Clarus Therapeutics, Inc. RSS has also received support from the NIH for testosterone studies in older men, and currently receives support from AbbVie as a study site for the Traverse Study of testosterone gel. He has also received support from NIH and NICHD for studies on male contraception where testosterone was a component of the experimental therapy. RED is employed by Clarus Therapeutics, Inc. (a private firm) and has an equity stake in the company.
Ethics statement
The studies described herein were conducted in compliance with all applicable country requirements for the conduct of clinical trials, including those outlined by the International Conference on Harmonization, Consolidated Guidelines on Good Clinical Practices, and the US Food and Drug Administration. Furthermore, both trials were approved by independent institutional review boards with jurisdiction over the respective study sites. Prior to participation in the trials, all patients signed informed consent documents.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
