Abstract
Use of testosterone replacement therapy (TRT) and anabolic–androgenic steroids (AAS) has increased over the last 20 years, coinciding with an increase in men presenting with infertility and hypogonadism. Both agents have a detrimental effect on spermatogenesis and pose a clinical challenge in the setting of hypogonadism and infertility. Adding to this challenge is the paucity of data describing recovery of spermatogenesis on stopping such agents. The unwanted systemic side effects of these agents have driven the development of novel agents such as selective androgen receptor modulators (SARMs). Data showing natural recovery of spermatogenesis following cessation of TRT are limited to observational studies. Largely, these have shown spontaneous recovery of spermatogenesis after cessation. Contemporary literature suggests the time frame for this recovery is highly variable and dependent on several factors including baseline testicular function, duration of drug use and age at cessation. In some men, drug cessation alone may not achieve spontaneous recovery, necessitating hormonal stimulation with selective oestrogen receptor modulators (SERMs)/gonadotropin therapy or even the need for assisted reproductive techniques. However, there are limited prospective randomized data on the role of hormonal stimulation in this clinical setting. The use of hormonal stimulation with agents such as gonadotropins, SERMs, aromatase inhibitors and assisted reproductive techniques should form part of the counselling process in this cohort of hypogonadal infertile men. Moreover, counselling men regarding the detrimental effects of TRT/AAS on fertility is very important, as is the need for robust randomized studies assessing the long-term effects of novel agents such as SARMs and the true efficacy of gonadotropins in promoting recovery of spermatogenesis.
Keywords
Introduction
The prevalence of male hypogonadism is increasing, 1 and it is projected that as many as 6.5 million men in the United States will have symptomatic hypogonadism by 2025. 2 This has led to a rise in the use of testosterone replacement therapy (TRT), with a 12-fold increase in worldwide sales of testosterone replacement preparations between 2000 and 2011, 3 with approximately 12% of men seeking TRT during their prime reproductive years. 4 Reflecting this trend, the expenditure on TRT in the United States quadrupled between 2007 (from $108 million) and 2016 (to over $400 million), 5 despite a slight reduction in TRT prescriptions in 2014 due to the U.S. Food and Drug Agency (FDA) issuing a safety communication regarding the use of TRT in men with cardiovascular disease. 6 Overall, this trend has coincided with more men presenting with infertility with a prior history of TRT use. A UK study showed 7% of men with a current or prior history of TRT use were requesting treatment for infertility. 6 In addition to this, the use of anabolic–androgenic steroids (AAS) has increased over the last 20–30 years largely among young men for muscle building and physique.7,8 While the detrimental impact of TRT and AAS on spermatogenesis is well recognized,7,9 awareness of these adverse effects among clinicians and patients is variable. Indeed, a survey of American urologists reported that 25% of participants would administer exogenous testosterone in cases of idiopathic male infertility. 10 Although this is a survey from 2012 and may not accurately reflect the contemporary practice of urologists, it is important to note such a perception has featured among trained urologists within the past decade. Hence, a closer evaluation of the potential adverse effects on male fertility along with better awareness of the effects of these agents and the search for more novel therapies for treating male hypogonadism are needed.
Conversely, it is also important to appreciate the rationale behind treatment of male hypogonadism and the proposed benefits TRT portends in men suffering from this condition. In this article, we will review the contemporary data for the pharmacotherapy of male hypogonadism, largely TRT and gonadotropins, and their effects on spermatogenesis. Moreover, we will describe the pharmacological and assisted reproductive strategies employed to address cases where there is a significant delay or failure of spermatogenesis to recover.
Background
To appreciate the rationale of the pharmacotherapy for male hypogonadism, it is important to understand the role of testosterone and other androgens in the maintenance of male reproductive physiology.
Physiological importance of androgens
The hypothalamic–pituitary–gonadal (HPG) axis
The level of intratesticular testosterone (ITT) is 50–100 times greater than serum testosterone and is achieved by stimulation of Leydig cells by luteinizing hormone (LH).11–13
Spermatocyte and spermatozoa maturation are heavily reliant on ITT and follicle-stimulating hormone (FSH). Moreover, FSH stimulates Sertoli cells, which are key in facilitating spermatogenesis.11,14 The hypothalamus produces gonadotropin-releasing hormone (GnRH), which stimulates the anterior pituitary gland to produce LH and FSH. This process is regulated by a negative feedback mechanism via the HPG axis. Both, serum testosterone and estradiol, provide negative feedback inhibition of the anterior pituitary and hypothalamus to inhibit the release of gonadotropins and GnRH, respectively. The use of exogenous testosterone and anabolic steroids suppresses male fertility by augmenting this negative feedback inhibition centrally. Consequently, there is inhibition of the pulsatile GnRH release: this leads to diminished FSH and LH, with subsequent decreased ITT. This results in hypogonadotropic hypogonadism (HH; Figure 1).

Hypothalamic–pituitary–gonadal axis.
Among the most common acquired causes of HH is previous use of TRT or AAS. Less common causes of HH are those where there is a primary defect in the synthesis and release of GnRH or gonadotropins from the hypothalamus or pituitary, respectively. This can be caused by ischaemic events, pituitary tumours or infections affecting the anterior pituitary gland/hypothalamus. 11
Mechanism of action of steroidal androgens
Androgens exert their direct effects via either the classical or the non-classical pathway. The classical pathway involves diffusion of testosterone across the phospholipid bilayer of cell membranes and binding to the cytoplasmic androgen receptor (AR), which in turn dimerizes with another AR. This enables the androgen–AR complex to enter the nucleus, where it regulates the transcription of specific genes associated with androgen response elements. The classical pathway typically takes 30–45 min to induce protein synthesis from the time of androgen stimulation. 16 The non-classical pathway involves an intracellular cascade of phosphorylation of tyrosine kinases, culminating in activation of a transcription factor called CREB. This results in transcription of various CREB-sensitive target genes and the effect occurs within seconds to minutes. 16 These two pathways are depicted in Figure 2.
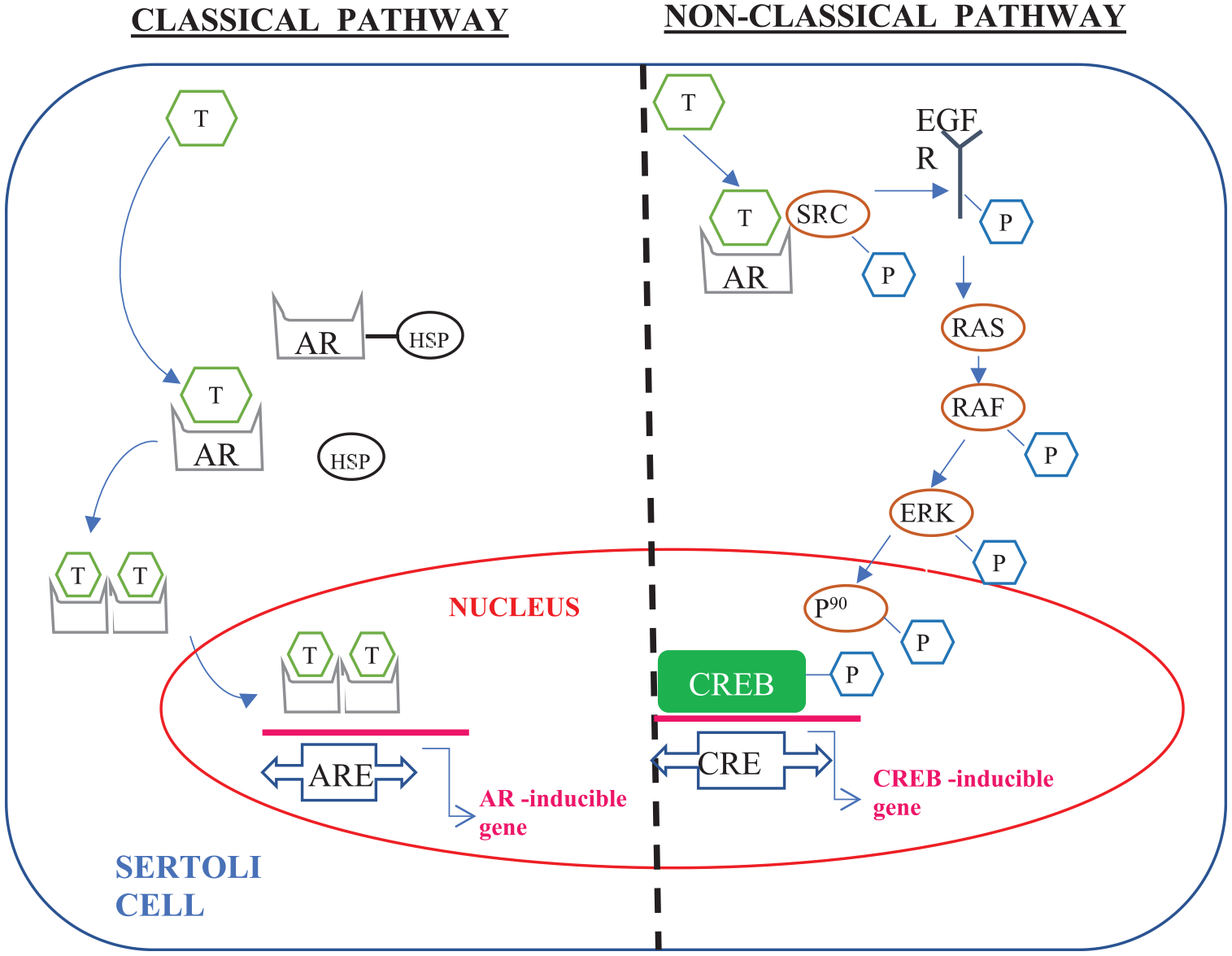
Classical pathway (left) and nonclassical pathway (right) of testosterone and other androgenic steroids acting on somatic cells.
Androgens and spermatogenesis
With regard to spermatogenesis, both the classical and the non-classical pathways are relevant for testosterone-mediated regulation. The adherence of maturing spermatids to the supporting Sertoli cells and release of fully mature spermatozoa from Sertoli cells into the lumen of the seminiferous tubules are mediated by the nonclassical signalling pathway.17,18
The role of androgens, in particular ITT, is critical in spermatogenesis. It is well established that the ITT concentration is 50- to 100-fold higher than serum testosterone.11–13 However, studies show that for spermatogenesis, germ cells are not the main testicular target for testosterone. 19 In fact, studies on male mice models have demonstrated that the absence of the AR in germ cells does not have a functional impact on fertility or sperm production.20,21 Moreover, germ cells in male mice lack expression of the AR. 22 Testosterone acts on other cellular and intercellular components of the testis in a paracrine and autocrine pattern such as the Sertoli, Leydig and peritubular myoid cells surrounding the seminiferous tubules. 17 The role of testosterone in this context includes maintaining the blood–testis barrier by stimulating transcription of proteins involved in the formation of tight junctions between supporting Sertoli cells; 23 stimulating meiosis of spermatocytes to form haploid spermatids;17,24 supporting the adherence of spermatids to Sertoli cells via a specific adherens–protein complex (ectoplasmic specialization) during the spermiation process;17,24 and the release of mature spermatozoa from the luminal surface of Sertoli cells into the lumen of seminiferous tubules. 25 These processes are all summarized in Figure 3.
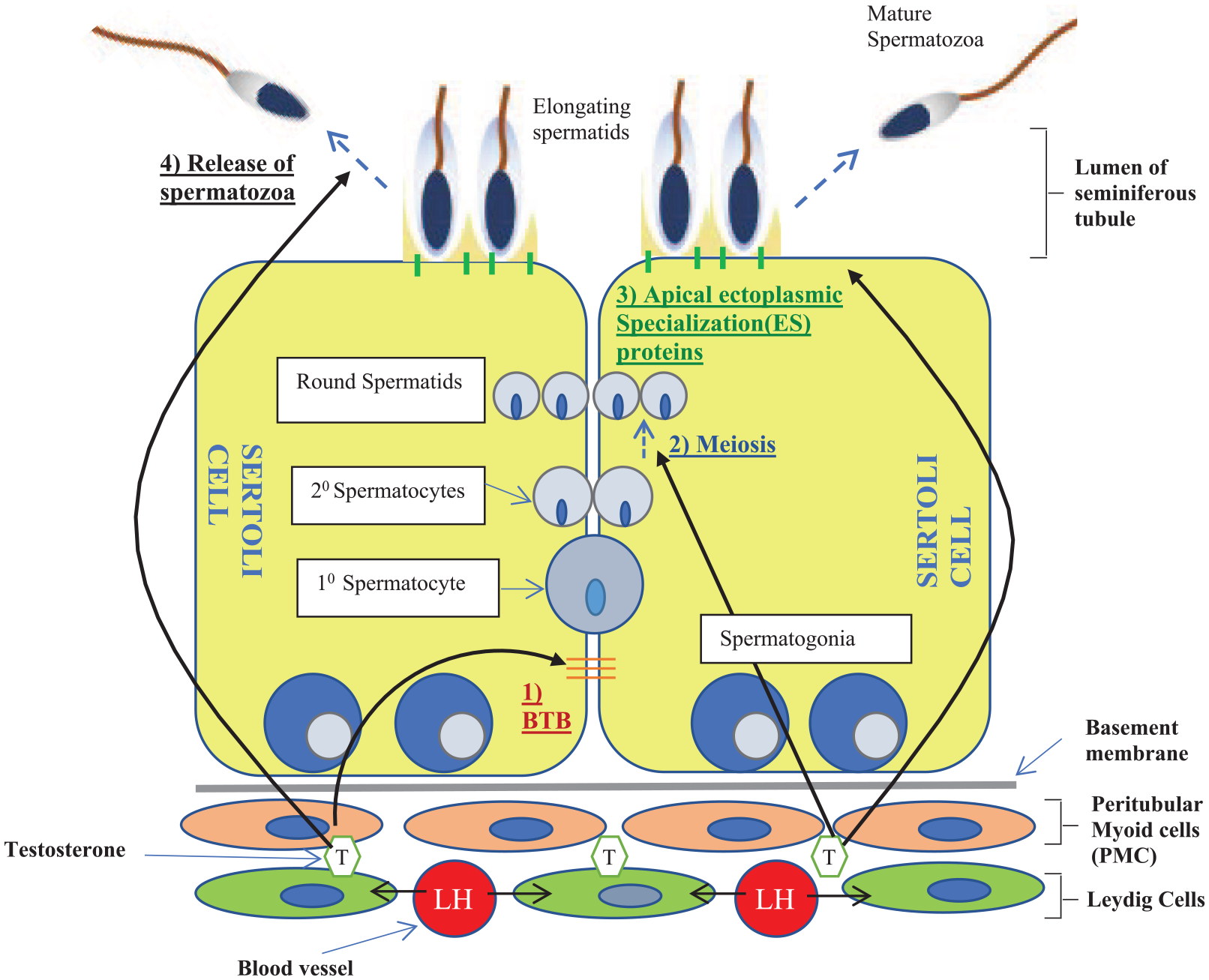
Cellular infrastructure of the seminiferous tubule and role of testosterone in spermatogenesis.
Figure 3 highlights the importance of ITT in spermatogenesis, but it is imperative to recognize this is distinct from circulating serum or exogenous testosterone. The use of exogenous testosterone (e.g. TRT/AAS) will suppress gonadotropin release by the anterior pituitary gland and thus decrease testosterone release by Leydig cells and Sertoli cell stimulation by FSH. 26 This consequently impairs spermatogenesis. The importance of ITT is corroborated by findings from early rodent studies showing that a fall in ITT concentration by more than 80% from normal values (equating to a concentration below 13 ng/ml) will result in an exponential fall in spermatozoa. 27 Further studies have demonstrated that spermatogenesis can be compromised below a higher ITT threshold of 20 ng/ml. 28
Dihydrotestosterone (DHT) is a more potent androgenic agonist of the AR compared with testosterone. 29 Testosterone is converted to DHT via the enzymatic action of 5-alpha reductase in the testes. The role of DHT in spermatogenesis is less well established but animal studies have demonstrated that inhibition of 5-alpha reductase in rats impairs the recovery of spermatogenesis from excess serum testosterone exposure. 30 Agents such as finasteride and dutasteride are 5-alpha reductase inhibitors (5ARIs) and are well-established pharmacotherapies for conditions such as benign prostatic hyperplasia (BPH) and androgenic alopecia (male-pattern baldness).
There are several randomized controlled trials in men comparing 5ARIs with placebo, but their results have shown differing effects on semen parameters dependent on dose of the 5ARI studied. Finasteride used for androgenic alopecia is prescribed at a lower dose (1 mg) than that for BPH (5 mg). Overstreet and colleagues 31 randomized men aged 19–41 years to 1 mg finasteride or placebo for 48 weeks, and reported no significant decrease in sperm concentration, motility or morphology between the two groups. However, another randomized placebo-controlled trial investigating a higher dose of finasteride (5 mg) and dutasteride (0.5 mg) found there was a reduction in sperm concentration and motility at 6 and 12 months with the 5ARIs (although the reduction at 12 months did not reach statistical significance). 32 It is unclear whether these are the direct effects of a reduced intratesticular DHT or whether the elevation in serum testosterone with 5ARIs causes a suppressive effect on spermatogenesis. 32 The authors also reported that restoration of spermatogenesis was seen when patients were reviewed 6 months after discontinuation of 5ARIs. 32 This is supported by prospective data showing that following cessation of finasteride there is an improvement in sperm parameters. 33 These findings clearly have implications on counselling men of reproductive potential contemplating treatment with 5ARIs.
Suppression and recovery of spermatogenesis
Exogenous testosterone/AAS results in an acquired form of HH. Other rarer causes of HH include structural/functional defects at the hypothalamus/pituitary (e.g. Kallmann syndrome), pan-hypopituitarism from tumours, cerebrovascular accidents, infection and radiation. Data on treatment of male infertility arising from these other causes of HH are scarce. Nevertheless, these data form the basis of understanding the effects of TRT/AAS, as well as the importance of gonadotropins in spermatogenesis. 7
The effects of exogenous testosterone and/or anabolic steroids on reducing ITT levels are well documented. 34 ITT concentration (usually 400–600 ng/g) is up to 100-fold higher than serum levels and must fall by more than 80% from its normal concentration before a decline in spermatogenesis occurs.12,13,28 Nevertheless, this threshold level of ITT is still higher than the circulating serum concentration. Interestingly, such reduced ITT concentrations would be sufficient to achieve AR stimulation in other peripheral tissues yet prove insufficient for spermatogenesis.
Therefore, treatment for male infertility resulting from HH is centred around increasing ITT. This is achieved by first addressing reversible causes or removing inhibitory agents, followed by administering agents which increase ITT. This includes human chorionic gonadotropin (HCG) and recombinant follicle-stimulating hormone (rFSH), both of which are gonadotropins acting directly on the testis, and selective oestrogen receptor modulators (SERMs) or aromatase inhibitors (AIs).
Recovery of spermatogenesis after TRT
Our understanding of the natural recovery of spermatogenesis following exogenous hormones is mainly derived from trials of men taking testosterone as a hormonal contraceptive. Liu and colleagues 35 performed a meta-analysis consisting of 30 trials investigating testosterone as a contraceptive and reported the likelihood of reaching a sperm concentration of 20 million/ml at 6, 12 and 24 months following cessation of testosterone was 67%, 90% and 100%, respectively. This is one of the most comprehensive datasets, analysing studies across 15 years 11990–2005] with a cohort of 1500 men. However, of importance, all these men were eugonadal at baseline before initiation of hormonal contraception, hence may be more likely to achieve restoration of spermatogenesis than hypogonadal men.
The findings from this meta-analysis are supported by a more recent meta-analysis of men with HH, in which 75% of men who were previously azoospermic had detection of sperm in the ejaculate with the aid of gonadotropin therapy. 36 The main benefit from gonadotropin therapy was seen in those with a pure form of secondary hypogonadism with postpubertal onset. Interestingly, prior use of TRT did not affect outcome. 36 Similarly, the role of gonadotropins, such as HCG and rFSH, in restoration of spermatogenesis has also been demonstrated in other studies.37,38 Two of the largest series including over 70 patients with acquired HH demonstrated successful induction of spermatogenesis in 90% 37 and pregnancy rates of 56% 38 following the use of HCG and FSH. However, limitations of these studies were the heterogeneous nature of patients (different aetiologies of HH, levels of baseline testicular function and semen parameters).
Anabolic–androgenic steroids
AAS cause male infertility by the same mechanism as described for TRT, as AAS are largely synthetic analogues of testosterone. 7 AAS facilitate muscle development and have been historically utilized to enhance success in sports. 39 For many other young men/non-athletes, this may be with a view to improving physique. These agents have previously been classified using a metric called ‘myotropic-androgenic’ (MA) index which describes the ability of the AAS to build muscle. For testosterone, the assigned MA index is 1:1. 40 Many of the more novel synthetic AAS have a much higher MA index (e.g. nandrolone: 11:1 and oxandrolone: 10:1). 41 This can theoretically alter the potency with which each AAS impacts on gonadal function. However, all AAS disrupt the HPG axis in the same way as testosterone – by increased negative feedback inhibition of gonadotropin release, hence suppressing spermatogenesis. This is supported by the fact that the ‘washout’ period after cessation of TRT is comparable with that for AAS and has been estimated to be 4 months.42,43 There is a paucity of data for infertility induced by AAS-mediated HH. 44
A recent study by Shankara-Narayana and colleagues 45 describes male gonadal/reproductive function in current or previous users of AAS. In this cross-sectional observational study, a total of 72 men were analysed – 41 current and 31 previous users of AAS. Moreover, in this study the authors compared reproductive function (hormonal and semen parameters) of AAS users with healthy nonusers. From the analysis of the prior AAS user cohort (mean time since last use: 300 days), there was no statistically significant difference between hormonal and semen parameters compared with those of healthy nonusers, suggesting full recovery of reproductive function after cessation of AAS. It was demonstrated that the recovery of sperm concentration after ceasing AAS took a mean of 10.4 months with a longer period of recovery for hormonal biomarkers of spermatogenesis (FSH: 19 months; serum inhibin B: 31 months). It was also concluded that a longer duration of AAS use was associated with slower recovery of sperm concentration and motility. This would be consistent with data on recovery of spermatogenesis after TRT use where duration of TRT strongly correlated with time to recovery. 35 This is further supported by a recent review concluding that in most cases with less than 1 year use of AAS, restoration of the HPG axis will occur within 1 year of cessation; 46 however, this can vary depending on dose, duration and type/regimen of AAS use. 44 The same review also suggested that in men who are infertile from prolonged AAS use (longer than 1 year), the use of clomiphene citrate (CC) or HCG may be beneficial. 46 This has been supported by the review by Rahnema and colleagues 44 which has reported on case reports/series describing successful use of SERMs (with/without HCG) to hasten recovery of semen parameters where a conservative approach of simply discontinuing the AAS was not effective.47–49 This review also included a report proposing a treatment algorithm incorporating the use of CC for managing AAS-induced hypogonadism. 50
Despite the aforementioned data, reports on recovery of spermatogenesis following AAS cessation are less well established and can be longer than that for TRT, with some reports documenting recovery after AAS use taking as long as 20 months. 43 This may seem paradoxical as most men taking AAS are likely to be eugonadal whereas those on TRT, by virtue of the need for TRT, are more likely to have pre-existing testicular dysfunction, which would confer a poor environment for spermatogenesis. Nevertheless, the longer recovery times following AAS may be explained by consumption of larger quantities, with doses amounting to more than 10–40 times what would be regarded physiological.48,49 Furthermore, the practice among bodybuilders of mixing various forms of AAS adds to the unpredictable nature of recovery after cessation.
The practice of ‘cycling’ among AAS abusers involves the use of medications like CC and other SERMs (see below) between courses of AAS as a way of mitigating the detrimental effects of AAS on fertility. 51 However, there have been no studies assessing the efficacy of such a regime. This not only adds to the obvious danger of adverse effects related to unmonitored use of such agents, but also increases the heterogeneity of data included in studies from this cohort of men.
Notwithstanding the negative impact on fertility, clearly the more global issue with the abuse of AAS is the elicit use of such agents by lay persons without careful monitoring by a clinician (c.f. TRT) and without a true understanding of the potential harms caused by AAS. Testament to this is the finding from a survey among prior AAS abusers that the biggest regret among these men was not understanding the repercussions of its use on fertility. 52 Although the current article specifically describes the adverse effects related to gonadal dysfunction, AAS can also cause cardiac, renal and hepatic complications.39,45
Selective androgen receptor modulators (SARMs)
SARMs are ligands of the AR hence mediate action in the same way as testosterone binding to the intra-cytoplasmic receptor and regulating nucleic transcription of target genes (Figure 4). However, they differ in their chemical composition and molecular structure to steroidal androgens. They were developed to offer many of the therapeutic anabolic effects of testosterone with a more tissue-specific mode of action, hence minimizing systemic androgenic effects. 53 Hence, their use has been investigated in the treatment of cancer-related cachexia, osteoporosis and low libido particularly in postmenopausal women. 54 SARMs have also been studied as a potential form of hormonal male contraception – a male version of ‘the pill’. 55 Chen and colleagues 55 demonstrated that the experimental SARM, C-6, induced a fall in sperm count by 25% compared with controls. A further study reported that a different SARM, S-23, induced azoospermia in rats. 56 Hence it is important to counsel men regarding the potential adverse effects SARMs may have on fertility when they are being contemplated for an indication other than contraception. Although none of the SARMs developed so far have been FDA approved for clinical use, the vast majority of experimental work in the last two decades has been performed to trial their use as fat-burning and muscle-building supplements for bodybuilders, as well as the other aforementioned therapeutic indications, 55 while offering a better side-effect profile than AAS. Like AAS, one would expect recovery of spermatogenesis after cessation of SARMs. This has been supported by a rodent study which investigated S-23 as a hormonal contraceptive and observed recovery of fertility in all patients within 4 months of discontinuing the SARM. 56

Mechanism of action of SARMs.
Factors affecting rate of recovery
The time needed for recovery of spermatogenesis following discontinuation of TRT/AAS is variable. The factors associated with a longer recovery time include a longer duration and higher dose of TRT/AAS use, Asian ethnicity, older age at initiation or cessation and baseline subfertility or poor testicular function prior to TRT/AAS use.7,40,45,57 Another parameter reported to predict success of recovery of spermatogenesis with aid of gonadotropic therapy is pretherapy testicular volume.37,38
Kohn and colleagues 57 demonstrated the age at cessation of TRT and duration of testosterone use both predicted time to recovery in men presenting with infertility after TRT use. The recovery of spermatogenesis was defined as a sperm concentration of more than 5 million motile sperms/ml. It was found that 70% of men overall achieved this target within 12 months. Approximately, 65% of men who were azoospermic during TRT managed to achieve recovery and this was more than 90% for those who were severely oligozoospermic during TRT. Age at cessation proved more of a consistent limitation on recovery compared with duration of TRT. 57
Results from a meta-analysis on inducing recovery of spermatogenesis with the aid of gonadotropins demonstrated better results for those with low/normal baseline serum gonadotropin levels. 36 Working on the premise that baseline gonadotropin levels are a reasonable surrogate for testicular function (raised gonadotropins would indicate primary testicular failure and poor intrinsic testicular function), the findings from this meta-analysis would support the inference that baseline testicular function is another variable affecting recovery of spermatogenesis following cessation of TRT/AAS. Similar suggestions have been made by others in the literature.58,59
By virtue of requiring TRT, hypogonadal men will have some degree of testicular failure – primary or secondary. A typical presentation of male infertility in the context of prior TRT use for hypogonadism is a Klinefelter syndrome patient. Indeed, the clear distinction between this group and those with HH is that Klinefelter syndrome patients initially present with hypergonadotropic hypogonadism consistent with primary testicular failure. Nevertheless, it represents a patient cohort in whom cessation of TRT alone may not be enough to promote recovery of spermatogenesis. Hence, these patients can be counselled for sperm cryopreservation before starting TRT if sperm is present in the ejaculate59,60 and azoospermic individuals should be counselled about the need to stop TRT before trying to conceive and the potential need for gonadotropin stimulation after cessation to promote spermatogenesis. The rationale for this may not entirely be to lift the inhibitory effects of TRT on FSH and LH (with FSH and LH already being elevated in cases of primary testicular failure), but it may be to stop the rise in estradiol via peripheral conversion of the exogenous testosterone, which is known to occur in men supplemented with TRT. 61 Estradiol may have direct effects on the testis to suppress spermatogenesis 62 in addition to a central effect of inhibiting FSH and LH release. Cessation of TRT alone may prove inadequate to produce sperm in the ejaculate and surgical sperm retrieval (SSR) may be necessary. Historically, an earlier (pubertal) sperm harvest was endorsed in azoospermic individuals prior to initiation of TRT. 63 However, more recent reviews of the literature have revealed a higher sperm retrieval rate at microdissection testicular sperm extraction after puberty in this cohort. 64 This higher rate of sperm retrieval is up to 50% 65 compared with 20% at the prepubertal stage. 64
Agents used to aid recovery of spermatogenesis
Although GnRH has been used in the treatment for Kallmann syndrome where the primary defect is failure of pulsatile GnRH release, this is rarely employed in most cases of acquired HH because it is expensive, requires an external pump for periodic release of GnRH and is not generalizable to all causes of HH as it requires an intact pituitary gland. 7 In the context of TRT/AAS-induced HH, more commonly used hormonal agents to stimulate spermatogenesis are HCG, rFSH, SERMs and AIs.
Studies evaluating the return of spermatogenesis in men with acquired HH specifically induced by TRT or AAS are scarce. Moreover, there is a paucity of studies evaluating the efficacy of gonadotropins in the treatment of HH. Wenker and colleagues 58 showed men on TRT with azoospermia or severe oligozoospermia (<1 million spermatozoa/ml) can have an improvement in sperm count to a mean of 22 million/ml within an average of 4.7 months after cessation of TRT when treated with HCG in combination with either AI (e.g. anastrozole), SERM (e.g. tamoxifen), or rFSH. Some series have shown that a combination of HCG and FSH can induce sperm in the ejaculate (>1.5 million/ml) from baseline azoospermic men with HH; however, this study excluded patients with a recent history (within last 5 weeks) of exogenous androgen use. 66 In support of these findings, Hsieh and colleagues 67 reported maintenance of semen parameters with use of HCG alongside concurrent TRT in hypogonadal men. Table 1 shows studies evaluating the efficacy of hormonal stimulation to promote spermatogenesis in the context of prior/concurrent TRT/AAS use.
Studies and series looking at role of gonadotropins, SERMs and AIs in recovery or maintenance of gonadal function in context of prior/concurrent TRT use.

AIs, aromatase inhibitors; CC, clomiphene citrate; EC, enclomiphene; FSH, follicle-stimulating hormone; HCG, human chorionic gonadotropin; HH, hypogonadotrophic hypogonadism; ITT, intratesticular testosterone; IU, international units; LH, luteinizing hormone; rFSH, recombinant follicle-stimulating hormone; SERM, selective oestrogen receptor modulator; T, testosterone; TRT, testosterone replacement therapy.
Gonadotropins: HCG and FSH
Produced by human placenta and isolated from the urine of pregnant women, HCG has a chemical structure very similar to that of LH and has been shown to act as an agonist of the LH receptor. 73 Therefore, HCG raises ITT and, in turn, promotes spermatogenesis. 74 Thus, not only can it be used as a therapy in infertile men but can also be used to treat symptoms of hypogonadism. 75 Hence, hypogonadal men wanting to preserve fertility should be considered for a trial of HCG. 75 Studies have shown that treatment with HCG at doses typically ranging between 1500 and 3000 international units (IU) administered intramuscularly or subcutaneously two to three times per week (alternate day dosing) alone or with rFSH can restore spermatogenesis in men with HH.58,76–78
The use of concurrent HCG stimulation in patients undergoing TRT has been shown to be effective at maintaining ITT by Coviello and colleagues. 72 This study measured ITT levels with incremental doses of HCG and used this as a surrogate marker of gonadal function. Hence, by observing maintenance of the ITT levels with HCG the suggestion was preserved spermatogenesis. This was later supported by Hsieh and colleagues 67 who demonstrated preserved semen parameters in hypogonadal men undergoing TRT with concurrent supplementation with low-dose HCG (500 IU). However, this was a retrospective study on a small sample of only 26 men. While the American Urological Association and the European Association of Urology guidelines advise against the use of TRT in men seeking fertility, this co-administration of HCG with TRT has been popularized as a strategy in managing hypogonadal men on TRT desiring fertility but not quite ready to discontinue TRT and forgo the other health benefits it confers.39,79
The analogue for FSH has been derived from the urine of postmenopausal women – referred to as human menopausal gonadotropin. However, this protein in its naturally occurring form is relatively inactive at the FSH receptor. This has led to the development of a more biologically active and specific form for the FSH receptor, primarily acting at the Sertoli cells. This is a refined form called rFSH.7,80 The molecular structures of LH, HCG and FSH are shown in Figure 5.

Molecular structure of LH, HCG and FSH – all have an identical alpha subunit with similarities in the beta-subunit between LH and HCG. All beta-subunits have varying degrees of N-linked glycosylations.
While rFSH has been used in HH men to promote spermatogenesis, it has proven unsuccessful in inducing or maintaining spermatogenesis when given alone with testosterone or alone in patients previously stimulated with a combination of HCG and FSH. 81 This reiterates the importance of maintaining ITT to permit spermatogenesis. A popular second-line regime is the addition of rFSH to HCG when inadequate semen parameters are obtained with HCG monotherapy.7,37,66 Hence most experts reserve rFSH as an addition to cases of unsuccessful HCG monotherapy.7,39,64,79 Two large series comprising 75 37 and 87 38 participants studied the use of a combination of HCG and rFSH in treating infertile men with HH and showed successful spermatogenesis in 90% 37 and pregnancy rates of 56%. 38
Selective oestrogen receptor modulators
SERMs were initially identified to stimulate ovulation in female animal models 82 and later in human subjects. 83 Their effect on increasing gonadotropin release suggested it may stimulate sperm production in men and this was supported in studies performed on oligozoospermic men using the largely centrally acting SERM CC.84,85 Both CC and tamoxifen are examples of nonsteroidal SERMs. As displayed in Figure 1, both act centrally by competitively binding to the oestrogen receptors at the anterior pituitary and hypothalamus thereby antagonizing the inhibitory effect oestrogen has on gonadotropin release. This results in an increased release of gonadotropins and hence stimulation of spermatogenesis. Oestrogen can also act within the testis on developing sperm to directly downregulate genes involved in spermatogenesis, causing apoptosis of spermatocytes. 62 Tamoxifen has both central and peripheral effects, hence it has been used for male infertility 86 among other indications such as treatment of gynaecomastia in men 87 and oestrogen receptor-sensitive breast cancer. 88
Despite CC being the most widely prescribed drug in the field of infertility, 39 it has not received FDA approval and remains an off-label therapy for use in male infertility. A recent review of studies evaluating its efficacy in male factor infertility has also shown mixed results with the authors concluding the need for more studies to further validate its use in this clinical setting. 89 Adverse effects reported with CC include headache, gynaecomastia, visual disturbances and mood instability. 89 Venous thromboembolism has also been reported in case studies of men using CC.90–92 However, a recent large retrospective analysis of 486 men on CC has found this risk to be less than 0.5%. 93 Although it is a rare side effect, it remains important to warn patients of before starting CC.
An often overlooked but important consideration is the reported paradoxical effect of SERMs on spermatogenesis. Pasqualotto and colleagues 94 reported three cases of severely oligozoospermic men rendered azoospermic after treatment with CC. The period of CC treatment prior to referral was a mean of 4.5 months. All three men were instructed to stop CC and were re-evaluated with semen analysis 3 months following cessation, where return of sperm in the ejaculate was observed in all men. 94 This paradoxical effect may be explained by the fact that CC is a racemic mixture of two isomers: enclomiphene (EC) and zuclomiphene. EC has a sole anti-estrogenic effect by antagonizing the oestrogen receptor hence having a pro-spermatogenic effect. However, zuclomiphene has been described as exhibiting estrogenic (agonist) effects thereby potentially opposing the action of EC. 95 This has formed the premise of isolating EC from the isomeric mixture of CC. Subsequent use of EC has been shown to concomitantly preserve sperm concentration while treating low testosterone levels compared with topical 1% testosterone gel in a study involving men with HH. 68 Furthermore, a proof-of-principle randomized study of 12 men previously treated with TRT showed all 7 men randomized to EC achieved restoration of spermatogenesis at 3 months and 6 men at 6 months, with all men having a sperm concentration of more than 75 million/ml. Whereas none of the five men randomized to testosterone gel managed to reach a sperm concentration of more than 20 million/ml at 3 months. Only EC was associated with increased sperm counts after 6 months follow up. 69
Like this study by Kaminetsky and colleagues, 69 there are limited studies investigating the ability of SERMs to restore spermatogenesis after TRT/AAS use. The data available are limited to small case series treating men with adult-onset HH with either azoo- or severe oligozoospermia.69,71 Whitten and colleagues 71 studied four men with adult-onset HH treated with 50 mg CC three times per week which produced an increase in sperm concentration and testosterone levels in three out of the four men. However, the other azoospermic patient in the same series who did not respond to the initial monotherapy with CC was subsequently treated with combination of HCG and rFSH and this yielded a normal sperm concentration and successful pregnancy with intra-uterine insemination. Wenker and colleagues 58 also studied SERMs in conjunction with gonadotropins in men taking TRT and showed over 95% had return of sperm in the ejaculate for azoospermic men or improvement in sperm concentration in those with severe oligozoospermia.
Aromatase inhibitors
AIs have also been used as empirical off-licence therapy in idiopathic male infertility and have not received FDA approval for this indication. The theorized benefit this agent confers is reducing the peripheral conversion of testosterone to oestrogen by inhibiting the enzyme aromatase, present in testes, liver, brain and adipose tissue. 7 The lower circulating estradiol levels dampen the negative feedback on the HPG axis, thus stimulating gonadotropin release.96,97 These agents are broadly divided into steroidal (e.g. testolactone) and nonsteroidal (e.g. anastrozole and letrozole), with the former causing irreversible inhibition of aromatase and latter mediating reversible inhibition. 98 The key metric to ascertain when assessing the potential benefit of AIs in male infertility is the serum testosterone/oestrogen (T/E) ratio, as studies have shown the greatest improvement in semen parameters with AIs are for those with a T/E ratio <10.99–101
While AIs have been shown by various studies to improve semen parameters in men with idiopathic infertility100,101 or hypogonadism, 102 their use in the context of HH such as with previous TRT/AAS use is very limited. As with the other agents used to hasten the recovery of spermatogenesis after TRT/AAS use, the data used to support use of AIs in this context are from retrospective series and simply an extrapolation from studies on men with acquired HH seeking treatment for infertility. The previously mentioned study by Wenker and colleagues 58 probably constitutes a group of men which most closely matches such a cohort and even in this study AIs were used as an adjunctive treatment to HCG rather than monotherapy. A prospective non-placebo-controlled randomized study published in the same year also compared efficacy of AI and CC with each being used as monotherapy for improving semen parameters in hypogonadal men. While serum testosterone levels were significantly higher in the CC group and T/E was higher in the AI group, there was no significant improvement in sperm concentration in either group, and no difference in semen parameters between both groups. 103 These two studies portray the weak and conflicting evidence base behind the use of AI in the setting of infertility from TRT/AAS-induced HH, and again, beckons larger and more robust study protocols.
Discussion
The growing prevalence of symptomatic male hypogonadism 104 and increasing number of prescriptions for TRT 4 have raised concerns for male fertility.7,9 Despite the well-documented detrimental impact of these drugs on fertility, there seems to be a lack of awareness of this effect among those using them and, in some cases, clinicians with the ability to prescribe them. 10 Alongside this, there is increasing use of AAS among young men seeking to improve muscle build and perceived physique.
Despite the relatively scant literature, natural recovery of spermatogenesis after TRT and AAS has been shown to occur in the vast majority of cases. 7 However, as a general rule men seeking conception should avoid TRT/AAS, or if they are taking such agents, they must be discontinued immediately. Following this, the recovery of spermatogenesis may be a slow process particularly in previously hypogonadal men with poor baseline testicular function, long history of TRT use or older age at cessation.7,40,57 This cohort of men or those with older female partners wanting to conceive as soon as possible will be suitable candidates for hormonal stimulation to speed up recovery of spermatogenesis. HCG, being an analogue of LH, has the theoretical benefit of directly stimulating the testicle to raise ITT and hence sperm maturation. Whereas SERMs largely rely on inhibition of the negative feedback effect of circulating oestrogen on the anterior pituitary and hence stimulating gonadotropin release. CC is the most widely prescribed agent for this use. 39 While the use of agents such as HCG and SERMs like CC has been well established to promote ovulation in women, their use in men to promote spermatogenesis is an off-licence treatment. Although there is some evidence demonstrating their efficacy in men, this largely stems from case series and non-randomized data.
While these agents have been used to aid recovery of spermatogenesis in men taking TRT or those previously having taken TRT/AAS, their use in this context is empirical. Although the theoretical basis is convincing, the data suggesting some efficacy of these agents in infertile men are mostly anecdotal and based on small retrospective series. There has yet to be a prospective randomized placebo-controlled trial that addresses the question of whether HCG ± rFSH or SERMs can effectively stimulate the recovery of spermatogenesis in this setting. Nevertheless, with the growing proportion of men who may not achieve spontaneous recovery after cessation of TRT/ASS, it is important for urologists and andrologists to counsel men thoroughly at the outset when contemplating TRT and be aware of these therapies, albeit with limited evidence base. Other strategies in those wishing to preserve fertility may be discussed and should include treatment with HCG ± rFSH in hypogonadal men (instead of TRT) or sperm cryopreservation in those with viable sperm in the ejaculate. Those who remain azoospermic after cessation of TRT/AAS and have older female partners with limited ovarian reserve may not be agreeable to initiate/complete a long trial of hormonal stimulation due to the limited time available for such couples to conceive. In such cases, consideration must be given to offering surgical sperm retreival (SSR) with a view to assisted reproductive techniques such as in vitro fertilization and intra-cytoplasmic sperm injection (IVF/ICSI).
Conclusion
The majority of men with a previous history of TRT and AAS use presenting with infertility will achieve recovery of spermatogenesis; however, this is not guaranteed and successful recovery after discontinuation of TRT/AAS is dependent on a number of patient-specific and treatment-specific factors. In those who do not achieve recovery of their fertility on cessation, additional agents such HCG ± rFSH, SERMs or AIs can be trialled. These men must be counselled that should this fail to yield sperm in the ejaculate, they may need to proceed with SSR. Thus, there is some merit in discussing cryopreservation of sperm from the ejaculate before embarking on TRT/AAS, which may later preclude the need for SSR. In cases where patients have not had sperm cryopreserved previously and become azoospermic following TRT/AAS, they would require attempts at SSR with a view to implementing assisted reproductive techniques (e.g. IVF/ICSI) and hence should also form a key part of the informed counselling process. Furthermore, more robust randomized placebo-controlled data are needed to ascertain the true efficacy of hormonal agents in aiding recovery of spermatogenesis after TRT/AAS as well as more novel agents such as SARMs.
Footnotes
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Author contributions
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
Availability of data and materials
All data presented are available to view in the published article or the individual referenced/cited studies, in which the data was originally reported.
