Abstract
Introduction:
Idiopathic retroperitoneal fibrosis (IRF) is a rare disease characterized by a fibrotic reaction that affects retroperitoneal organs, especially the urinary tract. In this review we analyze the current imaging techniques, morphological characteristics, clinical aspects and therapeutic aspects of idiopathic retroperitoneal disease.
Methods:
A PubMed search was conducted in December 2013 to find original articles, bibliographic reviews and series reports published in the past 15 years on idiopathic retroperitoneal fibrosis, its management and outcomes by combining terms like retroperitoneal fibrosis, periaortitis, treatment and autoimmune. A total of 89 articles were included in this review that referred strictly to IRF. We analyzed the imaging tools used for diagnostic and the decision making protocol used by physicians in the management of IRF.
Results:
A computerized tomography (CT) scan represents the most commonly used imaging technique for diagnosis. Magnetic resonance imaging (MRI) is unable to differentiate more accurately between benign and malignant retroperitoneal fibrosis (RF) than a CT scan. Biopsy remains the most reliable diagnostic tool for IRF. However, the histological characteristics of IRF are not yet well-defined and the protocol for biopsy is not standardized in terms of template, number of biopsies and the immunohistochemical panel needed for positive diagnosis. The most common treatment reported is corticosteroid therapy alone or in combination with other immunosuppressants, whereas surgical treatment is reserved for severe cases. Indwelling ureteric stents represent the most common procedure for renal drainage, but their efficacy is questionable. Open ureterolysis remains the gold standard for surgical treatment, but its purpose is only to resolve the ureteric obstruction, not to treat the retroperitoneal fibrosis. Laparoscopic and robotic approaches have been reported to be feasible, but no prospective, comparative trials have been performed due to the rarity of the disease. Surgical technique is not standardized and the outcome of the treatment only evaluates the recovery of the renal function.
Conclusions:
The imaging procedures available today are unable to accurately differentiate between idiopathic and malignant RF. A biopsy is mandatory to confirm the diagnosis, but there is no consensus regarding the template, timing and number of biopsies needed to exclude malignancy. Open ureterolysis represents the main surgical treatment for cases with severe IRF, and laparoscopic or robotic approach may be an option in selected cases. The recovery of the renal function is a surrogate for evaluating the success of the treatment. More clinical studies are needed in order standardize the protocol for diagnostic, treatment and follow up after medical or surgical management.
Keywords
Introduction
Retroperitoneal fibrosis (RF) is defined as a diffuse or localized fibroblastic proliferation accompanied by chronic inflammatory infiltrates, causing ureteral obstruction and compression to the vascular structures (abdominal aorta, inferior vena cava). Most cases are idiopathic, but can be secondary to infections, abdominal surgery, drugs or malignant diseases [Vaglio et al. 2006].
Ureteral entrapment represents the hallmark of this disease and the main reason for admittance in a urologic department; excretory urography or retrograde pyelography were used to outline the site and severity of obstruction [Vivas et al. 2000; Monev, 2002]. Nowadays, cross-sectional techniques, computerized tomography (CT) or magnetic resonance imaging (MRI) of the abdomen are used for diagnosis and evaluation of the extent and relationship to adjacent structures and organs [Lalli, 1977; Cronin et al. 2008a]. The role of nuclear scintigraphy, especially of 18F fluorodeoxyglucose (FDG) positron emission tomography (PET) has also been described, but its role in the management of idiopathic retroperitoneal fibrosis (IRF) has yet to be established [Drieskens et al. 2002; Vaglio et al. 2005].
Due to the poor prognosis of malignant RF [Johnson et al. 1995], its diagnosis is paramount in order to receive the appropriate treatment.
Despite the comprehensive evaluation of RF location, extent and effect on adjacent organs and vascular structures, the attempts to define imaging (CT/MRI) characteristics that may allow confident differentiation of benign from malignant RF have proven futile, thus making the biopsy (percutaneous or surgical) the only method to clearly define the diagnosis.
In this review we address the following issues:
analysis of current imaging techniques, highlighting the particular features of each type of RF;
clinical and therapeutic aspects of idiopathic retroperitoneal disease.
Evidence acquisition
A nonsystematic review search was conducted in December 2013 to find original articles, bibliographic reviews and series reports published in the last 15 years regarding IRF, its management and outcomes by combining terms such as ‘idiopathic retroperitoneal fibrosis’, ‘periaortitis’, ‘biopsy’, ‘ureterolysis’, and ‘diagnosis, medical and surgical treatment’. Figure 1 shows the flowchart of this nonsystematic review of the literature. The literature on certain subtopics was limited to case reports and small case series with fewer than five cases, and was included in the review due to the lack of large series on the specified research areas. A total of 89 articles were included in the review. We analyzed the imaging tools used for diagnostic and the decision making protocol in the management of IRF.
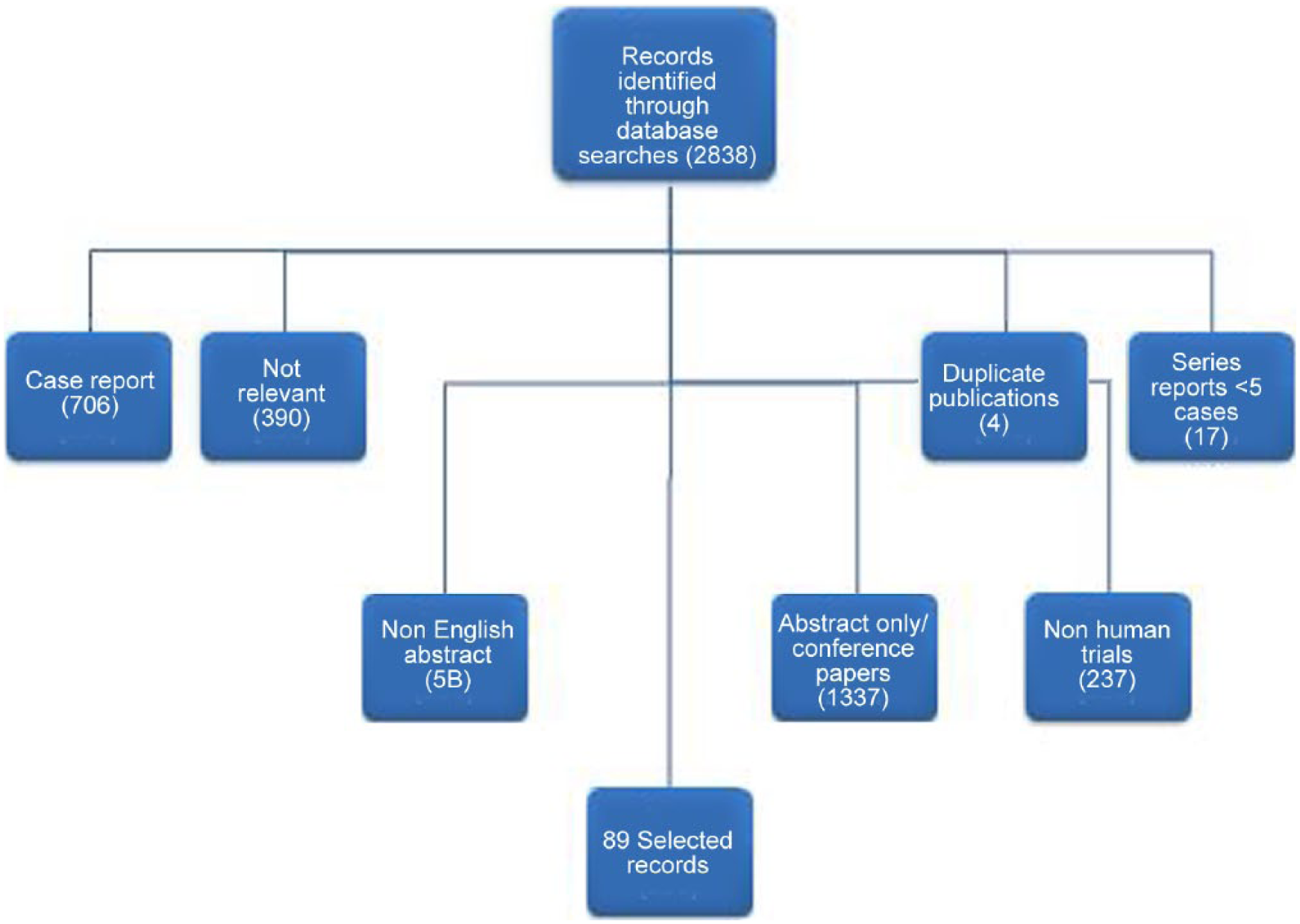
Flowchart of the nonsystematic review.
Imaging features of IRF
Imaging plays an important role in the diagnosis of retroperitoneal diseases and should be performed in patients with unexplained, persistent back, flank or abdominal pain [Vaglio et al. 2006]. Although ultrasound is the first imaging technique used, its findings are not specific, like various degrees of ureterohydronephrosis and in some cases hypoechoic or anechoic, irregularly contoured retroperitoneal mass [Rubenstein et al. 1986].
CT
Contrast-enhanced CT represents the most common cross-sectional technique used in the diagnosis of retroperitoneal masses. Despite the abundance of literature regarding its use in diagnosis and follow up, no sensitivity or accuracy in depicting IRF has been published, since most of the articles are case presentations or small series. IRF is most often described as a paraspinal, irregular retroperitoneal mass, isodense to surrounding muscle [George et al. 2013]. It is usually centered at the level of the fourth and fifth lumbar vertebrae and spreads down to the pelvis; rarely, it extends into the mesentery. Its evolution is centrifugal, with involvement of the ureters and in some cases over the diaphragm, causing fibrous mediastinitis (Figure 2). The major blood vessels (abdominal aorta and inferior vena cava) are usually not displaced anteriorly by the fibrotic process, in contrast to malignant RF, which presents a more cephalad distribution and displays a mass effect on the aorta; however, this rule cannot be used as a sign of malignancy [Brun et al. 1981; Feinstein et al. 1981; Rubenstein et al. 1986; Bullock, 1988; Mulligan et al. 1989; Amis, 1991].

(a) Axial contrast enhanced CT scan: low attenuation mass anterior and lateral to aorta, without anterior displacement of either aorta or inferior vena cava. Important medialization of right ureter. Single right kidney with nephrostomy tube present. (b) Coronar reconstruction of native CT scan: Retroperitoneal mass encasing both ureters. Bilateral JJ stents present with significant medialization of left ureter.
Despite these typical characteristics, Brun and colleagues reported that a third of cases with proven IRF (surgical or by biopsy) did not present any alterations on CT [Brun et al. 1981]. Different authors have proposed a degree of enhancement of 20–60 Hounsfield units (HU) in order to classify IRF as acute (with vivid enhancement after contrast administration) or chronic (with little or no enhancement), or to use soft tissue distribution to make a differential diagnosis between benign and malignant RF [Brun et al. 1981; Feinstein et al. 1981; Bullock, 1988; Mulligan et al. 1989; Brooks, 1990; Amis, 1991] (See Table 1). However, many patients present at first admission with obstructive renal failure due to ureteric involvement, prohibiting the usage of contrast agents and limiting the usage of these findings in clinical practice.
Utility of imaging techniques in the diagnosis of idiopathic retroperitoneal fibrosis.

CI, confidence interval; CT, computerized tomography; FDG PET, fluorodeoxyglucose positron emission tomography; MRI, magnetic resonance imaging; NPV, negative predictive value; PPV, positive predictive value; SPECT, single photon emission computed tomography.
A secondary contrast imaging evaluation is mandatory after recovery of the renal function to accurately assess the extent of the disease, but the timing is unclear and most of the patients are under medical treatment after ureteric stenting, making the results of the second imaging evaluation questionable.
MRI
Even though MRI is considered to have a higher spatial resolution and accuracy in delineating the retroperitoneal structures, its ability to differentiate between benign and malignant RF is similar to that of a CT scan [Arrive et al. 1989; Yuh et al. 1989; Brooks et al. 1990]. MRI brings an excellent assessment of the extent of the fibrotic process, its relationship with the adjacent retroperitoneal organs and with no need of contrast agents (Figure 3); it can be used in patients with moderate degree of renal failure and for guided biopsy [Zangos et al. 2006].

T1 weighted coronar fast spoiled gradient (FSPGR): low-signal-intensity para-aortic mass with no anterior displacement of the aorta or the inferior vena cava, and little soft-tissue enhancement of the fibrotic mass.
IRF has similar signal findings to other fibrotic process, with a soft tissue enhancement close to that found on CT scans. The IRF’s T2 signal varies as a reflection of the amount of inflammation process present in the tissue (active RF: high T2 signal; chronic: little or no signal). These characteristics were used as marker of favorable response to medical treatment by a number of authors as a decline of signal is to be expected, accompanied by a decrease in uptake of gadolinium in chronic fibrotic tissue [Yuh et al. 1989; Burn et al. 2002]. However, the signal intensity, degree of contrast enhancement and presence or absence of soft tissue edema are nonspecific [Cronin et al. 2008]. Secondly, MRI is unavailable for emergency use in most countries and the high cost limits its use in clinical practice. Also, caution must be used when using gadolinium-based contrast agents among patients with advanced kidney disease since these substances are associated with the development of nephrogenic systemic fibrosis [Agarwal et al. 2009].
Nuclear imaging
18F-FDG and 67Ga uptakes are similar; they mirror the metabolic activity inside different tissues, irrespective of their origin, benign or malignant, and thus limiting the use of PET as primary imaging tool for diagnosis. Several studies have shown the usage of FDG PET and single photon emission computed tomography (SPECT) in follow up after initiation of medical treatment and detection of occult neoplasias, recurrent disease or multifocal fibrosclerosis lesions that can cause secondary RF, with promising results [Hillebrand et al. 1996; Chander et al. 2002; Drieskens et al. 2002; Vaglio et al. 2003, 2005; Vaglio and Brun, 2005; Treglia et al. 2012]. However, due to the heterogeneity among the various studies for PET/SPECT analysis and inclusion criteria, a valid conclusion cannot be drawn regarding the utility of nuclear imaging in the management of IRF.
Management
Clinical evaluation
The diagnostic of IRF requires a high index of suspicion in the presence of a constant, nonlocalized pain in the back, flank or abdomen unchanged by posture. Systemic symptoms like fever, weight loss, anorexia, Raynaud’s phenomenon are less frequent. Ureteral colic, hematuria, lower extremity edema with venous thromboembolism, hydrocele, jaundice or spinal cord suppression have been reported in cases with severe IRF. Various degrees of unilateral or bilateral hydronephrosis and high blood urea nitrogen (BUN) levels are the earliest and most common findings for organ involvement of IRF [Vivas et al. 2000; Monev, 2002; Van Bommel, 2002; Vaglio et al. 2006].
Lab results
High erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) levels are present in approximately 80–90% of patients. Various degrees of anemia and hypergammaglobulinemia are also found in patients with mild IRF. Increased serum creatinine and BUN levels are present in cases with ureteral involvement [Ormond, 1948; Vivas et al. 2000; Van Bommel, 2002].
In cases of secondary IRF, the lab results are variable, mimicking the primary disease (high thyroid-stimulating hormone (TSH) with low levels of T4 and T3 in cases with Hashimoto thyroiditis, lymphocytosis in non-Hodgkin lymphoma or increased levels of autoantibodies – antinuclear antibody (ANA), smooth muscle antibody (SMA), extractable nuclear antigens (ENA), Rheumatoid factor (RF), etc.) in auto-immune diseases) [Nathwani et al. 1986; Vivas et al. 2000; Monev, 2002; Van Bommel, 2002; Vaglio et al. 2006].
Biopsy
The current imaging techniques cannot differentiate accurately between idiopathic and malignant retroperitoneal fibrosis, making a biopsy mandatory in order to confirm the diagnosis [Kottra et al. 1996; Drake et al. 1998; Drieskens et al. 2002; Vaglio et al. 2006]. However, there are several caveats regarding the clinical implications of a positive biopsy since the histological characteristics of IRF are not yet well-defined and the biopsy protocol is not standardized in terms of template, number of biopsies and immunohistochemical panel needed [Hellstrom et al. 1966; Mitchinson, 1970, 1984; Hughes and Buckley, 1993; Vaglio et al. 2003; Duvic et al. 2004]. These limitations of biopsy prohibit its use and most patients with mild symptoms undergo medical treatment without any histological evidence of IRF. Patients who undergo a biopsy (CT/surgical) are mainly those with limited response or failure to medical treatment.
The choice of biopsy technique depends on several factors, i.e. the size and anatomic location of the fibrotic mass, availability, type of percutaneous approach (CT or MRI) and the radiologist’s level of experience, number of biopsies needed, superficial or deep biopsies, risk of malignancy and, of course, the patient’s preference.
There have been reported open/laparoscopic/robotic biopsies, CT guided, transcaval retroperitoneal biopsy and fine needle aspiration cytology (FNAC), each with different results [Higgins and Aber, 1990; Pfammatter et al. 1998; Dash et al. 1999; Heidenreich et al. 2000; Mufarrij and Stifelman, 2006]. The largest series are presented in Table 2. The percutaneous approach is considered less reliable due to the small amount of tissue sampled compared with the other techniques [Bullock, 1988].
Biopsy results for the IRF mass using different approaches.

CT, computerized tomography; IRF, idiopathic retroperitoneal fibrosis; NA, not applicable.
Surgical biopsy remains the gold standard for diagnostic due to the possibility of multiple, deep biopsies that allow us to accurately diagnose malignant RF in which metastatic cell are usually dispersed and located profoundly in the fibrotic mass [Mufarrij and Stifelman, 2006; Cronin et al. 2008; Mufarrij et al. 2008].
A difficult dilemma appears in interpreting the negative results after biopsy, since a negative biopsy result does not rule out an underlying neoplasia. Several authors consider that in cases of negative results after biopsy and/or suboptimal results to medical treatment, surgical exploration is needed and further search for occult malignancies [Koep and Zuidema, 1977; Dash et al. 1999].
Medical treatment of IRF
The aim of medical therapy in IRF is to preserve the renal function and relieve symptoms given by other organ involvement. First reported in 1958 by Cosbie and Tinckler [Cosbie and Tinckler, 1958], corticosteroid therapy (CST) still represents the most popular therapy, either alone or in combination with other immunosuppressants such as tamoxifen, azathioprine, cyclophosphamide, mycophenolate mofetil, cyclosporine or progesterone (See Table 3).
Contemporary series regarding the medical treatment of IRF.
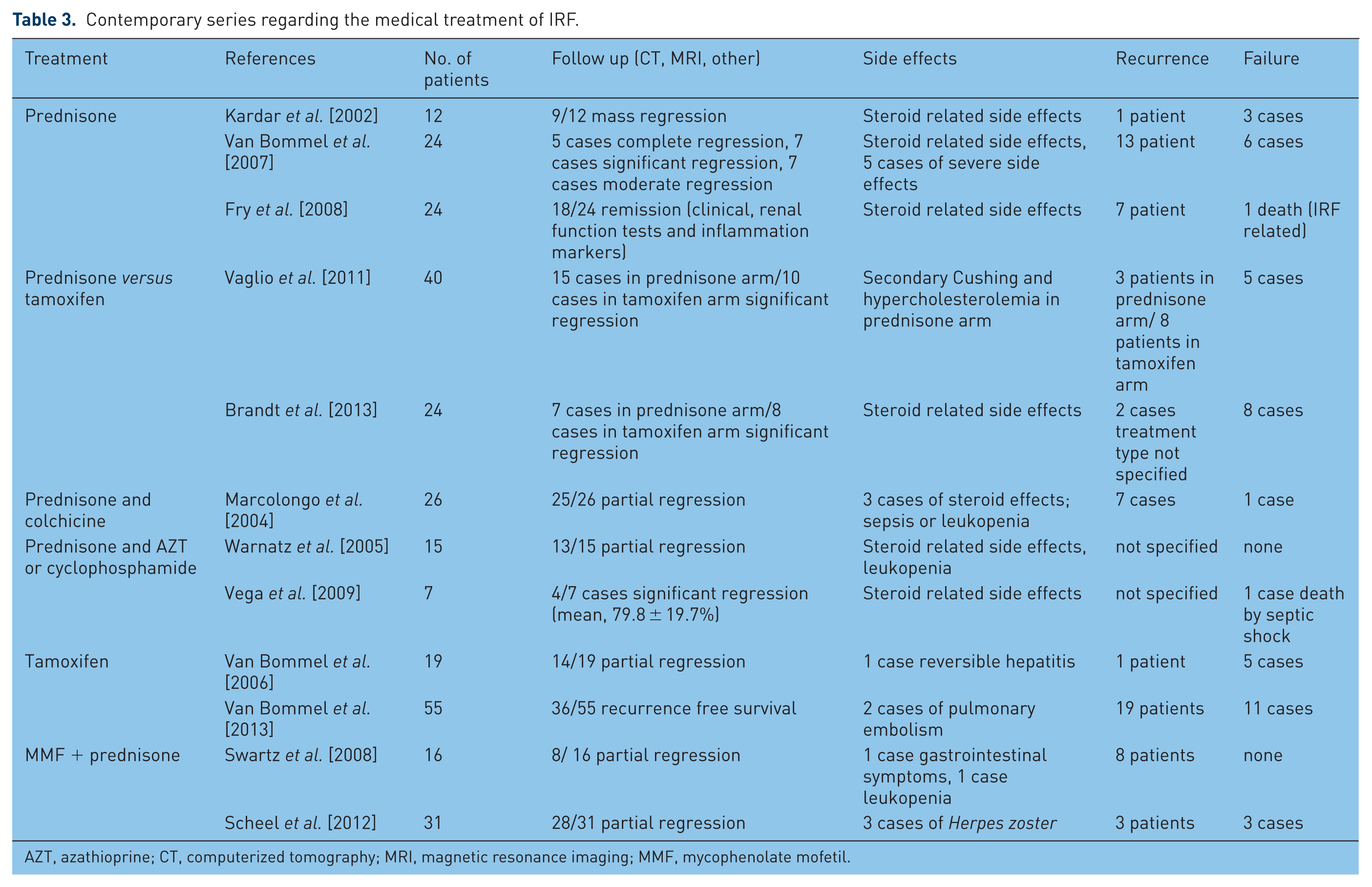
AZT, azathioprine; CT, computerized tomography; MRI, magnetic resonance imaging; MMF, mycophenolate mofetil.
The most frequently used protocol includes prednisolone administered 40–60 mg/day tapered to 10 mg/day in a time interval of 2–3 months followed by a gradually discontinuation after 12–24 months. In 2002, van Bommel conducted a study on 147 patients, which showed an improvement in 83% and a recurrence rate of 16% after discontinuation of CST [Van Bommel, 2002].
There has also been reported the use of a ‘pulse’ dose regimen by giving methylprednisolone 1 g/day intravenously for 3 three days combined with azathioprine, with similar results [Harreby et al. 1994].
Another immunosuppressant drug used in the treatment of IRF is tamoxifen, a nonsteroidal estrogen receptor modulator. The mechanism of action of tamoxifen is unknown, although no estrogen receptors have ever been identified in the fibrous tissue. Some authors advocate that it increases production and secretion of transforming growth factor β in fibroblasts and immune cells within the fibrous tissue and therefore decreasing the size of the fibrous plaque [Spillane and Whitman, 1995].
Tamoxifen is used in different regimens alone or in combination with corticosteroids in doses that vary between 10 and 40 mg for 6 months up to 3 years. Vaglio and colleagues conducted an open label, randomized, controlled trial on 40 patients in whom they compared prednisone with tamoxifen and analyzed their efficacy and adverse effects. After 18 months of follow up, the authors reported a relapse rate of 17% for prednisone and 50% for tamoxifen. The adverse effects, however, were significantly more severe in the prednisone group, such as therapy-related Cushing syndrome and grade 2 hypercholesterolemia. Even though tamoxifen appears to be a safer treatment option for IRF, the risk of thromboembolism and ovarian cancer still have to be taken into account when starting the treatment [Vaglio et al. 2011].
Azathioprine (AZT) represents an immunosuppressant drug used in organ transplantation to prevent acute organ reject [Cogan and Fastrez, 1985]. In 2006, Moroni published the results of a long-term follow up of 17 patients with IRF who were divided into three groups: CST+ surgery (group 1), CST + AZT (group 2) and surgery + tamoxifen (group 3). The response rates were 100% for the first two groups and 83% for group 3, but the recurrence rates reported were 40% for group 1, 16% for group 2 and no recurrence in group 3 [Moroni et al. 2006]. These results have shown that a multimodal treatment represents the best approach for severe cases, but there is a need for well conducted studies in order to establish the value of this therapy.
Mycophenolate mofetil (MMF) is another alternative in inducing immunosuppression alone or in association with CST. Adler reported a series of nine patients who received a regimen of MMF of 2 g/day and prednisone 1 mg/kg. All patients in this series had a radiological regression as well as a biological regression within 6 months of starting the treatment. CST was discontinued after a mean time of 7 months and MMF after a mean time of 27 months, with no recurrences after a mean follow-up of 55 months [Adler et al. 2008].
Marzano and colleagues described a case of IRF that showed no symptomatic or biochemical response after maximal CST and decided to administer oral cyclosporine (5 mg/kg), aiming to achieve a serum concentration of 200 ng/ml [Marzano et al. 2001]. They recorded a rapid response with biochemical regression, allowing the removal of ureteral stents after the patient’s recovery. The patient continued taking cyclosporine with no treatment-related toxicity.
In conclusion, in the past 20 years, no medical treatment seems to ‘cure’ IRF. Several medical treatments have been reported, but all carry a significant burden related to side effects such as corticosteroid complications that have a long life impact, not quantified by most of the studies published.
There are no controlled studies regarding any of the therapies described above, making the choice of the optimal drug subject to a matter of trial and error. Most of the cohorts assessed a small number of patients and the level of evidence is scarce.
The lack of consensus regarding the inclusion objective response to treatment criteria and therefore the variability of therapeutic objectives also contribute to the absence of a palpable treatment protocol. To this day there are no universally accepted criteria for defining clinical or imaging ‘remission’ and thus no objective criteria when to stop steroids or consider “failure” of medical treatment.
Surgical management
Surgical treatment is nowadays recommended in cases unresponsive to medical treatment or with severe ureteral entrapment [Elashry et al. 1996; Monev, 2002; Srinivasan et al. 2008]. The invasive therapy allows the performance of deep tissue biopsies in order to exclude malignant conditions, but it cannot prevent recurrence or progression of the disease and has no effect on systemic manifestation [Vivas et al. 2000; Vaglio et al. 2006; Cronin et al. 2008]. The recurrence rates reported range between 12 and 50%, irrespective of surgical approach (see Table 4) [Kavoussi et al. 1992; Puppo et al. 1994; Elashry et al. 1996; Monev, 2002; Srinivasan et al. 2008; Stifelman et al. 2008; Seixas-Mikelus et al. 2010].
Contemporary series regarding the surgical treatment of IRF.
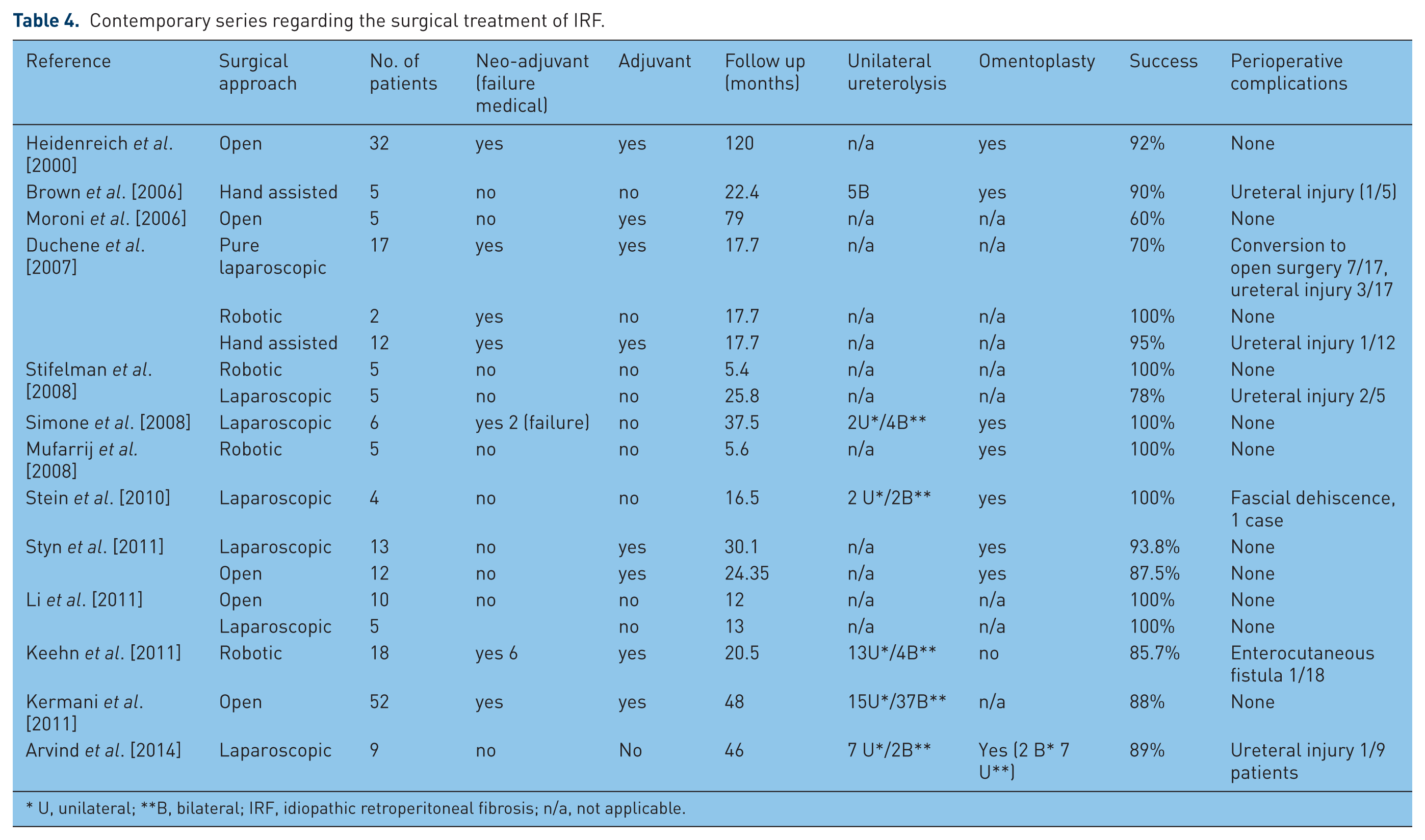
U, unilateral; **B, bilateral; IRF, idiopathic retroperitoneal fibrosis; n/a, not applicable.
The initial management consists of decompression of the obstructed renal units using ureteric stents and/or percutaneous nephrostomy and, if medical treatments are not successful, surgical ureterolysis. Indwelling ureteric stents are the most common form of renal drainage, but their efficacy is questionable. There are no studies comparing the results of various stents in term of stiffness, type of material and tolerance. The stents are usually removed at 3 months after medical or surgical treatment, but there are no objective criteria (clinical or biochemical) to sustain this decision. Also, the morbidity of indwelling stents has never been quantified in clinical trials nor their efficacy compared with the external drainage.
Open ureterolysis with deep tissue biopsy, repositioning the ureters laterally, with ‘intraperitonealization’ and/or omental wrapping are mandatory in order to restore the renal function [Heidenreich et al. 2000; Moroni et al. 2006; Vaglio et al. 2006] (See Figure 4). During the operation, the surgeon must evaluate the need for other procedures, like Boari Flap ureteroplasty, re-implantation, auto transplantation, nephrectomy, hysterectomy or bowel resection [Srinivasan et al. 2008; Stifelman et al. 2008; Seixas-Mikelus et al. 2010]. The urologist should be skilled in various reconstructive techniques, general and vascular surgery in order to face the intraoperative challenges that may be encountered.

(a) Open ureterolysis. (b) Intraperitoneal displacement of the right ureter with the closing of the parietal peritoneum.
The purpose of the operation is to resolve the ureteric obstruction and to exclude an underlying malignancy, not to treat the retroperitoneal fibrosis. The surgical technique is not standardized and the outcome of the treatment only evaluates the recovery of the renal function.
The complication rates reported vary between 8 and 16% and include ureteral devascularization, tears and ureteral strictures, ureteral leakage and urinary fistula, most of them amenable to a conservative approach [Elashry et al. 1996; Monev, 2002; Srinivasan et al. 2008; Stifelman et al. 2008; Seixas-Mikelus et al. 2010].
Since it was first reported by Kavoussi and colleagues in 1992 [Kavoussi et al. 1992], a number of reports regarding the efficacy and safety of laparoscopic ureterolysis in the treatment of IRF have been published [Matsuda et al. 1994; Puppo et al. 1994; Boeckmann et al. 1996; Elashry et al. 1996; Mattelaer et al. 1996; Nezhat et al. 1996; Castilho et al. 2000; Demirci et al. 2001; Fugita et al. 2002; Okumura et al. 2005; Mufarrij and Stifelman, 2006; Srinivasan et al. 2008; Stifelman et al. 2008; Seixas-Mikelus et al. 2010]. The authors did not find any variable that influenced conversion to open surgery and, moreover, complication and resolution rates were similar to the open approach [Elashry et al. 1996; Demirci et al. 2001; Fugita et al. 2002; Okumura et al. 2005; Seixas-Mikelus et al. 2010]. Recently, Mufarrij and Stifelman reported the first robot-assisted laparoscopic ureterolysis with laparoscopic omental wrap in the treatment of IRF [[Mufarrij and Stifelman, 2006]. Subsequently, several studies have proven the advantages offered by the robotic approach, including magnified three-dimensional, easier dissection of the ureter circumferentially and faster postoperative recovery [Mufarrij et al. 2008; Stifelman et al. 2008; Seixas-Mikelus et al. 2010].
However, most of the articles presenting the results of the minimally invasive procedures are retrospective series with small number of patients, most of them with secondary RF [Kavoussi et al. 1992; Matsuda et al. 1994; Puppo et al. 1994; Boeckmann et al. 1996; Elashry et al. 1996; Mattelaer et al. 1996; Nezhat et al. 1996; Castilho et al. 2000; Demirci et al. 2001; Fugita et al. 2002; Srinivasan et al. 2008; Stifelman et al. 2008; Seixas-Mikelus et al. 2010]. Most of the studies regarding the utility of minimal invasive approaches in the management of IRF have a limited follow up and most of the patients included present unilateral involvement of the ureter/small fibrotic mass. In addition, there is no prospective head-to-head comparison between open and lap/robotic ureterolysis in order to show the superiority of one approach to the other and also no agreement regarding the need for, and what type of, adjuvant treatment. To establish the role of minimally invasive treatment in the management of IRF, we need comparative randomized, controlled trials amongst these surgical procedures, but the perspective of ever having these studies conducted is minimal, due to the rarity of the disease and lack of reference centers for IRF.
Prognosis
Complete resolution of the IRF with ureteral involvement without therapy is rare. Malignant RF carries a poor prognosis, with a mean survival of 3–6 months [Vivas et al. 2000; Monev, 2002; Vaglio et al. 2006].
The follow-up protocol includes the evaluation of systemic symptoms, assessment of renal function, analysis of acute phase reactants and CRP levels at 3–6 months after remission/stabilization of the disease and evaluation of ureteral obstruction and the size of mass using CT or MRI [Kottra et al. 1996; Vivas et al. 2000; Monev, 2002; Vaglio et al. 2006] (See Figure 5).

Treatment algorithm of retroperitoneal fibrosis.
There are several questions that remain unanswered. First, is the renal function, which has been used as a surrogate of IRF improvement, accurate enough to define the success or failure of a treatment, either medical or surgical?
Second, if imaging is used to define the success/failure, do we need a complete resolution of the fibrotic mass to define success? Since the ureterolysis does not influence the fibrotic mass, how do we define the success of the surgical treatment? At present, there are no validated predictors of response to medical or surgical treatment and long-term assessment is mandatory, since recurrences have been reported up to 16 years [Cronin et al. 2008]. There are no published guidelines or cost-effective comparisons between different follow-up protocols [Kottra et al. 1996; Monev, 2002].
Conclusion
The imaging procedures, together with clinical and blood test features, often allow a diagnosis of IRF to be obtained, but a biopsy is mandatory to confirm it. However, there is no consensus regarding the template, timing and number of biopsies needed to exclude malignancy. Open ureterolysis represents the main surgical treatment for cases with severe IRF, and a laparoscopic or robotic approach may be an option in selected cases. The recovery of renal function is used as a surrogate for evaluating the success of the treatment. More clinical studies are needed to standardize the protocol for diagnostic, treatment and follow up after medical or surgical management.
Footnotes
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
