Abstract
Dysphagia is a serious symptom and requires urgent investigations and prompt diagnosis to initiate appropriate therapy. Dysphagia aortica is a rare entity described as difficulty in swallowing due to external compression by the aorta. The incidence of this entity is unknown. Herein we present a 90-year-old woman who presented with a progressive 3-month history of dysphagia, diagnosed with aortic aneurysm who then developed related complications and succumbed. Dysphagia aortica should be considered in the list of differential diagnoses in a patient with dysphagia although rare. Early diagnosis and management are vital to avoid poor outcomes.
Introduction
Dysphagia is difficulty in the passage of liquids or solids from the oropharynx to the stomach, and this is a subjective awareness. 1 It is considered a warning symptom for potentially serious underlying pathology, especially in the elderly population. 2 Dysphagia can be due to various causes including neuromuscular disease, obstructive lesions, or extrinsic compression. Dysphagia aortica is a rare cause of dysphagia defined by difficulty in swallowing by external compression by the aorta which can be ectatic, tortuous, or aneurismal.1,3
Herein, we report a case of dysphagia aortica in an elderly female who unfortunately could not receive definitive management.
Case presentation
A 90-year-old Black African woman presented to our surgical department with a 3-month history of progressive difficulty in swallowing which started with solid foods then followed by liquids thereafter even her saliva. This was associated with loss of appetite, significant unintentional weight loss, hoarseness of the voice, and intermittent dry cough. Furthermore, she reported a history of reduced urine output and changes in bowel habits. Her past medical history included a history of peptic ulcer disease for about 5 years for which she took oral Omeprazole. Otherwise, she denied a history of any chronic illnesses like diabetes mellitus or hypertension and no history of transfusion or major surgery. On family and social history, she was a businesswoman, married with eight children, no history of eating smoked food, and no history of alcohol or cigarette smoking. She also denied a family history of gastrointestinal cancer.
On clinical examination, she was alert, fully conscious with a Glasgow Coma Scale of 15/15, moderately dehydrated, wasted (body weight: 41 kg, body mass index (BMI): 16 kg/m2), not cyanotic, not pale, not jaundiced, and no peripheral lymphadenopathy. Her blood pressure on admission was 117/46 mm Hg, axillary temperature of 36.8°C, pulse rate of 60 beats per minute, blood glucose of 7.6 mmol/L, and saturating at 98% on 3 liters of oxygen and 92% on room air. On chest examination, her chest had normal skin contours with bilateral equal expansion, and bilateral air entry with bilateral basal fine crepitations. On the cardiovascular system, her JVP was not raised, she had a normal synchronized pulse of normal volume, warm extremities, and both heart sounds were heard with no murmurs. Her abdominal and nervous system examinations were unremarkable.
Laboratory investigations revealed hemoglobin of 12.5 g/dL; leukocytosis of 15.7 × 109/L predominant of neutrophils (84%); platelet count of 126 × 109/L; international normalized ratio of 1.89; erythrocyte sedimentation rate of 10 mm/h; elevated creatinine and urea of 299 µmol/L and 31.28 mmol/L, respectively; serum albumin of 29 g/L; serum sodium of 144 mmol/L; and serum potassium of 4.88 mmol/L. A plain chest X-ray revealed a significant mediastinal shift to the right with partial right lung collapse, trachea and esophagus deviated to the right, and no pleural collection (Figure 1).
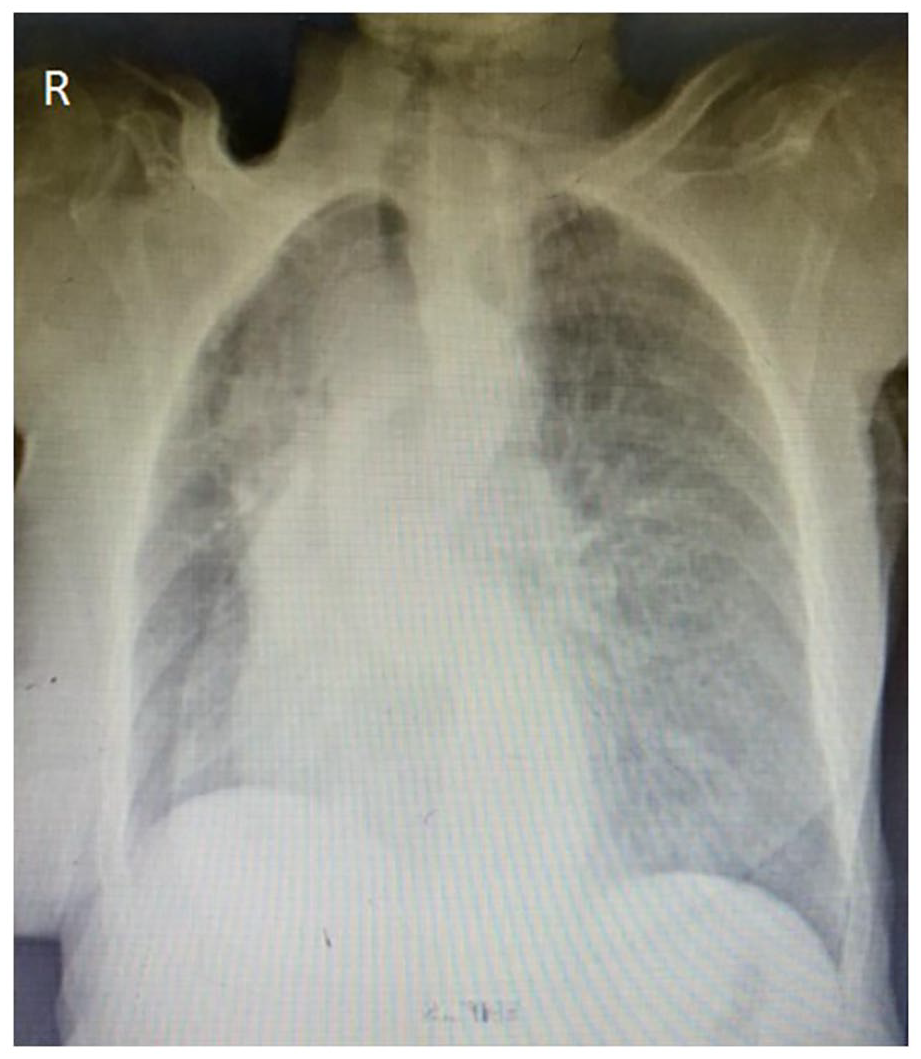
Mediastinal shift to the right with partial right lung collapse, trachea and esophagus deviated to the right, and no pleural collection.
Computed tomography (CT)-scan of the chest showed aneurysmal dilatation of the arch of the aorta and the proximal descending thoracic aorta measuring 5.1 cm in maximum diameter suggestive of an aortic aneurysm. Diffuse calcified atherosclerotic plaques were seen in the thoracic aortic wall. The tortuous aorta extrinsically compresses the mid-esophagus against the posterior wall of the right atrium. Consolidation was seen in the left lower lobe suggestive of aspiration pneumonia (Figure 2), and the CT scan of the abdomen and pelvis had no abnormalities. The patient was admitted, and given antibiotics, diuretics, intravenous fluids, with a plan of Naso-gastric tube or gastric feeding tube, and planned for esophagoscopy with biopsy.
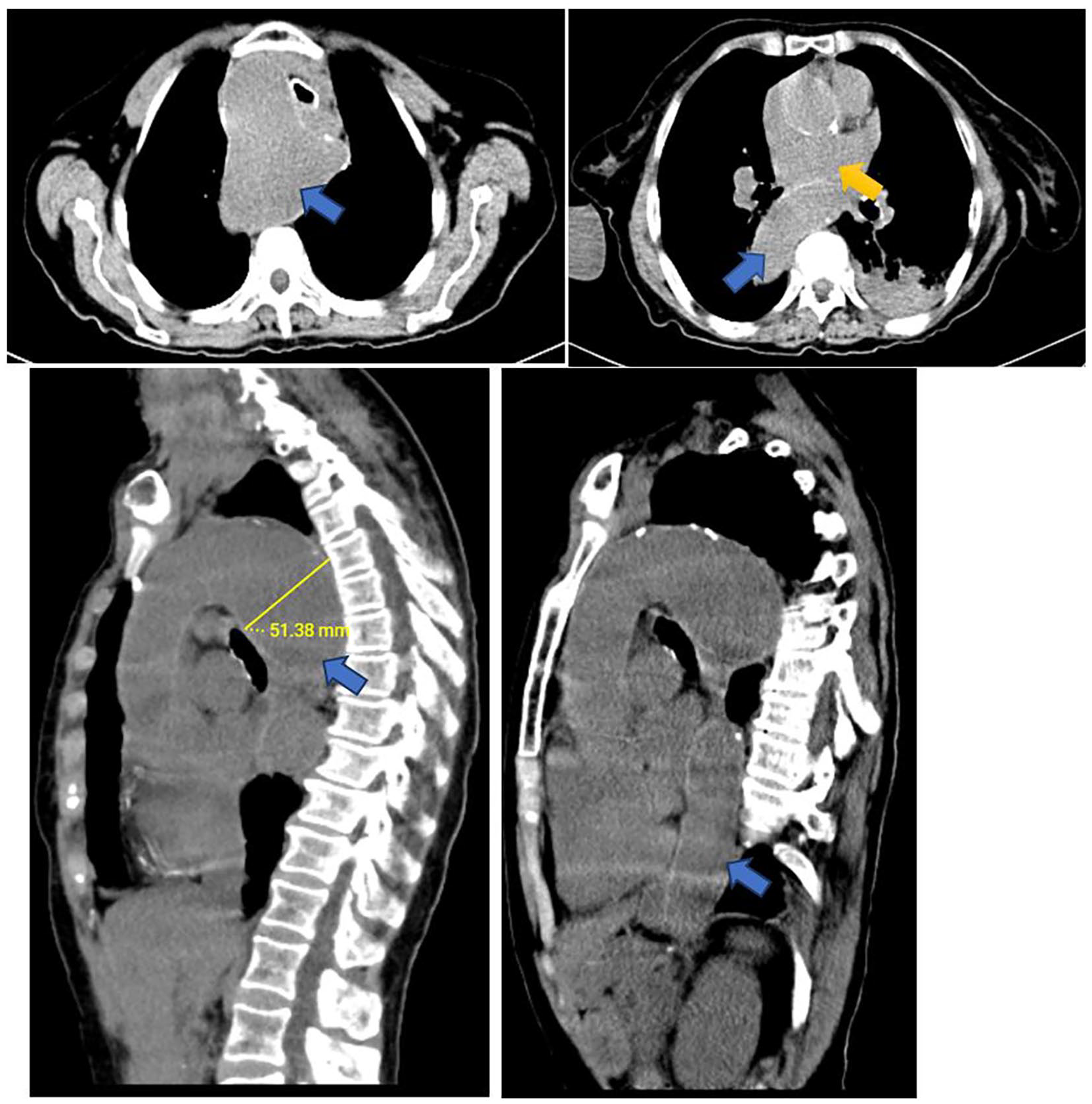
Non-contrast axial and sagittal images of the chest show aneurysmal dilatation of the arch of aorta and the proximal descending thoracic aorta (blue arrows) measuring 5.1 cm in maximum diameter. Diffuse calcified atherosclerotic plaques seen in the thoracic aortic wall. The tortuous aorta extrinsically compresses the mid-esophagus against the posterior wall of the left atrium (yellow arrow).
She developed deep venous thrombosis (DVT) on day 7 after admission diagnosed from Doppler ultrasound which revealed incompressible left femoral vein with thick echogenic material causing total occlusion to the distal third superficial femoral vein. She was then started on unfractionated heparin 5000 IU subcutaneous three times a day for 7 days. Esophagoscopy was not done due to financial constraints faced by the patient. She, unfortunately, passed away 9 days after admission; however, the cause of death was uncertain as postmortem was not done because the relatives did not consent due to traditional beliefs. However, the patient and caretakers were given two options of management which were the insertion of a gastric-feeding tube or referral to a specialized cardio-thoracic center.
Discussion
Dysphagia caused by a thoracic aortic aneurysm, also known as dysphagia aortic, is a rare entity encountered in clinical practice worldwide. It was first described by Pape in 1932 as dysphagia caused by an abnormally tortuous and dilated, atherosclerotic segment of the thoracic aorta due to age-related degeneration. 4 The true prevalence of dysphagia is unknown but studies reveal an estimated rate of prevalence of 16%–22% in patients above the age of 50 years as seen in our case. 5 As a rare condition, the true incidence of dysphagia aortica is unknown, and most cases are identified incidentally after radiologic investigations.4,6 The estimated incidence of thoracic aortic aneurysm is 5.3 per 100,000 individuals per year and the incidence of ruptured thoracic aneurysms is 1.6 per 100,000 individuals per year. 7
As aortic aneurysms are a rising global health challenge, several studies have been conducted to assess the epidemiology and burden of this disease. Twenty-two percent of people who suffer from aortic aneurysm syndromes die at home before reaching the hospital or getting any medical service, and among those getting medical services, 34% die within 30 days. Although men have a higher prevalence of thoracic aortic aneurysms, women have a higher aneurysm size together with higher mortality rates with a 40% increased risk of mortality and a three-fold increased risk of dissection as compared to men. 8 The annual mortality rate in sub-Saharan Africa ranged from 0.6 to 2.7 per 100,000 people. A study done in Nigeria reported a mortality rate of 0.7 per 100,000 people due to aortic aneurysms. 9
Other rare causes of dysphagia include dysphagia megalatriensis which involves a hypertrophied left atrium causing dysphagia,10,11 mediastinal tuberculous lymphadenitis, 12 and dysphagia lusoria which involves an aberrant subclavian artery resulting in a vascular ring around the esophagus causing dysphagia. 13
There is no gold standard diagnostic investigation for dysphagia aortica. Clinical symptoms together with radiologic and endoscopic imaging may aid in the diagnosis. Workup should include a chest X-ray, chest CT-scan with oral and intravenous contrast which show a dilated aortic arch and tortuous dilated aorta which aided in the diagnosis of our patient, and upper gastrointestinal endoscopy which may show stenosis and kinking of the esophagus which unfortunately was not performed in the index case, barium or video fluoroscopic swallowing study which may reveal partial esophageal obstruction and pulsatile movement of the barium synchronous with aortic pulsation, and esophageal manometry which may show a localized high-pressure band with superimposed pounding that is synchronous with the cardiac pulsation.5,6
Management depends on disease severity. Most patients are managed with diet modification to liquid diets and treated for underlying heart failure and arterial hypertension. Surgical modalities include percutaneous endoscopic gastrostomy, endoscopic esophageal dilation with a bougie, or esophageal self-expandable stents. Thoracic endovascular repair show symptom relief; however, it has a complication of esophageal erosion resulting in aorto-esophageal fistula. Other surgical procedures done to relieve esophageal compression include Heller myotomy.4,6 In our setting, the options were insertion of a gastric-feeding tube or referring the patient to a cardio-thoracic center of which both options were given to the caretakers of the patient.
Complications of thoracic aortic aneurysm include aortic dissection, compression, and erosion into an adjacent structure such as an aorto-esophageal fistula, leak or rupture, and thromboembolism such as disseminated intravascular coagulation, DVT, and pulmonary embolism as seen in our patient and could be the cause of death.5,6,14,15
Conclusion
Due to the rarity of dysphagia aortica, many patients encounter management delays and suffer catastrophic complications of thoracic aortic aneurysms although can be managed conservatively or surgically. Dysphagia aortica should always have a high clinical suspicion on differentials of dysphagia. Prompt and early investigations should be done to reduce morbidity and mortality at large including early referral to specialized centers whenever needed.
Footnotes
Acknowledgements
The authors would like to thank the patient and her son for permission to share her medical history for educational purposes and publication.
Author contributions
MS, JP, and JL reviewed medical records and conceptualized and drafted the manuscript. AS reviewed and reported the radiology films. All authors have read and approved the final manuscript.
Availability of data and material
We have not shared patient’s hospital records as they contain personal identification information.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from a legally authorized representative of the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal on request.
