Abstract
Intra- and extracranial tandem occlusions (TOCs) in acute ischaemic stroke (AIS) are defined by the coexistence of an intracranial anterior or posterior circulation large vessel occlusion together with an ipsilateral extracranial occlusion or significant stenosis in the corresponding proximal vascular axis. They represent a distinct subgroup of AIS requiring tailored diagnostic and therapeutic strategies and are commonly divided into anterior (ATOCs) and posterior (PTOCs) forms. This narrative review synthesises the current evidence base regarding diagnosis, management (medical and interventional) and prognosis of TOCs. ATOCs account for around 20% of anterior circulation large-vessel occlusions, while PTOCs appear proportionally more frequent in posterior circulation stroke. Atherosclerosis is the predominant mechanism (around 70%), followed by arterial dissection (around 20%–25%), with cardioembolism and rarer causes playing a smaller role. Diagnosis can be challenging, particularly due to pitfalls such as carotid pseudo-occlusion. Endovascular thrombectomy (EVT) is the cornerstone of treatment for TOCs. In ATOCs, emerging evidence suggests that emergent carotid artery stenting improves outcomes in selected patients, with large observational cohorts such as CERES-TANDEM reporting higher functional independence without increased bleeding risk. In PTOCs, EVT is feasible and technically effective, but evidence remains limited. Vertebral stenting may be beneficial in selected cases, though morbidity and mortality remain high. Periprocedural antithrombotic therapy varies widely between centres, and high-quality data are lacking. Bridging therapy with intravenous thrombolysis appears safe and should be performed in eligible patients with tandem lesions. Prognosis has improved substantially for patients with ATOCs in the EVT era, whereas outcomes in PTOCs are less well defined. Continued efforts to generate higher-quality evidence, particularly for PTOCs, will be essential to guide treatment strategies and improve patient outcomes.

Keywords
Introduction
Intra- and extracranial tandem occlusions (TOCs) in acute ischaemic stroke (AIS) are defined by the coexistence of an intracranial anterior or posterior circulation large vessel occlusion together with an ipsilateral extracranial occlusion or significant stenosis in the corresponding proximal vascular axis, although the specific stenosis severity threshold varies across studies.1–3 To date, anterior circulation TOCs (ATOCs) have been more extensively studied than posterior circulation TOCs (PTOCs). Consequently, multidisciplinary stroke teams currently have access to stronger and higher-quality evidence guiding the management of ATOCs, while the optimal treatment of PTOCs remains less well established.
The first reports describing emergent interventions for acute carotid artery occlusions appeared in the 1980s. 4 In 1996, Nesbit et al. 5 published the first case series of patients who underwent intracranial intra-arterial thrombolysis delivered through an occluded internal carotid artery (ICA). Following the widespread adoption of intravenous thrombolysis (IVT) as the cornerstone of AIS, early observational studies highlighted the challenges of achieving successful recanalisation in ATOCs and pointed to the frequently unfavourable long-term outcomes in this subgroup of patients.1,6 The paradigm shift introduced by mechanical thrombectomy, demonstrated by pivotal randomised controlled trials (RCTs), led to the gradual inclusion of ATOCs in pooled analyses. 7 These confirmed that acute-phase reperfusion therapies are both safe and effective in this setting. Currently, the main area of clinical uncertainty concerns the role of emergent carotid artery stenting (eCAS) in patients with ATOCs, particularly regarding its safety and efficacy. Closely linked to this issue is the unresolved question of optimal antithrombotic therapy, its type, timing and intensity, especially in patients requiring eCAS and in those with comorbidities necessitating anticoagulation (e.g. atrial fibrillation).
PTOCs have followed a markedly slower trajectory compared with their anterior counterparts. Although the first epidemiological and anatomical descriptions of PTOCs were published as early as 1961, 8 systematic clinical and therapeutic studies only began to emerge after 2014, following advances in endovascular therapy for large-vessel occlusion stroke, with the first comparative studies assessing endovascular approaches for vertebral artery (VA) occlusion in the setting of basilar artery (BA) thrombosis.8–10 The clear epidemiological, clinical and therapeutic differences between anterior and posterior TOCs suggest that they should be regarded as distinct clinical entities.
In this review, we provide an overview of TOCs across epidemiological, clinical, therapeutic and prognostic domains, highlighting the unresolved questions that impact decision-making in acute stroke care.
Epidemiology of TOCs
Anterior tandem occlusions
Epidemiological studies published after 2010 consistently report that ATOCs account for roughly 13%–16% of patients with AIS undergoing mechanical thrombectomy, although some multicentre series have reported rates approaching 20%.11–14 Beyond prevalence, these studies highlight distinct clinical characteristics in patients with ATOCs, including younger age, male predominance, higher rates of smoking, lower rates of atrial fibrillation and heart failure and longer onset-to-recanalisation times.
It is important to note that definitions of ATOCs vary across studies, particularly regarding the degree of ICA stenosis required for classification. In some series, no formal threshold is provided, while in others, the minimum degree of stenosis and the indication for endovascular intervention range between 70% and 90%, according to NASCET, ECST or ACAS criteria.15–17 These discrepancies directly affect the size of the ATOC subgroup and may influence both reported prevalence and outcome rates.
Posterior tandem occlusions
Although PTOCs have been classically underreported, recent epidemiological studies suggest that they may be more prevalent within posterior circulation AIS than the rates typically observed for ATOCs. In a cohort of 224 patients with acute BA occlusion, 18 the prevalence of PTOCs was 24.6%, with no significant differences in recanalisation rates, procedure times or clinical outcomes. 18 However, concerns were raised regarding the risk of early BA reocclusion when the respective VA lesion was not treated. More recently, a considerably larger joint retrospective analysis of the BASILAR and PERSIST registries 19 reported a 19.9% prevalence of PTOCs. 19 Patients with TOC who underwent stenting were found to be younger and to have a higher proportion of atherosclerotic and unknown aetiologies, while no other major baseline differences were observed. 19 A meta-analysis published in 202219,20 pooling 405 patients with posterior circulation stroke treated with endovascular thrombectomy (EVT; 81 PTOCs) found no differences between the subgroups (PTOC vs isolated vertebrobasilar occlusion) regarding clinical outcomes. 20
Overall, most available studies are limited by small sample sizes and heterogeneous methods, leaving important uncertainties regarding the epidemiology and management of PTOCs.
Table 1 summarises key studies on large stroke populations, including anterior (ATOCs) and posterior tandem occlusions (PTOCs), highlighting their epidemiological characteristics.
Frequency of anterior and posterior tandem occlusions across major clinical studies.

In both the German Stroke Registry and the study by Grigoryan et al., patients with anterior and posterior circulation AIS were included; the proportion of non-tandem anterior circulation cases was not detailed.
AIS, acute ischaemic stroke; ATOC, anterior tandem occlusions; BAO, basilar artery occlusion; EVT, endovascular treatment; LVO, large-vessel occlusion; MeVO, medium-vessel occlusion; PTOC, posterior tandem occlusions; VBO, vertebrobasilar occlusion.
Pathophysiology of TOCs
Most TOCs, both anterior and posterior, originate from proximal vessel pathology. The two most frequent mechanisms are progressive stenosis due to large-vessel atherosclerotic disease and sudden stenosis/occlusion due to arterial dissection. Other causes, although considerably less common, include cardioembolic sources, patent foramen ovale and carotid webs. 23
Anterior tandem occlusions
Evidence from large multicentre registries, particularly the TITAN study (2017) suggests that atherosclerotic lesions account for approximately 60%–70% of ATOCs, followed by arterial dissections in 20%–30%. 24 A subsequent large retrospective study 25 of 482 patients with ATOCs treated with EVT under different medical regimens (2018) reported a similar distribution, although with a slightly higher proportion of cardioembolic aetiologies (10%; Figure 1(a)–(c)). 25

Tandem occlusions: types and main aetiologies. (a) CT scan showing anterior circulation tandem occlusion. (a1) CT angiography showing occlusion of the left ICA at its origin (red arrow), with calcifications signalling a possible atherosclerotic plaque. (a2) Tandem intracranial M1 occlusion (red arrow). (b) Tandem occlusion of the posterior circulation. (b1, b2) Retrograde filling of the distal BA occurs through the posterior communicating arteries; proximal V4 dissection with a visible false lumen (b1, red arrow), caused vertebral occlusion and was likely the source of the distally migrated thrombus; absence of flow in the BA on TOF MRA (b2, red arrow). (a) Atherosclerosis represents the main aetiology in both anterior and posterior tandem occlusions. (b) Dissection is the second most common cause. (c) Cardioembolism is the third cause of tandem occlusions.
Patients with atherosclerosis are typically older, more often male and have a higher burden of vascular risk factors; they also tend to present with more robust collateral circulation. The male predominance in ATOCs may partly reflect the overall higher prevalence of atherosclerosis in men. In contrast, arterial dissections are more common in younger patients with fewer vascular risk factors, frequently triggered by cervical or head trauma and are also associated with more competent circles of Willis (Figure 2).23,26 Importantly, dissections often have a relatively favourable natural course, with spontaneous healing of the vessel wall in up to 70% of cases, usually within the first 9 months. 27

Tandem occlusion after carotid dissection. Forty-three years-old, male, with a carotid occlusion and a tandem M2 occlusion; approached by anterograde endovascular technique. (a) Initial run shows the characteristic sharp occlusion of the ICA (white arrows). (b) After crossing the occlusion with a microcatheter, the dissection flap and the true and false lumina are clearly visible. (c) Once careful catheterisation of the true lumen is performed, a stent was deployed with successful recanalisation of the cervical carotid. (d) Allowing the progression of an aspiration catheter up to the M2 segment, with a complete intracranial recanalisation.
The nature of the proximal lesion is also relevant for EVT outcomes. Occlusive atherosclerotic lesions present greater technical challenges than dissections, owing to thicker and less compliant vessel walls. Moreover, EVT for complete occlusions differs technically from procedures in severe stenosis, as the distal lumen cannot be visualised or filled with contrast, requiring a ‘blind’ approach. 26 Despite these challenges, current evidence suggests that EVT may provide greater benefit in atherosclerotic lesions than in dissections, largely due to the high risk of recurrent embolisation from unstable atherosclerotic plaques. 2 A rare additional mechanism is carotid stump syndrome, in which a chronically occluded ICA becomes a source of recurrent embolisation, typically microemboli, but occasionally macroemboli in settings of turbulent flow, leading to repeated cerebral or retinal ischaemic events. 28 This has been more often reported in Asian populations where atherosclerotic disease is particularly prevalent. 28
Posterior tandem occlusions
In PTOCs, proximal VA atherosclerosis, especially at the ostium, represents a common aetiology, with unstable plaques acting as a source of embolisation to the distal intracranial VA or the BA.10,29 Individual patient data pooled from the BASILAR and PERSIST registries 19 has confirmed this predominance, reporting that about 75% of PTOCs are related to extracranial VA atherosclerosis, followed by cardioembolic sources (11%) and less common causes such as dissection (Figure 1(a)–(c)). 19
Although rare, vertebral artery dissection has been described as a mechanism of PTOC. In these cases, AIS may result from distal embolisation of thrombus arising from the dissected artery, from complete local vessel occlusion with collapse of the vertebrobasilar system or from a combination of the two processes (TOC). 30
Another recognised mechanism is vertebral stump syndrome, a posterior counterpart to the carotid stump phenomenon, in which a chronically occluded vertebral artery continues to produce recurrent embolisation into the vertebrobasilar system. This is thought to occur when fragments of thrombus detach under turbulent haemodynamic conditions created by cervical collateral flow.31,32
The intrinsically lower collateral capacity and reduced vasomotor reactivity of the posterior circulation have been emphasised as additional factors that may predispose PTOCs to more severe ischaemic injury compared with their anterior counterparts. 33
Diagnosis
The diagnosis of TOCs relies on the integration of clinical and radiological findings, as in any case of AIS.
Clinically, patients with ATOCs or PTOCs present in a manner similar to those with isolated anterior or posterior circulation large vessel occlusions. However, the characteristic ‘double hit’ phenomenon of TOC compromises haemodynamic adaptation, increasing tissue vulnerability and often resulting in a more severe clinical presentation. 22
In practice, the diagnostic pathway for suspected TOCs follows the standard protocol for suspected acute ischaemic stroke: non-contrast CT to rule out haemorrhage, followed by CT angiography (CTA) of the head and neck to identify vessel occlusions and guide further management. 34 Multiphase CTA has been shown to provide a higher diagnostic yield than single-phase CTA, as it allows estimation of thrombus length and improves sensitivity for both intra- and extracranial thrombi. 35 Brain MR combined with MR angiography is an alternative first-line approach, particularly in centres where MR is routinely available or in patients with contraindications to iodinated contrast administration. 34
In PTOCs, CTA or magnetic resonance angiography (MRA) provides the additional advantage of characterising the vertebral arterial axis. Identification of vertebral dominance is crucial to plan endovascular intervention, as TOCs typically arise from thrombosis of the dominant vertebral artery. 29 Vertebral dominance also influences BA configuration, which often bends contralaterally, creating a straighter path between the dominant vertebral artery and the basilar trunk. 36
TOCs caused by cervical artery dissection can be diagnostically challenging. Clinical suspicion is higher in younger patients, especially when there is a history of recent infection, cervical trauma or manipulation, connective tissue disease or associated cervical pain and headache. 37 In this setting, brain MR with a 3D T1-weighted black-blood sequence can help distinguish intramural haematomas from intraluminal occlusions. 38 CTA or MRA may show the typical ‘flame-shaped’ lumen and crescent-shaped intramural thrombus, while non-contrast CT can reveal watershed infarctions in patients with severe ICA stenosis or occlusion. 23
Beyond confirming vessel occlusion, digital subtraction angiography (DSA) provides valuable prognostic information in TOCs, particularly through reliable assessment of collateral circulation and venous drainage. It is also useful when CTA or MRA findings are atypical or inconclusive. 39
Certain pitfalls require special attention. One important diagnostic challenge is the phenomenon of carotid pseudo-occlusion, which refers to the apparent occlusion of the cervical ICA on CT angiography or DSA, caused by a stagnant column of unopacified blood proximal to a terminal (carotid-T) occlusion. This finding may mimic a true TOC, although the cervical ICA is in fact patent (Figure 3). 40 The flame or beak sign, characterised by a post-bulbar flame-shaped tapering of contrast filling in the cervical ICA, is often interpreted as indicating pseudo-occlusion; however, its accuracy remains uncertain, as it may also occur in true cervical ICA occlusions. Absence of ophthalmic artery opacification when the ipsilateral common carotid artery is injected during DSA has been identified as a possible indirect sign of intracranial ICA occlusion, suggesting cervical pseudo-occlusion.41,42 Diagnostic accuracy can be improved by combining imaging techniques such as carotid or vertebral ultrasound with delayed CTA or MRA.23,43 Multiphase CTA, with its delayed phase, can further aid in distinguishing extracranial ICA true occlusions from pseudo-occlusions. 35

Pseudo-occlusion of the ICA. (a–c) CT and CT angiography demonstrating left ICA occlusion mimicking tandem occlusion (red arrows). (a) Axial CT showing intracranial ICA terminus occlusion. (b, c) Axial and sagittal views showing apparent cervical ICA occlusion. (d) DSA of the same patient (red arrow) reveals delayed filling of the cervical ICA, confirming pseudo-occlusion rather than true tandem occlusion.
Treatment strategies in ATOCs
The management of ATOCs remains challenging, with several aspects still unresolved despite their inclusion in major registries and RCTs. Key issues include the optimal sequence of treating the extracranial and intracranial lesions, the safety of emergent carotid stenting, the role of IVT and the appropriate antithrombotic regimen. High-quality data are also scarce regarding the best treatment options in patients with large ischaemic cores and minor clinical presentations.
Endovascular strategies
One of the central technical questions in EVT for ATOCs is whether to treat the intracranial or extracranial lesion first. In the TITAN registry, the retrograde (‘head-first’) approach (Figure 4, (1)), addressing the intracranial occlusion first, has been associated with faster groin-to-reperfusion times, an independent predictor of good outcome, without differences in reperfusion success or complications, when compared to the anterograde (‘neck-first’) approach (Figure 4, (2)).44,45 A meta-analysis including over 1500 patients also favoured the retrograde strategy, reporting higher rates of successful reperfusion and better functional independence at 90 days, with similar safety outcomes. 46 More recently, a simultaneous approach has been described, combining stent retriever deployment with angioplasty in a monorail fashion, potentially reducing procedure time but remaining highly operator-dependent and in need of validation in larger prospective studies. 47

Tandem occlusions: endovascular techniques approach. (1) Retrograde endovascular technique (1a-1c) – Guide catheter was advanced across the carotid occlusion (1d), and a triaxial system was used to aspirate the M1 clot (1e). A carotid stent was then advanced inside the guide catheter, and the guide catheter was retrieved, leaving the stent in place for deployment (1f). Final run (1g) showing complete recanalisation of the MCA, and of the cervical ICA. There was residual spasm of the distal cervical ICA, which was reverted with 5 mg IA verapamil. (2) Anterograde endovascular technique (2a-2c) – a pre-occlusive carotid stenosis (2d), with a tandem distal M1 occlusion (2e). The stenosis was severe and could not be crossed with the triaxial thrombectomy system. Therefore, a proximal approach was chosen, with a primary carotid angioplasty and stenting (2f), followed by distal aspiration of the M1 thrombus (2g), with TICI 3 recanalisation.
Another important question is whether to immediately treat the proximal ICA lesion or defer its management. Data from the STRATIS registry 48 has shown that in practice, treatment of the cervical ICA lesion is frequently postponed. 48 The decision to defer treatment is often driven by concerns about increased haemorrhagic risk, both from sudden hyperperfusion after recanalisation and the need for intensified antithrombotic therapy, as well as the risks of acute in-stent thrombosis and acute micro- and macroembolisation due to stent manipulation at the lesion site.49,50 On the other hand, immediate treatment may help eliminate the embolic source and restore cerebral perfusion. 26 The European Stroke Organisation (ESO) guidelines on endarterectomy and stenting for carotid artery stenosis recommend performing carotid endarterectomy in patients with symptomatic carotid stenosis of 70%–99% within the first 2 weeks post-index event, while carotid artery stenting should be considered in young populations (<70 years old) with carotid stenosis of 50%–99%. These recommendations, however, derive from studies focused on atherosclerotic carotid disease and are not directly applicable to TOCs.26,51
In ATOCs, some authors posit that carotid angioplasty alone can be sufficient in selected groups to reach the intracranial occlusion, making it possible to delay the carotid stenting. 52 Theoretically, angioplasty alone would bypass the risks of acute antiplatelet therapy (APT) in patients submitted to thrombolysis or with large core strokes. However, a 2022 systematic review and meta-analysis addressing this particular question revealed higher odds of good functional outcome (modified Rankin Scale (mRS)) in acute carotid stenting and in the retrograde approach groups; no differences were found regarding mortality, ICH or thrombolysis status; therefore, favouring acute carotid stenting over balloon angioplasty alone in patients with ATOCs. 53
An individual participant data meta-analysis from the IRIS collaboration, including 340 ATOC patients from a cohort of 2267 patients with anterior circulation large-vessel occlusion, found that acute ICA stenting was associated with improved 90-day outcomes (adjusted common odds ratio (OR) 1.60, 95% confidence interval (CI) 1.03–2.47), independent of prior IVT administration as bridging therapy. Although patients undergoing acute stenting had numerically higher rates of any ICH (44% vs 35%) and symptomatic ICH (6.3% vs 3.7%), these differences were not statistically significant. 54
Recently, the results of CERES-TANDEM, a large international multicentric retrospective cohort of patients with ATOCs, were presented at the ESO Conference. 55 This international retrospective study included 4053 patients, of whom 2522 underwent eCAS and 1531 were treated with direct EVT without stenting. In adjusted analyses, eCAS was associated with significantly better outcomes, including an improved shift in the mRS distribution at 90 days (common OR 1.31, 95% CI 1.17–1.47), higher rates of functional independence (47% vs 35%) and greater successful recanalisation (91% vs 76%). Importantly, these benefits were not accompanied by an excess risk of symptomatic intracranial haemorrhage (sICH; 11% vs 9%), although asymptomatic ICH was slightly more frequent in the stenting group. Early neurological deterioration was also less common with eCAS (19% vs 22%). Together, these results provide the strongest real-world evidence to date supporting eCAS in the management of ATOCs. However, as CERES-TANDEM was observational, confirmation from prospective randomised trials remains essential. The ongoing START (NCT05902000, completion expected in 2025) and PICASSO (NCT05611242) trials will provide critical insights into the role of eCAS in this population.
Regarding the endovascular management of ATOCs with underlying carotid artery dissection, a STOP-CAD substudy of 328 patients found that eCAS was associated with higher odds of successful intracranial reperfusion (mTICI 2b-3) but similar rates of 90-day functional independence and sICH compared with EVT without eCAS. Recurrence rates were low in both our study groups. These findings are consistent with a 2020 pooled analysis from the TITAN and ETIS registries.56,57 However, causation cannot be established, as patients achieving favourable intracranial reperfusion may be more likely to undergo stenting. Importantly, angioplasty and stenting of complete occlusive ICA dissections present multiple technical challenges and risks. Catheter advancement through a complete occlusion is technically demanding and risks false lumen passage, potentially extending the dissection. Thrombus within the false lumen may embolise, causing additional cerebral ischaemia. Furthermore, a long dissection may complicate stent placement. Finally, sudden reperfusion, especially in poorly collateralised territories, may precipitate ICH.58,59
Embolic protection devices may be used during eCAS to prevent distal embolisation with both anterograde and retrograde techniques. These devices are routinely employed in elective carotid revascularisation for symptomatic and asymptomatic disease; however, evidence supporting their use in acute settings remains limited, particularly when urgent intracranial reperfusion is required.60–63
Table 2 summarises international recommendations on acute management of TOCs. The 2023 European Society for Vascular Surgery (ESVS) guidelines recommend performing eCAS after EVT in selected cases, including poor antegrade ICA flow, inadequate collateral circulation or small infarct core with low bleeding risk, while deferral is suggested when intracranial reperfusion is incomplete, collateral compensation is adequate or when infarct volume or bleeding risk are high. 64
Recommendations on the management of tandem occlusions in major stroke guidelines.
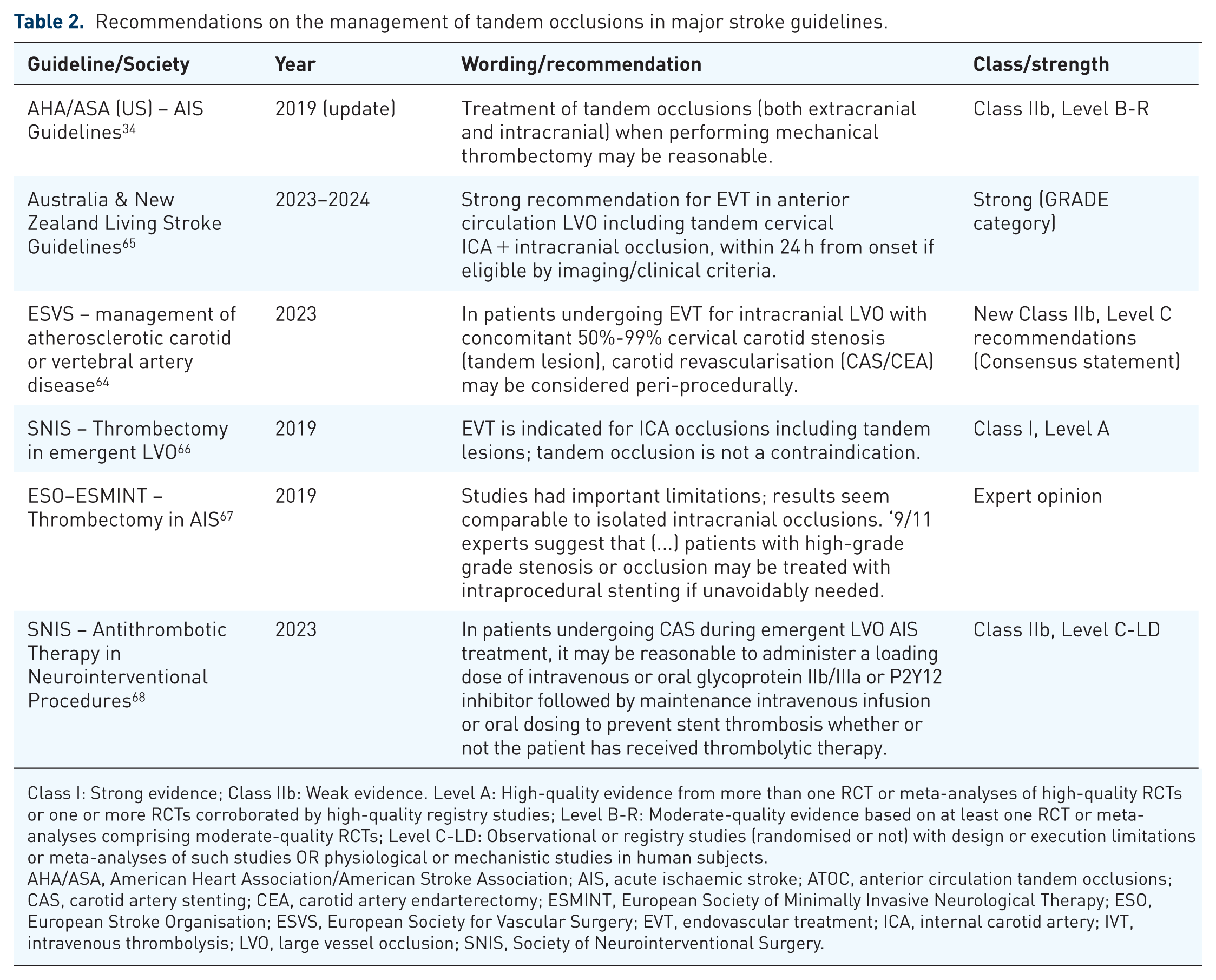
Class I: Strong evidence; Class IIb: Weak evidence. Level A: High-quality evidence from more than one RCT or meta-analyses of high-quality RCTs or one or more RCTs corroborated by high-quality registry studies; Level B-R: Moderate-quality evidence based on at least one RCT or meta-analyses comprising moderate-quality RCTs; Level C-LD: Observational or registry studies (randomised or not) with design or execution limitations or meta-analyses of such studies OR physiological or mechanistic studies in human subjects.
AHA/ASA, American Heart Association/American Stroke Association; AIS, acute ischaemic stroke; ATOC, anterior circulation tandem occlusions; CAS, carotid artery stenting; CEA, carotid artery endarterectomy; ESMINT, European Society of Minimally Invasive Neurological Therapy; ESO, European Stroke Organisation; ESVS, European Society for Vascular Surgery; EVT, endovascular treatment; ICA, internal carotid artery; IVT, intravenous thrombolysis; LVO, large vessel occlusion; SNIS, Society of Neurointerventional Surgery.
Bridging therapy
While initially debated, current evidence indicates that IVT prior to EVT can be safely administered in patients with ATOCs when indicated. In addition to the findings on acute stenting described earlier, 53 the IRIS collaboration evaluated bridging therapy with IVT in TOCs.71,72 The observed treatment effect with IVT plus EVT versus EVT alone corresponded to a −2.0% (95% CI −10.5 to 6.5) lower absolute risk of functional independence (mRS 0–2) in patients with tandem lesions, with no heterogeneity of treatment effect between patients with and without tandem lesions. Rates of intracranial haemorrhage and symptomatic haemorrhage were comparable between treatment groups. The safety profile is consistent with a prior meta-analysis of 15 observational studies (n = 1857), which similarly reported comparable safety with bridging therapy versus EVT alone. 73 Taken together, these findings suggest that the presence of tandem lesions does not modify the efficacy or safety of IVT, which should not be withheld solely on this basis.
Recent studies have also explored the use of tenecteplase (TNK) as the thrombolytic agent in bridging therapy in TOCs. TNK appears to be both safe and effective compared with alteplase. A post hoc analysis of the EXTEND-IA TNK study found similar reperfusion rates, clinical outcomes and intracranial haemorrhage rates between TNK and alteplase in patients with TOC. 74 Registry data from ETIS and TETRIS in patients with ATOC further suggested higher recanalisation rates (OR 4.21; 95% CI 2.69–6.61) and lower mortality (OR 0.59; 95% CI 0.40–0.87) with TNK in comparison to alteplase, and while the rate of any ICH was higher in the tenecteplase group, no differences were found in sICH or parenchymal haematoma rates. 75 A substudy from the AcT trial including 128 patients with ATOC reported a higher proportion of favourable outcomes (46.0% vs 32.6%, adjusted OR 3.21; 95% CI 1.06–9.71) with TNK versus alteplase, with no difference in mortality, reperfusion success or safety outcomes. 76
Antithrombotic therapy
Periprocedural antithrombotic therapy is a critical component of TOC management, particularly when eCAS is performed. However, practice varies considerably across centres in both agent selection and timing of administration, reflecting the absence of standardised protocols. Table 3 summarises the antithrombotic regimens employed in major studies of acute extracranial stenting for TOCs.
Periprocedural antithrombotic regimens in clinical studies of acute extracranial stenting for tandem occlusions.

In the ETIS and TETRIS pooled analysis, 256 of 753 patients underwent emergent ICA stenting; however, the specific antithrombotic regimen used in this subgroup was not reported.
AP, antiplatelet; AT, antithrombotic therapy; ATOC, anterior circulation tandem occlusions; ICA, internal carotid artery; TITAN, Thrombectomy In TANdem occlusions; tPA, tissue plasminogen activator.
Among the most informative datasets is the TITAN registry (Thrombectomy In TANdem occlusions), an international multicentre collaboration that collected real-world data between 2012 and 2016. A 2019 analysis of 295 patients with ATOCs suggested that EVT combined with eCAS and periprocedural APT achieved higher rates of successful reperfusion and favourable outcomes, without a major excess in bleeding risk. 77 Even in patients also treated with bridging IVT, safety remained acceptable: the rate of sICH in the ‘quadruple therapy’ group (EVT + eCAS + IVT + APT) was 2.4%. In comparison, the HERMES collaboration, which pooled individual patient data from five landmark randomised thrombectomy trials in AIS, reported sICH rates just above 4% in patients with large-vessel occlusion treated with EVT. 7 Building on these findings, the ongoing TITAN trial, launched in 2020, is designed to establish whether EVT with eCAS and APT provides superior outcomes to EVT alone. Results are expected in 2026 and are anticipated to provide more definitive guidance on this issue. 80
Glycoprotein IIb/IIIa inhibitors are also often considered in the periprocedural setting. 81 Abciximab has consistently been associated with higher rates of sICH and mortality, particularly in older patients, and its use has therefore declined.49,82 By contrast, tirofiban appears to have a more favourable safety profile: in a 62-patient cohort, isolated tirofiban use was associated with lower mortality and better recanalisation rates compared with dual APT (DAPT), although functional outcomes were similar. 83 Eptifibatide has also shown promise, with early studies suggesting acceptable safety even after IVT, and later analyses indicating possible improvements in clinical outcomes IVT.84–86 Intravenous cangrelor, a fast-acting reversible P2Y12 inhibitor, has more recently been reported as a feasible periprocedural option with a reassuring safety profile, though evidence remains limited to retrospective series.3,87
After stenting, most centres initiate DAPT with aspirin and clopidogrel once a control CT has excluded early haemorrhage.21,77,81 The optimal duration remains uncertain. A recently published multicentre study including 223 patients with ATOCs from 12 German centres examined the safety and efficacy of different regimens. 88 The study confirmed that antithrombotic regimens differed in timing (preprocedural; periprocedural; postprocedural after day 1 or day 2; after discharge), type of medication (intravenous tirofiban; heparin; single APT usually with aspirin or clopidogrel; DAPT with aspirin and a P2Y12 inhibitor, such as clopidogrel or ticagrelor). Their findings suggested that DAPT, initiated from day 1 and maintained to day 90, was associated with reduced risk of stent occlusion and sICH as well as better functional outcomes. In contrast, single APT or periprocedural regimens alone were not associated with significant benefit. A nationwide Korean cohort study including over 12,000 patients found that short-duration DAPT (90–180 days) was as safe and effective as longer regimens (>180 days). 89 The 2024 ESC Guidelines for the management of peripheral arterial and aortic diseases recommend a minimum of 1 month of DAPT after stenting, which remains the most widely adopted approach in clinical practice. 90
For patients with non-cardioembolic ATOCs who are not stented, single APT with aspirin is standard, usually started within 24–48 h, but withheld for 24 h in those who received IVT. In minor ischaemic strokes, short-term DAPT for 21 days, followed by SAPT, is recommended.91,92
Beyond APT, anticoagulation also has a role in selected subgroups. In carotid dissections, anticoagulants are considered reasonable, especially in high-risk patients.34,93,94 An individual participant data meta-analysis combining data from the CADISS and TREAT-CAD trials showed a non-significant reduction in the composite primary endpoint, ischaemic stroke and major bleeding, among participants randomised to anticoagulation (OR 0.33, 95% CI 0.08–1.05, p-value = 0.06). Additionally, when focusing solely on ischaemic stroke as an outcome, anticoagulation showed a significant benefit (OR 0.14, 95% CI 0.02–0.61, p-value = 0.01), suggesting a potential advantage of anticoagulation in the setting of stroke prevention. 95 Assuring secondary prevention measures is also key; smoking cessation and blood pressure control are recommended in all patients who have suffered strokes in the past, and high-intensity statin therapy is recommended in patients with noncardioembolic AIS, most particularly in patients with underlying carotid stenosis.64,96 In patients with atrial fibrillation, early initiation of direct oral anticoagulants (OAC) is favoured. 97 In cases where patients require both eCAS and long-term anticoagulation, triple therapy with DAPT and OAC is discouraged given the high risk of bleeding and mortality; shorter periods of overlap may be considered but should be kept to a minimum. 98
Treatment strategies in PTOCs
Compared with ATOCs, the evidence base for the acute management of PTOCs is even more limited, with current recommendations largely derived from small cohorts and expert consensus. Most treatment strategies for PTOCs are extrapolated from anterior circulation occlusions, but the high prevalence of vertebral atherosclerosis in this setting introduces specific technical challenges for intervention.99,100 Access to the BA can be achieved through two main routes: the ‘clean-road’ pathway via the patent vertebral artery, or the ‘dirty-road’ pathway through the occluded/stenotic vertebral artery.99,101 The clean-road route may facilitate faster reperfusion and, by temporarily arresting flow, improve aspiration efficacy. However, it can also critically compromise blood flow to the basilar circulation, particularly during prolonged interventions, requiring general anaesthesia for these procedures.10,102,103
Despite the theoretical advantages, the clean-road approach is not always feasible. Unilateral vertebral hypoplasia or distal aplasia are common anatomical variants, and in many cases, the dominant vertebral artery is the one affected by the proximal lesion. This often leaves the dirty-road approach as the only or most practical option. Although technically more challenging, it allows for direct treatment of the culprit vertebral lesion, typically through angioplasty or stenting, albeit requiring periprocedural APT.10,29,101
Within the dirty-road approach, two main strategies are described, resembling those used in ATOCs. In the anterograde approach, vertebral angioplasty or stenting is performed first, followed by basilar thrombectomy. 10 This sequence allows earlier stabilisation of the proximal atherosclerotic plaque, reducing the risk of further distal embolisation and improving antegrade flow to the BA. However, it typically results in longer time-to-reperfusion, which can worsen outcomes by delaying basilar recanalisation and increasing the risk of haemorrhagic transformation of posterior fossa infarcts. By contrast, the retrograde approach prioritises early basilar recanalisation, with vertebral stenting performed only afterwards. This reduces delays in restoring intracranial flow but carries the risk that subsequent stent placement may trigger distal embolisation. 101
Evidence from cohort studies examining EVT in PTOCs remains inconsistent, with no clear superiority demonstrated for either clean-road or dirty-road strategies.9,10,18,29,101,103–106 Interpretation is limited by small sample sizes, anatomical bias (since the dominant vertebral artery is often the culprit and thus constrains the feasibility of clean-road access) and marked heterogeneity across studies. A 2022 meta-analysis pooling 7 studies and 405 patients reported no significant difference in safety or functional outcomes between approaches, but important limitations included lack of detail on endovascular techniques (e.g. thrombectomy device, angioplasty vs stenting) and substantial methodological variability, which restricts firm conclusions. 20
Evidence from randomised trials in posterior circulation stroke has historically excluded patients with isolated vertebral artery occlusion, but the recent VERITAS individual patient data meta-analysis (BEST, BASICS, ATTENTION, and BAOCHE trials) did provide a specific estimate for vertebral artery occlusions extending into the BA. 107 In this subgroup, which comprised 46 of 833 patients (6%), endovascular therapy was associated with a larger apparent benefit than for other occlusion sites (adjusted common OR for mRS 0–3 at 90 days: 8.25, 95% CI 1.58–42.93), although the very wide CIs underline the limited precision of this estimate. Given the small sample size these results are hypothesis-generating.
Beyond VERITAS, additional insights come from observational series. An analysis of the BASILAR and PERSIST registries including 1320 patients with posterior circulation large-vessel occlusion identified 217 (19.9%) with PTOCs. 19 Among them, 84 underwent acute vertebral stenting (with periprocedural IV tirofiban) and 133 did not. Patients treated with stenting had more favourable outcomes at both 90 days and 1 year, with higher odds of functional independence and lower mortality, without an excess of sICH. These findings suggest that acute-phase vertebral stenting can be both beneficial and safe in selected PTOC patients. Importantly, this study also indicated that IVT appears to be safe when used in combination with EVT for PTOCs. Together, these data reinforce the long-standing view that PTOCs, like their anterior counterparts, derive meaningful benefit from EVT.20,101,108
Evidence on IVT before EVT in PTOCs is very limited. The BASILAR/PERSIST registry analysis is one of the few studies to specifically report on this question. In PTOC patients, no significant differences were observed between bridging therapy and direct EVT regarding functional outcomes, complications or mortality. 19 Therefore, IVT should be performed in patients with PTOCs who otherwise have no contraindications. 109
Periprocedural therapy in acute VA stenting is highly variable across centres but broadly mirrors anterior circulation practice. The main concern is haemorrhagic transformation, although posterior circulation infarcts appear to carry a somewhat lower bleeding risk compared to anterior ones. 19 Intravenous tirofiban is among the most commonly used periprocedural antiplatelet agents. In a retrospective series of 105 patients with posterior circulation occlusion (not specific to PTOCs), tirofiban use was associated with faster reperfusion, lower mortality and lower sICH rates, although no clear differences in long-term functional outcomes were observed. 110
Prognosis
Anterior tandem occlusions
The advent of EVT and eCAS has markedly improved outcomes for patients with ATOCs, both in terms of functional recovery and acute mortality reduction. In older IVT-only cohorts, favourable outcomes (mRS 0–2 at 90 days) were achieved in only around 20% of patients. 25 Contemporary data suggest that approximately 40%–45% of patients achieve functional independence (mRS 0–2) at 90 days, with excellent outcomes (mRS 0–1) in about one quarter. In the CERES-TANDEM 55 cohort, which represents the largest dataset to date (4053 patients), overall rates of independence and excellent recovery were 42.5% and 25.7%, respectively. 55 sICH occurred in around 11% of patients, similar to what has been reported in other large EVT cohorts. These figures highlight the progress achieved with modern endovascular approaches, although outcomes remain less favourable than in non-tandem large vessel occlusions, and 90-day mortality continues to range between 15% and 25%.2,111,112
Posterior tandem occlusions
Posterior circulation AIS has long been associated with worse outcomes and higher mortality compared with anterior circulation stroke, owing to the critical involvement of the brainstem and the limited capacity of the posterior fossa to accommodate oedema. Prognosis in PTOCs reflects both these inherent risks and the technical challenges of endovascular therapy. 18 Successful recanalisation is achieved in most cases, with recent series reporting rates of around 85%–90% of cases. Despite this, favourable functional outcome at 90 days (mRS 0–2) is attained in only about 35%–40% of patients.19,108
Anatomical factors such as vertebral artery dominance also influence outcome. Patients with stenosis or occlusion of a dominant VA generally have a poorer prognosis, whereas lesions affecting a hypoplastic or co-dominant artery are associated with milder presentations and better recovery. Bilateral VA occlusion, although less frequent, carries a poor prognosis. 112 Mortality remains substantial, with most contemporary studies reporting 90-day rates of 30%–40%.19,108
