Abstract
Headache is a frequent accompaniment of acute ischaemic stroke. The predisposing factors and underlying mechanisms are currently incompletely defined. We analysed prospectively collected data relevant to headache occurring at ischaemic stroke onset in consecutive patients included in the Henry Ford Hospital Stroke Data Bank. Patients with headache (HA+) and without headache (HA–) were compared for demographic factors, medical history, medications, examination findings, laboratory findings, and stroke localization and subtype. Group comparisons for categorical data were performed with χ2 test, and for continuous variables with two-sample t-tests. Stepwise logistic regression analysis, including all variables with P < 0.25, was used to define the independent predictors of onset headache. Three hundred and seventy-five patients had complete headache and clinical datasets and were included in the analysis (HA+, N = 118; HA–, N = 257). Multivariate analysis revealed that the independent predictors of HA+ were: infarct in the distribution of the posterior circulation [P = 0.0076, odds ratio (OR) 2.15, 95% confidence interval (CI) 1.23, 3.77], absence of history of hypertension (P = 0.0106, OR 0.48, 95% CI 0.27, 0.84), and treatment with warfarin at the time of the index stroke (P = 0.0135, OR 4.89, 95% CI 1.39, 17.21). The occurrence of headache at onset of ischaemic stroke is determined by posterior circulation distribution of the ischaemic event, absence of history of hypertension and treatment with warfarin at the time of the index stroke. These results suggest that preserved elasticity and maintenance of the intracranial vasculature in a relaxed state, in combination with coagulation system derangements, and activation of dense perivascular afferent nerves, play a role in the pathogenesis of onset headache.
Introduction
Headache often accompanies acute ischaemic stroke. Several prospective observational studies indicate a frequency of 17–34%(1–10). Despite this clinical observation, most aspects of headache occurring at the onset of ischaemic stroke (also called onset headache) are still controversial and incompletely defined. For example, the incidence of onset headache varies widely among epidemiological studies, and depends on a variety of factors, including criteria used for definition, method of patient selection, ischaemic stroke aetiology, affected arterial territory and infarct location (1–10). Additionally, there is lack of consensus as to what factors contribute to the development of this type of headache, and its pathophysiology remains unknown, despite theoretical suggestions that mechanisms related to migraine and tension-type headache are involved.
Headaches with a variety of characteristics have been observed in association with ischaemic stroke (1, 2). The onset headache, occurring in close temporal proximity with onset of the neurological deficit, still lacks an exact definition. Several prospective studies used different temporal definitions for onset headache (1–10). Some studies defined as stroke-related headache the one occurring from 48 h prior to 24 h after onset of neurological deficit, others from 3 days prior to 3 days after, and others avoided specific temporal definitions. The International Headache Society (IHS) in its recent classification (11) has not defined the exact timing of headache in connection with neurological deficit onset, but certainly it is clear that more than one type of headache may be observed in ischaemic stroke. The onset headache, still lacking a precise definition, may be different from delayed headache in ischaemic stroke. The former may be a reflection of the initial vascular occlusion and resultant ischaemia; the latter may be due to a variety of factors, including oedema, haemorrhagic conversion, delayed effects of products of thrombosis and ischaemia, or delayed derangement in the function of the trigeminovascular system.
Defining the factors which contribute to the development of onset headache, and clarifying their associations with this form of headache could lead to better understanding of the pathophysiology and mechanisms involved in its production. To accomplish this, we analysed prospectively collected clinical, laboratory and radiological data of consecutive patients with acute ischaemic stroke enrolled in the Henry Ford Hospital Stroke Data Bank (HFHSDB) (12).
Methods
Patient population
All patients with the diagnosis of acute ischaemic stroke, who were admitted to the Acute Stroke Unit of Henry Ford Hospital from 1987 until 1993, were entered into the computerized HFHSDB. Patients admitted to the general neurological wards or to other services were not enrolled in the HFHSDB. The criteria for admission to the Acute Stroke Unit were: (i) carotid territory stroke within 48 h of symptom onset or vertebrobasilar territory stroke within 72 h of symptom onset; (ii) stroke in the young (defined as <50 years of age); (iii) ischaemic stroke with fluctuating or progressing neurological deficit even after the above-mentioned time window; (iv) transient ischaemic attack(s) (TIA) (excluded from this study). The diagnosis of ischaemic stroke and TIA was made according to the criteria of the ad hoc committee on the classification of cerebrovascular disease (13).
The HFHSDB consisted of seven different forms where all data from each enrolled patient were systematically recorded. These forms were linked together by the medical record number and the symptom date. Each form contained the following information: Form 1, demographics; Form 2, history of the presenting stroke or TIA, including the presence or absence of headache at the onset of the ischaemic event history, past medical and surgical histories, alcohol and drug use, family history of stroke, neurological history, and medications on admission; Form 3, findings of the initial neurological examination; Form 4, initial laboratory and diagnostic evaluation; Form 5, medications and treatment while in hospital; Form 6, assessment at discharge; Form 7, discharge neurological examination. Forms 1, 2, 4 were collected on all subjects. The remaining four forms were collected on subjects until January 1991. Any inconsistencies were identified and corrected during the final clean-up process.
Inclusion criteria
In this analysis, we included all patients with acute ischaemic stroke, entered into the HFHSDB, for whom all clinical datasets, including information on the occurrence or not of headache at stroke onset, were available.
Exclusion criteria
We excluded patients unable to provide reliable information on the presence or absence of onset headache; the main reasons were aphasia, confusion, or depressed level of consciousness. We also excluded patients with incomplete or unavailable data on the location, mechanism(s) and type of the ischaemic event, and patients with diagnoses other than ischaemic stroke (i.e. haemorrhagic stroke, TIA, migraine with aura or migraine equivalents, arterial dissection not leading to cerebral ischaemia).
Headache
Headache related to ischaemic stroke was arbitrarily defined as headache occurring 24 h prior to 24 h after the onset of the neurological deficit. We did not follow the criteria set by the IHS because the HFHSDB forms were designed prior to the availability of the IHS classifications. Additionally, information on the presence, or not, of prior history of migraine was also recorded.
Clinical assessment
All patients underwent: detailed physical and neurological examination, head computed tomographic scan and/or magnetic resonance imaging scan, carotid duplex ultrasonography, echocardiography, laboratory tests, including complete blood count (CBC), platelet count, prothrombin time (PT), partial thromboplastin time (PTT), and, subsequently, transcranial Doppler ultrasonography (in selected cases) or cerebral angiography (in selected cases). The diagnostic evaluation was empirical, not standardized, and was tailored according to the individual patient's history and physical examination and the results of the initial standardized evaluation.
Anticardiolipin antibodies
Screening for anticardiolipin (aCL) was performed by a single laboratory (Division of Immunopathology, Henry Ford Hospital), using an internationally standardized ELISA method. IgG and IgM levels were reported in GPL and MPL, respectively. One GPL (PL = phospholipid) unit was defined as the cardiolipin binding activity of 1 μg/ml of an affinity-purified IgG aCL preparation from a standard serum (14). MPL units were defined in a similar manner. Sera whose aCL activity exceeded 100 GPL or 60 MPL could not be directly measured by ELISA method and had to be diluted with a single normal donor human serum. aCL positivity (aCL+) was defined as GPL > 10 or MPL > 7.5.
Stroke classification
The aetiological categories of ischaemic stroke were based on the Pilot Stroke Data Bank (15) with modifications. The following mechanisms were considered: (i) large-vessel atherosclerosis, (ii) cardiogenic embolism, (iii) lacunar infarct, (iv) other aetiology, (v) infarct of undetermined aetiology. The topographical classification was made according to the arterial territory of the cerebral infarction (carotid, vertebrobasilar or both).
Factors potentially associated with onset headache that were included in the analysis
In the analysis, we included multiple potential risk factors for development of onset headache, including: demographic and medical history factors (age, gender, race, hypertension, cigarette smoking, diabetes mellitus, history of myocardial infarction, angina, congestive heart failure, cardiac catheterization or coronary artery bypass grafting surgery, atrial fibrillation, valvular heart disease, history of TIA, history of stroke, history of migraine, cancer, and family history of stroke), medications (warfarin, antiplatelet agents, calcium channel- and β-blockers, digoxin, and insulin), physical examination and laboratory data (systolic and diastolic blood pressure on initial presentation, haematocrit, white cell count, and aCL, mechanism(s) of ischaemic stroke, and affected vascular territory (ies).
Statistical analysis
Statistical analysis was performed using the SAS version 6.12 (SAS Institute, Cary, NC, USA). Two-sample t-tests were used for continuous variables. χ2 tests were used for categorical variables. Stepwise logistic regression, including all variables with P < 0.25, was done to assess which variable or combination of variables were most likely to differentiate patients with and without headache at stroke onset (16). The testing level for significance was set at 0.05.
Results
Patients included in the HFHSDB totalled 1203. Of those, 570 patients had all form sets completed. Twenty-four patients were excluded because of incomplete data on stroke subtype. Of the remaining 546 patients with all data available, 116 patients were excluded because the final diagnosis was other than ischaemic stroke [intraparenchymal haemorrhage (n = 19), subarachnoid haemorrhage (n = 4), subdural haematoma (n = 2), TIA (n = 61), other (n = 30)]. Fifty-five additional patients were excluded either because data on headache occurrence at stroke onset were missing (n = 22) or because the patient was unable to provide the necessary relevant information (n = 33). Patients with acute ischaemic stroke and complete headache and clinical datasets (N = 375) were considered for the present analysis (Fig. 1). Of these, 118 (31.5%) experienced onset headache, and 257 (68.5%) did not.

Patient population enrolled in the Henry Ford Hospital Stroke Data Bank and included in the present analysis.
Univariate analysis
Patients with headache were significantly younger than their non-headache counterparts (age 61 ± 14 years vs. 66 ± 12 years; P = 0.001). The two groups did not differ in other demographic aspects, including gender or race (Table 1). Headache patients were less frequently hypertensive compared with the non-headache patients (65% vs. 75.3%; P = 0.039) (Table 1). Headache patients were more often migraineurs by history (8.8% vs. 3.3%; P = 0.026). There was no clear difference between the two groups in terms of other risk factors for ischaemic cerebrovascular disease, including diabetes mellitus, cigarette smoking, atrial fibrillation, prior stroke or TIA, family history of stroke, valvular heart disease, congestive heart failure, or cancer. In particular, there was no difference regarding angina, myocardial infarction or ischaemic heart disease in general, although a trend towards more frequent presence of angina in the headache patients was noted (26.1% vs. 17.8%; P = 0.069) (Table 1).
Risk factors for ischaemic cerebrovascular disease

Statistically significant difference.
The headache group was on treatment with warfarin at the time of the index stroke more frequently than the non-headache cohort (6.9% vs. 2.0%; P = 0.017) (Table 2). Analysis for use of potentially antimigrainous drugs at the time of stroke onset (e.g. β adrenergic blockers, calcium-channel blockers, aspirin) revealed no significant difference between the two groups. A trend towards less frequent use of either β-blockers or calcium-channel blockers was noted in the headache patients (20.8% vs. 29.5%; P = 0.102) (Table 2). There was no difference in the use of other medications including diuretics, oral hypoglycaemic agents, insulin or digoxin.
Medications

Statistically significant difference.
The levels of systolic and diastolic blood pressure at the time of presentation to the hospital did not differ between the two groups. Similarly, the levels of haematocrit were comparable in both groups. Headache patients more often tested positive for IgG aCL antibodies (32.1% vs. 12.1%; P = 0.045). The status of IgM aCL antibody seroreactivity was comparable in both groups (Table 3).
Ischaemic stroke in the posterior circulation was recorded in 44.3% of headache patients and in 23.7% of the non-headache patients (P = 0.001) (Table 4). The ischaemic stroke subtype did not play a significant role as the groups were similar for the proportion of patients with large vessel atherosclerosis, cardiogenic embolism, lacunar infarction, cryptogenic infarction or infarction of other aetiological mechanisms.
Physical examination and laboratory factors
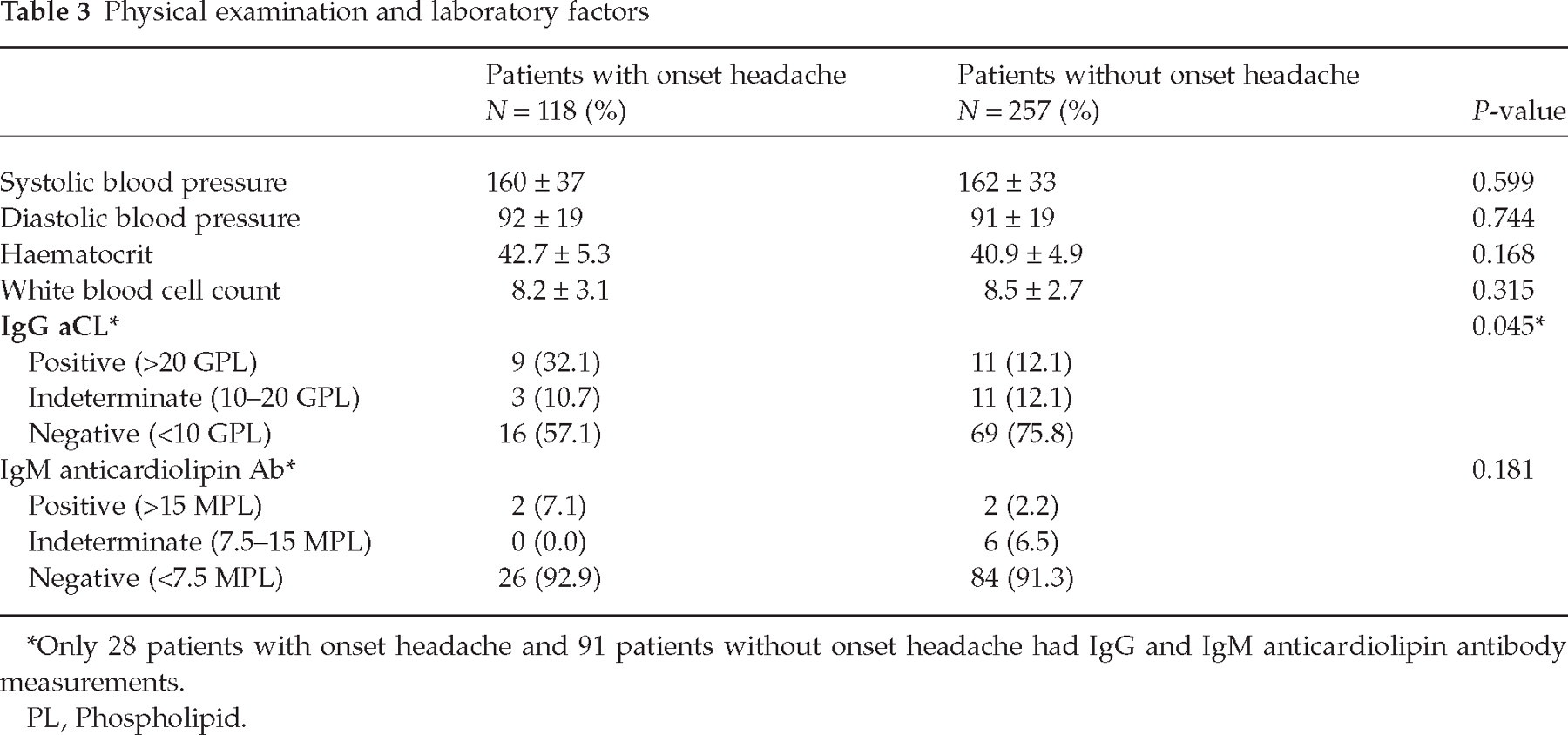
Only 28 patients with onset headache and 91 patients without onset headache had IgG and IgM anticardiolipin antibody measurements.
PL, Phospholipid.
Cerebral vascular territory and ischaemic stroke subtype
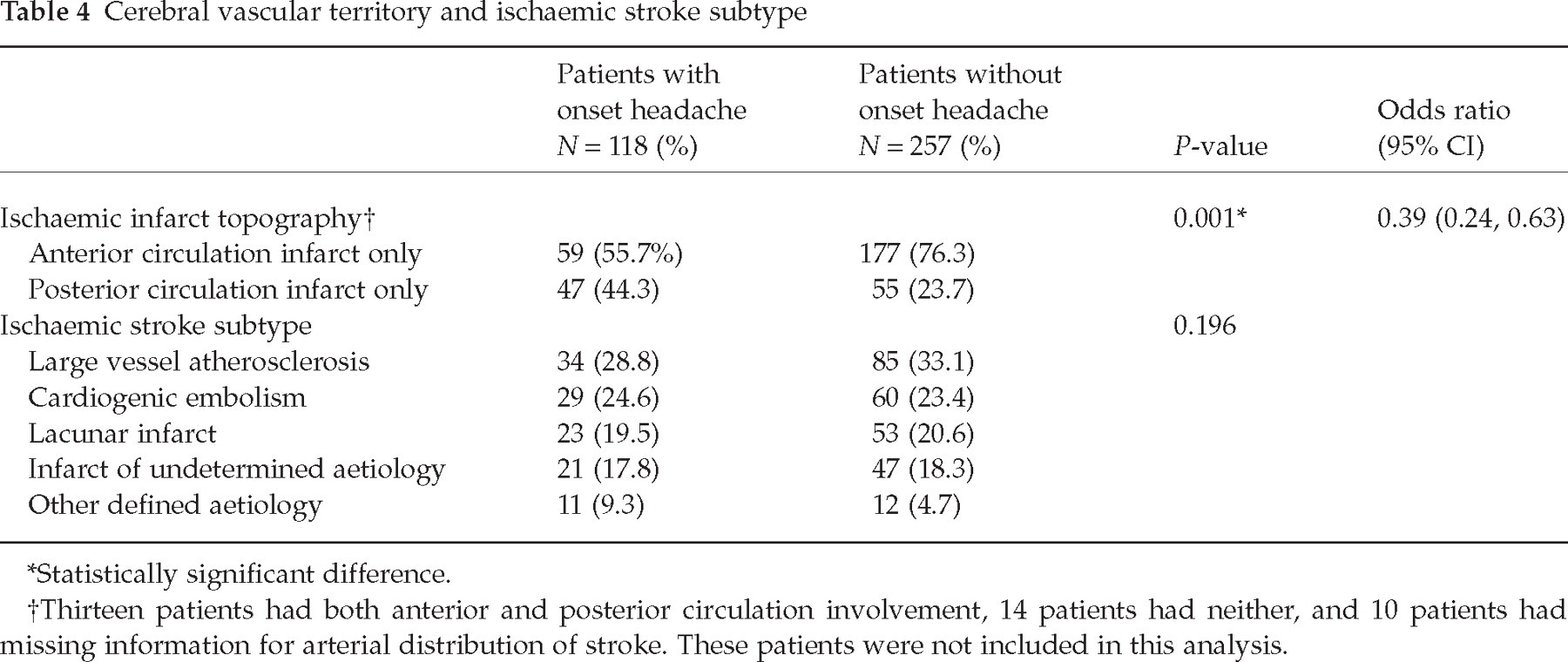
Statistically significant difference.
Thirteen patients had both anterior and posterior circulation involvement, 14 patients had neither, and 10 patients had missing information for arterial distribution of stroke. These patients were not included in this analysis.
Logistic regression analysis
All variables with a P-value < 0.25 were included as potential candidates in the stepwise logistic regression procedure. These variables included age, hypertension, angina, myocardial infarction, all ischaemic heart disease, history of migraine, current treatment with warfarin, diuretics, digoxin, β-blockers, calcium-channel blockers, haematocrit, infarct in the posterior circulation distribution, and stroke mechanism. IgG and IgM aCL antibodies were not included, even though their P-values were < 0.25, because large numbers of patients had missing values for these variables, and this testing was not done routinely but only in selected patients, with either undefined stroke aetiology or young patients with stroke.
Treatment with warfarin at the time of the index stroke [P = 0.0135, odds ratio (OR) 4.89, 95% confidence interval (CI) 1.39, 17.21], absence of hypertension (P = 0.0106, OR 0.48, 95% CI 0.27, 0.84), and infarct in the distribution of the posterior circulation (P = 0.0076, OR 2.15, 95% CI 1.23, 3.77) were the factors independently associated with headache at stroke onset (Table 5).
Independent risk factors for onset headache (logistic regression analysis)

Discussion
We found that posterior circulation distribution of the ischaemic stroke, absence of history of hypertension, and warfarin use at the time of the ischaemic event were the independent factors determining headache at onset of ischaemic stroke.
Patients with headache at the onset of stroke were more likely to have infarcts in the distribution of the posterior rather than the anterior circulation (P = 0.0076, OR 2.15, 95% CI 1.23, 3.77). In agreement with previous reports, this finding indicates that posterior circulation ischaemic events are more often accompanied by headache (5–10, 17). The reasons for this special predilection of posterior circulation ischaemic strokes to be accompanied by headache still remain incompletely understood. A denser perivascular innervation of the extracranial and intracranial vessels of the posterior circulation (18), ischaemia of the trigeminal nucleus leading to dysfunction and activation of the trigeminovascular system (19), ischaemia and dysfunction of the serotoninergic nuclei of the brainstem (20), or even ischaemia of the dura (a pain-sensitive structure), which may be partially supplied by the posterior cerebral artery (21), are potential explanations. Unfortunately, we do not have more specific imaging data for lesion localization that would allow correlation of headache production with specific ischaemic lesion location.
Patients with headache at stroke onset were less likely to have a history of hypertension compared with those without headache (P = 0.0106, OR 0.48, 95% CI 0.27, 0.84), a finding that stands in contrast to several previous prospective studies which did not detect such an association. Previous studies (3–5, 8), however, indicated that headache is less frequent in lacunar infarctions, which are more often encountered in hypertensive patients. One could thus consider that the association between hypertension and absence of onset headache is a result of a higher frequency of lacunar infarction in the hypertensive population. We detected no difference in the frequency of lacunar infarction (19.5% vs. 20.6%), or other stroke subtypes, between the two groups (Table 2) and therefore this explanation seems very unlikely. Chronic hypertension may result in decreased elasticity of the intracranial arteries, in part due to concentric intimal thickening and disrupted internal elastic lamina (22), and in part due to decreased nitric oxide (NO) production as a result of endothelial dysfunction (23, 24). NO generation from the endothelium is important to maintain the vasculature in a relaxed state (25). It is thus conceivable that normotensive patients with acute stroke are more prone to develop onset headache because the preserved elasticity and maintenance of the intracranial vasculature in a relaxed state could more easily activate perivascular nerve fibres, leading to headache production.
One intriguing finding was the association between onset headache and treatment with warfarin at the time of the index stroke. Despite the small numbers of patients, which raises suspicion that this may be a chance association and thus an unimportant finding, this factor was an independent predictor of headache at stroke onset, on logistic regression analysis (OR 4.49, 95% CI 1.39, 17.21, P = 0.0135). We are unaware of any prior studies having revealed a similar association. It is possible that during the acute vascular occlusion, pre-existing anticoagulation with warfarin may have prevented extension of a large vessel-occluding clot into the vasa nervorum, thus allowing activation of perivascular trigeminal fibres by circulating products of the ischaemic process or by the clot itself.
In contrast to previous studies (5, 7, 9, 10), we did not find any association between onset headache and female gender, angina or ischaemic heart disease, non-smoking status, or cortical localization of the ischaemic stroke. Previous reports also indicated that migraineurs are more likely to develop headache at stroke onset (6, 9, 10). In our study, migraine and younger age were significantly more frequent in patients with onset headache on univariate analysis, but they were not independent predictors of onset headache after multivariate analysis.
IgG aCL were more frequently found in patients with onset headache, while the other isotypes were equally present in the headache and the non-headache groups. No other previous large study on headache in stroke investigated this association (1–10). In the present multivariate analysis, however, we did not include aCL immunoreactivity, because not all patients were consistently tested for this. Patients were tested only after the initial diagnostic studies failed to define the stroke subtype, thus skewing the results towards stroke of undetermined aetiology or young patients without traditional stroke risk factors. A previous report indicated that 37% of patients with cerebral ischaemic events associated with high titres of IgG aCL had concurrent migraine-like headaches (26). The mechanisms by which aCL contribute to the development of headache are unclear and may involve a complex activation of endothelial cells resulting in NO and nitric superoxide production (27, 28) and enhanced thrombosis (29).
Our results indicate that ischaemic stroke in the distribution of the posterior circulation, absence of prior hypertension, and warfarin use at the time of the ischaemic event, are independent factors determining headache at ischaemic stroke onset. We hypothesize that cerebral vasculature with preserved elasticity and maintained in a relatively relaxed state, denser perivascular innervation and coagulation system derangements are important for headache production at ischaemic stroke onset.
Footnotes
Acknowledgements
Supported by NIH/NINDS Grant PO1 NS 23393.
