Abstract
An association between migraine and ischaemic vascular events, particularly ischaemic stroke, has been debated for many years. The pathophysiology of migraine has been explored in detail, and it is known that a dysfunction of brain cells and arteries is a major component of this disorder. The involvement of cerebral arteries during the migraine attack as well as the high prevalence of migraine among young individuals with ischaemic stroke has led to the hypothesis that migraine may be a risk factor for ischaemic stroke. Furthermore, there is evidence that the vascular nature of migraine is not limited to meningeal blood vessels and that migraine and overall cardiovascular disease may share aetiological pathways. The aim of this review is to summarize the epidemiological evidence that links migraine with ischaemic stroke and ischaemic heart disease and to discuss potential biological mechanisms.
MIGRAINE AND RISK OF ISCHAEMIC STROKE
Migraine is a very common neurovascular disorder (1,2). Over the last decades, an increasing body of publications has linked migraine, specifically migraine with aura, with ischaemic stroke. Several retrospective case-control (3–12), three prospective (13–15), as well as one cross-sectional cohort study (16), and some studies using data from stroke registries (17,18), have been published on the association between migraine and stroke risk. Three of the case-control studies found increased risk of ischaemic stroke among women <45 years of age who reported a history of migraine with aura (6,7,10), with risk estimates ranging from 3.8 (7) to 8.4 (10), and two additional case-control studies (3,5) found increased risk for migraineurs with aura among both genders. In one case-control study, migraine without aura was associated with increased risk of ischaemic stroke (6).
A meta-analysis of 11 case-control and three cohort studies published before 2004 indicated that the risk of stroke is increased in people with migraine (pooled relative risk 2.16, 95% confidence interval (CI), 1.89–2.48). This risk was consistent in people who had migraine with aura (relative risk, 2.27; 95% CI, 1.61–3.19) and migraine without aura (relative risk, 1.83; 95% CI, 1.06–3.15) (19).
Since the publication of the meta-analysis, two large-scale prospective cohort studies (14,15) and one population-based case-control study (12) were published. The first prospective cohort study used data from the Women's Health Study, which included over 39 000 apparently healthy women 45 years of age or older who were followed for an average of 9 years (14). This study found a 1.7-fold increased risk for ischaemic stroke (relative risk, 1.71; 95% CI, 1.11–2.66) for women who reported migraine with aura when compared with women without migraine. This risk was stronger in those who were 45–55 years of age (relative risk, 2.25; 95% CI, 1.30–3.91) and was not seen in older women. Migraine without aura was not associated with an increased risk of ischaemic stroke (relative risk, 1.01; 95% CI, 0.63–1.62 14).
The second prospective study used data from the Atherosclerosis Risk in Communities Study and included over 12 000 men and women aged 55 and older (15). Compared with participants without migraine or other headache, migraineurs had a 1.8-fold increased risk of ischaemic stroke (relative risk, 1.84; 95% CI, 0.89–3.82). The fact that the risk estimates did not reach statistical significance may be due to the headache and aura classification. Specifically, the category of ‘other headache with aura’, which showed a significant increased risk of ischaemic stroke (relative risk, 2.91, 95% CI, 1.39–6.11), probably contains participants with migraine with aura because the only other headache form that has an aura, i.e. cluster headache, is rare in population-based studies (20). Again, migraine or headache without aura was not associated with increased risk. This study also had a retrospective component, which showed in general higher risk estimates and also found increased risk for migraineurs without aura. As both prospective studies found an association between migraine and ischaemic stroke in participants older then 45 and 55, respectively, the migraine–stroke association may not be limited to younger individuals.
The Stroke Prevention in Young Women Study matched 386 women aged 15–49 years with first ischaemic stroke with 614 age- and ethnicity-matched controls (12). Subjects were classified as having no headache, probable migraine without visual symptoms, and probable migraine with visual symptoms. Compared with women without headache, those who reported probable migraine with visual symptoms had a 1.5-fold increased risk of ischaemic stroke (odds ratio, 1.5; 95 CI, 1.1–2.0), which slightly attenuated after control for stroke risk factors. This risk further increased for women who reported a probable migraine frequency of at least 12 per year (odds ratio, 2.3; 95% CI, 1.5–3.5) and for those who had begun having migraines during the prior year (odds ratio, 6.7; 95% CI, 2.3–19.2). Women with probable migraine without visual symptoms were not at increased risk for ischaemic stroke.
In the existing literature, when the interaction between migraine and other risk factors for stroke was evaluated, the risk was more than tripled by smoking (odds ratio = 10) (6), and quadrupled by oral contraceptive use (odds ratios = 13.9–16.9) (6,7). The combination of migraine, oral contraceptives and smoking further increased the risk (7,12).
In several studies, migraine has been associated with clinically silent brain lesions, mostly in the white matter (21–24). In a meta-analysis of neuroimaging studies, the odds ratio between migraine and white-matter lesions was 3.9 (95% CI, 2.3–6.7) (25). A large nested case-control study randomly selected patients with migraine with aura, with migraine without aura, and controls, who were frequency matched to cases for age, sex and place of residence and were studied by MRI (26). The authors found no significant difference between patients with migraine and controls in overall infarct prevalence (8.1% vs 5.0%). However, in the cerebellar region of the posterior circulation territory, patients with migraine had a higher prevalence of silent infarcts than controls (5.4% vs 0.7%; P = 0.02; adjusted odds ratio, 7.1; 95% CI, 0.9–55). The adjusted odds ratio was 13.7 (95% CI, 1.7–112) for patients with migraine with aura when compared with controls. In patients with migraine with a frequency of attacks of one or more per month, the adjusted odds ratio was 9.3 (95% CI, 1.1–76). The highest risk was in patients with migraine with aura with one attack or more per month (odds ratio, 15.8; 95% CI, 1.8–140).
The association between migraine and ischaemic stroke from retrospective and prospective studies is summarized in Table 1.

RR denotes relative risk. CI denotes confidence interval.
Presented are the relative risks of the fully adjusted multivariable model in cases when several multivariable models were presented.
Prospective indicates migraine assessment prior to the occurrence of a stroke event.
Retrospective indicates that migraine was assessed after a stroke event occurred.
Study included also men but a relative risk for the entire study and men was not shown.
MIGRAINE AND RISK OF ISCHAEMIC HEART DISEASE
The association between migraine and increased prevalence of cardiovascular risk factors and the observation that the vascular dysfunction of migraine may also extend to coronary arteries (27,28), has led to speculation that migraine may not only be associated with increased risk of stroke but also with ischaemic coronary events. Case reports and clinical data suggest an association between migraine and angina (27–29), especially the variant type and migraine. However, although several case reports (27–29) and large-scale cohort studies (30,31) found an association between migraine and chest pain that in some cases was associated with documented ischaemic electrocardiographic changes (28,29), an association between migraine and subsequent coronary events could not be firmly established (32).
Some studies have suggested a link between migraine and prevalent coronary heart disease. In a Spanish case-control study of 40 patients with acute myocardial infarction and matched controls, patients with myocardial infarction had a higher prevalence of migraine (25% vs 18.5%) (33). Two larger studies found associations between migraine and existing cardiovascular disease (34,35). The first was a community-based study of 3654 men and women from Australia aged 49 or older, which showed an approximately two-fold increased risk of prevalent myocardial infarction, stroke and angina for migraineurs (34). The second was a population-based study from the Netherlands, which identified 620 patients with migraine and 5135 controls without migraine. Migraineurs with aura had a four-fold increased risk of self-reported history of coronary heart disease prior to age 46 (35). In a large prospective matched cohort study, patients with migraine were at increased risk of subsequent non-myocardial infarction ischaemic heart disease (relative risk, 1.29; 95% CI, 1.15–1.44). The association between migraine and myocardial infarction demonstrated slight increased risk, which was not statistically significant (relative risk, 1.15; 95% CI, 0.96–1.38 36). In addition, one large study found an association between migraine and coronary events only in the subgroup of women with a family history of myocardial infarction (30).
In contrast, results from large cohort studies did not find an association between overall migraine (37) or migraine with aura (31) and coronary events. In another study of 905 women who were referred for coronary angioplasty, 24% reported a history of migraine (38). Compared with women without migraine, migraineurs had lower angiographic coronary severity scores and less severe coronary artery disease. After a mean of 4.4 years of follow-up of 873 of the participants, women with a history of migraine were not statistically significantly more likely to experience subsequent vascular events, although the relative risk estimate for any cardiovascular event was increased (relative risk, 1.21; 95% CI, 0.93–1.58). Information about migraine aura was not recorded in this study.
Recently, however, two large-scale prospective cohort studies found associations between migraine and ischaemic heart disease. Data from the Women's Health Study indicated an association between overall migraine and major ischaemic cardiovascular disease, including coronary heart disease, after a mean of 10 years of follow-up (39), which was not apparent with shorter follow-up (40). In this prospective cohort of 27 840 apparently healthy women, overall migraine was associated with a significantly increased risk of ischaemic major cardiovascular disease (non-fatal myocardial infarction, non-fatal ischaemic stroke, ischaemic cardiovascular disease death), myocardial infarction, coronary revascularizations, angina and cardiovascular disease death when compared with women with no history of migraine (39). More specifically, the results of the Women's Health Study indicate that the increased risk for any ischaemic vascular event is only apparent for women with migraine with aura (Table 2). Compared with women who did not report any history of migraine and after adjustment for traditional cardiovascular disease risk factors, women who reported migraine with aura had a significantly approximately two-fold increased risk of major cardiovascular disease, myocardial infarction, coronary revascularization, angina and cardiovascular disease death. Women who reported migraine without aura did not have increased risk for any ischaemic vascular events (39). The shape of the cumulative incidence curves for major cardiovascular disease showed a striking increased risk after approximately 6 years of follow-up (Fig. 1). Potential explanations for this interesting observation, however, are speculative and would have to involve a differential effect for migraineurs with aura. Besides chance, such explanations may include the design of the Women's Health Study, underlying biological mechanisms that increase the risk of CVD only after some time period, or a change in environmental factors during the 10-year follow-up of the study. The shapes of the association between migraine and aura status and risk of specific vascular events were similar (39).
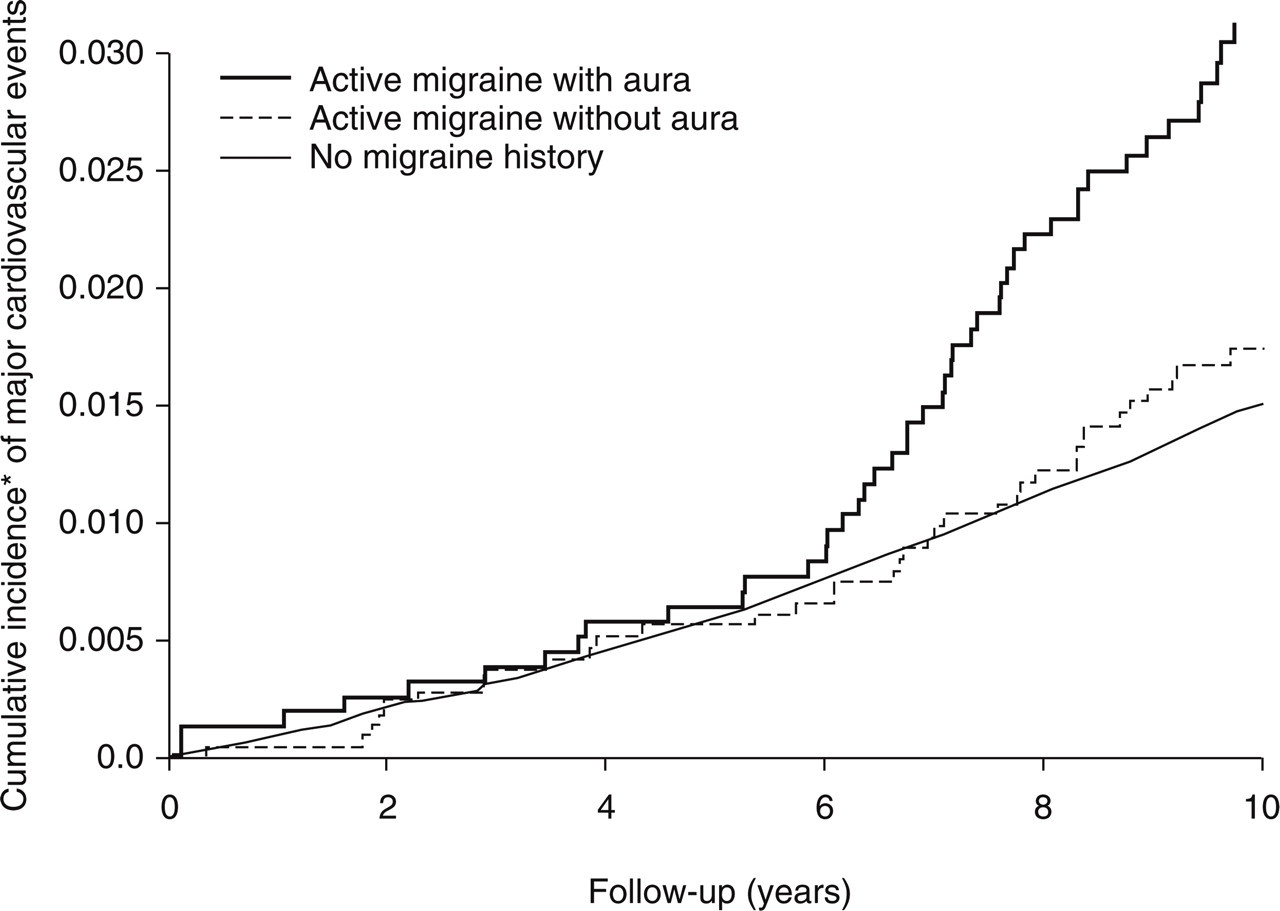
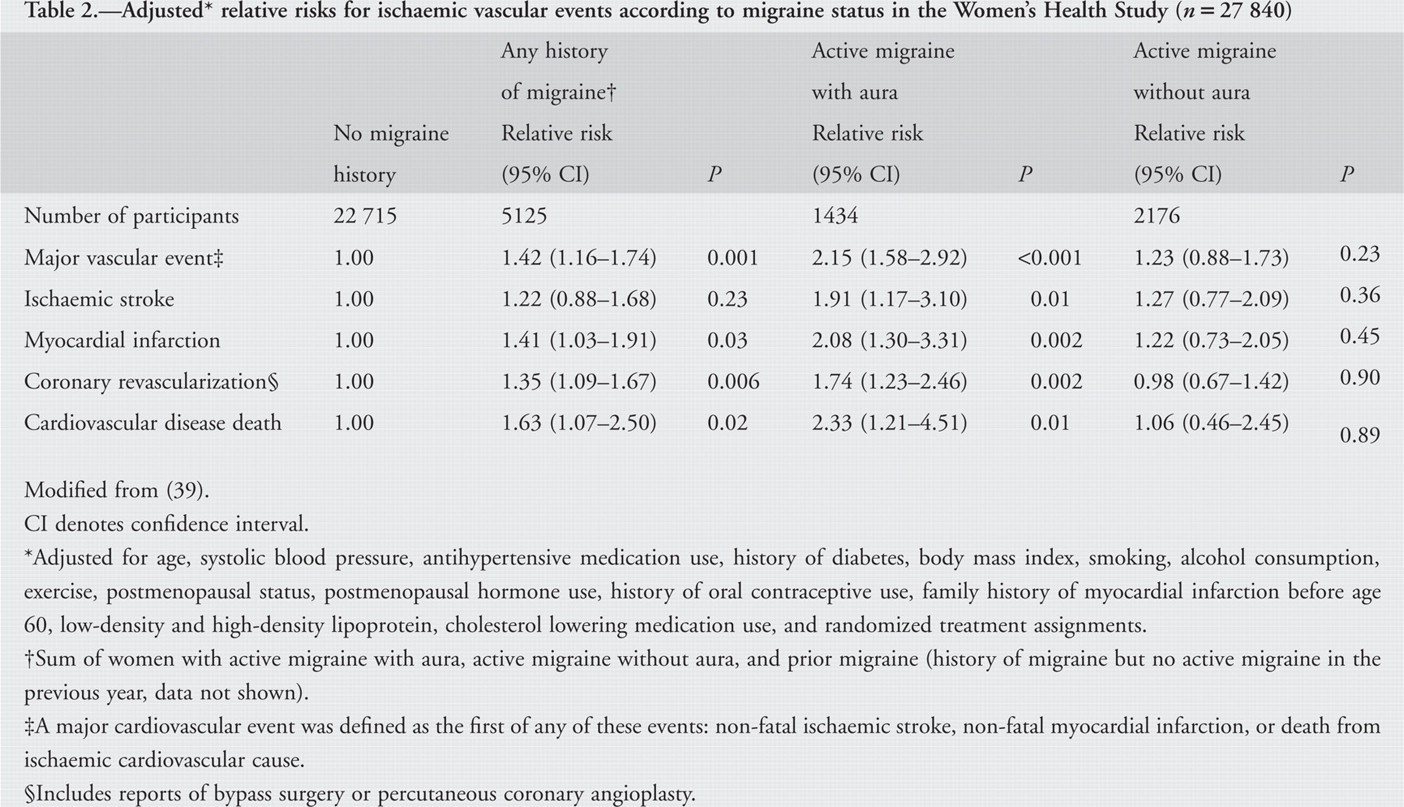
Modified from (39).
CI denotes confidence interval.
Adjusted for age, systolic blood pressure, antihypertensive medication use, history of diabetes, body mass index, smoking, alcohol consumption, exercise, postmenopausal status, postmenopausal hormone use, history of oral contraceptive use, family history of myocardial infarction before age 60, low-density and high-density lipoprotein, cholesterol lowering medication use, and randomized treatment assignments.
Sum of women with active migraine with aura, active migraine without aura, and prior migraine (history of migraine but no active migraine in the previous year, data not shown).
A major cardiovascular event was defined as the first of any of these events: non-fatal ischaemic stroke, non-fatal myocardial infarction, or death from ischaemic cardiovascular cause.
Includes reports of bypass surgery or percutaneous coronary angioplasty.
With regard to men, recent data from the Physicians’ Health Study indicate an association between overall migraine and major cardiovascular disease, which was driven by a significant 42% increase in the risk of myocardial infarction (41). In this study, 20 084 apparently healthy US male physicians were followed for a mean of 15.7 years, during which a total of 1449 major cardiovascular disease events occurred. Compared with non-migraineurs, men who reported migraine had an adjusted relative risk (95% CI) of 1.24 (1.06–1.46; P = 0.008) for major CVD, 1.12 (0.84–1.50; P = 0.43) for ischaemic stroke, 1.42 (1.15–1.77; P < 0.001) for myocardial infarction, 1.05 (0.89–1.24; P = 0.54) for coronary revascularization, 1.15 (0.99–1.33; P = 0.068) for angina, and 1.07 (0.80–1.43; P = 0.65) for ischaemic cardiovascular death. The cumulative incidence curves for major CVD are shown in Fig. 2. With regard to stroke, there was a significant modification by age, indicating that the men who were younger than 55 years of age had increased risk of stroke (relative risk, 1.84; 95% CI, 1.10–3.08), which was not apparent in the older age group.

POTENTIAL BIOLOGICAL MECHANISMS
The precise mechanisms by which migraine may lead to ischaemic vascular events are currently unknown and likely to be complex. Several hypotheses can be envisioned: (i) migraine may directly cause an ischaemic event (i.e. a migrainous infarct); (ii) migraine pathophysiology may affect the endothelial function and by this alone or in combination with existing local vascular pathologies increase the risk of stroke outside of a migraine attack; (iii) migraine is associated with an increased prevalence of risk factors for ischaemic vascular events; (iv) the link is caused by migraine-specific drugs; and (v) migraine and ischaemic vascular events are linked via a genetic component. With regard to ischaemic stroke, congenital heart defects, in particular patent foramen ovale, have also been discussed as potential biological mechanisms.
A migrainous infarct is a rare event since the International Headache Society introduced strict criteria in 1988 (42,43) and 2004 (44). The definition includes one or more aura symptoms associated with ischaemic brain lesion in an appropriate territory demonstrated by neuroimaging. This ischaemic brain lesion must occur in conjunction with a migraine attack in a patient with migraine with aura, which is typical of previous attacks except that one or more aura symptoms persists for >60 min. Other causes for ischaemia have to be ruled out (44). Migrainous infarcts that are caused by severe hypoperfusion during a migraine attack with aura, however, are rare and likely to be over-diagnosed (45). In addition, most ischaemic strokes among migraine patients occur between attacks and not during or shortly after a migraine attack with aura (4,6); therefore, a migrainous stroke is unlikely to explain the overall migraine–stroke association.
There is increasing evidence that migraine effects the vascular system not just in the brain but outside as well (46–48). For example, in a cross-sectional study of 50 patients with migraine and an equal number of matched controls without migraine, brachial artery diameter as well as brachial artery and femoral artery compliance were decreased in migraine patients. Furthermore, aortic augmentation index was increased in migraine patients (46). Recent results from the Atherosclerosis Risk in Communities Study indicate that middle-aged persons with migraine and other headaches were more likely to have retinopathy signs (48). At this point, however, it remains unclear whether endothelial dysfunction is a cause or a consequence of migraine or whether they coexist for other reasons.
Recently, migraine has been associated with a more unfavourable cardiovascular risk profile. The Genetic Epidemiology of Migraine (GEM) study found that compared with controls, migraineurs are more likely to smoke and are more likely to have a parental history of early myocardial infarction (35). The study also found that migraineurs with aura are more likely to have an unfavourable cholesterol profile, elevated blood pressure, report a history of early onset coronary heart disease or stroke, and have a two-fold increased risk of a high Framingham risk score-predicted 10-year risk of coronary heart disease even after adjusting for age (35,49). Furthermore, a recent study linked migraine frequency and severity with increased body mass index (50).
However, in most studies that evaluated the migraine–ischaemic vascular event association, the estimated relative risks were not attenuated after controlling for traditional cardiovascular risk factors (6,14–16,39,41). In addition, several studies indicated that the migraine–stroke association was particularly present in the absence of traditional cardiovascular risk factors (3,12,14,18). This may also explain why the association between migraine and stroke diminishes with increasing age in most studies: other major risk factors for ischaemic stroke, such as hypertension and diabetes, acquire greater importance with increasing age or interact with the mechanism by which migraine may lead to stroke. This would result in a relatively lesser influence of migraine as a risk factor for ischaemic stroke in the elderly. Furthermore, these data may suggest that migraine can increase the risk of ischaemic stroke only among those with intact vasculature.
Concerns have been raised regarding the cardiovascular safety of the use of migraine medications, especially the triptans, due to their vasoconstrictive ability and the occurrence of sensations that include chest pressure and tightness shortly after their use. Indeed, the use of ergots has been associated with white-matter lesions (26). However, recent reviews of the clinical, pharmacological and post-marketing cardiovascular safety data of triptans currently do not support a direct association of these drugs and ischaemic vascular events (36,37,51–55). It is important to note, however, that most of the data from clinical trials and clinical practice regarding the safety of triptans have been derived from patients without known cardiovascular disease (53). Contraindications for triptans already include existing coronary artery and cardiovascular disease and uncontrolled hypertension. With regard to an increased risk of ischaemic events due to triptan therapy, two large-scale studies from a health-care provider in the USA (37) and from the General Practice Research Database in the United Kingdom (36) found no association between triptans and ischaemic vascular events. Recently, a large claims-based study from the Netherlands evaluated whether overuse of triptans or ergotamin was associated with increased risk of ischaemic vascular events (56). The results of this study indicate that overuse of triptans even among patients concomitantly using cardiovascular drugs did not increase the risk of vascular events. In contrast, overuse of ergotamin was associated with an increased risk (odds ratio 2.55; 95% CI, 1.22–5.36) that was further increased for those patients who concomitantly used cardiovascular drugs. Therapeutic doses of either triptans or ergotamins were not associated with increased risk of vascular events (56). In addition to these data, all migraine patients are utilizing migraine-specific drugs and thus, unless there is a biological interaction between these drugs and migraine aura, use of these medications is an unlikely explanation for the association between migraine with aura and ischaemic vascular events.
In some patients, migraine and ischaemic stroke occur commonly as part of a distinct disorder, including, for example, cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL) (57,58) and mitochondrial myopathy, encephalopathy, lactacidosis and stroke (MELAS) (59). These, however, are distinct diseases only affecting very few migraine patients. These disorders are autosomal dominant diseases, pointing to a potential genetic component of the migraine–stroke association. Furthermore, the familiar hemiplegic form of migraine is also an autosomal dominant disorder (60–62). However, genetic aspects underlying the common forms of migraine have not been established. Because of the broad clinical spectrum of migraine, the interaction of several polymorphisms is likely to determine the manifestation and severity of migraine, and the effect of a single genetic mutation is expected to be small.
Migraine, in particular migraine with aura, has been associated with MTHFR C677T genotype (63,64), which is associated with increased homocysteine levels, a risk factor for vascular events. In recent years, a large number of studies have investigated the genetic basis underlying cerebro- and cardiovascular diseases, and numerous polymorphisms have been detected that show a firm association. For example, the following physiological systems are involved: beta-adreno-receptors (65), G-protein-coupled intracellular signal transduction cascades (66), the renin-angiotensin-aldosterone system (67), lipoprotein metabolism (68), coagulation cascades (69), and inflammatory cascades (70). Some of the prophylactic treatments for migraine, such as beta-blockers and calcium channel blockers, involve the cardiac system and thus a potential link between genetic markers of cardiovascular disease and migraine is possible, however, currently speculative.
There is increasing evidence that migraine, and particularly migraine with aura, is associated with increased prevalence of patent foramen ovale (PFO) (71). This link may be a potential explanation for the migraine–ischaemic stroke association but does not explain the association between migraine and coronary heart disease. In small case-control studies, PFO was about three times more frequent in migraine patients with aura compared with controls without migraine (72–74). In addition, migraine was associated with large PFO (75). However, these studies should be interpreted carefully because potential biases such as selection and detection bias are plausible and prospective population-based data are still lacking. It has also been reported from observational studies that closure of a PFO is associated with reduced migraine frequency (72,76). However, the only randomized and controlled trial of PFO closure in migraineurs does not support a causal link (77). The primary endpoint, cure of migraine, was not significantly different between PFO closure and control groups. There was a trend towards reduction of migraine frequency in the active-treatment group, which, however, was not significant after adjustment for the imbalance in migraine frequency at baseline. In addition, the procedure was associated with some serious adverse events.
In summary, there is good epidemiological evidence that migraine with aura is associated with an increased risk of ischaemic stroke, which appears to be stronger among the young but may persist in the elderly. There is recent evidence that migraine, and again migraine with aura, is associated with increased risk of any ischaemic vascular event, including coronary heart disease. This may lead to the hypothesis that migraine with aura is associated with a systemic vascular disorder. However, the precise mechanisms by which migraine with aura may lead to ischaemic vascular events are currently unknown and likely to be complex. At least from prospective data, there is no evidence that migraine without aura is associated with increased risk of any ischaemic vascular events. As the absolute risk of increase in ischaemic vascular events for patients with migraine with aura is considerably low, it is currently impossible to identify those at particular increased risk. Until there is further evidence unveiling the association between migraine and ischaemic vascular events, physicians should screen for and modify traditional major cardiovascular risk factors.
