Abstract
Therapeutic plasma exchange (PLEX) is a powerful and fast-acting immunomodulating therapy that is underutilized for autoimmune neurological disorders. Here, we present the largest collection of real-world experiences with PLEX procedures to date in the treatment of autoimmune neurological conditions, supporting its safety and clinical benefits with patient cases and corresponding patient videos. Our collective real-world experience with PLEX spans over 67 years, 90,210 procedures, and includes nine double-blind randomized controlled and unblinded studies serving as principal investigators. Case histories and videos of our patients demonstrate when and how PLEX should be used, identify barriers to using PLEX, and ways to overcome these barriers. Specific protocol details are shared of how to treat an acute or chronic phase of a disease. If used appropriately and early in the disease course for both acute and chronic progressive phases, PLEX can safely change the trajectory of many autoimmune neurological disorders in both outpatient and inpatient settings.
Plain language summary
What is plasma exchange (PLEX)?
PLEX is a procedure that removes and replaces plasma to eliminate harmful antibodies and immune factors in autoimmune and neurological diseases. It can work quickly and does not suppress or deplete the immune system.
What did we look at?
Our team combined real-world experience from our three centers, over nearly seven decades, including more than 90,000 PLEX procedures in 2,625 people with autoimmune neurological conditions. Most treatments (~89%) were safely treated in the outpatient setting.
Who may benefit?
We use PLEX for several conditions. Certain clues suggest a higher chance of improvement, such as being younger, having a shorter disease duration, and MRI scans showing active or ring-enhancing lesions.
Is PLEX safe?
We did not see any major complications caused by PLEX, including in the very young, very old, and critically ill patients. The most common issue was temporary low blood pressure during the procedure, which was easy to treat. Problems are far more likely when permanent central lines are used; we recommend peripheral veins and (when needed) short-term, temporary radial-artery access instead.
Why isn’t PLEX used more often?
Many people who could benefit do not receive PLEX because of insurance denials, limited local access to the procedure, uncertainty about the “right” protocol, and misunderstandings about safety.
How does PLEX compare with treatments?
PLEX is often less expensive than many biologic drugs used in these conditions. For example, one PLEX session at our center cost about $5,000 in 2021, compared with much higher per-infusion charges for some biologics.
What’s the takeaway?
Used early and appropriately and continued as maintenance, PLEX can change the course of autoimmune neurological diseases for many patients, with a safety record that supports outpatient care. It acts fast, avoids long-term immune suppression, and can be tailored to the individual.
Keywords
Introduction
Therapeutic plasma exchange (PLEX) has been part of neurology for nearly a century, yet in day-to-day practice, it remains underused for autoimmune disorders that flare or smolder despite standard care. Drawing on more than six decades of experience across three centers, over 90,000 procedures, published work, and our own experiences, we will discuss when and how to use PLEX, what to expect clinically, and how we keep patients safe in both outpatient and inpatient settings. We include cases, along with illustrative videos, to demonstrate common situations (steroid-refractory relapses, crises, and progressive disease), practical protocols for acute courses and maintenance, and information about how to identify patients who are likely to respond. We also discuss common obstacles (insurance coverage denials, misconceptions about risk, and variability in technique) and offer strategies to expand access to patients who would benefit from PLEX. Our aim is to make it easier to use PLEX as an effective, fast-acting therapy earlier and more confidently, so more patients can avoid preventable disability.
Method for reporting real-world experiences
In this review, we include real-world observations, recommendations, and present exemplar patients who were treated with PLEX at three centers and enrolled in our pilot, double-blind randomized controlled, and long-term studies. The patients presented here are representative of the clinical trials we and others have performed.
Inclusion criteria
The major requirement to utilize PLEX was a failure to respond and/or objective clinical deterioration while on conventional therapies. This documentation (in patients’ medical records) was required and thoroughly scrutinized by insurance companies to authorize PLEX for our patients. Copies of medical records summarizing clinical progress while on PLEX were required by insurance companies to authorize maintenance PLEX. It was, in essence, used as a last resort. In some instances, it was necessary to write multiple appeal letters, do peer-to-peer reviews, get attorney’s and media involved to exert pressure by publicizing the denial, all to get insurance company to authorize PLEX.
Objectives and outcomes
The objectives and outcomes in our studies, which are part of this report, are summarized in Table 1.
Nine double-blind randomized controlled and open-label PLEX studies.

Patient evaluation
A video exam was provided for each of the patients while on conventional therapies to show their condition before and after PLEX.
Reporting standards
We used the same protocols as we used in our published controlled trials and as summarized in Table 1.1–9
Ethics
The patients provided written informed consent for publication, consistent with CARE recommendations.
Data analysis
No statistical analysis for our real-world experience; however, statistical analysis was done in all the trials published by the authors. We report our overall impression based on our retrospective chart reviewed.
Role of PLEX in multiple forms of multiple sclerosis
In addition to its established role in acute, steroid-refractory cases, growing evidence supports the potential utilization of PLEX in multiple forms of MS. The following sections explore the current evidence base for PLEX in both acute and progressive MS, highlighting clinical outcomes, patient selection, and evolving treatment strategies. These and other related studies are summarized in Table 1.
Acute multiple sclerosis
Intravenous methylprednisolone, administered at doses of 500–1000 mg daily over 3–5 days, remains the standard first-line therapy for acute exacerbations of MS, owing its ability to inhibit pro-inflammatory cytokine production, limit immune cell infiltration, and suppress broader inflammatory pathways. However, a substantial proportion of patients, estimated at 25%–40%, fails to achieve meaningful recovery following corticosteroid treatment. 10 In such steroid-refractory cases, escalation to PLEX is warranted.
PLEX is often effective for steroid-refractory MS relapses with studies showing improvements in a significant percentage of patients, including those for whom standard high-dose corticosteroid therapy fails to control an MS relapse. 11 A total of 73.8% of severe MS relapses showed marked clinical improvement after PLEX, 7.1% showed mild improvement, and 19.0% had no improvement. The median expanded disability status score (EDSS) significantly decreased from 6.0 indicating a high level of disability to 4.0 by the time patients stopped PLEX. This improvement was sustained at the 6-month follow-up appointment.12,13 Two double-blind, sham-controlled randomized trial of PLEX in acute MS relapses established its efficacy. The investigators reported 42% of patients receiving true plasma exchange demonstrated moderate-to-marked functional improvement, compared with only 6% in the sham group. 14 Notably, differences became apparent as early as the third treatment session, supporting PLEX as a valuable rescue therapy for steroid-refractory exacerbations. Additionally, a North American multicenter, double-blind, randomized controlled study of 116 MS patients with an acute exacerbation measured the time to functional recovery using the Kurtzke EDSS. 2 All patients were treated with adrenocorticotropic hormone (ACTH) (40 Units (U) intramuscularly twice a day for 7 days, then 20 U twice a day for 4 days, followed by 10 U twice a day for 3 days) and cyclophosphamide (2 mg/kg orally for 12 weeks), while half also received PLEX (1.5 plasma volume exchanges carried out five times in 2 weeks then every 6 weeks). The median time to recovery was significantly shorter in the PLEX-treated patients compared to sham-treated patients (4 weeks vs 13 weeks). Additionally, EDSS scores remained increased by 0.79 in the sham-treated group at 3 months. This indicates that if PLEX is added to ACTH and cyclophosphamide to treat an acute attack, then the disability-recovery is hastened by 9 weeks. Based on these results, European and American guidelines consider patients with MS who have not responded to treatment with intravenous pulse methylprednisolone as candidates for PLEX as a second-line therapy.15,16
Progressive MS
While its use is well established in acute forms of MS, emerging evidence suggests that PLEX may also provide therapeutic benefit in progressive forms of MS, particularly in patients experiencing superimposed relapses or those with specific inflammatory profiles. We first reported positive results of our double-blind randomized sham-controlled study of PLEX in 1985. 17 Efficacy was well sustained in a long-term extension trial and finally a 25-year follow-up trial.5,18 Treatment benefits were more pronounced in patients with shorter disease duration and cerebellar-dominant symptoms. A meta-analysis further supported the potential benefit of PLEX in progressive MS. 19 By analyzing data from six clinical trials, the study found that patients receiving PLEX had a significantly reduced risk of neurological decline at 12 months compared to those who received standard of care or sham treatment.
A double-blind, randomized sham-controlled study conducted by the Canadian Cooperative MS group evaluated the efficacy of PLEX in patients with progressive MS. 20 The primary analysis compared the rates of treatment failure (worsening of EDSS score) among the groups. The study found no significant differences in treatment failure rates or other outcome measures among the groups. However, there was frequent and uncontrolled use of corticosteroids, thereby confounding the interpretation of the results. Patients who received corticosteroids due to clinical worsening of symptoms were classified as “treatment failure.” A total of 21/56 patients in the placebo group received corticosteroids, whereas only 8/56 in the treatment group did. When corticosteroids were controlled for in the analysis, the Canadian Study acknowledged that PLEX significantly (p = 0.005) delayed the time to treatment failure. Based on these study results, we offered PLEX to patients who had failed conventional therapies, that is, cortico-steroids or disease modifying therapies. Clinical outcomes were measured by using EDSS. We did not use MRI imaging for outcomes since this was not a funded study and also because MRIs were done infrequently and as needed at different times. Labs were drawn before and at each PLEX and included CBC, CMP. HIV and hepatitis panels were checked every 12 months. Complications were recorded in the progress notes as they occurred. For those patients who were not part of the study, a healthcare provider recorded their neurological examination and disability status, and we reviewed their medical records retrospectively. At one center (BK), baseline videos were recorded at the initiation of PLEX and at every 10 PLEX procedures in all patients. We used the same protocol as described in our controlled trials and they are listed in Table 1 for each of the disease state. The clinical outcomes we observed were consistent with those observed in our clinical trials, and we report them as a percentage of patients who improved (Table 2).
Experience with PLEX for each of the authors in patients with auto-immune neurological disorders.

Bhupendra O. Khatri, Milwaukee, WI.
Jay S. Raval, Albuquerque, NM, Chapel Hill, NC and Pittsburgh, PA.
Regina Berkovich, Los Angeles, CA.
Acute MS, acute multiple sclerosis; ADEM, acute disseminated encephalomyelitis; Anti-NMDARE, anti-N-methyl-d-aspartate (NDMA) receptor encephalitis; CIDP, Chronic inflammatory demyelinating polyneuropathy; CRPS, complex regional pain syndrome; GBS, Guillain–Barre syndrome; LEMS, Lambert–Eaton myasthenic syndrome; MG, Myasthenia Gravis; MS, multiple sclerosis; NMOSD, neuromyelitis optica spectrum disorder; NORSE, new onset refractory status epilepticus; Progressive MS, progressive multiple sclerosis.
In our experience, progressive MS patients need ongoing maintenance PLEX for a very long time. We currently manage nine patients who have been continuously treated with PLEX for the past 30 years (frequency once every 6–8 weeks) without any major safety issues. These patients were severely disabled for an extended period despite trying all conventional treatments, but with initiation of PLEX therapy they became, and remain, fully ambulatory. An example of such a patient is shown Supplemental Video 5.
Comparison of PLEX and approved high-efficacy therapies for auto-immune neurological disorders
When evaluating the safety in treating MS, it is appropriate to consider the rates and severity of adverse events (AEs) associated with PLEX in the context of other approved and/or widely used MS drug therapies. Some of the most-used immunotherapies for MS, include B cell-depleting drugs such as ocrelizumab, ublituximab, and ofatumumab, while other classes of drugs include natalizumab and cladribine.21–23
Ocrelizumab was the first drug in that B cell-depleting class, approved in 2017, while natalizumab, cladribine, ofatumumab, and ublituximab were approved in 2004, 2019, 2020, and 2022 respectively. Since the first drug approvals, there have been robust reporting on AEs and complications arising from their use (Table 3). 24 Some of these drugs when used in other immunomodulating conditions such as MG and CIDP can cause significant lymphopenia and hypogammaglobulinemia putting the patients at the risk for serious infections.25–27 There are known risks of reactivation of latent or occult infections and published recommendations advise careful screening and monitoring for potentially serious disease mediators such as herpes virus, John Cunningham virus (JC virus), human papilloma virus, hepatitis B (HBV), hepatitis C (HBC), tuberculosis, and cytomegalovirus (CMV). 28 Rare but serious side effects of immunotherapy include the risk of progressive multifocal leukoencephalopathy (PML), which can be fatal with no known therapy.29–31 Additionally, acute liver injury and reactivation of HBV are listed risks for several common immunosuppressive therapies. 32 As such, hepatic screening tests and regular monitoring during therapy is recommended. Additionally, there is insufficient guidance based on long-term data to inform how long a patient may safely receive these drugs. In contrast, PLEX has no such risk factors and can be safely used on a long-term basis.
Drug manufacturer published risks.

Published warnings for FDA-approved drugs for MS include increased risk of hematologic toxicity, hepatotoxicity, and malignancy. Data obtained from manufacturers’ websites, accessed 01 September 2025.
HBV, hepatitis B virus; MS, multiple sclerosis; PML, progressive multifocal leukoencephalopathy; TRALI, transfusion-related acute lung injury.
Finally, PLEX is less expensive than currently used biologics. The charge for one PLEX procedure in 2021 at our institution was approximately $5000.00. In comparison, one single ocrelizumab infusion was $106,720.00, one natalizumab infusion was $24,170.00 and one eculizumab infusion was $82,500.00.
Response rate and predictors of PLEX responsiveness
In the practice of medicine, it is always ideal to be able to determine responders to a therapy based on the disease state and patient-specific demographics using evidence-based medicine. The question is, is there a way to predict responders to PLEX? Based on our previously published study and the work of others,5,33 knowing the MS patient’s age, duration of disease, and progression of EDSS over the previous 2 years, it is possible to predict the degree and chance of improvement with PLEX. Younger patients with shorter duration of disease and on-going clinical worsening are more likely to improve with PLEX. Disability of patients who have been stable for a year or more are not likely to improve. Preserved deep tendon reflexes and early symptomatic improvement during PLEX sessions are associated with better outcomes. 34 MRI features, particularly gadolinium-enhancing (Gd+) magnetic resonance imaging (MRI) lesions, strongly predict PLEX response. Patients with large (>2 cm) tumefactive or ring-enhancing lesions are more likely to benefit. Among these, nearly 52% exhibited ring-enhancing lesions on MRI, which were significantly associated with favorable outcomes.35,36
Real-world experience with PLEX
Safety and effectiveness of therapeutic PLEX in autoimmune neurological disorders have been demonstrated in randomized controlled and uncontrolled studies.2–6,17,37–43 Our 67 years of real-world collective experience with 90,210 procedures in 2625 patients confirms these findings. In addition, our experience includes nine double-blind randomized controlled and open-label PLEX studies (Table 1). The most common diagnoses we have treated include multiple sclerosis (MS), chronic inflammatory demyelinating polyneuropathy (CIDP), myasthenia gravis (MG), Guillain–Barre syndrome (GBS) and other autoimmune neurological disorders as listed in Table 2. Eighty-nine percent of the 90,210 PLEX procedures were performed on an outpatient basis.
We believe PLEX remains underutilized for autoimmune neurological disorders and discuss the reasons why and how to overcome them. The most common barriers when utilizing PLEX including insurance denial, lack of adequate and safe vascular access, and inappropriate treatment protocols, among others discussed below. Specific protocol details are shared of how to treat an acute or chronic phase of a disease.
Underutilization of PLEX
We have come to view PLEX as a viable and effective first line of defense rather than a “last resort” as is commonly used in the United States. It is important to note, due to the chronic nature of some autoimmune conditions, some patients will require ongoing maintenance therapy. In addition, we currently manage nine patients who have been continuously being treated with periodic PLEX for the past 30 years (frequency once every 6–8 weeks) without any major safety issues. These patients were severely disabled for an extended period despite trying all conventional treatments, but with initiation of PLEX therapy they became, and remain, fully ambulatory.
Reasons for the underutilization of PLEX
The underutilization of PLEX as a therapeutic option is likely due the following obstacles:
Lack of widespread knowledge about the value of PLEX as a therapeutic option
Requirement for long-term and ongoing utilization of PLEX for durable effects
Insurance coverage denial when PLEX is medically necessary
Vascular access issues
Misconceptions about the safety of PLEX
Lack of understanding of PLEX mechanism of action
Lack of access to the PLEX procedure
The following representative clinical scenarios of real patients exemplify each of the reasons why PLEX is underutilized and ways to overcome them (Table 4). In each of these instances, PLEX completely changed the trajectory of their disease-courses, from being bedridden to being able to walk unassisted and, in some cases, becoming well enough to return to work and come off or reduce their immunosuppressive drugs.
Representative clinical scenarios of real patients.
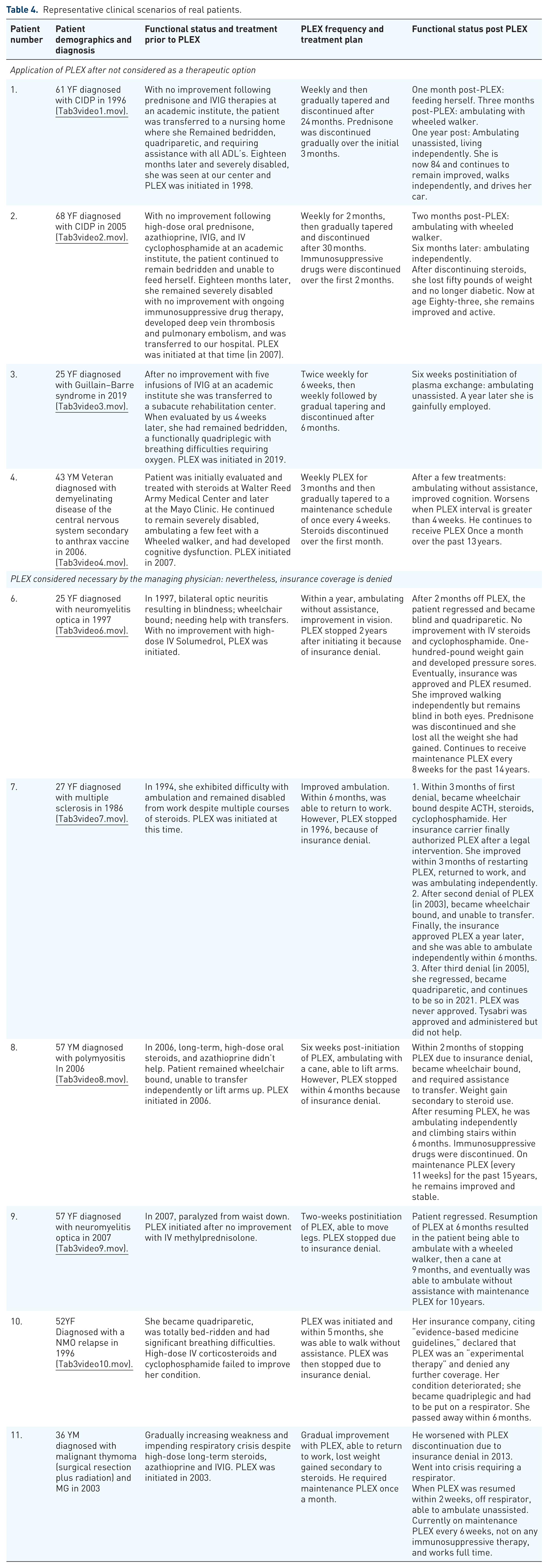
Lack of widespread knowledge of the value of PLEX as a therapeutic option
PLEX is a practical choice when relapses persist despite corticosteroids, IVIg, biologic therapy, or when those agents are contraindicated. It offers rapid, reversible control by clearing circulating pathogenic factors and can be delivered safely in outpatient programs, allowing teams to stabilize patients, bridge to longer-acting treatments, or maintain function when ongoing therapy is needed. The four cases that follow, representing several different neurologic disorders, illustrate these uses in practice.
In 1997, a 61-year-old African American woman experienced progressive, widespread weakness due to CIDP (Supplemental Video 1). Despite IVIG and high-dose long-term corticosteroid treatment, she remained bedridden. Her doctors conveyed that no further therapeutic options were available, and she was transferred to a nursing home. Eleven months later, her son learned about a patient with a similar diagnosis who had recovered with PLEX, and he brought his mother to our clinic. PLEX was initiated in September 1998. Her video examination just prior to first PLEX shows her cushingoid features due to chronic high-dose prednisone and severely disabled needing help with all activities of daily living. Gradual improvement followed each weekly PLEX treatment. By 3 months, she could walk with a walker and could walk unassisted within a year. Her steroids dosage was gradually decreased and discontinued 6 months after starting PLEX. Her final PLEX session was in September 2000. At 84 years old now, she remains fully functional.
A 68-year-old Caucasian woman with CIDP was treated with high-dose oral prednisone and azathioprine (Supplemental Video 2). Within 18 months, she was confined to a wheelchair and admitted to a teaching hospital. IVIG, steroids, and IV cyclophosphamide were administered without success. She was eventually discharged home in a bedridden state with pressure sores. While at home, she developed a pulmonary embolus and was transferred to our hospital for further care. She was cushingoid, diabetic, and quadriparetic (as shown in her video exam just prior to PLEX). PLEX was initiated in December 2008. Following 2 months of weekly PLEX, she could walk using a walker. In 6 months, she could walk without assistance, discontinued prednisone, lost weight, and was no longer diabetic. PLEX frequency was gradually reduced and discontinued 36 months after initial treatment. Now, 83 years old, she walks unassisted, drives her car, and lives independently.
In 2019, a 25-year-old Hispanic woman was hospitalized at a teaching hospital due to progressing ascending weakness and paresthesia (Supplemental Video 3). GBS diagnosis was confirmed. Although she had slight improvement post-IVIG infusion, she relapsed off IVIG, leading to near quadriplegia and confinement to bed. She was discharged to an acute rehabilitation facility but was transferred within a few days to our hospital because if of an impending respiratory crisis. Her video examination just prior to PLEX shows her quadriplegic. After six initial PLEX sessions (twice a week), she was able to walk unassisted. PLEX was discontinued within 6 months. She is currently doing well and holding a full-time employment.
In 2007, the Milwaukee Journal Sentinel newspaper reported on the hardships of a 43-year-old veteran diagnosed with a “demyelinating disease of the nervous system” at Walter Reed Army Medical Center in 2004 (Supplemental Video 4). The cause of his illness was Anthrax Vaccine Absorbed he had received. Long-term high-dose corticosteroids were ineffective, and with no other treatment offered, he remained cognitively and physically significantly disabled.
We contacted the patient, consulted his physician at the Walter Reed, and offered to treat him with PLEX. He agreed to a weekly PLEX trial. His video examination prior to PLEX shows him walking slowly with a cane. Within five treatments, he began thinking clearly, his strength improved, and he could walk without assistance. He has continued to receive PLEX once a month over the past 13 years. Decreasing the frequency of PLEX causes measurable physical decline. According to him, “PLEX gave me my life back.” He was able to enroll in a law school and is now a practicing lawyer.
Each of the above four cases indicates the importance of recognizing PLEX as a viable therapeutic option for autoimmune disorders, which have not responded to other therapies. The patients were coincidentally identified as candidates for PLEX, suggesting a more systematic assessment for candidacy should be considered when other treatments have failed.
Requirement for long-term and ongoing utilization of PLEX for durable effects
Acute courses of PLEX can deliver rapid improvement, but for some patients, the benefit remains durable only when treatment is continued on a scheduled, long-term basis. The case that follows describes a patient who has maintained neurologic stability and day-to-day function for more than 25 years with maintenance PLEX, underscoring the role of ongoing therapy when the clinical course warrants it.
A 26-year-old male was diagnosed with severe MS in 1996 (Supplemental Video 5). Despite injectable interferon and frequent IV methylprednisolone, he stayed wheelchair-bound, unable to transfer or feed himself. Choking led to aspiration pneumonia and extended hospital stays. Because he was getting worse despite being treated with an approved disease-modifying interferon beta-1a therapy and frequent IV steroid therapies, PLEX was offered. Thirty months later, in Nov 1999, he was treated with PLEX (initially twice a week for 3 weeks, then tapering to once every 6 weeks). By 6 years, he was able to walk without assistance, could do tandem gait, could hop on one leg and returned to work fixing roofs. He has been on maintenance PLEX for the past 25 years. Decreasing the frequency brings about objective physical decline.
For chronic autoimmune diseases such as MS, there currently is no cure, and patients typically require ongoing conventional therapies for decades. This is true for PLEX as well to achieve long-term durable effects. PLEX will not cure chronic autoimmune conditions, but if patients are appropriately managed on a PLEX maintenance program, patients can experience long-term disease control. PLEX protocol recommendations are in Table 5.
Typical PLEX protocol recommendations.
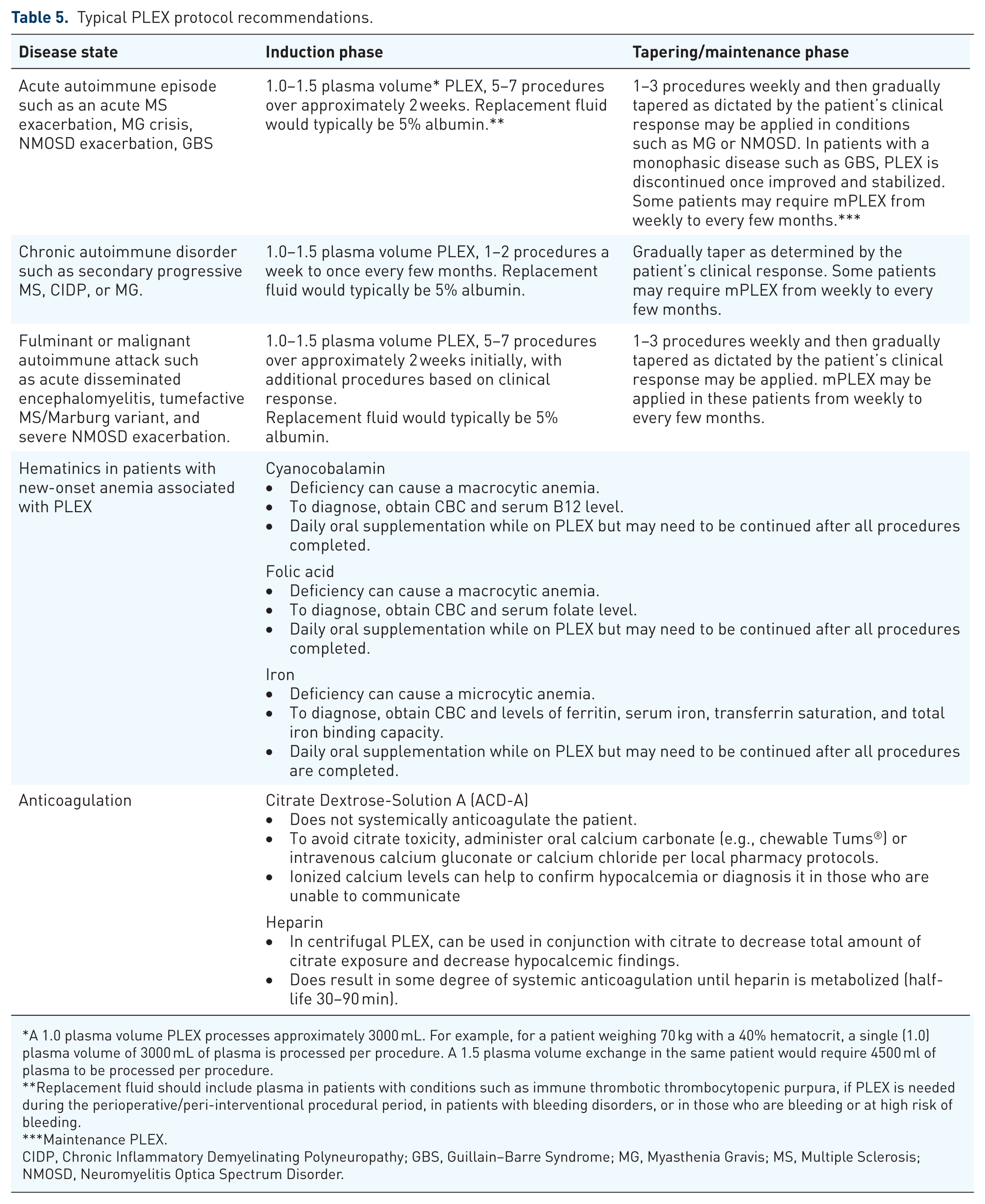
A 1.0 plasma volume PLEX processes approximately 3000 mL. For example, for a patient weighing 70 kg with a 40% hematocrit, a single (1.0) plasma volume of 3000 mL of plasma is processed per procedure. A 1.5 plasma volume exchange in the same patient would require 4500 ml of plasma to be processed per procedure.
Replacement fluid should include plasma in patients with conditions such as immune thrombotic thrombocytopenic purpura, if PLEX is needed during the perioperative/peri-interventional procedural period, in patients with bleeding disorders, or in those who are bleeding or at high risk of bleeding.
Maintenance PLEX.
CIDP, Chronic Inflammatory Demyelinating Polyneuropathy; GBS, Guillain–Barre Syndrome; MG, Myasthenia Gravis; MS, Multiple Sclerosis; NMOSD, Neuromyelitis Optica Spectrum Disorder.
Insurance coverage denial when PLEX is medically necessary
Despite the emphasis on evidence-based medicine in physician training, insurance approval can often be a significant limitation as to how patients are treated. Even with convincing published data, insurance companies either deny PLEX or approve it only for an acute exacerbation when other treatments have failed. Maintenance PLEX therapy, which could potentially allow patients to discontinue long-term immunosuppressive drugs and avoid their side-effect profiles, is usually not approved even if the patient initially responds well to PLEX. This often leads to patients having to take chronic steroid medication, which can cause complications such as cushingoid features, osteoporosis, hypertension, and diabetes, as seen in patients with CIDP and MG, whereas maintenance PLEX therapy (typically once a month) could potentially allow them to discontinue steroids and avoid such complications.
In 1997, a 25-year-old African American woman presented with bilateral optic neuritis-induced blindness and quadriparesis with significant demyelination in her brain and spinal cord (Supplemental Video 6). Initial MS diagnosis led to high-dose IV solumedrol treatment yielding no improvement. Twice-weekly PLEX was initiated, and this frequency was gradually reduced as she improved. Within 6 months, she was able to walk unassisted, and her vision improved. While on maintenance therapy and doing well for 2 years, her insurance company denied coverage for PLEX. Within 2 months off PLEX, she regressed and became blind in both eyes as well as quadriparetic. Intensive steroids and cyclophosphamide were ineffective, causing substantial weight gain and infected pressure sores.
Video evidence (her clinical examination before, during, and after stopping PLEX) was provided to her insurance company, and after multiple pleadings, PLEX was reapproved. Subsequent response to PLEX was gradual. Within 2 years and while on maintenance PLEX, she was able to walk without assistance. Unfortunately, her vision never improved, which may have been prevented if her insurance did not deny PLEX. Over the past 21 years, she has been receiving PLEX once every 6 weeks. In 2004, her serum tested positive for anti-aquaportin-4 autoantibodies (AQP4-IgG; measured using a cell-based assay with flow cytometric detection at Mayo Clinic Laboratories, Rochester, MN). She was able to discontinue steroids soon after resuming PLEX and lost all the weight she had gained.
In 1994, a 27-year-old Caucasian woman was significantly disabled due to worsening of her MS despite cyclophosphamide and multiple courses of cortico-steroids (Supplemental Video 7). She was started on weekly PLEX with remarkable improvement. She returned to work and based on qualitative objective assessment by a qualified neurologist, continued to improve while on maintenance PLEX until 1996 when it was discontinued due to insurance denial. This led to regression, and while off PLEX for 2 years, she remained confined to a wheelchair despite ACTH, steroids, and cyclophosphamide. Multiple appeals led to reapproval of PLEX, and she was able to walk independently and return to work within 8 months. In 2003, PLEX was again denied, and she became wheelchair bound. After nearly 10 months, PLEX reapproved in 2004. She gradually improved and was able to walk across a room independently. She continued to remain stable for 2 years while on PLEX when it was once again denied by her insurance company. She regressed off and is now (as of 2025) quadriparetic. Maintenance PLEX was never approved. Natalizumab was approved, but strength in her legs did not improve.
In 2005, a 57-year-old Caucasian man diagnosed with biopsy-confirmed polymyositis began long-term high-dose oral steroids causing significant weight gain (Supplemental Video 8). By September 2006, he was wheelchair-bound. PLEX was initiated at this time while prednisone was gradually tapered to 5 mg daily. Within 6 weeks of weekly PLEX, he was able to walk with a cane and could raise his arms. By March 2007, he walked unassisted until insurance denied PLEX. Off PLEX therapy, he began to decline. Within 2 months, he was confined to a wheelchair. Despite peer-to-peer appeal and videos demonstrating his improvement with PLEX, reapproval was not granted until legal intervention was taken. PLEX was restarted in June 2007. By December 2007, he was able to walk unassisted and could climb stairs. Now, 14 years later, he continues to receive PLEX once every 11 weeks, while not being on any immunosuppressive therapy.
In 2007, a 57-year-old Caucasian woman with seropositive NMOSD experienced a severe relapse causing her to be paraplegic (Supplemental Video 9). A course of IV solumedrol did not help. Twice-weekly PLEX was initiated, and within a month, she was able to take a few steps with a walker. Despite appeals and peer-to-peer talks, outpatient PLEX was denied by her insurance. She regressed, became confined to bed and it was then, with the patient’s consent, the local newspaper was contacted to report her story in the hopes that public pressure would help. They informed her insurance company that they were publishing her story in the newspaper the following day. The insurance medical director called the treating physician at home that evening before the story went public telling him that they had decided to approve PLEX. The patient resumed her therapy; within 6 months, she was able to walk with a walker and in 9 months, with a cane. In 2019, while on PLEX once every 3 weeks, she was able to take a few steps unassisted. She stopped PLEX and switched to eculizumab in 2019 (mainly because of vascular access issues) and subsequently to ravulizumab in 2024. She now ambulates with a walker.
A 52-year-old Caucasian female was bedridden, quadriplegic, and had significant breathing problems due to a NMO relapse (Supplemental Video 10). High-dose IV corticosteroids and cyclophosphamide failed to improve her condition. She was transferred to our hospital to receive PLEX. Within 5 months, she was able to walk without assistance. The video recording of her examinations before and after PLEX is remarkable. As she walked toward the video camera, she proudly proclaimed that her wish “to walk down the aisle was about to come true.” Sadly, her wish was not granted. Her insurance company, citing “evidence-based medicine guidelines,” declared that PLEX was an “experimental therapy.” Her condition deteriorated, she again became quadriplegic, had to be put on a respirator, and passed away within 6 months. Despite the video evidence and peer-to-peer appeals, PLEX was not approved.
In 2013, a 36-year-old Caucasian male with a diagnosis of MG was transferred to our care (Supplemental Video 11). Despite being on high-dose steroids, azathioprine, and IVIG, he remained bedridden with an impending respiratory crisis. An aggressive management with PLEX (three in a week) brought about significant improvement. He could talk, chew, walk, and lift his arms up. Four years later, while on maintenance PLEX (once every 6 weeks), he had lost all the weight he had gained due to steroids and was off all immunosuppressive drugs, and he was able to return to work. At this time, his insurance company, citing “evidence-based medicine,” declared that there was no role for “maintenance PLEX” in MG and, thus, denied coverage.
Off PLEX, the patient significantly worsened and was put on a respirator. He was then transferred again in an ambulance to our hospital. As an inpatient, PLEX once again brought about a marked improvement. Within 2 weeks, he was walking unassisted and discharged home. Once again, the insurance company denied outpatient “maintenance therapy” for him.
A peer-to-peer appeal was not successful until an academic MG specialist intervened and called the insurance company, stating the patient may die off PLEX. The insurance company finally gave approval, and the patient was transferred to our hospital for ongoing PLEX. The patient continues to do well on maintenance PLEX once every 6 weeks and minimal immunosuppressive drugs (prednisone 5 mg daily) and is working full-time.
The above cases illustrate that insurance barriers are a major issue for patients to access PLEX. With any type of specialty medication, it can be challenging to obtain insurance approval. Usually, there are several steps required before insurance approves a specialty medication or PLEX. In some instances, using the tactics described above, we have had a good success rate in obtaining insurance approval for PLEX.
Vascular access issues
PLEX requires adequate vascular access for removing blood from circulation (usually at the rate of 60–100 ml per minute) and typically, with another venous access to return cells and replacement fluid to the circulation (Supplemental Videos 12 and 13). PLEX in the United States is typically performed under the direction of nephrology or transfusion medicine. While peripheral vascular access is preferred, many patients usually require an indwelling central line or embedded ports for vascular access to perform PLEX.
This approach is a leading cause of serious complications related to PLEX. 44 One of the authors (JR) strictly uses all central lines for vascular access and has experienced complications in 20% of their patients. These complications include infection, thrombosis, bleeding, and catheter kinking. At another center, outflow vascular access is successfully accomplished by placing a 16-gauge angio-catheter in an antecubital vein. A tourniquet is placed above the venous access; a heating pad is placed over it; and the patient is asked to squeeze off and on, ensuring adequate blood flow. However, in about one-third of the patients, this approach fails. In these patients, a temporary 18-gauge arterial catheter is inserted in the radial (preferred) or brachial artery which usually allows a blood flow of 80–120 ml per minute. 45 (Supplemental Video 12). The catheter is removed at the conclusion of the PLEX procedure and pressure is applied at the site until it stops bleeding (rarely, it may require an ice bag over the site to stop bleeding). A pressure bandage is applied, which can be removed by the patient typically 4 h later. The placement of an arterial catheter takes just a few minutes and may be facilitated with use of an ultrasound machine for easier placement. The inflow access is via an 18-gauge angio-catheter placed peripherally in a vein, not on the same arm as the out-flow access. One of the physician-assistants (PA), who had never inserted an arterial catheter before, learned how to do this procedure within a week. She now routinely inserts them via ultrasound guidance in patients, and it takes less than 5 min (Supplemental Video 13). We have thus far inserted over 20,000 temporary radial or brachial artery catheters without any major complications. In a few patients who required long-term maintenance PLEX, a temporary catheter is inserted in the radial or brachial artery over 400 times, in each, without any complications.
Based on our experience and that of others, and in keeping with the American Society for Apheresis Choosing Wisely recommendations, we recommend avoiding the use of central venous vascular access. 46 Indwelling catheters in these blood vessels are associated with serious complications, such as infection and thrombosis. In a handful of patients over the years, in whom peripheral venous and arterial access was difficult, an indwelling central line using Permcath™ chronic silicone oval catheter is used for a long-term vascular access. It provides an average blood flow rate of 150-300 mL/min. While ports or fistula for vascular access may be utilized, radial artery catheterizations should be the preferred way.
Misconceptions about the safety of PLEX
Concerns about hypotension, bleeding, infections, and “risky lines” often reflect early experience or the use of long-term central venous access rather than the procedure itself ().47,48 In modern programs, using peripheral access when possible, citrate anticoagulation, and standardized monitoring, AEs are uncommon and typically self-limited 49 ; the section below outlines practical steps to prevent safety issues, based on our clinical experience with PLEX.
We have not had any fatalities, or any major complications related to PLEX. The youngest patient we have treated was 18 months old and the oldest 93 years old. We have treated severely debilitated patients, patients in septic-shock, those on ventilators, as well as those who were severely immunocompromised. Some of our patients have been on PLEX maintenance therapy for more than 30 years. Most of them were initially prescribed long-term immunosuppressive drugs, which were able to be discontinued once they started receiving PLEX.
The most common problem is transient hypotension occurring in approximately 33% of patients at some point during their PLEX therapies. This is readily corrected by rapid infusion of normal saline and/or 5% albumin solution and by placing the patient in Trendelenburg position. If hypotension occurs during subsequent procedures, we consider pretreating the patient with 0.3 mg of atropine IM. Anticoagulants, such as citrate dextrose-solution A (ACD-A), used during PLEX can cause mild/moderate AEs (usually tingling and numbness around lips and in extremities). These are typically temporary and manageable with calcium supplementation or flow rate adjustments. Patients on systemic anticoagulants require close monitoring. When central indwelling lines are used for vascular access, infection, and venous thrombosis are the most common and serious AEs. Mild iron deficiency anemia with increased PLEX procedures can occur due to the small but additive blood losses that occur during each procedure. After a series of 12 weekly PLEX procedures in 12 randomly selected patients, there was a significant decline in vitamin B12 (mean decrease of 496 pg/mL; p<0.001) and folate levels (mean decrease of 5.3 ng/ml; p < 0.001). 18 We now routinely administer supplemental folic acid and Vitamin B12 following PLEX (Table 5). A transient increase in prothrombin time (mean change of 20.7 s) followed each PLEX when using albumin as a replacement fluid. 18 However, no bleeding, cardiac complications, or other major medical problems were encountered as a result of the procedures.
Lack of understanding of PLEX mechanism of action
PLEX is an immunomodulatory therapy with multiple modes of action that does not involve depleting immune cells, as supported by growing scientific evidence. 50 PLEX effectively and rapidly removes large molecular weight substances from the plasma, including circulating antibodies (about 60%–70% with a 1.0–1.5 plasma volume procedure), complement components, immune complexes (leading to improvement in monocyte/macrophage function), cytokines, chemokines, endotoxin, lipoproteins, and many other different molecules from circulation that may be pathogenic (Figure 1). PLEX’s therapeutic effect can occur within hours. For example, one of our patients remained significantly disabled for several months while on pyridostigmine, steroids, and azathioprine. Halfway through her first PLEX procedure, her ptosis improved (Figure 2), and she could lift her arms. While such a rapid response is atypical, it illustrates the kinetic variability of clinical responses to PLEX therapy.

Possible mechanisms of PLEX. (a) Pathological antibody removal, (b) stimulation of the proliferation of B and plasma cells, (c) removal of immune complexes, (d) missing plasma component replacement, (e) cytokine removal, lymphocyte number alterations, (f) T and B cell counts, (g) T regulatory cell number and T suppressor activity increases, (h) T-helper cell type 1/2 correction.

Significantly disabled MG patient while on pyridostigmine, steroids, and azathioprine. Halfway through her first PLEX, her ptosis improved.
Other viable mechanistic hypotheses include possible alterations in lymphocyte proliferation and function that could sensitize these cells to immunosuppressant and chemotherapeutic agents, as well as other alterations in the immune system, including changes in B and T cell numbers and activation, increased T-suppressor function alteration in T-helper cell type 1/2 (Th1/Th2) ratio, and suppression of IL-2 and IFN-γ production.51–54 PLEX has been shown to downregulate activated monocytes and restore regulatory T cells,34–36 which are important controllers of autoimmune responses.
Multiple sclerosis
Understanding the mechanism of action in PLEX, which involves removing and replacing plasma, is crucial for explaining its efficacy in treating MS. Most disease-modifying therapies (DMTs) for MS work by modulating the immune system, primarily by suppressing the activity of autoreactive immune cells that attack the myelin sheath in the CNS, thereby reducing inflammation and slowing disease progression. This is achieved through mechanisms like interfering with immune cell migration into the brain, altering the balance of T cell subsets, or directly depleting specific immune cell populations depending on the drug used. The immune-driven nature of MS makes PLEX a valuable treatment option. PLEX removes harmful proteins (like antibodies) from the blood plasma that are attacking the myelin sheath, while DMTs work to prevent the immune system from attacking the myelin sheath. By removing these proteins, PLEX aims to stop the attack on the myelin sheath and potentially reduce the severity and duration of MS relapses (flare-ups). Thus, PLEX is more effective in minimizing damage from an ongoing attack as opposed to preventing future attacks.
In contrast, steroids alone often insufficiently resolve autoantibody-mediated pathologies. 55 Regulatory T cells (Tregs), which normally suppress autoreactive immune cells, are functionally impaired in MS. This impairment contributes to the autoimmune attack on the central nervous system characteristic of MS. Tregs are crucial for maintaining immune system balance and preventing autoimmune diseases, making their dysfunction in MS a significant factor in disease progression. 56 PLEX can restore regulatory immune cell populations, such as regulatory T cells (Tregs), and favorably modulate the balance between Th1 and Th2 helper T cell responses, contributing to a more regulated immune environment. These changes may contribute to the observed clinical benefits of plasmapheresis in some MS patients. 57
Sero positive or negative myasthenia gravis
Several treatment options are available for people with MG; however, based on our experience, PLEX has been the most effective not only in crisis but also in treating significant weakness affecting activities of daily living. PLEX offers a quick improvement in muscle strength, usually within a few hours (Figure 2) to a few days after treatment. PLEX improved symptoms in a varying degree in 100% of our patients. Some of these patients required maintenance therapy. PLEX protocol for MG is described in Table 5.
PLEX in MG brings about multiple immunological changes due to removal of harmful components from the plasma such as autoantibodies and particularly the acetylcholine receptor (AChR) antibodies and muscle-specific kinase (MuSK) antibodies, which interfere with neuromuscular transmission in MG. It also eliminates circulating immune complexes, which contribute to inflammatory and autoimmune processes. PLEX can remove cytokines and other inflammatory mediators from the plasma, leading to a reduction in the inflammatory response associated with MG exacerbations. PLEX also has an impact on the complement system (inhibition due to removal of AchR antibodies) and by decreasing the levels of complements which are involved in inflammatory responses, including in MG.58–60
One study reported that PLEX led to decreased levels of pro-inflammatory cytokines like IL-17A and IFN-γ, while increasing levels of the anti-inflammatory cytokine IL-10. This suggests a potential shift toward a more balanced immune response. 61
Neuromyelitis optica spectrum disorder
PLEX is a well-established treatment for acute attacks of NMOSD. It is often used when patients do not respond adequately to initial treatments like corticosteroids or intravenous immunoglobulin. The rapid removal of these immune system components by PLEX often leads to a quick improvement in neurological disability. 62 In fact, PLEX effectiveness was associated with the duration between disease and the initiation of PE, and the optimal timing for PE initiation is 8–23 days after the onset of the disease. 63 A short delay to initiate plasma exchange is the strongest predictor of outcome in severe attacks of NMO spectrum disorders early initiation of PLEX during severe attacks of NMO-SD. Perceiving PLEX as a rescue therapy only after steroid failure could be deleterious. 62 Our real-world experience over the years has been similar and is reported in Table 2. In our long-term study of PLEX, seven patients met the criteria for NMO diagnosis, and all were positive for antibodies against aquaporin-4. Over a mean of 7.1 years (range, 2–16), these patients received between 21 and 154 PLEX treatments (mean, 76). Although treated with maintenance PLEX, five out of the seven patients improved by more than one point on the Expanded Disability Status Scale. Interruption of PLEX in five patients resulted in clinical worsening. When PLEX was restarted in three out of the five patients who experienced a cessation of PLEX, these patients either stabilized or improved. The two patients who did not restart the PLEX protocol (due to insurance denial) died. Patients treated with PLEX also experienced a reduction in the number of NMO exacerbations. Finally, stabilization of the retinal nerve fiber layer thickness was observed while on PLEX 4 (Supplemental Video 6).
PLEX induces immunological changes in NMOSD patients’ removal of AQP4-IgG antibodies; reduction of inflammatory mediators; and removal of other inflammatory factors involved in NMOSD pathogenesis, such as complement components and various pro-inflammatory cytokines. These mediators contribute to the damage of the optic nerve and spinal cord that is characteristic of NMOSD. Interleukin (IL)-22 secreting CD4+ T (Th22) cells and IL-22 are involved in the pathogenesis of autoimmune disease. The proportion of Th22 cells, Th17 cells and serum IL-22 were increased in patients with NMO and play an important role in the pathogenesis of NMO and MS. 64 PLEX helps to reduce these elevated levels.
Guillain–Barre syndrome
PLEX is a well-established treatment for Guillain–Barré syndrome (GBS). BK was one of the principal investigators in a study that established for the first time an effective therapy for GBS. (North American Study of Plasmapheresis in Acute Guillain–Barre Syndrome. Neurology 1985;35(8):1096-104). The treatment’s efficacy has been demonstrated in multiple studies, showing significant benefits in terms of muscle strength improvement, reduced need for mechanical ventilation, and faster recovery times in patients with severe GBS. Some studies suggest that PLEX can be more effective in patients who do not respond adequately to IVIG. 65 (Supplemental Video 3).
GBS is characterized by the presence of autoantibodies that target the myelin sheath or axons of peripheral nerves. These antibodies can activate the complement system, leading to further nerve damage and inflammation. PLEX physically removes these circulating autoantibodies and complement components from the blood, reducing the immune system’s attack on the nerves. PLEX also causes an increase in T suppressor lymphocytes and Fc receptor-bearing lymphocytes alongside a decrease in B lymphocytes following plasmapheresis in a GBS patient, suggesting a restoration of the balance among peripheral lymphocytes. 66 PLEX also removes inflammatory mediators like cytokines, which play a role in GBS pathogenesis. Reducing these factors may help to mitigate nerve inflammation. 67 Some studies suggest PLEX may influence the frequency and function of regulatory T cells, which are important in maintaining immune homeostasis. 68 We and others have reported PLEX to be a safe and effective therapy in children (19 months–16 years) with acute inflammatory polyneuropathy.7,69
Chronic inflammatory demyelinating polyneuropathy
PLEX is a recognized treatment for CIDP. 70 Removal of pathogenic factors by PLEX is thought to be responsible for the observed improvement in nerve conduction velocity and clinical symptoms in patients with CIDP. 71 Our collective experience over the years is similar to what has been reported. Long-term maintenance PLEX may be required in some, and it also allows for reduction in cortico-steroids and immunosuppressive drugs (videos Patient 1.wmv; Patient 2.wmv).
The precise mechanisms of its action in CIDP are still under investigation; however, PLEX effectively removes circulating autoantibodies, such as immunoglobulin G (IgG), IgA, and IgM, which are implicated in the pathogenesis of CIDP. It also reduces the levels of inflammatory mediators, such as cytokines (e.g., TNF-α, IL-6), complement components, and adhesion molecules that contribute to nerve inflammation and damage in CIDP. PLEX modulates the immune system, including influencing immune cell populations and function, potentially by inducing regulatory T cells and altering cytokine patterns. The reduction in inflammatory cytokines achieved by PLEX may help create an environment conducive to the induction of regulatory T cells.57,68,72
Lack of access to the PLEX procedure
Gaining access to PLEX is a challenge, and this remains one of the most common causes of underutilization of this therapy (because it requires specialized equipment, facility, and trained staff, and it is therefore it is usually available at tertiary centers). Over 95% of the 70,000 PLEX procedures performed by BK were on an outpatient basis. Even though the majority of therapeutic PLEX performed in the United States are on patients with neurological disorders, very few neurologists run their own outpatient PLEX units. Evaluation and management of patients requiring PLEX can be performed much more efficiently and easily when this procedure is offered in-house or in proximity to the neurology clinic.
Discussion
Across three centers and 67 years (>90,000 procedures), we found that PLEX is a fast-acting immunomodulator with a dependable safety profile that fits routine outpatient care. Used early for steroid-refractory attacks in MS, NMOSD, MG, and GBS and continued as maintenance therapy in selected progressive cases, PLEX restores function for many patients; response is more likely with shorter disease duration, active MRI lesions, preserved reflexes, and prompt initiation. Complications are uncommon when programs rely on peripheral venous access and short radial-artery catheters with citrate anticoagulation; the serious events we see largely track with long-term central lines. Practical barriers, not biology, often limit its use: inconsistent insurance approval, unfamiliarity with workable protocols, and lingering misconceptions about risk and mechanism. In this review, we have summarized basic protocols, suggest simple predictors of benefit, and present steps to build safe outpatient services, noting that costs compare favorably with several high-efficacy biologics.
PLEX is a procedure covered by most health insurance plans in the United States when deemed medically necessary. Denial rates can be influenced by factors such as the insurance plan, the reason for treatment, and the accuracy of the claim. It is recommended to confirm coverage before undergoing PLEX. In addition to providing diagnosis, documentation of clinical deterioration despite being treated with conventional therapies is an important consideration for approval. Occasionally, when everything fails for insurance companies to cover PLEX, a letter from the patient’s attorney, or having media expose the patient’s story works. It is, however, easier to get approval for PLEX now than it was about 5 years ago. A 2023 analysis of qualified health plans on HealthCare.gov showed a combined average denial rate of 20% for all claims. In-network claims were denied at a rate of 19%, while out-of-network claims were denied 37% of the time. The reasons for denial vary, with lack of medical necessity accounting for a small percentage (around 6% for in-network claims), and other reasons including administrative errors, excluded services, and lack of prior authorization. Specific data on the percentage of insurance denials for plasmapheresis procedures across Europe is unavailable. However, coverage depends on the medical condition and individual insurance plans. The cost of PLEX can be an affordability issue, especially for those with lower incomes or in countries with less robust healthcare systems. Reimbursement restrictions could also limit access. Evidence suggests that PLEX may be more affordable than IVIG in some Asian settings, particularly for conditions like GBS.73,74 In India, for instance, a study found that the out-of-pocket cost for the IVIG group was significantly higher than the PLEX group. 75
There are limitations to the current retrospective real-world experience analysis. For example, our primary outcome with PLEX was clinical disability improvement rather than “slowing the disability progression” as is normally reported in such disorders. We demonstrated that to be the case in the majority of our patients; these patients had failed conventional therapies at not only one center but at all of the three centers. This degree of initial clinical disability was also observed in our randomized controlled trials.
Based on our long-term and extensive collective experience with PLEX, we conclude that PLEX is safe and, when used with the appropriate protocol, effective as an acute and chronic therapy (Box 1). If PLEX is clinically indicated, we have yet to identify an evidence-based contraindication for patients to receive PLEX. Patients in septic shock; hepatic, renal, or heart failure; acute stroke; respiratory crisis; and of practically any age have all responded well to PLEX. It is a fast-acting immunomodulator, does not cause immunosuppression, and has a highly favorable long-term safety record in both an outpatient and inpatient settings. In patients with significant comorbidities or who are acutely ill, or in those who have a fulminant autoimmune attack, PLEX would be our first choice over drugs which bring about immunosuppression or inhibit complement to prevent end-organ damage. It is worthwhile reiterating that when patients with chronic and lifelong progressive disease improved with PLEX, a maintenance or ongoing PLEX (usually once every 4–8 weeks) is needed. The weaning process or extending the time in between PLEX is gradual. If patients have noticeable worsening just before the next PLEX, the time interval between treatments is shortened.

In our clinical experience, 18 patients with a higher rate of disease progression in the previous 2 years are more likely to respond positively to PLEX. If the age, disease duration, and severity of progression is known, it is possible to predict the degree of improvement on EDSS and also the percentage of patients who will improve by one step on disability status scale. Several predictors of favorable response have been identified, including:
Time to Initiation: A shorter interval between relapse onset and PLEX initiation significantly improves outcomes. Most responders exhibit improvement within a median of 4 days or after the third PLEX procedure. 37
Baseline Disability: Patients with lower pretreatment EDSS scores are more likely to achieve better recovery. Shorter disease duration prior to PLEX initiation correlates with better outcomes, reflecting the importance of early intervention. 35
MRI Features: Radiographic features, particularly gadolinium-enhancing (Gd+) MRI lesions, strongly predict PLEX response. Patients with large (>2 cm) tumefactive or ring-enhancing lesions are more likely to benefit. Among these, nearly 52% exhibited ring-enhancing lesions on MRI, which was significantly associated with favorable outcomes35,36
Additional Clinical Predictors: Preserved deep tendon reflexes and early symptomatic improvement during PLEX sessions are associated with better outcomes. 34
It is important to use a disease state appropriate PLEX protocol and, in particular, not undertreat conditions that are responding to PLEX therapy. PLEX is less expensive than currently used biologics and advantages in clinical impacts with PLEX may be due to its unique mechanisms of action. Along with insurance denials, lack of appreciation of how PLEX can benefit autoimmune neurology patients is a main reason for its underutilization. Most of the complications associated with PLEX are related to central line vascular access. We cannot overemphasize the importance of avoiding an indwelling central line access whenever possible. Lastly, if you are a neurologist with an interest in treating auto-immune neurological disorders, invest in having your own PLEX program or closely partner with a team that operates an existing one.
Footnotes
Acknowledgements
Dr. Michael P. McQuillen for supporting my work with PLEX from the time when I was his neurology resident to present. To all my patients who put their trust in me and in this therapy and for giving me their consent to use their videos and photos in scientific publications and meetings. John Kramer PA-C, for taking care of patients undergoing PLEX. Dr. Ryan Metzger for editing the initial draft of this manuscript. Dr. Elliot and Teresa Frohman for editing the initial draft of this manuscript. Pamela Harvey, PhD (Acumen Medical Communications) for medical writing and editorial support.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
