Abstract
Background:
There is no standardized treatment for acquired amyloid polyneuropathy (AAP) in domino liver transplant (DLT) recipients.
Objectives:
Our objective is to analyze the efficacy and safety of patisiran for the treatment of AAP in DLT recipients.
Design:
We performed a postauthorization prospective longitudinal study of DLT recipients with AAP who received patisiran treatment for 22 months.
Methods:
The primary endpoint was change in the Neuropathy Impairment Scale (NIS) from baseline. Other assessments included neurophysiologic study, quantitative sensory testing, 10 m walking test, and quality of life and disability questionnaires. As safety parameters we analyzed evidence of graft rejection, immunosuppression levels, and renal and cardiac adverse effects.
Results:
Four patients were recruited. The mean NIS at baseline was 8.5 ± 2.08. All patients presented clinical improvement after 22 months of treatment, with a mean NIS of 4.75 ± 2.27 points. The mean change from baseline in the NIS was −3.75 ± 0.71 (95% CI: −0.47 to 7.97). The use of patisiran was not associated with cardiovascular or renal side effects. No patient presented relevant changes in immunosuppression levels or graft rejection.
Conclusion:
Our study suggests that patisiran may improve neurological manifestations in DLT recipients with AAP, producing no relevant adverse effects.
Plain language summary
Hereditary transthyretin amyloidosis is a genetic disease where a protein called amyloid builds up in different organs of the body. One of the most affected parts are the peripheral nerves in the arms and legs, causing a condition called polyneuropathy. Since 90% of that amyloid protein is produced in the liver, the main treatment for the disease has been a liver transplant: removing the liver with the genetic defect and replacing it with a new one. However, these livers with the genetic defect still work properly, and they started being transplanted into other patients with end-stage liver damage who were at the bottom of the transplant waiting list. This is called a domino liver transplant. But over the years, these patients also develop the same nerve damage in their arms and legs. In recent years, medical treatments have appeared as alternatives to transplant to prevent amyloid from building up in organs. One of them is patisiran, which has proven to be useful and safe in patients with hereditary amyloidosis. However, there is no data on its usefulness and safety in patients who received domino liver transplants and developed nerve damage. The goal of this study is to follow up on four of these patients, tracking the effect of the treatment in stabilizing nerve damage, and its safety (checking for side effects in the kidneys, heart, or transplanted liver). Over 22 months of follow-up, none of the four patients’ nerve damage got worse, and none had any significant side effects. This means that treatment with patisiran could be useful for treating these patients.
Introduction
Hereditary transthyretin amyloidosis (ATTRv) is an autosomal dominant disorder caused by mutations in the transthyretin (TTR) gene,1,2 resulting in the production of an unstable protein that forms amyloid fibrils, which accumulate extracellularly in multiple organ systems.2,3 The ATTRv phenotype may be predominantly neurological, cardiac, or mixed, depending on the type of mutation.2,3
Given that TTR is mainly synthesized in the liver, orthotopic liver transplant has been the main therapeutic strategy.3,4 However, the liver of ATTRv patients is functionally healthy and can be donated to others with hepatic failure, a procedure known as domino liver transplant (DLT). 5 Over the past decade, several medical therapies have been developed for patients with ATTRv, aimed either at stabilizing TTR tetramers (diflunisal and tafamidis)6,7 or blocking TTR synthesis (inotersen, eplontersen, patisiran, and vutrisiran).8–11 Patisiran is a small interfering RNA that binds to the 3’ region of TTR mRNA, inhibiting the synthesis of both wild type and mutant TTR. Its efficacy as a treatment for ATTRv with polyneuropathy (ATTRv-PN) was demonstrated in the APOLLO clinical trial in 2018, 10 and its use was approved for stages I and II of the disease by the FDA and the EMA.
After transplantation to the DLT recipient, the grafted liver will continue to synthesize mutated TTR, and over time, this can result in acquired amyloid polyneuropathy (AAP).12–15 In the event of this complication arising, liver re-transplantation may be considered in an attempt to stabilize or slow symptom development,16,17 but in practice, this is often limited by the patient’s age and comorbidities. In terms of medical treatments for these patients, there are isolated reports of DLT recipients with AAP being treated with diflunisal, tafamidis, or patisiran.18–22 In one series of seven DLT recipients with AAP who received diflunisal for 12 months, stabilization of neurological symptoms (increase <2 points in the Neuropathy Impairment Scale (NIS)) was observed in only 25% of patients, and there was a high incidence of adverse effects. 23 However, in another series of seven DLT recipients with AAP who were treated with tafamidis, stabilization of neurological symptoms was achieved in 85.7% of patients at 18 months of treatment. 24
Although the pathophysiology of AAP in DLT recipients is the same as in patients ATTRv-PN, gene-silencing therapies have not been tested in long-term clinical trials with the former group. In this article, we present efficacy and safety results from a series of DLT recipients with AAP who were treated with patisiran for 22 months in a tertiary hospital setting.
Materials and methods
Study design
This was a postauthorization prospective longitudinal study of DLT recipients with AAP who received patisiran treatment for 22 months (May 2020–March 2023) in our multidisciplinary unit for familial amyloidosis. In all cases, the diagnosis of AAP was confirmed by a sural nerve biopsy showing amyloid deposits, with other causes of peripheral neuropathy being ruled out or corrected. The full set of inclusion and exclusion criteria are listed in Supplemental Material 1.
At the start of the study, we collected data on patients’ age, sex, any concomitant disease(s) that could contribute to the development of polyneuropathy, and the immunosuppressive therapy employed. We also recorded the indications for DLT, the time between transplant and the onset of neurological symptoms, and any prior treatments for AAP.
Dose administration
Patisiran 0.3 mg/kg was administered intravenously (IV) every 21 days over a period of 22 months, on each occasion in a day hospital setting. Premedication (paracetamol 1 g IV, ranitidine 10 mg or famotidine 20 mg IV, dexchlorpheniramine 5 mg IV, and methylprednisolone 40 mg IV) was administered 60 min prior to the patisiran infusion. Patients’ vital signs were monitored prior to and 1 h after the infusion, and all patients received vitamin A supplementation (2660–3600 international unit daily).
Efficacy parameters
The primary endpoint was the change from baseline in the NIS at 22 months of treatment (range 0–244). Secondary efficacy outcomes were as follows: change from baseline in the Neuropathy Impairment Scale-Lower Limbs (NIS-LL) at 22 months of treatment; neurophysiologic study (sensory nerve conduction study of sural and ulnar nerves and motor nerve conduction study of ulnar, peroneal, and tibial nerves); score on quantitative sensory testing (QST); and the 10 m walking test (10MWT), (Supplemental Material 2). In the neurophysiologic study, a reduction of 50% in the amplitude of compound motor action potential (CMAP) and sensory nerve action potential in the explored nerves was considered clinically significant.
We also assessed changes in (1) quality of life using the Norfolk Quality of Life Scale (range: −4 to 136, with higher scores indicating poorer quality of life), (2) disability with the Rasch-built Overall Disability Scale (R-ODS; range: 0–48, with lower scores indicating greater disability), and (3) autonomic symptoms using the Composite Autonomic Symptom Score (COMPASS-31; range: 0–100, with higher scores indicating more autonomic symptoms).
To assess patients’ functional status, we recorded polyneuropathy disability (PND) stage (0: no symptoms; I: sensory symptoms but preserved walking capacity; II: unassisted walking but with difficulty; IIIA: one stick or crutch required for walking; IIIB: two sticks or crutches required for walking; IV: bedridden or wheelchair bound) and familial amyloid polyneuropathy (FAP) stage (0: no symptoms; I: mild symptoms with unassisted walking; II: assistance required for walking; III: bedridden or wheelchair bound).
Safety parameters
A baseline cardiological evaluation was performed, including the New York Heart Association (NYHA) classification, electrocardiogram (ECG), and measurement of troponin T and proBNP levels. Additionally, patients underwent a baseline assessment with 24-h Holter ECG, echocardiography, and cardiac scintigraphy with technetium, performed within the 6 months preceding or following the initiation of patisiran treatment, to provide a more precise evaluation of cardiac amyloid involvement. During follow-up, periodic assessments were conducted using the NYHA classification and measurements of troponin T and proBNP levels (at 6, 12, and 22 months), with echocardiography performed at 1 year and at the end of the study. Additional Holter ECG monitoring and cardiac scintigraphy during follow-up were performed only based on cardiological clinical criteria.
Abdominal ultrasound was performed at the start of the study to confirm that each liver graft was morphologically and functionally normal. Follow-up involved testing of liver function parameters and immunosuppression levels at the start of treatment and at 3, 6, 12, and 22 months. A further abdominal ultrasound was performed at 12 (±3) and 22 (±3) months.
Cell-mediated immunity (CMI) specific to viral and allogeneic antigens was assessed before initiation of treatment with patisiran and at 12 months in two patients. An IFN-Y ELISPOT assay was used to measure CMI specific to three different viruses: cytomegalovirus (CMV), Epstein–Barr virus (EBV), and influenza, as well as total allogeneic cellular stimuli.25,26 Briefly, 3·105 peripheral blood mononuclear cells (PBMCs) in a 100 µL volume were stimulated in duplicates with 6 different allogeneic fully HLA-mismatched cellular stimuli (CD3+ depleted PBMCs) and overlapping peptide pools of two main immunogenic CMV antigens (IE-1 and pp65; Oxford Immunotherapy limited, Oxford, United Kindom), EBV (Oxford Immunotec Ltd), and influenza (Autoimmun Diagnostik®, Strasberg, Germany) for 22 h. We detected IFN-γ spots using a biotinylated antihuman IFN-γ antibody developed by the addition of alkaline phosphatase conjugate substrate. The resulting spots were counted semiautomatically with an ELISPOT reader (7th generation Autoimmun Diagnostik). Medium alone was used as a negative control, and pokeweed mitogen (Autoimmun Diagnostik) as a positive control.
Renal function was also monitored periodically (at baseline, 3, 6, 12, 22 months) with deterioration being defined as an estimated glomerular filtration rate (eGFR) value below 60 mL/min and a decrease from baseline of 10 mL/min or greater in the eGFR. Vitamin A levels were determined at baseline and at 6 and 12 months of treatment.
All adverse effects during patisiran treatment were recorded, regardless of causality (Supplemental Material 3).
Statistical analysis
Counts and percentages were calculated for categorical variables, while for numerical variables, we computed either the mean and standard deviation or the median with the first and last quartiles.
Results
Clinical and demographic characteristics
Four DLT recipients with AAP initiated treatment with patisiran 0.3 mg/kg every 21 days (Table 1). Their mean age at the start of the study was 69.6 years (SD: 8.48; range: 59–77 years). All liver grafts came from donors with pure neurological phenotype and V30M mutation.
Clinical and demographic characteristics of patients at baseline.

AAP, acquired amyloid polyneuropathy; AI, autoimmune; DLT, domino liver transplant; DM, diabetes mellitus; F, female; FAP, familial amyloidotic polyneuropathy; HbA1c, glycated hemoglobin; HCV, hepatitis C virus; IS, immunosuppressive therapy; LC, liver cirrhosis; M, male; NIS, Neurological Impairment Scale; NIS-W, Neurological Impairment Scale Weakness; PND, polyneuropathy disability score.
Mean time from DLT to onset of neurological symptoms was 9.5 years (range: 5–15), with all patients reporting painful dysesthesias in the lower limbs and impaired distal thermoregulation consistent with small fiber neuropathy. The mean interval between the onset of neurological symptoms and the initiation of patisiran treatment was 11.75 months.
Mean baseline NIS and NIS-LL was 8.5 ± 2.08 (range: 6–12). Three patients (75%) were at PND stage I and one at PND stage II (25%). All four patients were classified as FAP stage I. One patient (25%) had type 1 diabetes, but blood glucose was strictly controlled at the start of patisiran treatment and throughout. The presence of amyloid deposits was confirmed in all patients by sural nerve biopsy. One patient (25%) had previously received treatment with diflunisal, but this was suspended after a few days due to cholestasis, 2 months before the initiation of patisiran treatment. None of the other three patients had previously received treatment for AAP.
Treatment efficacy
Primary endpoints
During the first 6 months of treatment all patients showed an improvement in the NIS, with a mean decrease from baseline of 1.5 points (mean −1.5; 95% CI: −3.74 to 6.74; Table 2, Figures 1 and 2). Improvement was even more evident after 12 months of treatment, with the mean decrease from baseline now reaching 4.75 points (mean −4.75; 95% CI: −0.62 to 10.12), and it was maintained at 22 months, at which point the NIS showed a decrease from baseline of 3.75 points (mean −3.75; 95% CI: 0.47–7.97).
Efficacy parameters at baseline and at 6, 12, and 22 months of treatment with patisiran and mean differences from baseline and 95% CI.
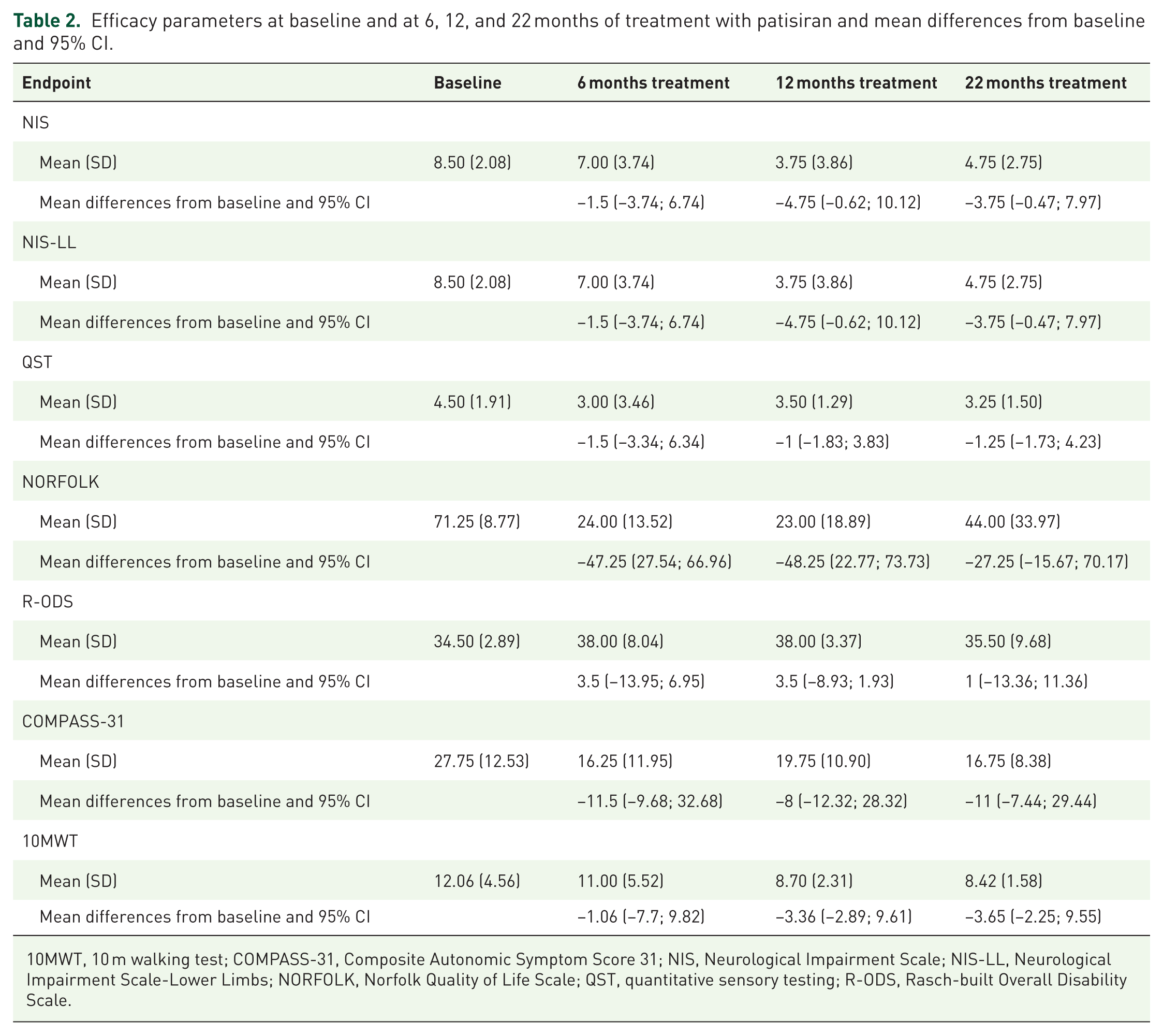
10MWT, 10 m walking test; COMPASS-31, Composite Autonomic Symptom Score 31; NIS, Neurological Impairment Scale; NIS-LL, Neurological Impairment Scale-Lower Limbs; NORFOLK, Norfolk Quality of Life Scale; QST, quantitative sensory testing; R-ODS, Rasch-built Overall Disability Scale.

NIS at baseline and at 6, 12, and 22 months of treatment with patisiran in domino liver transplant recipients with acquired amyloid polyneuropathy.

Plot showing changes in the mean and 95% CI for the longitudinal efficacy variables at each assessment (0, 6, 12, and 22 months). (a) NIS. (b) QST. (c) 10MWT.
Secondary endpoints
Since none of the patients exhibited clinical involvement in the upper limbs, changes in the NIS-LL followed the pattern observed with the NIS (Figures 2). All patients remained at the same PND stage (75% stage I, 25% stage II) and FAP stage (100% stage I) during treatment.
Only one patient showed a decrease of more than 50% in the amplitude of the peroneal nerve CMAP in the context of L5 radiculopathy with axonal degeneration. No significant changes in motor and sensory nerve conduction were observed in the other three patients.
A trend toward improvement at 22 months (compared with baseline) was also observed in scores on QST (−1.25; 95% CI: −1.73 to 4.23) and the 10MWT (−3.65; 95% CI: −2.25 to 9.55), Figure 2.
The same pattern of results (trend toward improvement compared with baseline) was obtained for the subjective parameters assessed with the Norfolk Quality of Life Scale (−27.25; 95% CI: −15.67 to 70.17), the R-ODS (+1; 95% CI: −13.36 to 11.36), and the COMPASS-31 (−11.00; 95% CI: −7.44 to 29.44), Figure 3.

Plot showing changes in the mean and 95% CI for the longitudinal efficacy variables at each assessment (0, 6, 12, and 22 months). (a) NORFOLK. (b) RODS. (c) COMPASS-31.
Tolerability and adverse effects of treatment
All adverse effects during treatment with patisiran were recorded, irrespective of causality (Supplemental Material 3). Only one adverse effect related to the use of patisiran was identified, with one patient repeatedly reporting generalized pain and atypical chest pain at the start of the infusion. This was resolved by reducing the infusion rate or briefly halting the administration. There were no serious adverse events during the follow-up of our patients.
None of the four patients had evidence of cardiac amyloidosis on echocardiography or cardiac scintigraphy with technetium at the start of treatment with patisiran (Table 3). No deterioration in cardiac status (NYHA classification) was observed during follow-up. Troponin T levels showed after 22 months treatment a mean decreased from baseline of 6.58; 95% CI: −11.23 to 24.39), while mean proBNP levels increased from baseline by 121.5 ng/L at 22 months (95% CI: −1846.11 to 1603.11). Patients with the greatest increase in proBNP levels had underlying valvular heart disease. Cardiac amyloidosis was not observed during the follow-up period.
Cardiac assessment before and after treatment with patisiran.

Perugini Grading Scale in cardiac scintigraphy with 99mTc-DPD (grade 0: no cardiac uptake and normal bone uptake; grade 1: cardiac uptake which is less than bone uptake; grade 2: cardiac uptake with intensity similar to or greater than bone uptake; grade 3: cardiac uptake with much reduced or absent bone signal).
AF, atrial fibrillation; AoE, aortic stenosis; BNP, NT-proBNP (ng/L, normal <300 ng/L); ECG, electrocardiogram; EF, ejection fraction; LV, left ventricle; MI, mitral insufficiency; SR, sinus rhythm; TI, tricuspid insufficiency.
Regarding the DLT, none of the four patients exhibited graft rejection. There was no relevant fluctuation in liver function or immunosuppression levels from baseline, and serial liver ultrasound examination revealed no functional or morphological alterations in the graft. The study of adaptive alloimmunity in two of our patients through the assessment of circulating alloreactive T-cells using a panel of six complete HLA mismatched B-cell lines showed no pre- versus post-treatment differences. Similarly, no increased activation of circulating T-cells was observed.
During the 22 months of treatment with patisiran, none of the four patients showed worsening renal function.
In all patients, vitamin A levels remained below the normal threshold despite supplementation, although this was not accompanied by the development of clinical signs.
Discussion
According to the Domino Liver Transplant Registry, a total of 1264 DLTs involving donors with ATTRv had been performed by the end of 2019. 27 Although the procedure has been much less widely used in recent years, 19 there remain a considerable number of patients worldwide who have developed (or will develop) AAP following DLT.
Among medical treatments for DLT recipients with AAP that have been investigated, diflunisal achieved stabilization of neurological symptoms in only a small proportion of patients (25%), and it was associated with a high incidence of adverse effects and drug cessation, 23 considerably greater than in the pivotal clinical trial of diflunisal with ATTRv-PN patients. 6 A recent study reported a higher rate of neurological stabilization in DLT recipients with AAP treated with tafamidis for 18 months (85.7%), without a statistically significant improvement in NIS. 24
In the APOLLO trial, patisiran was found to be effective in treating the multiple clinical manifestations of ATTRv, with 74% of patients having a less than 10-point increase from baseline in the NIS + 7 at 18 months, and 56% showing improvement in polyneuropathy at the same time point. 10 Long-term data at 3, 4, and 5 years postapproval further support the efficacy and safety of patisiran, as initially demonstrated in the APOLLO trial.28–30
The pathophysiological mechanisms underlying AAP are similar to those observed in ATTRv-PN. Although DLT recipients with AAP were not included in clinical trials of TTR gene silencing therapies, the Pharmacy and Therapeutics Committee in our region (Catalonia, Spain) has authorized the use of these treatments in this patient group. 31 In this study of patisiran, all four patients (100%) showed clinical improvement at 22 months, compared with 56% of ATTRv-PN patients at 18 months in the APOLLO trial. A possible explanation for this difference is that our patients presented much milder neurological symptoms (mean NIS for weakness 2 ± 2.16 vs 32.7 ± 25.2 for patients in the APOLLO trial). 10 In our AAP patients treated with patisiran, sustained improvement in NIS scores from baseline was observed at 22 months. However, following the initial improvement at 1 year, a slight increase in NIS was noted at 22 months in three patients (Figure 1), two points in one patient, and one point in the other two. These findings highlight the need for long-term follow-up to confirm durability of response in these patients. This is likely because patisiran reduces serum TTR levels by approximately 81% but does not eliminate it completely. 10 Therefore, treatment is expected to substantially slow the progression of neurological symptoms but not achieve definitive stabilization. Similar trends have been reported in ATTRv-PN patients treated with patisiran during long-term follow-up after its approval, with mean NIS score increases of less than 5 points after 4 years of treatment and more than 10 points after 7 years of therapy, in contrast to an estimated NIS worsening of 14.3 points per year in the natural course of the disease.28–30
In addition to the NIS, our patients also showed improvement in all the secondary parameters assessed, both objective (QST, 10MWT) and subjective (Norfolk Quality of Life Scale, R-ODS, COMPASS-31), and importantly this was sustained at almost 2 years of follow-up. However, the comorbidity and adverse effects of polypharmacy in our patients may have confounded the measurement of quality of life and autonomic symptoms, particularly regarding gastrointestinal symptoms, potentially leading to an underestimation of improvement in these scales. Similarly, results on the 10MWT may have been affected by other motor symptoms in some patients (radiculopathy in one patient and polyarthritis in another).
For comparison with the series of DLT recipients with AAP treated with tafamidis, patients treated with patisiran showed better outcomes, with stabilization or even improvement observed in 100% of cases, compared to 85.7% in the tafamidis group. Additionally, a greater mean reduction in NIS score was observed in the patisiran group: 4.75 points at 12 months versus 3.25 points, and 3.75 versus 2.75 points beyond 18 months. Moreover, patients with AAP treated with patisiran demonstrated improvement from baseline in NIS score at 22 months in 100% of cases, whereas only 14.28% of patients receiving tafamidis maintained improvement after 18 months of treatment. However, it should be noted that the patisiran series showed milder baseline neurological involvement, with a mean NIS of 8.5 ± 2.08 (range: 6–12), in contrast to the tafamidis cohort, which had a mean NIS of 18.43 (range: 5–33 points). 24 These findings highlight the critical importance of early diagnosis and initiation of therapy in patients with AAP, as is also observed in ATTRv-PN, to optimize clinical outcomes. Once treatment has been initiated, it is essential to monitor the therapeutic response using different tools, such as NIS, neurophysiological studies, and quality of life questionnaires, as proposed for the follow-up of ATTRv. 32 Up to one-third of ATTRv patients in large postapproval follow-up studies continue to experience mild disease progression despite treatment with patisiran. 30 The management of AAP patients who exhibit worsening despite therapy with TTR gen silencers remains an unmet clinical challenge.
Regarding the autoimmune adverse events reported in the inotersen clinical trial (thrombocytopenia in 3% and glomerulonephritis in 3% 8 ), the same was not reported in patisiran since the mechanism of drug delivery is different. Treatment with patisiran produced no immune-mediated adverse effects in our patients. Furthermore, the study of acquired immunity that we performed in two patients showed no significant change at 12 months from the start of treatment.
Concerning graft safety in liver transplant recipients, data are available from a clinical trial of patisiran in patients with ATTRv-PN who experienced neurological worsening after orthotopic liver transplantation. None of the 23 treated patients showed deterioration in liver function, and only one case of hepatic rejection was reported, which was related to improper use of immunosuppressive therapy and not associated with patisiran treatment. 33 Similarly, none of our patients experienced deterioration in liver function, interaction with immunosuppressive therapy, or graft failure.
We only identified one adverse event related to patisiran consisted of generalized pain associated with the infusion.
This is consistent with data from real-world studies involving large cohorts of ATTRv-PN patients, in which adverse events related to patisiran were mild and infrequent (10%), most commonly back pain or infusion reactions. 30
The safety and efficacy results obtained here with patisiran suggest that treatment with vutrisiran, which has shown a similar safety profile and efficacy to patisiran in ATTRv patients, 11 might also be an alternative therapeutic strategy for DLT recipients with AAP. Moreover, vutrisiran has recently shown efficacy in the HELIOS-B clinical trial involving ATTRv patients with cardiomyopathy, indicating potential utility in the management of patients with mixed phenotypic presentations. 34
Limitations
The main limitation of this study is the small sample size, which is difficult to avoid given the relative rarity of the disease. Furthermore, the total NIS + 7 score for neurological impairment was not calculated. No follow-up data beyond 22 months are available to assess the persistence of the neurological response.
Conclusion
The results of this study suggest that patisiran may improve neurological manifestations in DLT recipients with AAP, producing no relevant adverse effects and achieving high treatment retention rates at 22 months.
Supplemental Material
sj-docx-1-tan-10.1177_17562864251396030 – Supplemental material for Efficacy and safety of patisiran for the treatment of acquired amyloid polyneuropathy in domino liver transplant recipients
Supplemental material, sj-docx-1-tan-10.1177_17562864251396030 for Efficacy and safety of patisiran for the treatment of acquired amyloid polyneuropathy in domino liver transplant recipients by Velina Nedkova-Hristova, Laura Donadeu, Carmen Baliellas, José González-Costello, Laura Lladó, Emma González-Vilatarsana, Miosés Morales de la Prida, Valentina Vélez-Santamaría, Oriol Bestard and Carlos Casasnovas in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-2-tan-10.1177_17562864251396030 – Supplemental material for Efficacy and safety of patisiran for the treatment of acquired amyloid polyneuropathy in domino liver transplant recipients
Supplemental material, sj-docx-2-tan-10.1177_17562864251396030 for Efficacy and safety of patisiran for the treatment of acquired amyloid polyneuropathy in domino liver transplant recipients by Velina Nedkova-Hristova, Laura Donadeu, Carmen Baliellas, José González-Costello, Laura Lladó, Emma González-Vilatarsana, Miosés Morales de la Prida, Valentina Vélez-Santamaría, Oriol Bestard and Carlos Casasnovas in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-3-tan-10.1177_17562864251396030 – Supplemental material for Efficacy and safety of patisiran for the treatment of acquired amyloid polyneuropathy in domino liver transplant recipients
Supplemental material, sj-docx-3-tan-10.1177_17562864251396030 for Efficacy and safety of patisiran for the treatment of acquired amyloid polyneuropathy in domino liver transplant recipients by Velina Nedkova-Hristova, Laura Donadeu, Carmen Baliellas, José González-Costello, Laura Lladó, Emma González-Vilatarsana, Miosés Morales de la Prida, Valentina Vélez-Santamaría, Oriol Bestard and Carlos Casasnovas in Therapeutic Advances in Neurological Disorders
Footnotes
Appendix
Acknowledgements
We express our gratitude to the patients for their collaboration. We thank the CERCA Programme/Generalitat de Catalunya for institutional support. Four of the authors of this publication (Velina Nedkova-Hristova, Miosés Morales de la Prida, Valentina Vélez-Santamaría, and Carlos Casasnovas) are members of the European Reference Network for Neuromuscular Diseases.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
