Abstract
Background:
Hereditary transthyretin amyloidosis (hATTR) is caused by mutations in the transthyretin (TTR) gene, which lead to the aggregation of misfolded TTR protein and amyloid accumulation in the peripheral nerves, heart, and gastrointestinal tract. Recently, RNA therapeutics, including small interfering RNAs (siRNAs) and antisense oligonucleotides (ASOs), have been approved for treating patients with hATTR.
Objectives:
To assess the neurological efficacy and safety of RNA therapeutics in hATTR patients.
Design:
Systematic review and meta-analysis.
Data sources and methods:
A systematic literature search was conducted on PubMed, Cochrane, and ClinicalTrials.gov from inception to August 14, 2024. Outcomes included changes from baseline in the Norfolk Quality of Life–Diabetic Neuropathy (Norfolk QOL-DN) score and the modified Neuropathy Impairment Score +7 (mNIS + 7), modified body mass index (mBMI), adverse effects, serious adverse events, and all-cause mortality.
Results:
Our study included four RCTs with 842 patients (568 in the RNA therapeutics group and 274 in the placebo group). RNA therapeutics significantly improved Norfolk QoL-DN (mean difference (MD), −18.79; 95% CI, −22.32 to −15.25; p < 0.00001; I2 = 28%) and mNIS + 7 scores (MD, −26.90; 95% CI, −31.67 to −22.13; p < 0.00001; I2 = 61%), with significant preservation of mBMI (MD, 114.98; 95% CI, 90.64–139.32; p < 0.00001; I2 = 59%) compared to placebo. There were no significant differences between the two groups regarding the risk of adverse effects (risk ratio (RR), 0.89; 95% CI, 0.69–1.15; p = 0.36; I2 = 34%), serious adverse effects (RR, 0.70; 95% CI, 0.31–1.58; p = 0.39; I2 = 20%), and all-cause mortality (RR, 0.70; 95% CI, 0.31 to 1.58; p = 0.39; I2 = 20%).
Conclusion:
RNA therapeutics are effective and well-tolerated in patients with hATTR, significantly improving quality of life and the progression of neurological impairment. siRNAs demonstrate better outcomes compared to ASOs.
Trial registration:
PROSPERO (CRD42024568346).
Introduction
Hereditary transthyretin amyloidosis (hATTR) or variant ATTR amyloidosis (ATTRv) is an autosomal dominant, progressive, and fatal disease arising from mutations in the transthyretin (TTR) gene, which results in misfolding and aggregation of TTR proteins, generating amyloid fibrils that accumulate in many tissues and organs, especially the peripheral nerves, heart, and gastrointestinal tract. 1 The tetrameric TTR protein is mainly derived from the liver. Patients usually present with polyneuropathy, cardiomyopathy, and cachexia, with gradual worsening of symptoms and decline in quality of life. 2 It has a 2- to 15-year survival rate following the onset of neuropathy and a 2- to 5-year survival rate for patients with cardiomyopathy. 3
hATTR has been treated with orthotopic liver transplantation, which lowers mutant TTR levels, or tetramer stabilizers (tafamidis or diflunisal), which stabilize the tetrameric TTR protein. However, many patients still experience disease progression following these treatments, underscoring the need for novel, disease-modifying therapies.3 –5
A relatively modern therapeutic approach is TTR gene silencing, which targets and degrades TTR messenger RNA (mRNA) in the liver. RNA therapeutics include small interfering RNAs (siRNAs) such as patisiran and vutrisiran, and antisense oligonucleotides (ASOs) such as inotersen and eplontersen. These FDA-approved drugs act at the post-transcriptional level, targeting mRNA rather than proteins. 6 siRNAs are double-stranded RNA molecules that attach to the RNA-induced silencing complex and break down targeted mRNA, leading to the downregulation of gene expression. ASOs are single-stranded oligonucleotides with RNA or DNA-based structures that use complementary base pairing to attach to their target mRNA to block translation or activate RNase H to cleave Mrna.7,8 Several clinical trials have demonstrated the efficacy of these drugs in improving the quality of life and decreasing the progression of polyneuropathy in patients with hATTR.2,3,9,10
Despite multiple trials on the use of RNA therapeutics in hATTR, to our knowledge, no meta-analysis has been carried out so far. Therefore, this systematic review and meta-analysis was conducted to evaluate the neurological efficacy and safety of RNA therapeutics in patients with hATTR.
Methods
This systematic review and meta-analysis was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 11 criteria and registered with PROSPERO (CRD42024568346), accessible at https://www.crd.york.ac.uk/prospero/display_record.php?ID=CRD42024568346.
Search strategy
A systematic literature search was conducted on PubMed, Cochrane, and ClinicalTrials.gov from inception to August 14, 2024. MeSH (Medical Subject Heading) terms and relevant keywords like “RNAi Therapeutics,” “RNA Small Interfering,” “Oligonucleotides Antisense,” “Patisiran,” “Vutrisiran,” “Inotersen,” “Eplontersen,” and “Amyloidosis Hereditary Transthyretin-Related” were used. A manual evaluation of the bibliographies of relevant articles was conducted to find pertinent articles.
Study selection and eligibility criteria
All the articles retrieved by our search were imported into Rayyan, and duplicates were removed. 12 The titles and abstracts of all the retrieved articles were independently screened by two authors (A.E. and H.Q.), followed by the full-text screening of the included articles. The articles that failed to meet the eligibility criteria were removed. A third author (M.S.) was consulted to resolve any conflicts. The eligibility criteria were as follows: (1) population: patients diagnosed with hATTR, (2) intervention: siRNAs or ASOs, (3) comparison: placebo, (4) outcomes: changes from baseline in the Norfolk Quality of Life–Diabetic Neuropathy (Norfolk QOL-DN) score and modified Neuropathy Impairment Score +7 (mNIS + 7), and (5) study design: randomized controlled trials (RCTs). Only studies with a minimum 12-month follow-up period were included.
Data extraction and outcomes
Two independent authors (R.B. and H.I.) used a standard Microsoft Excel spreadsheet for data extraction, and conflicts were settled by a third author (R.A.). Data on study characteristics (author name, publication year, study design, sample size, intervention, comparison), patient baseline characteristics (age, sex, prior therapies, duration of hATTR), and follow-up duration were extracted. The primary outcomes were changes from baseline in the Norfolk QOL-DN score and mNIS + 7. The secondary outcomes were adverse effects, serious adverse events, all-cause mortality, and modified body mass index (mBMI).
Risk of bias assessment
The Cochrane Risk of Bias 2 (RoB 2.0) tool developed by the Cochrane Collaboration was used to evaluate the risk of bias in the included RCTs. 13 Two authors (U.N. and A.A.) independently conducted the quality assessment, and conflicts were settled by consulting with a third author (M.K.). RoB 2.0 is divided into five domains: (1) bias arising from the randomization process, (2) bias due to deviations from intended interventions, (3) bias due to missing outcome data, (4) bias in the measurement of the outcome, and (5) bias in the selection of the reported result.
Data analysis
Mean differences (MDs) and risk ratios (RRs) with 95% confidence intervals (CIs) were pooled for continuous and dichotomous outcomes, respectively. The inverse variance random effects model and the Mantel-Haenszel random effects model were used to pool MDs and RRs, respectively. Data from the final follow-up time point were used for all analyses. Forest plots were generated, and p < 0.05 was deemed significant. Heterogeneity among the studies was evaluated using the I² statistic and chi-square test, considering p < 0.1 as statistically significant. We used leave-one-out sensitivity analysis, eliminating one study at a time, to ascertain whether a single study impacted the results. Subgroup analysis was conducted based on the mechanism of action of RNA therapeutics (siRNAs vs ASOs). Statistical analysis was done using Review Manager (RevMan, Version 5.4; The Cochrane Collaboration, Copenhagen, Denmark) software.
Results
Literature screening
A systematic search from inception to August 14, 2024, retrieved 511 articles. After deleting duplicates, 418 studies were included for title and abstract screening. After careful evaluation and screening, 16 were selected for full-text screening. Five were excluded due to lack of outcomes, three due to wrong study design, two due to wrong intervention, and two due to insufficient follow-up. Four studies were finally included in our analysis. A detail of the search results is provided in the PRISMA flow diagram (Figure 1).

PRISMA flowchart of the study selection process.
Study characteristics
We selected four RCTs2,3,9,10 with a total of 842 patients (568 in the RNA therapeutics group and 274 in the placebo group) for inclusion in our study. Most of these patients were males (70%). Two of the four multicenter RCTs were double-blind, and two were open-label. APOLLO 3 and NEURO-TTR 2 trials used a conventional placebo group. However, HELIOS-A 10 used the placebo group of the APOLLO trial, 3 and NEURO-TTRansform 9 used the placebo group of the NEURO-TTR trial 2 as a historical control. HELIOS-A had reported outcomes for both patisiran and vutrisiran, so we included data from both interventions separately in our analysis. The follow-up ranged from 15 to 18 months. A summary of the study and baseline characteristics is provided in Table 1.
Characteristics of included studies.
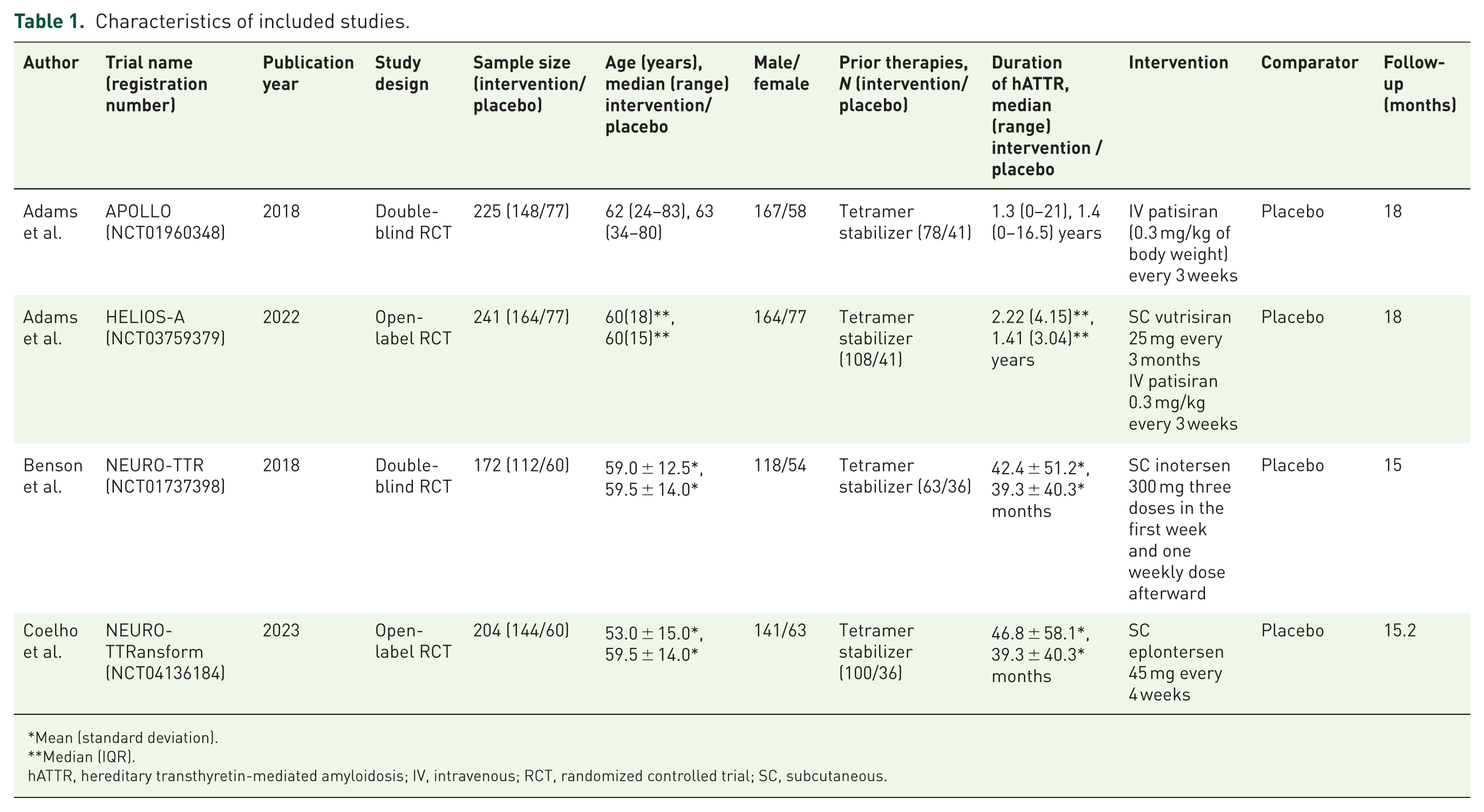
Mean (standard deviation).
Median (IQR).
hATTR, hereditary transthyretin-mediated amyloidosis; IV, intravenous; RCT, randomized controlled trial; SC, subcutaneous.
Risk of bias assessment
We used the Cochrane Risk of Bias Tool RoB 2.0 to assess the risk of bias in the included RCTs. 13 All four studies showed some concern for bias. This risk of bias was due to the domains of measurement of the outcome and the randomization process. All four studies showed a low risk of bias in the domains of deviations from intended interventions, missing outcome data, and selection of the reported result. Supplemental Figure 1 provides a graphical summary of the risk of bias assessment.
Meta-analysis
Primary outcomes
Norfolk QoL-DN
Norfolk QoL-DN was assessed in four studies with a total of 743 patients (495 RNA therapeutics vs 248 placebo). Patients who received RNA therapeutics were associated with a significant improvement in Norfolk QoL-DN score as compared to those who received a placebo (MD, −18.79; 95% CI, −22.32 to −15.25; p < 0.00001; I2 = 28%; Figure 2).

Forest plot with estimates of the MD and 95% CI of Norfolk QoL-DN score improvement of patients receiving RNA therapeutics or placebo.
A subgroup analysis according to the mechanism of action of RNA therapeutics showed that no significant difference in Norfolk QoL-DN score improvement was identified between siRNAs and ASOs (MD, −20.90; 95% CI, −24.79 to −17.01 vs MD, −15.74; 95% CI, −23.60 to −7.88; p = 0.25; I2 = 24.9%; Supplemental Figure 8).
Modified Neuropathy Impairment Score +7
Four studies were included in the analysis of mNIS + 7 with a total of 755 patients (498 RNA therapeutics vs 257 placebo). There was a significant improvement in the mNIS + 7 score in the RNA therapeutics group as compared to the placebo group (MD, −26.90; 95% CI, −31.67 to −22.13; p < 0.00001; I2 = 61%; Figure 3).

Forest plot with estimates of the MD and 95% CI of mNIS + 7 score improvement of patients receiving RNA therapeutics or placebo.
Our findings showed a high degree of heterogeneity among the included studies. We employed leave-one-out sensitivity analysis to determine if a single study affected the outcomes, eliminating one study at a time. We determined that Adams et al. 3 was the cause of the heterogeneity in the sensitivity analysis. Heterogeneity was reduced following the exclusion of this study from the analysis (MD, −25.06; 95% CI, −29.01 to −21.11; p < 0.00001; I2 = 27%; Supplemental Figure 6).
A subgroup analysis according to the mechanism of action of RNA therapeutics showed that a significant difference in mNIS + 7 score improvement was identified between siRNAs and ASOs (MD, −30.17; 95% CI, −34.38 to −25.96 vs MD, −22.26; 95% CI, −27.23 to −17.29; p = 0.02; I2 = 82.4%; Supplemental Figure 9).
Secondary outcomes
MBMI
Three studies reported mBMI with a total of 619 patients (414 RNA therapeutics vs 205 placebo). The RNA therapeutics group was associated with a significantly less change in mBMI as compared to the placebo group (MD, 114.98; 95% CI, 90.64–139.32; p < 0.00001; I2 = 59%; Supplemental Figure 2).
A sensitivity analysis was conducted to explain the high heterogeneity by excluding the study by Coelho et al. The exclusion of this study resulted in a noticeable reduction in heterogeneity (MD, 126.47; 95% CI, 108.16–144.78; p < 0.00001; I2 = 0%; Supplemental Figure 7).
A subgroup analysis according to the mechanism of action of RNA therapeutics showed that siRNAs result in a significantly higher change in mBMI than ASOs (MD, 126.47; 95% CI, 108.16 to 144.78 vs MD, 82.70; 95% CI, 53.15–112.25; p = 0.01; I2 = 83.6%; Supplemental Figure 10).
Adverse effects
Four studies reported data related to adverse effects, including a total of 919 patients (568 RNA therapeutics vs 351 placebo). There was no significant difference in the risk of adverse effects between the two groups (RR, 0.89; 95% CI, 0.69–1.15; p = 0.36; I2 = 34%; Supplemental Figure 3).
A subgroup analysis according to the mechanism of action of RNA therapeutics showed that no significant difference in adverse effects was identified between siRNAs and ASOs (RR, 1.00; 95% CI, 0.97–1.03 vs RR, 0.99; 95% CI, 0.96–1.01; p = 0.57; I2 = 0%; Supplemental Figure 11).
Serious adverse effects
Four studies reported serious adverse effects with a total of 927 patients (576 RNA therapeutics vs 351 placebo). No significant difference was observed in the risk of serious adverse effects between RNA therapeutics and placebo (RR, 0.70; 95% CI, 0.31–1.58; p = 0.39; I2 = 20%; Supplemental Figure 4).
A subgroup analysis according to the mechanism of action of RNA therapeutics showed that no significant difference in serious adverse effects was identified between siRNAs and ASOs (RR, 0.82; 95% CI, 0.65–1.03 vs RR, 1.06; 95% CI, 0.53–2.13; p = 0.50; I2 = 0%; Supplemental Figure 12).
All-cause mortality
Four studies, including a total of 927 patients (576 RNA therapeutics vs 351 placebo), were included to analyze the relationship between RNA therapeutics and all-cause mortality. There was no significant difference in the risk of mortality in both groups (RR, 0.70; 95% CI, 0.31–1.58; p = 0.39; I2 = 20%; Supplemental Figure 5).
A subgroup analysis according to the mechanism of action of RNA therapeutics showed no significant difference in all-cause mortality between siRNAs and ASOs (RR, 0.55; 95% CI, 0.26–1.14 vs RR, 3.50; 95% CI, 0.43–28.11; p = 0.10; I2 = 63.1%; Supplemental Figure 13).
Discussion
In this meta-analysis of four RCTs involving patients with hATTR, RNA therapeutics emerged as a promising treatment option, showing significant benefits over placebo. Patients receiving RNA therapeutics experienced notable improvements in key clinical measures, including quality of life (Norfolk QoL-DN), neurological function (mNIS + 7), and nutritional status (mBMI) scores. Importantly, despite these marked efficacy gains, the safety profile of RNA therapeutics remained comparable to placebo, with similar incidence rates of adverse events, serious adverse events, and all-cause mortality. These findings highlight the potential of RNA therapeutics to provide meaningful clinical benefits without compromising patient safety in hATTR.
hATTR is a rare and progressive disorder driven by TTR gene mutations that severely impact the peripheral nervous system, causing both somatic and autonomic dysfunction.14,15 hATTR symptoms worsen over time without treatment, highlighting the need for early intervention. 16 Emerging RNA interference (RNAi)-based therapies like short interfering RNAs (siRNAs) and other oligonucleotide drugs like ASOs target specific mRNAs for degradation, offering precise control over gene expression.8,17 Long-term studies show that initiating these therapies early in hATTR patients with polyneuropathy can significantly improve outcomes, emphasizing the potential of these gene-targeting approaches in disease management.18,19
The primary outcome of this meta-analysis, the Norfolk QoL-DN questionnaire, comprehensively assesses symptoms of diabetic neuropathy associated with large, small, and autonomic nerve fibers.20,21 Our meta-analysis found that RNA therapeutics significantly improved the Norfolk QoL-DN score, with an MD of −18.79 (95% CI, −22.32 to −15.25; p < 0.00001; I2 = 28%). Further subgroup analysis revealed that both siRNAs and ASOs significantly improved Norfolk QoL-DN scores, with siRNAs showing a higher MD than ASOs. This finding aligns with previous trials, notably the APOLLO trial, 3 which demonstrated that patisiran, a siRNA, led to a significant reduction in Norfolk QoL-DN scores compared to placebo (MD, −21.1; 95% CI, −27.2 to −15.0; p < 0.001). Similarly, a trial evaluating inotersen, 2 an ASO, reported an improvement over placebo with a reduction of −11.7 points (95% CI, −18.3 to −5.1; p < 0.001). Despite differences in mechanisms of action, both RNA therapeutics provide meaningful clinical benefits in managing quality of life for patients. The more substantial reduction in Norfolk QoL-DN scores observed with patisiran, a siRNA, suggests it may offer a superior impact on quality of life compared to inotersen, especially in patients with polyneuropathy. This comparative insight reinforces the growing body of evidence that RNA therapeutics, whether via RNAi or antisense mechanisms, can be effective treatment strategies for improving patient outcomes. Ultimately, these findings underscore the therapeutic promise of RNA-based approaches in enhancing quality of life, supporting their continued development and refinement as treatments for diabetic neuropathy and potentially other chronic conditions.
The earlier NIS + 7 measured muscle weakness and reflexes effectively, but was limited in assessing sensation loss and autonomic dysfunction. The mNIS + 7, a refined and quantitative tool, was developed to address these limitations, offering a more comprehensive and reproducible assessment that correlates strongly with disability and disease severity in hATTR polyneuropathy.22 –24 In our meta-analysis, we observed a significant reduction in the mNIS + 7 score with RNA therapeutics compared to placebo, with an MD of −26.90 (95% CI, −31.67 to −22.13; p < 0.00001; I2 = 61%). This finding aligns with results from Adams et al., 10 where 50.4% of patients treated with vutrisiran (an RNAi therapeutic) demonstrated improvement in mNIS + 7 at month 9, compared to 18.2% in the external placebo group. However, the heterogeneity in our analysis was notably high (I2 = 61%), which may be attributed to differences in the mNIS + 7 scales used across trials. Specifically, the APOLLO trial, 3 which assessed patisiran, used the mNIS + 7Alnylam, while the NEURO-TTR trial, 2 assessing inotersen, employed the mNIS + 7Ionis. The mNIS + 7Alnylam score relies on somatotopic quantitative sensation testing (S ST QST) and postural hypotension for autonomic dysfunction. By contrast, the mNIS + 7Ionis includes additional NIS sensation measures and uses heart rate decrease with deep breathing (HRdb) for autonomic assessment. 25 To explore the impact of these variations, we conducted a sensitivity analysis excluding the APOLLO trial, 3 which reduced the heterogeneity to I² = 27%. This suggests that differences in the scales used in these trials may contribute to the observed variability, underscoring the need for standardized outcome measures in future research on hATTR patients. Further subgroup analysis evaluating the mNIS + 7 score improvement showed a significant difference between siRNAs and ASOs. The pooled MD for siRNAs was −30.17 (95% CI, −34.38 to −25.96), indicating a substantial improvement in neuropathy scores. In comparison, ASOs demonstrated an MD of −22.26 (95% CI, −27.23 to −17.29), also showing a notable effect but with a comparatively lower reduction. The statistically significant p value of 0.02 suggests that the difference between these two mechanisms is unlikely due to chance, indicating that siRNAs may offer greater improvements in mNIS + 7 scores than ASOs.
In our meta-analysis, we observed a significant increase in mBMI, with an MD of 114.98 (95% CI, 90.64–139.32; p < 0.00001; I2 = 59%). This result indicates that RNA therapeutics have a pronounced effect on improving mBMI, a crucial marker of nutritional status in patients with hATTR. Studies have shown that patients with a lower mBMI have a shorter median time to death, highlighting the potential role of nutritional status in survival outcomes. Higher mBMI is associated with better overall health and quality of life, as malnutrition in hATTR can lead to poor outcomes and poor survival. 26 Thus, by improving mBMI, RNA therapeutics may offer a survival advantage beyond their primary mechanism of action, which targets the underlying pathology of amyloid accumulation. Subgroup analysis highlights that siRNAs may have better results than ASOs in enhancing nutritional status, which could affect patient management and survival outcomes in hATTR. Moreover, beyond mBMI, recent studies have utilized bioelectrical impedance analysis (BIA) and handgrip strength to evaluate more direct functional outcomes, such as muscle mass and motor strength. Notably, a study demonstrated that patisiran significantly increased muscle mass and preserved motor function after 9 months of treatment in patients with hATTR, as measured by BIA and handgrip strength assessments. 27 These findings suggest that RNA therapeutics, particularly siRNAs like patisiran, may offer broader functional and nutritional benefits beyond what is reflected by mBMI alone. Including objective measures such as BIA and handgrip strength in future trials could further clarify the full clinical benefits of RNA-based therapies in hATTR.
Our meta-analysis found no significant difference in the risk of adverse effects, serious adverse effects, or all-cause mortality between patients receiving RNA therapeutics and those receiving a placebo. Subgroup analyses also showed no significant differences in these outcomes between siRNAs and ASOs. Comparing these findings with TTR stabilizers like tafamidis, a significant decrease was observed in all-cause mortality when tafamidis was administered in fixed doses, with an odds ratio of 0.52 (95% CI, 0.39–0.70; p ⩽ 0.0001; I2 = 0%). 28 In siRNA studies, peripheral edema and infusion-related reactions (such as back pain, abdominal pain, flushing, and nausea) were reported more frequently. 3 By contrast, ASO studies reported common adverse events such as nausea, pyrexia, chills, vomiting, anemia, diarrhea, urinary tract infections, and vitamin A deficiency. 9 Despite these adverse events, the incidence of serious adverse effects remained low, suggesting that both siRNAs and ASOs are generally well-tolerated therapeutic options for patients with hATTR.
In addition to the neurological outcomes, it is important to understand the cardiac effects of RNA therapeutics in hATTR, as heart involvement significantly influences prognosis. Several RCTs have shown improvements in cardiac health with RNA-based therapies. For instance, the APOLLO trial demonstrated that patisiran reduced left ventricular wall thickness, improved left ventricular longitudinal strain, and lowered N-terminal pro-brain natriuretic peptide (NT-proBNP) levels compared to placebo. 3 Likewise, the NEURO-TTR trial found that inotersen either stabilized or improved cardiac biomarkers and echocardiographic parameters in patients with cardiac involvement. 2 These findings suggest that RNA therapeutics could provide dual benefits by targeting both neuropathy and cardiomyopathy in hATTR. However, because different cardiac endpoints were evaluated across trials, and at least two comparable outcomes are necessary for a quantitative synthesis, a meta-analysis of cardiac outcomes was not possible. 29
This meta-analysis has several limitations that should be considered. First, as it focuses on a rare disease, the statistical power is inherently limited by the relatively small sample size of available studies, which may reduce the robustness of conclusions. In addition, only RCTs were included, excluding potentially informative cohort studies that could offer a broader perspective on long-term outcomes. Furthermore, variations in trial design, with some RCTs being double-blind and others open-label, introduce potential bias due to differences in blinding protocols. Another limitation is the presence of high heterogeneity in certain outcomes. Sensitivity analysis was conducted to address this issue, excluding studies that contributed most to heterogeneity, which led to a decrease in overall variability. Despite these adjustments, subgroup analyses of certain outcomes continued to show elevated heterogeneity, suggesting that factors such as patient characteristics or intervention specifics may be influencing the results. Finally, although cardiac involvement is the main factor affecting prognosis in hATTR, a meta-analysis of cardiovascular outcomes was not performed because the trials reported different cardiac-related endpoints. These limitations emphasize the need for more comprehensive and diverse studies to strengthen the evidence base.
Conclusion
This systematic review and meta-analysis of four RCTs suggests that RNA therapeutics may effectively slow neurologic disease progression in hATTR patients while preserving nutritional status and enhancing quality of life. Subgroup analysis based on the mechanism of action indicated that siRNA-based therapies might yield more favorable results than ASOs. In addition, adverse event rates were comparable between patients receiving RNA therapeutics and those in placebo groups, supporting RNA-based treatments as a potentially safer, noninvasive option for hATTR management. These findings underscore the promise of RNA therapeutics as a targeted approach for treating this challenging condition.
Supplemental Material
sj-docx-1-tak-10.1177_17539447251390782 – Supplemental material for Neurological efficacy and safety of RNA therapeutics in hereditary transthyretin amyloidosis: a systematic review and meta-analysis of randomized controlled trials
Supplemental material, sj-docx-1-tak-10.1177_17539447251390782 for Neurological efficacy and safety of RNA therapeutics in hereditary transthyretin amyloidosis: a systematic review and meta-analysis of randomized controlled trials by Maha Sajjad, Rabia Ashraf, Riya Bhagwan, Hassan Ijaz, Ayesha Ejaz, Hifza Qadeer ud din, Amna Hussain, Muhammad Mohsin Khan, Umaimah Naeem and Aizaz Ali in Therapeutic Advances in Cardiovascular Disease
Supplemental Material
sj-docx-2-tak-10.1177_17539447251390782 – Supplemental material for Neurological efficacy and safety of RNA therapeutics in hereditary transthyretin amyloidosis: a systematic review and meta-analysis of randomized controlled trials
Supplemental material, sj-docx-2-tak-10.1177_17539447251390782 for Neurological efficacy and safety of RNA therapeutics in hereditary transthyretin amyloidosis: a systematic review and meta-analysis of randomized controlled trials by Maha Sajjad, Rabia Ashraf, Riya Bhagwan, Hassan Ijaz, Ayesha Ejaz, Hifza Qadeer ud din, Amna Hussain, Muhammad Mohsin Khan, Umaimah Naeem and Aizaz Ali in Therapeutic Advances in Cardiovascular Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
