Abstract
Background:
Hereditary transthyretin amyloidosis (ATTRv; v for variant) with polyneuropathy is a rare, progressive, and fatal autosomal dominant disorder. Therapies such as liver transplantation and TTR stabilizations have limitations. Patisiran is a small interfering RNA (siRNA), offering potential as a genetic-level therapy for hereditary transthyretin amyloidosis with polyneuropathy (ATTRv-PN). However, evidence on patisiran’s efficacy and safety for ATTRv-PN remains limited.
Objectives:
This study aimed to further clarify patisiran’s efficacy and safety for ATTRv-PN by meta-analysis.
Design:
Systematic review and meta-analysis.
Methods:
After literature searches in PubMed, Ovid MEDLINE, Embase, JBI EBP, Cochrane, and ClinicalTrials.gov databases on 7 June 2024, 11 studies with 503 patients were included and clinical data were extracted.
Results:
Results showed an 88% (95% confidence interval (CI): 81%–94%) pooled responsiveness rate. The standardized mean difference of modified Neuropathy Impairment Score plus 7 nerve tests (mNIS + 7) scores was −0.18 (95% CI: −0.32 to −0.03, p-value 0.018) and Norfolk Quality of Life–Diabetic Neuropathy was −0.21 (95% CI: −0.35 to −0.08, p-value 0.002). In total, 413 adverse events (AEs) (84.8%), 158 serious AEs (32.4%), and 37 deaths (7.6%) were recorded. Most of AEs were mild to moderate. No deaths were attributed to patisiran. However, there is no statistically significant improvement in Neuropathy Impairment Scores.
Conclusion:
In conclusion, patisiran was effective and safe for patients with ATTRv-PN. More large-scale clinical trials and long-term studies are necessary to further validate patisiran’s efficacy and safety.
Trial registration:
PROSPERO registration ID: CRD42023428838.
Keywords
Introduction
Hereditary transthyretin amyloidosis (ATTRv; v for variant) with polyneuropathy (ATTRv-PN), also known as transthyretin familial amyloid polyneuropathy (TTR-FAP), is a rare, progressive, and fatal autosomal dominant disorder. It is caused by mutations in the transthyretin (TTR) gene, which results in a misfolded TTR protein. 1 The formation of insoluble amyloid fibrils accumulates in a multisystem manner that includes the peripheral nervous system, gastrointestinal tract, and heart.2,3
Previous treatments for patients with ATTRv-PN included liver transplantation and TTR stabilizations (diflunisal or tafamidis). However, many cases with continuous disease progression were reported after these therapies.4–8 Recently, with a deeper comprehension of the mechanism of ATTR amyloidosis, additional therapies have been recommended as superior treatment options. Patisiran is a small interfering RNA (siRNA), which is targeted to the 3′ untranslated region of transthyretin mRNA to reduce the production of variant and wild-type TTR at the first stage of the natural history. 9 Its efficacy in ATTRv-PN has been shown in the APOLLO trial (a multicenter, randomized, double-blind, placebo-controlled clinical trial) and an open-label extension study.9–11 Since the Food and Drug Administration (FDA) and European Medicines Agency (EMA) approved patisiran (0.3 mg/kg via intravenous infusion every 3 weeks) for adults suffering from ATTRv-PN, the first approved RNA interference therapy in patients with ATTRv-PN had initiated a new age of ATTRv-PN treatments.12,13 Nowadays, there has been an increase in the number of relevant studies being conducted in the ATTRv-PN space. However, the current findings on the efficacy and safety of patisiran for ATTRv-PN remain understudied. We have performed a systematic review and meta-analysis to further clarify the efficacy and safety of patisiran for ATTRv-PN. Additionally, we examined its efficacy in populations who had liver transplantation, with cardiomyopathy and with long-term use of patisiran.
Methods
This systematic review and meta-analysis was completed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. 14 The protocol was registered with PROSPERO, an international database of systematic review protocols (CRD42023428838).
Search strategy and study selection
The English medical and scientific literature was examined using PubMed, Ovid MEDLINE, Embase, JBI EBP, Cochrane, and ClinicalTrials.gov databases on 7 June 2024. Additionally, references cited in the included studies were also screened. The keywords included in the search were: (1) “TTR” OR “transthyretin” OR “ATTR,” (2) “amyloid” OR “amyloidosis,” (3) “neuropathy” OR “peripheral neuropathy” OR “nerve” OR “PN” OR “FAP,” and (4) “patisiran” OR “onpattro” OR “ALN-TTR02.”
Inclusion and exclusion criteria
Eligibility criteria included (1) case reports, case series, prospective or retrospective cohort studies, randomized controlled trials (RCTs) and (2) patients diagnosed with ATTRv-PN with pathogenic TTR mutations. Ineligibility criteria included (1) reviews, books, and documents, secondary sources of literature, (2) irrelevant disease, treatment, (3) insufficient information on the outcomes, and (4) duplicate data in several studies.
Data extraction
Data extracted from each study included the first author, publication year, age, gender, treatment regimen, and disease conditions (Table 1).
Clinical characteristics of 11 studies with 503 patients.

CADT, Compound Autonomic Dysfunction Test; CHLT, Combined Heart-Liver Transplantation; COMPASS-31, The Composite Autonomic Symptom Score; EQ-5D-5L, European Quality of Life 5 Dimensions 5 Level Version; FAP, Familial Amyloid Polyneuropathy stage; INCAT, Inflammatory Neuropathy Cause and Treatment disability score; iv, intravenous infusion; LT, Liver Transplantation; mBMI, modified body mass index; MRC, Medical Research Council; IVIG, Intravenous immunoglobulin; KPS, Karnofsky Performance Status; 10MWT, 10 Meter Walk Test; NCS, nerve conduction study; NIS, Neuropathy Impairment Score; mNIS + 7, modified Neuropathy Impairment Score plus 7 nerve tests; NIS-LL, Neuropathy Impairment Score in the lower limbs; Norfolk QoL-DN, Norfolk Quality of Life–Diabetic Neuropathy Questionnaire; PND, Polyneuropathy Disability Score; q3w, every 3 weeks; R-ODS, Rasch-built Overall Disability Scale; 6MWT, 6 Meter Walk Test.
Quality assessment
The quality of the included randomized studies was evaluated using the Cochrane Risk of Bias 2.0 Tool (RoB 2), which categorized the studies as having low risk, some concerns, or high risk of bias. 23 Included non-randomized studies were assessed by the Newcastle–Ottawa Scale (NOS) ranging from 0 to 9 points. 24 Studies with less than 5 points were identified as at high risk of bias. 25 The included randomized study ranked at low risk (Supplemental Figure S1). All included non-randomized studies scored 6 points (Table 2).
NOS for the quality of included non-randomized studies.
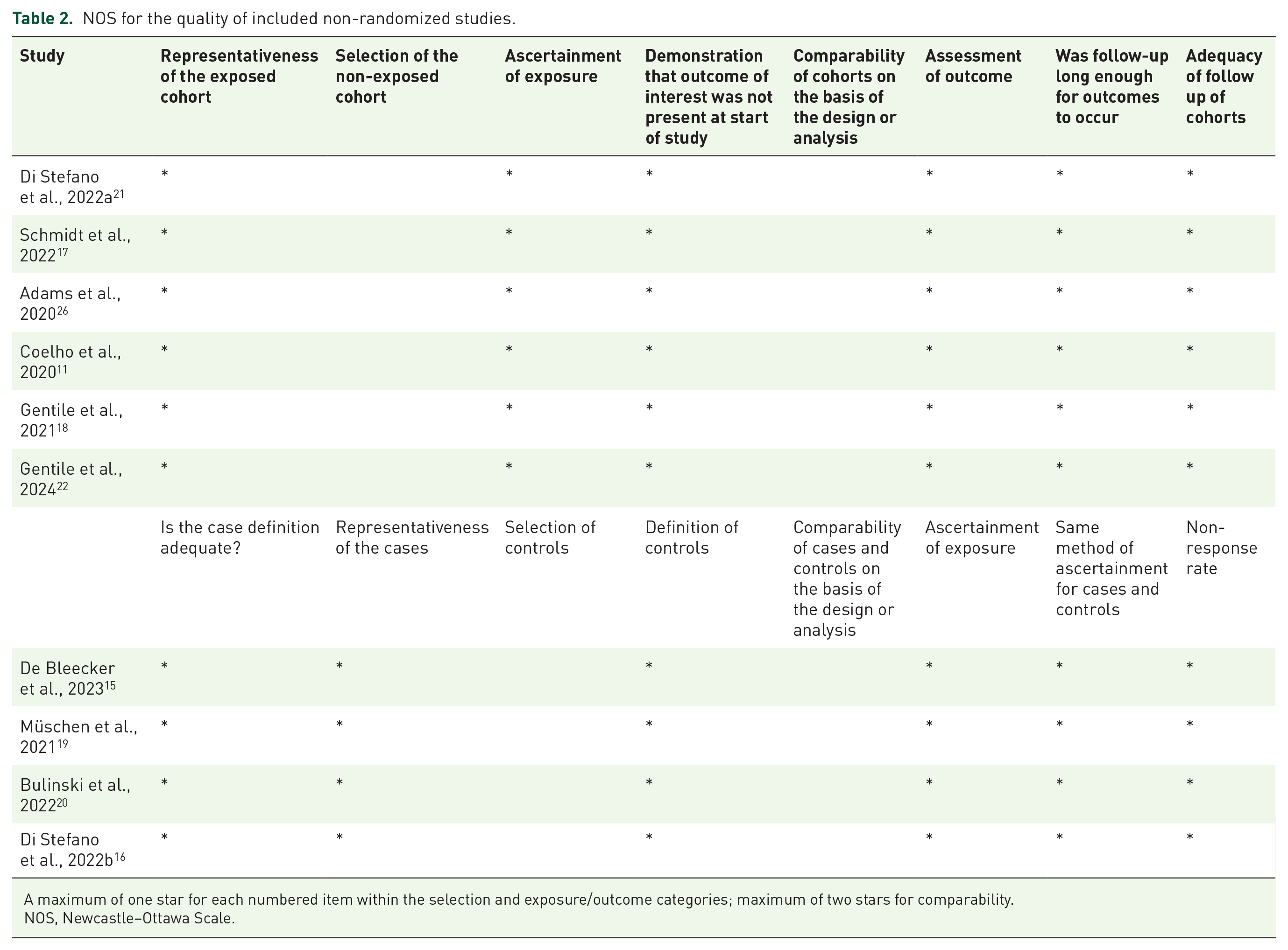
A maximum of one star for each numbered item within the selection and exposure/outcome categories; maximum of two stars for comparability.
NOS, Newcastle–Ottawa Scale.
Statistical analysis
To identify the responsiveness of patisiran, we utilized a random-effect model to calculate the pooled proportion and 95% confidence interval (95% CI). FAP staging system, polyneuropathy disability (PND) scoring system, and clinical characterstics are used to measure the responsiveness of patisiran because they were simple to evaluate and general to all studies. FAP stage and PND score indicate the extent of disability which has been a longstanding measure of neuropathic burden. Patients with stable and improved FAP stage and PND score, or assessed by experienced clinical physicians as improving or stable defined as responding to patisiran. The changes in the Neuropathy Impairment Score (NIS), the modified Neuropathy Impairment Score plus 7 nerve tests (mNIS + 7), and the Norfolk Quality of Life–Diabetic Neuropathy (Norfolk QoL-DN) were measured by the standardized mean difference (SMD) and 95% CI to demonstrate the nerve function and the quality of life. More detailed descriptions of FAP, PND, and Norfolk QoL-DN are shown in the Supplemental Information. Heterogeneity was assessed by Cochrane’s Q, while the I2 was calculated to gage the level of heterogeneity. A meta-regression was carried out to evaluate the association between treatment response and several variables such as the mean age, male ratio, mean disease duration, and intervention period. Safety outcomes included the ratio of any adverse events (AEs), serious AEs (SAEs), and death. The “meta” package of the R language was used in this meta-analysis.
Results
Study characteristics
After comprehensive literature searches using PubMed, Ovid MEDLINE, Embase, JBI EBP, Cochrane, and ClinicalTrials.gov, a total of 410 studies were initially identified. Following the removal of 137 duplicates, 206 studies were excluded during the title and abstract screening stage due to the study type and unrelated subjects. Following a full-text screening, 56 studies with insufficient outcomes and repetitive data were excluded, leaving 11 studies with 503 patients for inclusion in this meta-analysis.10,11,15–22,26 Of these studies, one was a case series, one was an RCT, two were single case reports, and seven were cohort studies. The PRISMA chart provided a clear overview of the selection (Figure 1).

The overview of study selection in the PRISMA chart. From 410 initial studies, 137 duplicates were removed. After title and abstract screening, 206 studies were excluded. Full-text screening excluded 56 studies, leaving 11 studies with 503 patients for the final analysis.
Among 471 patients from 9 studies with sex specified, 72.4% were men and 27.6% were women. The mean age of the 503 patients from 11 studies was 61.5 years of age. The mean disease duration of the 425 patients from 7 studies was 3.3 years. The mean intervention period of the 503 patients from 11 studies was 17.4 months. Two hundred fifty-six of 503 patients (50.9%) were characterized as having cardiac symptoms. Twenty-four patients (4.8%) had liver transplantation before the patisiran treatment. One hundred ninety-five patients (38.8%) used patisiran for more than 30 months. The patisiran regimen for all the patients mentioned was intravenous at 0.3 mg/kg every 3 weeks.
Effectiveness of patisiran treatment
Of the 503 patients included in the study, the pooled responsiveness rate was 88% (95% CI: 81%–94%) (Figure 2). Five studies assessed the changes in NIS before and after patisiran treatment. The results demonstrated no statistically significant improvement in NIS scores, with a standardized mean difference (SMD) of 0.01 (95% CI: −0.28 to 0.31) and a p-value of 0.92 (Figure 3). Three studies, from which one study with one patient was excluded, measured the changes in modified Neuropathy Impairment Score plus 7 nerve tests (mNIS + 7) before and after patisiran treatment. The results showed a statistically significant improvement in mNIS + 7 scores, with a SMD of −0.18 (95% CI: −0.32 to −0.03) and a p-value of 0.018 (Figure 4). The I2 value of 0 demonstrated low heterogeneity. Meanwhile, no significant correlation between the changes in mNIS + 7 scores and the following variables was found through the meta-regression as follows: mean age (p = 0.75, 95% CI: −0.16 to 0.11), male ratio (p = 0.97, 95% CI: −7.76 to 8.05), mean intervention period (p = 0.75, 95% CI: −0.03 to 0.04).

Forest plot showing the pooled responsiveness of patisiran for patients with ATTRv-PN. The size of the squares indicates the number of patients in each study. Pooled responsiveness rate among 503 patients included in the study was 88% (95% CI: 81%–94%).

Forest plot showing the improvement of NIS score of patisiran for patients with ATTRv-PN. The size of the squares indicates the number of patients in each study. Results showed no statistically significant improvement in NIS scores, with a SMD of 0.01 (95% CI: −0.28 to 0.31) and a p-value of 0.92.

Forest plot showing the improvement of mNIS + 7 score of patisiran for patients with ATTRv-PN. The size of the squares indicates the number of patients in each study. Results showed a statistically significant improvement in mNIS + 7 scores, with a SMD of −0.18 (95% CI −0.32 to −0.03) and a p-value of 0.018.
Additionally, seven studies measured the changes in Norfolk QoL-DN Questionnaire before and after patisiran treatment. The results illustrated a statistically significant improvement in Norfolk QoL-DN, with a SMD of −0.21 (95% CI −0.35 to −0.08) and a p-value = 0.002 (Figure 5). The I2 value of 0 demonstrated low heterogeneity. Meanwhile, no significant correlation between the changes of Norfolk QoL-DN and the following variables was found through the meta-regression for the following variables: mean age (p = 0.39, 95% CI: −0.028 to 0.07), male ratio (p = 0.53, 95% CI: −0.004 to 0.002), mean disease duration (p = 0.57, 95% CI: −0.18 to 0.34), and mean intervention period (p = 0.34, 95% CI: −0.007 to 0.02).

Forest plot showing the improvement of Norfolk QoL-DN score of patisiran for patients with ATTRv-PN. The size of the squares indicates the number of patients in each study. Results showed a statistically significant improvement in Norfolk QoL-DN scores, with a SMD of −0.21 (95% CI: −0.35 to −0.08) and a p-value of 0.002.
Safety of patisiran treatment
Nine studies with 487 patients recorded 413 AEs (84.8%) during patisiran treatment (Table 3). Most of the AEs were mild to moderate which may interfere but not prevent daily activities. One hundred fifty-eight SAEs (34.0%) and 113 severe AEs (23.2%) were reported. Common AEs were reported in greater than 10% of patients sorted by frequency including diarrhea (23.0%), peripheral edema (17.5%), infusion-related reaction (IRR) (13.8%), urinary tract infection (12.7%), fall (11.5%), and nasopharyngitis (10.1%). Common SAEs were reported in greater than 1% of patients sorted by frequency including diarrhea (2.1%), cerebrovascular accident (1.4%), cardiac failure (1.4%), cardiac failure congestive (1.2%), conduction disorder (1.2%), and pneumonia (1.2%). Thirty-seven deaths (7.6%) were recorded, but none of them were related to patisiran. The most common causes were cardiovascular events and it was as expected in hereditary transthyretin amyloidosis patients. A detailed summary of deaths was depicted in the Supplemental Information (Supplemental Table S1).
Safety and side effects of 9 studies with 487 patients.

AE, adverse event.
Publication bias
The Egger’s test was used to measure the publication bias, and the results indicated that the bias was acceptable (p = 0.408) (Figure 6).

Funnel plot showing the publication bias of included studies through the distribution pattern of the dots. Egger’s test for publication bias indicated acceptable bias with a p-value of 0.408.
Discussion
ATTRv-PN is characterized by mutations in the transthyretin (TTR) gene resulting in the damage of various systems, such as polyneuropathy and cardiomyopathy.27,28 Nowadays, there have been reports involving 140 mutations in the TTR contributing to the heterogeneity in the clinical presentation, with the mutation (p. Val50Met) being the most prevalent.28–30 Although the global prevalence ranges from 5000 to 10,000 individuals, the findings of the present study suggest that the actual prevalence may have been significantly underestimated. 31 ATTRv-PN imposes a substantial burden as it has the potential to be fatal within 7–12 years from the onset of symptoms.31,32 These discoveries raise a call for diagnosis and treatment of ATTR amyloidosis at an early stage.
Previous therapeutic options for ATTRv-PN such as liver transplantation and TTR stabilizers (diflunisal or tafamidis) have some limitations. Liver transplantation has shown long-term survival rates, especially for early-onset TTR (p. Val50Met) patients, but it comes with disease progression associated with wild-type TTR accumulation, procedure-related risks, reliance on donors, and ongoing immunosuppression.5,28,33,34 Diflunisal has shown efficacy in clinical trials but has limited effectiveness and notable side effects.7,35,36 Tafamidis has shown promise in slowing disease progression, but conflicting results have been reported.6–8,37 Hence, there is an urgent need for innovative therapeutic methods such as gene silencers and gene-editing therapies.
Patisiran, as the pioneering siRNA developed and licensed for ATTR amyloidosis, has brought about a revolutionary change in the treatment landscape. Patisiran is encapsulated within a lipid nanoparticle and targets hepatocytes where it splits into single-stranded RNAs that bind to complementary mRNA, thus activating the Argonaute slicer protein and inhibiting TTR synthesis.9,38 The treatment regimen of patisiran (0.3 mg/kg via intravenous infusion every 3 weeks) was established in a phase II trial with 29 ATTRv-PN adults and showed improved or stabilized disease progression in an open-label extension study.9,39 The subsequent phase III APOLLO trial involving 225 patients with ATTRv-PN demonstrated remarkableefficacy in all the endpoints including the improvement in nerve function, quality of life, walking ability, and modified body mass index (mBMI), leading to FDA and EMA approval in 2018.12,13 A global open-label extension (OLE) study with 211 ATTRv-PN patients further confirmed the long-term efficacy of patisiran. 26 Ongoing trials, such as APOLLO-B for ATTR cardiac amyloidosis patients, aim to expand its applications. AEs were generally mild to moderate in all trials.9–11,26 Although the efficacy and safety of patisiran have been demonstrated in consistent trials, further studies are necessary to gain broader recognition and understanding.
Herein, this meta-analysis and systematic review provided substantial evidence that patisiran treatment is effective for patients with ATTRv-PN. The improvement in scores of mNIS + 7 and Norfolk QoL-DN demonstrated its clinical significance. Nevertheless, there was no statistically significant improvement in NIS scores. Besides the sample sizes of 100 patients in 5 studies (vs 187 patients assessed by mNIS + 7), the possible reasons for this observation may be that mNIS + 7 encompasses additional domains and captures more comprehensive and detailed information compared to the traditional NIS, indicating that mNIS + 7 may serve as a more suitable and improved scale. 40 More detailed comparisons between NIS and mNIS + 7 are demonstrated in the Supplemental Information.
Our meta-analysis and systematic review also illustrated the safety of patisiran treatment. It is worth noting that IRRs were more frequent in the initial 3 months after patisiran treatment and tended to decrease over time.10,26 The causes of death observed in the study were in line with the expected progression of hereditary transthyretin amyloidosis. 26 Most patients who passed away had identifiable risk factors associated with a poor prognosis, including non-V50M genotype, older age, advanced disease stage, prolonged disease duration, and notable involvement of neuropathic and cardiac manifestations. 26 These findings suggest the importance of initiating treatment at an early stage.
In the population who underwent liver transplantation and polyneuropathy progression (23 from Schmidt et al. 17 and 1 from Bulinski et al. 20 ), the efficacy of patisiran was demonstrated through significant median percent reductions in serum TTR levels by 91.0% after 12 months, as well as improvements in NIS, mNIS + 7, and Norfolk QoL-DN scores and stabilization in R-ODS and mBMI. For the safety analysis of the 23 patients, 100% AEs, 21.7% SAEs and 0 death were reported, which aligns closely with our study results. 17 The most commonly reported AEs were diarrhea (34.8%) and IRRs (26.1%), which were mostly mild to moderate in severity. 17 Only one IRR, identified as an SAE (dizziness), was considered patisiran-related, but resolved without any intervention. 17 Attributed to the mechanism of action, patisiran has the potential to reduce the burden of wild-type TTR deposition and slow down the disease progression by inhibiting the synthesis of TTR in the hepatocytes, solving the root cause of disease progression in post-liver transplantation patients. The results above suggested the potential utility of patisiran in managing progressive ATTRv-PN post-liver transplantation. However, further studies with larger sample sizes and longer intervention periods are anticipated to provide more comprehensive insights.
In the 228 patients cohort with cardiomyopathy, patisiran also showed a great benefit to them including the decreases or stabilization in N-terminal prohormone of brain natriuretic peptide (NT-proBNP) as well as the improvement or stabilization in left ventricular wall thickness and left ventricular ejection fractions.10,11,15–17,19,20,26 The safety profile was similar to the overall study population.10,11,15–17,19,20,26 These observations are consistent with the expectation that patisiran delays cardiac involvement by reducing TTR production, supporting the application of patisiran for ATTRv amyloidosis patients with cardiac involvement. Soon, the APOLLO-B trial is expected to yield substantial evidence, further strengthening these findings.
In the 195 patients cohort who received patisiran over 30 months, consistent improvement or stabilization was observed in nearly all the efficacy assessments including the NIS, mNIS + 7, Norfolk QoL-DN, COMPASS-31, and mBMI.22,26 Meanwhile, the safety evaluation revealed similar results to those observed in the overall study population.22,26 The final results of the global OLE and more long-term studies are needed to verify these findings.
This systematic review and meta-analysis has limitations. The small sample size, variations in assessment indexes and definitions, and lack of clear data descriptions make it challenging to conduct subgroup analyses. Moreover, ongoing clinical trials and long-term studies are necessary to provide further insights into the efficacy and safety of patisiran for ATTRv-PN.
Conclusion
In conclusion, the efficacy and safety of patisiran for patients with ATTRv-PN were demonstrated in our meta-analysis. More large-scale clinical trials and long-term studies are needed to further clarify the efficacy and safety of patisiran for ATTRv-PN.
Supplemental Material
sj-docx-1-tan-10.1177_17562864241273079 – Supplemental material for Efficacy and safety of patisiran for ATTRv-PN: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tan-10.1177_17562864241273079 for Efficacy and safety of patisiran for ATTRv-PN: a systematic review and meta-analysis by Xinyue Huang, Chong Sun, Haofeng Chen, Chongbo Zhao and Jie Lin in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-2-tan-10.1177_17562864241273079 – Supplemental material for Efficacy and safety of patisiran for ATTRv-PN: a systematic review and meta-analysis
Supplemental material, sj-docx-2-tan-10.1177_17562864241273079 for Efficacy and safety of patisiran for ATTRv-PN: a systematic review and meta-analysis by Xinyue Huang, Chong Sun, Haofeng Chen, Chongbo Zhao and Jie Lin in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
