Abstract
Background:
Drug-resistant epilepsy (DRE) imposes a heavy disease burden and urgently requires new, effective, and safe treatment options.
Objective:
To evaluate the efficacy and safety of transcutaneous auricular vagus nerve stimulation (ta-VNS) in patients with DRE.
Design:
Ongoing, single-center, prospective real-world study.
Data sources and methods:
Patients diagnosed with DRE and undergoing ta-VNS treatment at Beijing Tiantan Hospital, affiliated with Capital Medical University, were prospectively enrolled in this study between January 2023 and December 2024. The follow-up period lasted 1–2 years. The frequency of seizure reduction was assessed using seizure diaries, with a 50% decrease in seizure frequency deemed indicative of efficacy. Adverse reactions and comorbidities were recorded concurrently. All patients were maintained on stable anti-seizure medications during this study.
Results:
Ninety-nine patients were enrolled, among whom 16 were lost to follow-up and 18 refused follow-up. Ultimately, 65 patients were successfully followed up for analysis. The overall efficacy rate was 61.54%. Specifically, 15 patients experienced seizure reductions >90%, 8 achieved a reduction between 75% and 90%, 17 demonstrated a reduction ranging from 50% to 75%, and 1 exhibited a reduction of <50%. There were no severe adverse events, although 10 patients reported mild side effects (e.g., ear tingling and tinnitus). The efficacy rate was found to be independent of variables such as age, sex, treatment frequency, and type of epilepsy. However, it demonstrated an association with baseline seizure frequency prior to treatment and the etiology of epilepsy. Patients exhibiting a higher baseline frequency of seizures (>30 episodes per month) demonstrated significantly improved response rates, with an efficacy rate of 90.9%. Etiologies, including posttumor surgery, congenital brain dysplasia, and genetic mutations, demonstrated efficacy rates >90%.
Conclusion:
The efficacy of ta-VNS was 61.54% in patients with DRE. Patients with high baseline seizure frequency or identifiable etiologies tended to experience greater therapeutic benefits. Owing to its noninvasive nature and favorable safety profile, it presents a promising alternative for the management of DRE.
Plain language summary
Keywords
Introduction
Epilepsy is a chronic neurological disorder that transcends geographic, social, and racial boundaries and affects individuals across all age groups. 1 The World Health Organization classifies epilepsy as a major neuropsychiatric disease that requires focused prevention and management strategies. With more than 50 million people suffering from epilepsy worldwide, epilepsy has increasingly become a serious public health and social concern. 2 Even if patients with epilepsy are treated with regular antiseizure medications (ASMs), approximately one-third of them continue to experience incomplete seizure control.3,4 When two appropriately chosen and tolerated ASMs (monotherapy or combination) fail to achieve seizure freedom, the condition is defined by the International League against Epilepsy (ILAE) as drug-resistant epilepsy (DRE). 5 Currently, the main alternative treatment options for patients with DRE are surgical excision and neuromodulatory therapy. However, surgical resection for epilepsy requires systematic preoperative evaluation. Following preoperative evaluation, certain patients may face challenges in precisely identifying the epileptogenic zone; the epileptogenic region may be situated adjacent to a functional area or the epileptogenic focus may be extensive, resulting in an inoperable condition or poor surgical prognosis. In recent years, emerging neuromodulatory therapies, such as deep brain stimulation, responsive nerve stimulation, and vagus nerve stimulation (VNS), have emerged as promising interventions to improve the quality of life of these patients. 6 In previous clinical trials, invasive stimulation of the cervical branch of the vagus nerve was considered to be effective, with an efficacy rate of approximately 40%–60%.7,8 However, the many side effects associated with invasive VNS (i-VNS) cannot be ignored. First, the implantation of stimulators and electrodes is an invasive procedure that requires general anesthesia, and the patient will have risks associated with surgery and anesthesia. 9 Second, i-VNS can lead to various surgical and technology-induced postoperative complications such as mild voice hoarseness (38.8%), dysphonia (<5%), cervical pain (<5%), exertional dyspnea (<5%), cough (~5%), snoring (<5%), hypersialorrhea (<5%), and other side effects associated with the stimulation period.10–14 The incidence of complications necessitating urgent removal or deactivation of the VNS device varies across studies, with reported rates ranging from 2.7% to 9.4%.11–14 A particular study reported that 16.8% of the complications were associated with the surgical procedure, while 16.8% pertained to hardware malfunctions. 12 In this series, deep infections (3.5%) and vocal cord palsy (5.6%) were the most frequent surgery-related events. Complications related to the device included electrode fracture (11.9%), disconnection (2.8%), extinction (1.4%), and pacemaker failure (1.4%). 12 In addition to surgical risks and complication rates, i-VNS has limitations in magnetic resonance imaging (MRI) compatibility and requires regular field device inspection. 15 Given these limitations and risks, transcutaneous VNS (t-VNS) offers a safer alternative with the advantage of being noninvasive.
There are two t-VNS categories: transcutaneous auricular VNS (ta-VNS) and transcutaneous cervical VNS (tc-VNS). The cervical branch of the vagus nerve is stimulated by tc-VNS, which is primarily used to treat migraines and cluster headaches, whereas ta-VNS stimulates the auricular branch of the vagus nerve (ABVN) and is primarily used to treat epilepsy, depression, chronic pain, and anxiety. 16 The ABVN, which supplies the cymba conchae, could be stimulated using an external device attached to bipolar electrodes on the skin of the left ear conch. Therefore, ta-VNS is considered noninvasive, safe, and convenient for the treatment of epilepsy and is a new therapy to overcome the disadvantages of i-VNS. Preliminary studies of ta-VNS in the treatment of epilepsy have shown that the efficacy (⩾50% reduction in seizures) and average seizure reduction rates could reach 53.85% and 54.21%, respectively.17,18 Bauer et al. 19 reported that the response rate (⩾50% reduction in seizures) was 27.0% and 25.6% in the high- and low-frequency stimulation groups, respectively. Other studies from earlier years showed that the average seizure frequency reduction by ta-VNS treatment could reach approximately 40%.19–21 A randomized, double-blind, controlled clinical trial in 2023 showed that after 20 weeks of effective stimulation, the efficacy rate (seizure reduction ⩾50%) in 76 patients with DRE receiving ta-VNS was 44.74%, and was significantly higher than that of the control group (16.67%). Moreover, there was no significant increase in the incidence of adverse events. 22 Clinical trials for the treatment of DRE with ta-VNS have been conducted both domestically and internationally. However, no real-world studies have been conducted. Therefore, we conducted the first single-center prospective real-world study to evaluate the efficacy of self-administered ta-VNS before and after treatment and to observe adverse reactions and the effect of ta-VNS on patients’ comorbidities. This study also aimed to investigate the differential therapeutic effects of ta-VNS across various etiologies, frequencies of seizures, age, sex, treatment frequency, and type of epilepsy in DRE.
Materials and methods
Patients and study methods
Between January 2023 and December 2024, patients diagnosed with medically intractable epilepsy who attended the outpatient clinic at Beijing Tiantan Hospital, affiliated with Capital Medical University, were prospectively recruited to participate in this study, which aimed to evaluate the use of ta-VNS therapy. We determined the sample size using the formula shown below and consulted previous studies in the literature to ascertain the efficacy rate, which was identified as 44.74%.
22
The efficacy rate in the experimental group in the pre-experiment was 60.08%. The sample size was calculated using the formula for comparing sample rates with known rates (
General patient information, including age, sex, age of onset, duration of disease, type of epilepsy, etiology, interictal electroencephalogram (EEG), MRI, and baseline seizure frequency at the first visit, was obtained by interview (Table 1). The parameters for ta-VNS stimulation were as follows: a pulse width of 0.2 s, with the stimulation frequency progressively increased to exceed 30 Hz. The stimulation was then maintained at a stable frequency exceeding 30 Hz, yet below the threshold at which the patient experienced significant discomfort, with the maximum frequency reaching 50 Hz. The treatment was administered four times daily, with each session lasting 30 min. During the biannual telephone follow-up, the frequency of seizures was documented based on entries in the patients’ seizure diaries. We reduced the influence of subjective factors by increasing the recorder’s awareness of the attack diary’s significance for condition analysis and treatment and providing specialized training and guidance for recorders. Medical staff provided prior training to family members or caregivers on how to identify epileptic seizures and how to distinguish different types of seizures, which can ensure the acquisition of more accurate clinical seizure diaries. The frequency of seizure reduction was assessed using the participants’ seizure diaries. The primary efficacy variable was the response rate at the end of treatment, defined as the percentage of patients with a ⩾50% reduction in the average seizure frequency from baseline to the end of treatment. Concurrently, adverse reactions and comorbidities were observed. In this study, ASMs were consistently maintained at a uniform level for all patients.
Basic characteristics of the patients.
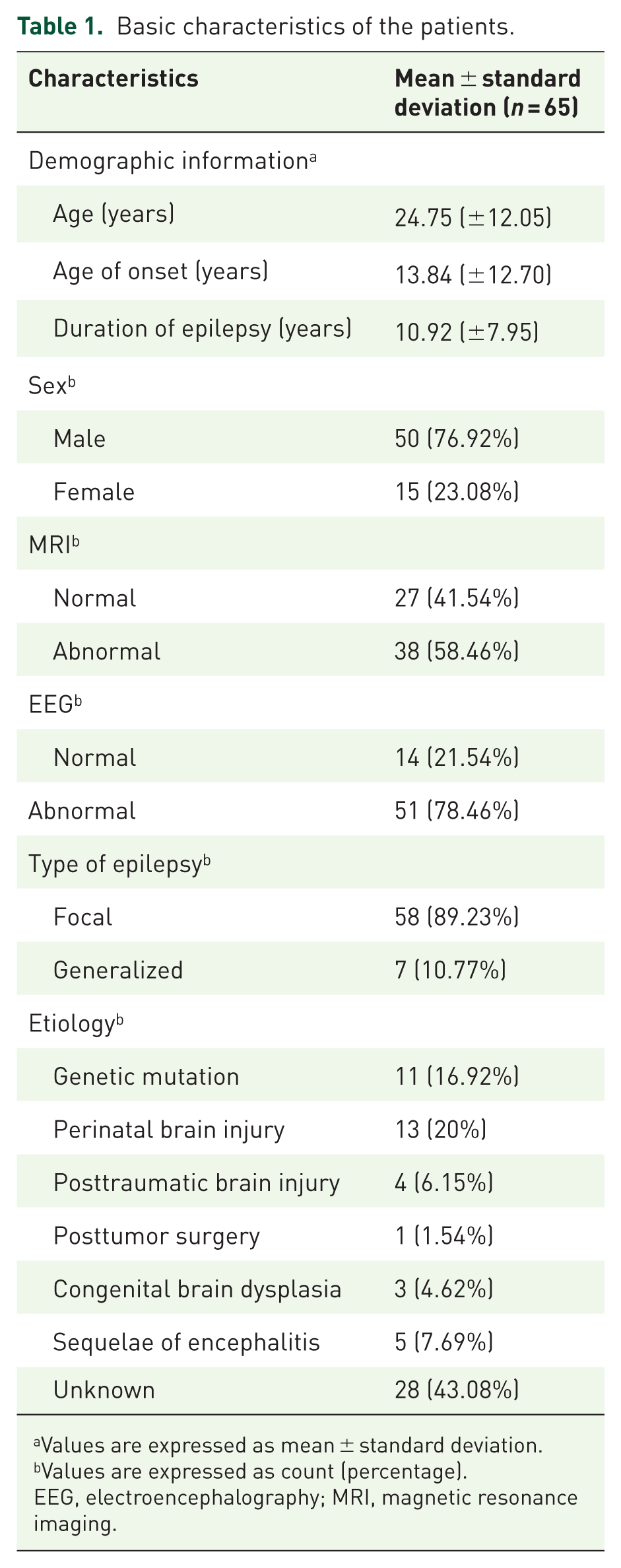
Values are expressed as mean ± standard deviation.
Values are expressed as count (percentage).
EEG, electroencephalography; MRI, magnetic resonance imaging.
Statistical analysis
SPSS 27.0 statistical software was used for statistical analysis (IBM Corp., Armonk, N.Y., USA). Count data were represented as frequencies (%). The Chi-squared test or Fisher’s exact test was used for comparisons between groups. The measurement data were represented as mean ± standard deviation, and one-way analysis of variance was used for comparison.
Results
This research project initially aimed to enroll 99 patients with a follow-up duration ranging from 1 to 2 years. However, 16 patients were lost to follow-up, and 18 refused to continue with the follow-up. Loss to follow-up indicates that the individuals did not answer subsequent multiple phone calls. Refusal to continue with the follow-up means that individuals were unwilling to provide additional personal information or assess treatment effectiveness. Ultimately, 65 patients with medically intractable epilepsy were included in the analysis (Figure 1). Among these patients, 50 were male and 15 were female, with ages ranging from 8 to 60 years (average age, 24.75 (±12.05) years). The duration of the disease ranged from 1 to 40 years, with an average of 10.92 (±7.95) years. The age of onset ranged from 0 to 58 years, with an average of 13.84 (±12.70) years. The average follow-up period was 14.69 months. Among these patients, 57 had a follow-up period of more than 1 year.

Flowchart of the study.
Efficacy
Among the 65 patients included in the study, 15 exhibited a reduction in seizure frequency exceeding 90%, 8 demonstrated a reduction between 75% and 90%, 17 showed a reduction ranging from 50% to 75%, 1 experienced a reduction of <50%, and 24 showed no change in seizure frequency following treatment. The efficacy rates of ta-VNS in patients with DRE aged <18 and 18–60 years were 55.0% and 64.44%, respectively, with no significant difference between the two groups. The efficacy rates of ta-VNS in the male and female populations were 60.0% and 66.67%, respectively, and there was no significant difference between the two groups. The treatment frequency of ta-VNS per day (1–4 times/day) showed no significant difference in efficacy among patients with DRE. There was no significant difference in the efficacy of ta-VNS for different types of epilepsy (focal and generalized epilepsy).
The rates of seizure frequency ⩽1, 2–4, 5–30, and >30 times/month in patients with DRE before treatment with ta-VNS were 66.67%, 43.48%, 63.16%, and 90.91%, respectively, and the differences were significant (
Influence of baseline seizure frequency on therapeutic effect.

Influence of etiology on curative effect.

Subgroup analysis of gene mutations.

DEE, developmental epileptic encephalopathy; DRPLA, dentatorubral-pallidoluysian atrophy; EE, epileptic encephalopathy; LGS, Lennox–Gastaut syndrome; PME, progressive myoclonic epilepsy.
Influence of a clearly identified or unknown etiology on efficacy.


There was no significant difference between the two different etiology groups (
In this study, there were four cases of epilepsy co-occurring with anxiety, two cases of epilepsy co-occurring with depression, three cases of epilepsy combined with cognitive impairment, and seven cases of epilepsy co-occurring with sleep disorders. Following the administration of ta-VNS, the participants reported subjective improvements.
Adverse reactions
In this study, no apparent adverse reactions were observed in 55 patients. However, 10 patients (15.38%) experienced adverse reactions. Specifically, six patients reported ear tingling, one experienced ear burning, one suffered from an ear burn, one reported tinnitus, and one experienced symptoms of dizziness.
Discussion
The vagus nerve, the longest and most extensively distributed cranial nerve in the human body, plays a crucial role in the regulation of peripheral organ and brain functions. 23 The nucleus tractus solitarius (NTS) is thought to play an important role in VNS in the treatment of epilepsy. The outer ear region is primarily innervated by the ABVN, commonly known as the Alderman or Arnold nerve. 24 Studies have shown that ABVN projects directly to the NTS in rats, which is thought to be the anatomical basis of the auricular vagal junction. 25 Therefore, stimulation of the auricle mitigates seizures by inhibiting the activation of neurons in the NTS. Once ABVN is stimulated, the cerebral cortex, thalamus, brain stem, and other associated regions receive the signal and transition into an activated state to achieve a therapeutic effect. Based on this theory, we conducted a real-world prospective study. We found that the efficacy of ta-VNS in patients with refractory epilepsy was not associated with variables such as age, sex, treatment frequency, or epilepsy type. However, it was significantly associated with the pretreatment seizure frequency and the underlying etiology of epilepsy.
Clinical trials have demonstrated the efficacy of ta-VNS in the treatment of epilepsy. Rong et al.
26
found that ta-VNS was effective in reducing the frequency and severity of seizures in patients with DRE. Aihua et al. found that following a 12-month treatment period, the treatment group exhibited a reduced monthly seizure frequency compared to the control group, which was associated with baseline seizure frequency and duration of epilepsy. However, it was not associated with age, seizure type, number of ASMs, family history of epilepsy, MRI and EEG abnormalities, or the initial stimulus intensity.
20
This study found that ta-VNS can effectively reduce the seizure frequency in DRE, and this reduction in seizure frequency was associated with the baseline seizure frequency, which was consistent with the results of Aihua et al.
20
Although Rong et al. and Yang et al. indicated that the reduction in seizure frequency was not related to the baseline seizure frequency, we found that the baseline seizure frequency in Rong’s study (65 times per month) was higher than that in the Huajun study (4 times per month), and the observed effectiveness was correspondingly greater. In the study by Rong et al., the efficacy rate in the treatment group reached 54% after 24 weeks (with a ⩾50% reduction in epileptic seizures), whereas in the study by Yang et al.,
22
the efficacy rate in the treatment group was 44.74% at 20 weeks (with a ⩾50% reduction in epileptic seizures).
26
We conducted a subgroup analysis of the baseline seizure frequency. We found that the efficacy rate was as high as 90.91% in patients experiencing more than 30 seizures per month, compared to an efficacy rate of only 43.48% in patients with 2–4 seizures per month, and this difference was statistically significant (
Previous studies have not paid attention to the impact of different causes of DRE on the efficacy of ta-VNS. This study is the first to perform an etiological subgroup analysis to observe its effects on therapeutic outcomes. In this study, the causes of DRE included gene mutations, perinatal brain injury, posttraumatic brain injury, posttumor surgery, congenital brain dysplasia, and sequelae of encephalitis. Owing to the extensive nature of the epileptogenic region resulting from the aforementioned causes, radical excision surgery was typically deemed unfeasible; consequently, noninvasive neuroregulatory interventions were selected as the preferred approach. In this study, ta-VNS was found to be effective in patients with DRE caused by these six causes, with efficacy rates ranging from 53.85% to 100%. Among them, the efficacy rate exceeded 90%, which is attributable to factors such as tumor resection, congenital brain development abnormalities, and genetic mutations. A linear mixed-effects model, with the frequency of seizures at follow-up as the dependent variable, revealed that etiology had an impact on the frequency of seizures at follow-up. Compared to patients with an unknown etiology, individuals with gene mutations, posttumor surgery, and congenital brain developmental abnormalities exhibited a reduced frequency of seizures following treatment (β = −14.14,
Previous studies have shown that the type of epilepsy does not significantly influence the efficacy rate of ta-VNS,19,20 which is consistent with the results of this study. In this study, there were seven cases of generalized epilepsy; the frequency of seizures was reduced by >90% in 3, 75%–90% in 1, and 50%–75% in 2 patients; only 1 patient had no change in seizure frequency. This reveals an efficacy rate that was as high as 85.71%, whereas the efficacy rate was 58.62% in focal epilepsy, and there was no significant difference between the two groups. This may be due to the small sample size of generalized epilepsy in this study, which can be expanded in the future to further study the efficacy of ta-VNS for different types of epilepsy.
Previous animal studies and clinical trials have confirmed the beneficial effects of VNS on mood disorders, such as anxiety and depression.27–30 VNS mainly stimulates afferent fibers that are anatomically and functionally linked to target areas of the brain, such as the limbic system, to improve mood. Regarding its mode of action, VNS can regulate the concentration of neurotransmitters, such as serotonin, norepinephrine, glutamate, and GABA, and their metabolites, while causing functional alterations in the central nervous system, which allows VNS to produce effects similar to most antianxiety and antidepressant drugs. 31 Several studies have shown that ta-VNS can improve anxiety/depression conditions in addition to its antiepileptic effects19,20,29; ta-VNS has been approved in Europe for the treatment of epilepsy and depression. 32 However, most of this effect takes a relatively long time (up to 12–24 months) to become apparent. In the study by Yang et al., 22 patients treated with ta-VNS showed no significant improvement in their anxiety and depression scores. One possible reason for this is the limited sample size and short follow-up time. Another possible reason is that the level of emotional disorder was relatively low. In 2025, Xu et al. 33 discovered that ta-VNS could effectively alleviate the depressive symptoms of patients with epilepsy and may be an effective and safe treatment method for patients with epilepsy and mild-to-moderate depression. In this study, four patients had epilepsy and anxiety and two had epilepsy alongside depression; the follow-up period exceeded 1 year for both patients. Both experienced subjective improvement after ta-VNS treatment. Unfortunately, we did not conduct an objective anxiety and depression scale assessment, and subsequent studies will be further improved.
Evidence for the benefit of VNS on cognitive function is relatively limited, mainly from animal studies.34–36 Although a small number of clinical studies have reported improvements in cognitive function with VNS,37,38 a randomized, double-blind clinical trial conducted in 2022 demonstrated the efficacy and safety of ta-VNS for mild cognitive impairment. 39 In this study, three patients with epilepsy accompanied by cognitive impairment exhibited subjective improvement following ta-VNS treatment; however, no cognitive function scale assessment was conducted. The effect of ta-VNS on cognitive improvement in patients with epilepsy combined with cognitive impairment needs to be further confirmed in the future.
The results of a single-center randomized clinical trial in 2024 showed that ta-VNS significantly alleviated the severity of insomnia, and its efficacy lasted for 20 weeks. 40 In this study, seven patients with epilepsy and comorbid sleep disorders exhibited improvement following treatment with ta-VNS, and future multicenter clinical trials with a large sample size are needed to verify its effectiveness in different populations.
Our study had some limitations. First, the dropout rate in this study was relatively high (34.34%). Most patients who were lost to follow-up or refused to undergo follow-up underwent ta-VNS treatment, which could potentially influence the outcomes. Owing to ethical considerations, we refrained from conducting additional multiple follow-ups with these patients. The exclusion of these patients possibly resulted in the loss of key information. However, there were no differences in age, sex, baseline seizure frequency, disease duration, age of onset, and seizure type between the 34 patients who were lost to follow-up and the 65 patients who were not lost (
Conclusion
This study found that high-frequency ta-VNS can reduce the frequency of epileptic seizures. We believe that it is an effective, safe, and convenient treatment method for DRE. Moreover, we discovered that ta-VNS may be most effective for patients exhibiting a high frequency of seizures and a well-defined etiology of epilepsy. Etiologies with an efficacy rate exceeding 90% were posttumor surgery, congenital brain dysplasia, and gene mutations. However, the sample sizes for posttumor surgery and congenital brain dysplasia were limited, necessitating further confirmation of the efficacy rates by increasing the sample size. In addition, this study found that ta-VNS also showed certain subjective improvements in epilepsy complications such as anxiety, depression, sleep disorders, and cognitive disorders.
Supplemental Material
sj-docx-1-tan-10.1177_17562864251396022 – Supplemental material for Efficacy and safety of transcutaneous auricular vagus nerve stimulation in drug-resistant epilepsy: a single-center prospective real-world study
Supplemental material, sj-docx-1-tan-10.1177_17562864251396022 for Efficacy and safety of transcutaneous auricular vagus nerve stimulation in drug-resistant epilepsy: a single-center prospective real-world study by Jin Liao, Jingwen Zuo, Yingyue Dai, Jiaying Zhang, Qun Wang, Tao Cui, Maomao Liu and Ruijuan Lv in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
