Abstract
Background:
Highly purified cannabidiol (CBD), recently approved for various neurological disorders, is explored as a potential therapeutic avenue for drug-resistant epilepsy (DRE) among adult people with epilepsy (PWE) in this systematic review and meta-analysis.
Objectives:
To conduct an extensive literature review and meta-analysis of CBD use for DRE in adult PWE.
Design:
Systematic review and meta-analysis.
Data sources and methods:
We conducted a systematic review of the literature according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines and two electronic resources; we searched Ovid MEDLINE and Scopus using appropriate keywords until August 2023. Data were presented as standardized mean difference (SMD) and odds ratio with confidence interval (CI) via random effect. We appraised the risk of bias of the included studies using the Joanna Briggs Institute critical appraisal tool while their strength of evidence with the Oxford Centre for Evidence-Based Medicine (OCEBM) and Grading of Recommendations Assessment Development and Education (GRADE) Levels of Evidence.
Results:
We identified 16 studies, 3 of which were randomized controlled trials and 3 prospective cohort studies, while the rest were expanded access programs, deriving a total of 668 participants receiving CBD for seizure control. CBD was used concomitantly with antiseizure medications in all studies. There was a reduction in seizures in the group receiving CBD therapy compared to the placebo group [SMD: −1.50, 95% CI (−3.47, 0.47)], though this did not reach statistical significance.
Conclusion:
The evidence on CBD use in adult patients with DRE demonstrates a moderate level of certainty according to GRADE level and OCEBM level 2. Further prospective studies involving multiple centers are encouraged to study both the efficacy and safety of CBD in adult patients with DRE.
Trial registration:
International Prospective Register of Systematic Reviews (PROSPERO) 2023 CRD42023449955.
Keywords
Introduction
Epilepsy constitutes a heterogeneous group of neurological disorders characterized by a predisposition to recurrent, unprovoked seizures with multifaceted implications affecting neurobiological, cognitive, psychological, and social domains. 1 Globally, approximately 70 million people across all age groups grapple with epilepsy, with a disproportionate burden experienced in low- to middle-income countries. 2 The global lifetime prevalence of epilepsy is estimated at 7.60 per 1000 persons. 3
People with epilepsy (PWE) are typically treated with antiseizure medications (ASMs) to prevent seizure recurrence. While current ASMs prove effective in controlling seizures for 60%–70% of individuals, about one-third still experience ongoing epileptic seizures despite appropriate treatment, resulting in adverse psychological and social ramifications, compromised educational and occupational attainment, diminished quality of life, and heightened mortality risk.4–7 Drug-resistant epilepsy (DRE) is presently defined as the failure of two ASM schedules, either as monotherapies or in combination, that were appropriately selected, tolerated, and utilized but failed to achieve sustained seizure freedom. 8
Cannabis (Cannabis sativa) contains various compounds, with phytocannabinoids being prominent among them. Of particular interest is cannabidiol (CBD), notable for its lack of psychoactive properties, absence of euphoria or undesirable side effects, and minimal risk of abuse.9,10 While further studies are needed, the anticonvulsant effects of CBD, acting on multiple molecular targets, have been identified to be mediated through various mechanisms, including both agonist and antagonist effects on ionic channels, neurotransmitter transporters, and multiple 7-transmembrane receptors. 10 Negligible activity has been observed at cannabinoid receptors. Moreover, it is suggested that a combination of interactions with cannabinoid receptors, particularly CB1 receptor-mediated and CB1 receptor-independent pathways, contributes to the anti-epileptiform and antiseizure effects of CBD. 11
Recent legislative advancements in the United States and Australia have legalized the use of highly purified CBD for medicinal purposes, encompassing its diverse applications, including the management of rare epilepsy syndromes such as Lennox–Gastaut syndrome (LGS) and Dravet syndrome (DS). This addresses chronic noncancer pain and cancer-related symptoms, mitigating anxiety, and treating other neurological disorders.12,13 Similarly, in Europe, CBD is not classified as a controlled substance, and its use is not restricted by specific laws. 14 The European Medicines Agency authorized CBD in 2019 as an adjunctive therapy for seizures specifically associated with LGS, DS, and tuberous sclerosis complex. 15
Given the nascent nature of research on the use of CBD for DRE management, we would like to investigate CBD as adjunctive therapy for adults with DRE. We aim to delve into the specifics of highly purified CBD interventions in DRE and analyze the outcomes in adult patients following its treatment.
Materials and methods
We conducted this systematic review in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). We registered the study protocol in the International Prospective Register of Systematic Reviews (PROSPERO; ID #CRD42023449955).
Eligibility criteria
We applied the Population, Intervention, Comparison, Outcome, and Study design (PICOS) framework in compliance with the PRISMA 2020 guidelines to define the eligibility criteria for studies included in this review:
Population (P): Studies which documented quantifiable adult patients and their respective sex; adult patients were defined as 18 years of age and above; sample size of at least two participants or one participant in each study group for interventional and observational studies; with confirmed diagnosis of DRE.
Intervention (I): Studies using CBD-only formulations without any tetrahydrocannabinol (THC) or other psychoactive cannabinoids for managing seizures of DRE. We excluded studies that used CBD in combination with THC or other cannabinoids to maintain the focus on the effects of pure cannabidiol in the treatment of DRE. The majority of studies used highly purified CBD, such as Epidiolex®, a plant-derived, pharmaceutical-grade formulation of cannabidiol with a concentration of 100 mg/mL.
Comparison (C): Studies which reported pre- and post-CBD therapy seizure frequency.
Outcomes (O): Studies which reported CBD regime and participants’ outcomes. CBD regime details included the brand and molecular names of CBD, types of CBD used, dosage, frequency, and duration of CBD therapy. Participants’ outcomes included seizure control and adverse events of CBD usage.
Study design (S): Reviews, abstracts, book chapters, patents, symposiums, oral and poster presentations, case reports, and publications whereby full texts cannot be obtained or out of scope were excluded.
Information sources and search strategy
We conducted a comprehensive search at Ovid MEDLINE and Scopus in August 2023 using keyword combinations of (“adult drug-resistant epilepsy” OR “drug-refractory epilepsy” OR “intractable epilepsy” OR “medication-resistant epilepsy” OR “treatment-resistant epilepsy”) AND (“cannabidiol”). We limited our search to studies published in the English language.
Selection process
All relevant articles acquired from the search strategy were imported to the website-based systematic review software, Covidence, with the duplicates withdrawn. Two reviewers independently performed a two-step review of the imported studies. The titles and abstracts of the articles were first screened for inclusion, followed by a full-text review of the chosen studies. A third reviewer resolved any conflicts between the two reviewers.
Data collection process and data items
The data were extracted from the selected studies using the data extraction features of Covidence. The data were then exported and cross-checked using Microsoft Office 365 Excel. The data extracted includes study design, participant demographics, CBD regime, and participant outcomes. Participant demographics included age, sex, geography, and ethnicity. Details of epilepsies included therapies prior to CBD treatment, the diagnosis of DRE, pre- and post-CBD therapy seizure frequency. CBD regime includes the brand and molecular names of CBD, types of CBD used, dosage, frequency, and duration of CBD therapy. Participants’ outcomes included seizure control and adverse events of CBD usage. For controlled studies, we extracted relevant information along with their variances. For uncontrolled studies, we extracted similar information but without control group comparisons. The final Excel format was then converted to an R-compatible format for analytical implementation on the R interface.
Risk of bias assessment
Three independent reviewers assessed the risk of bias in the included studies using the Joanna Briggs Institute (JBI) critical appraisal tools tailored to their study designs. The cumulative scores from domains queried in the tools reflect the overall risk of bias, where higher scores indicate a lower risk. This scoring methodology allows categorization into high (<50%), moderate (50%–79%), or low (⩾80%) bias risk based on the percentage of selected criteria. Any discrepancies in scoring were resolved through consensus by a third reviewer, ensuring a comprehensive assessment of bias across the included studies.
Synthesis methods
We performed descriptive summaries for the characteristics of included studies, participants, and key findings.
We employed a random-effects model using the R 4.3.1 software with meta version 7.0-0 package to synthesize the results. 16 The random-effects model accounts for both within-study and between-study heterogeneity of participant demographics, CBD regimes, and outcomes, providing more conservative estimates of effect sizes as we combined diverse study designs. Effect sizes were calculated as standardized means and standard deviations for continuous outcomes and odds ratios for dichotomous outcomes with a confidence interval (CI) of 95% using the Cochran–Mantel–Haenszel test and visualized in forest plots. The width of the squares in the forest plot corresponds to the weight of each study in the analysis. Heterogeneity among studies was assessed using both the I2 statistic (>50% indicates significant heterogeneity) and Cochran’s Q test (p < 0.1 indicates significant heterogeneity).
Studies where quantitative data are limited or heterogeneity is too high to be included in the meta-analysis are discussed in a narrative synthesis.
Certainty assessment
Two independent reviewers performed a certainty assessment for each included study, utilizing the Grading of Recommendations Assessment Development and Education (GRADE) Levels of Evidence and the Oxford Centre for Evidence-Based Medicine (OCEBM) Levels of Evidence frameworks. Any discrepancies between the assessments of the two reviewers were resolved via a third reviewer.
Results
Selection of studies
Figure 1 shows the search and selection of studies based on the PRISMA flowchart. The electronic search retrieved a total of 2594 studies from Scopus and 143 studies from OVID Medline. Of these studies, we removed 136 studies due to duplicates. We further excluded 2526 studies based on the title and abstract content. We retrieved the remaining 75 studies for full-text evaluations and excluded another 59 for not meeting the eligibility criteria. Finally, 16 studies were included in this review. Ten of the 16 studies were included in the meta-analysis. The search algorithm utilised can be referred to our Supplemental Material.

Flow chart.
Participant demographics
Of the 16 studies, 10 are expanded access programs (EAPs), 3 are prospective cohort studies, and 3 are randomized controlled trials (RCTs). Eleven out of 16 studies were conducted in the United States of America. Across all studies, 668 adult PWE treated with CBD were identified, who included 340 males and 328 females. Therapies before initiating CBD trials include ASMs, vagus nerve stimulation, ketogenic diet, and epilepsy surgeries such as callosotomy and lobectomy. Common concomitant ASMs used were levetiracetam, clonazepam, carbamazepine, valproic acid, and lamotrigine. The subjects’ demographics and their clinical information are summarized in Table 1.
Profile of recruited participants.
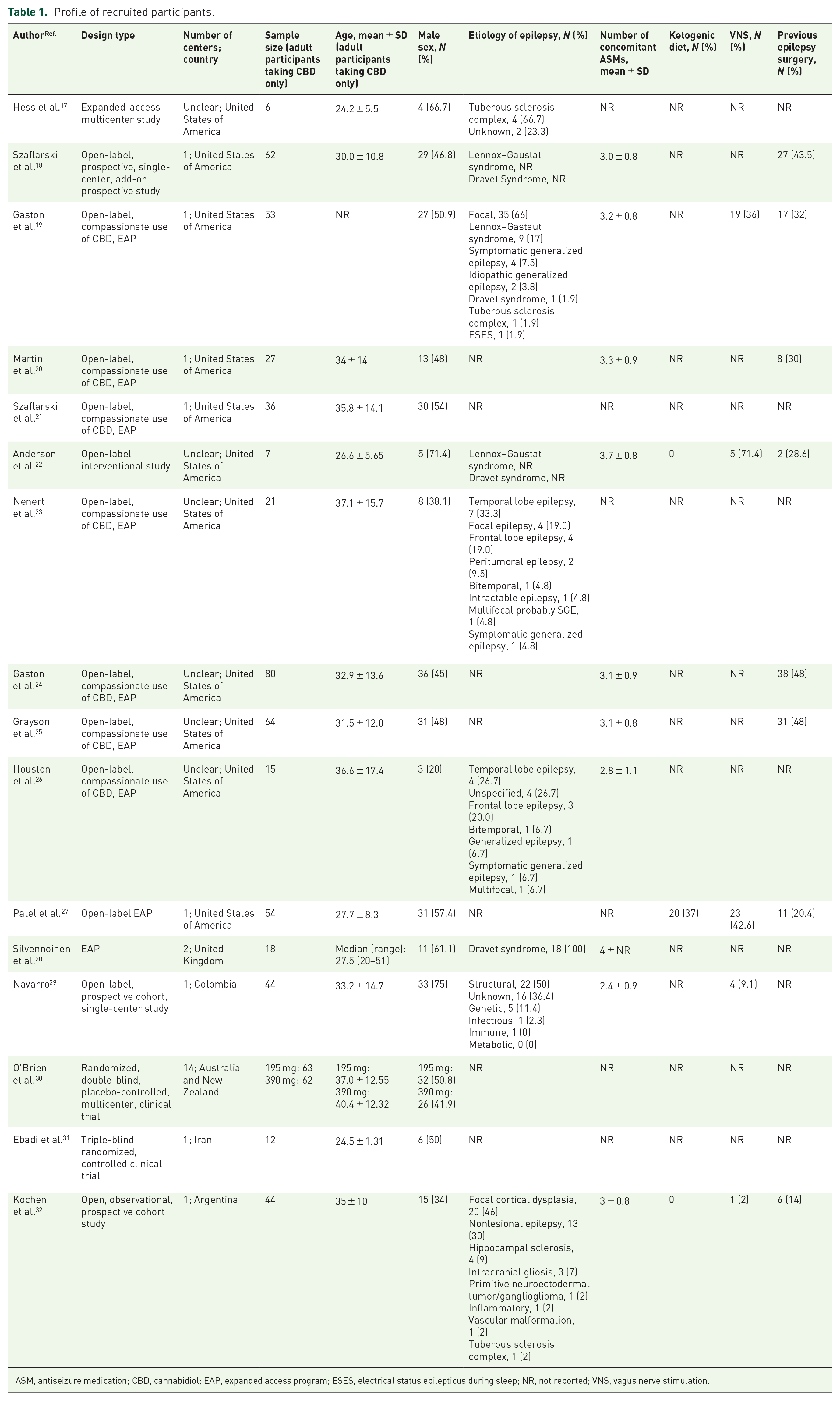
ASM, antiseizure medication; CBD, cannabidiol; EAP, expanded access program; ESES, electrical status epilepticus during sleep; NR, not reported; VNS, vagus nerve stimulation.
CBD regime and outcomes
CBD was used concomitantly with ASMs in all studies. Twelve studies used a plant-derived highly purified CBD solution named Epidiolex, which contains 100 mg/mL of CBD solution administered orally. In terms of the route of CBD administration, 13 were administered orally, 2 sublingually, and 1 transdermally. Ten studies used 50 mg/kg/day as the maximum CBD dosage. The CBD dosing across studies was homogenous at 6- and 12-month points, as illustrated by forest plots in Figures 2 and 3, with an I2-value of 0%.18,19,21–25

CBD dosage (mg/kg/day) at 24 weeks/6 months, illustrated by forest plots.
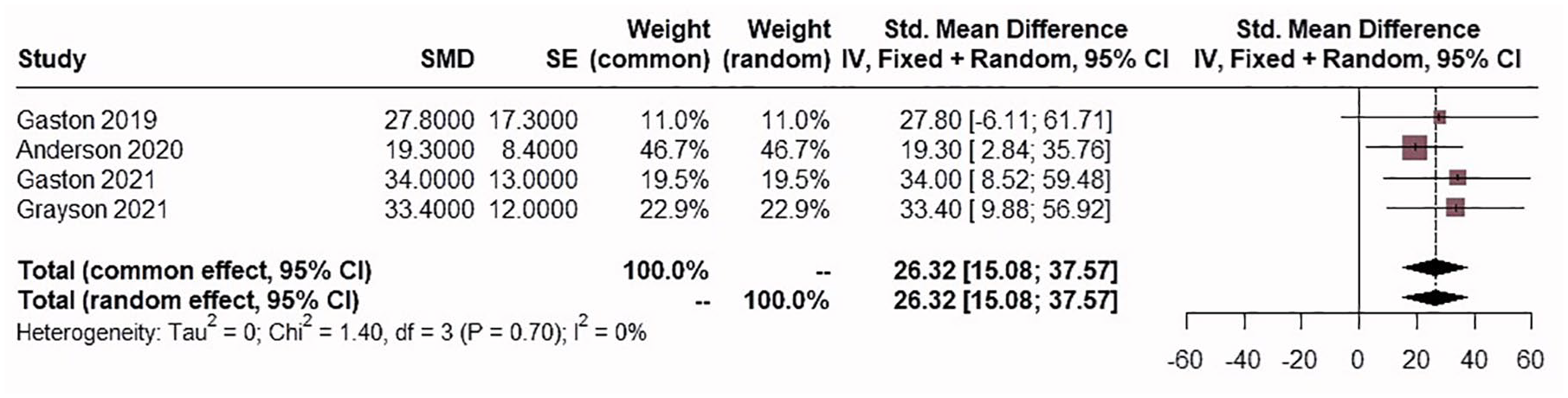
CBD dosage (mg/kg/day) at 12 months, illustrated by forest plots.
Seizure reduction was observed in all studies but was reported to be statistically significant in the studies by Gaston et al. 19 and Ebadi et al. 31 Three studies compared changes in seizure frequency between the CBD-treated and placebo/control groups. These studies include 162 participants receiving CBD therapy and 194 placebo/control participants. Using a random-effects model, as depicted in Figure 4, there was a reduction in seizure frequency in the group receiving CBD therapy compared to the placebo/control group [SMD: −1.50, 95% CI (−3.47, 0.47)], though this did not reach statistical significance.
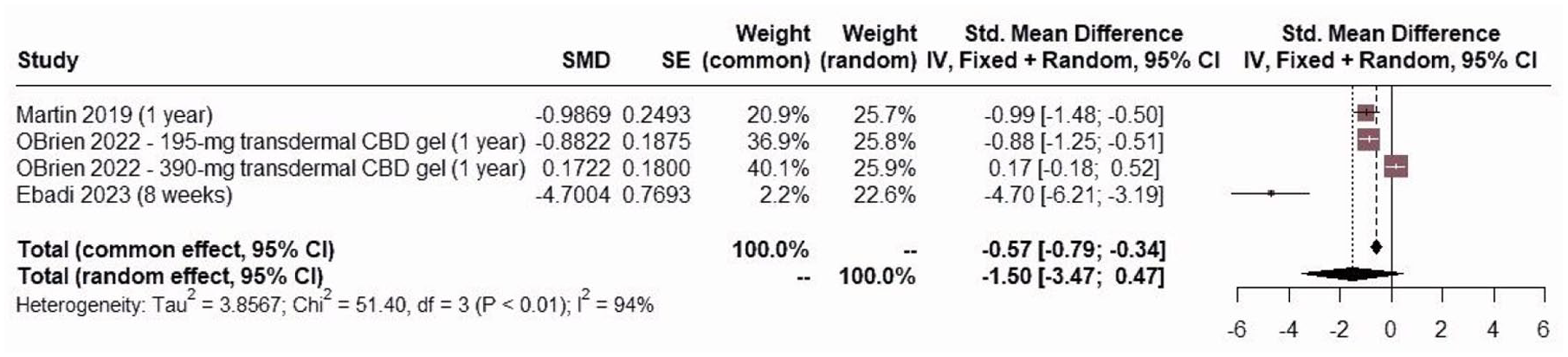
The pooled results of comparing the change in seizure frequency between the groups receiving CBD therapy to the placebo/control group, illustrated by forest plots.
The studies reported adverse events and patient outcomes differently, resulting in heterogeneity. Of the 16 studies, only 11 looked into adverse events related to CBD administration. Common adverse events reported following CBD treatment are gastrointestinal and neuropsychiatric manifestations. One study reported the three most common adverse events: diarrhea, sedation, and decreased appetite. 24 Regarding patient outcomes, two studies reported improved quality of life and mood profiles. The CBD regime and outcomes of each study are further described in Table 2.
CBD regime and outcomes of included studies.

AD, axial diffusivity; AEP, adverse events profile; ASM, anti-seizure medication; CBD, cannabidiol; CBMF, cannabis-based-magistral formulation; FA, fractional anisotropy; LFT, liver function test; MD, mean diffusivity; NR, not reported; POMS, Profile of Mood States; QoLI-31, quality of life; QOLIE, Quality of Life in Epilepsy; RD, radial diffusivity; SD, standard deviation; THC, tetrahydrocannabinol. Bolded values indicate statistical significance.
Risk of bias assessment
Tables 3 and 4 summarize the quality assessment for each study using the JBI critical appraisal tools tailored to their respective study designs. Two case series were judged to be at a low risk of bias, while the other two were judged to be at moderate risk of bias, primarily due to lack of methodology description.
JBI critical appraisal checklist for cohort studies.
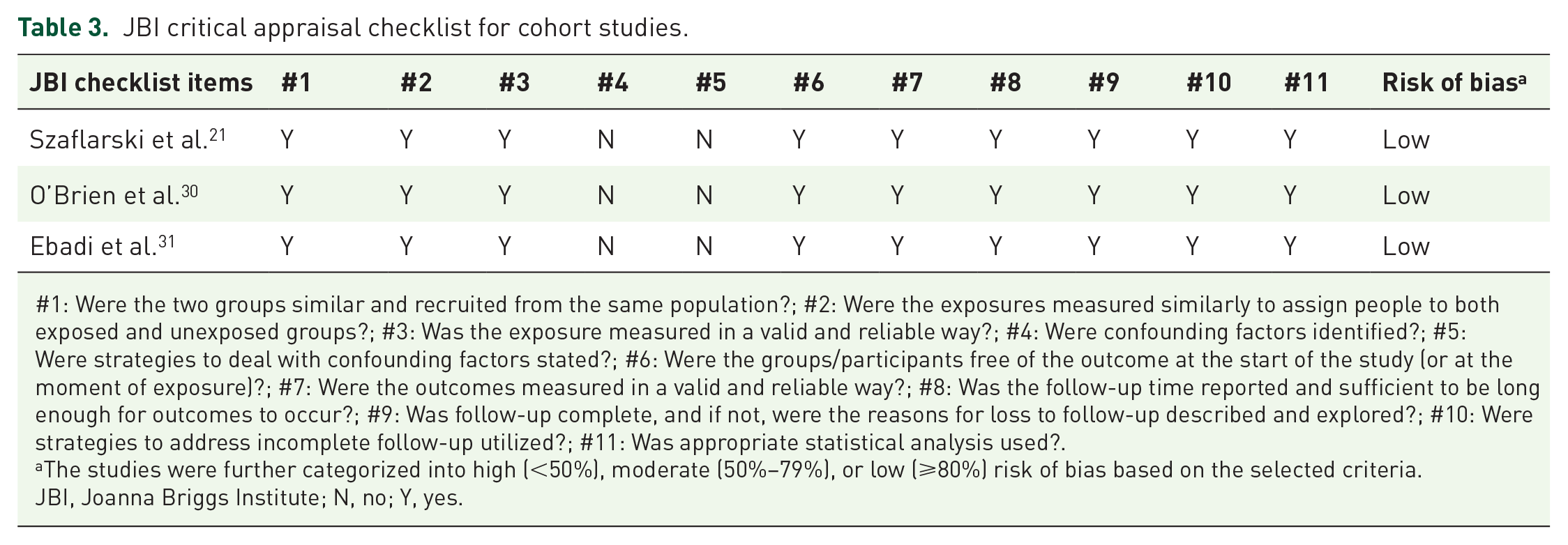
#1: Were the two groups similar and recruited from the same population?; #2: Were the exposures measured similarly to assign people to both exposed and unexposed groups?; #3: Was the exposure measured in a valid and reliable way?; #4: Were confounding factors identified?; #5: Were strategies to deal with confounding factors stated?; #6: Were the groups/participants free of the outcome at the start of the study (or at the moment of exposure)?; #7: Were the outcomes measured in a valid and reliable way?; #8: Was the follow-up time reported and sufficient to be long enough for outcomes to occur?; #9: Was follow-up complete, and if not, were the reasons for loss to follow-up described and explored?; #10: Were strategies to address incomplete follow-up utilized?; #11: Was appropriate statistical analysis used?.
The studies were further categorized into high (<50%), moderate (50%–79%), or low (⩾80%) risk of bias based on the selected criteria.
JBI, Joanna Briggs Institute; N, no; Y, yes.
JBI critical appraisal checklist for case series.

#1: Were there clear criteria for inclusion in the case series?; #2: Was the condition measured in a standard, reliable way for all participants included in the case series?; #3: Were valid methods used for identification of the condition for all participants included in the case series?; #4: Did the case series have consecutive inclusion of participants?; #5: Did the case series have complete inclusion of participants?; #6: Was there clear reporting of the demographics of the participants in the study?; #7: Was there clear reporting of clinical information of the participants?; #8: Were the outcomes or follow-up results of cases clearly reported?; #9: Was there clear reporting of the presenting site(s)/clinic(s) demographic information?; #10: Was statistical analysis appropriate?
The studies were further categorized into high (<50%), moderate (50%–79%), or low (⩾80%) risk of bias based on the selected criteria.
JBI, Joanna Briggs Institute; N, no; Y, yes.
Certainty of evidence for CBD in DRE
A total of 16 manuscripts were systematically reviewed, revealing a spectrum of evidence quality across the studies. Many of the studies were open-label, introducing potential biases such as reporting bias and placebo effects, which may have influenced subjective outcomes like seizure reduction and quality of life improvements. The lack of blinding reduces the certainty of these results, and they should be interpreted with caution.
Among the included studies, three had a moderate GRADE level of evidence and 2b OCEBM level, indicating sound evidence derived from individual RCTs due to their use of blinding and placebo controls. Concurrently, six studies were categorized as having a low GRADE level of evidence and 3b OCEBM level, while seven were designated as having a very low GRADE level of evidence and 4 OCEBM level. Overall, the certainty of evidence for the utilization of CBD in DRE emerges as a combination of moderate, low, and very low-quality studies. The distribution of evidence levels is presented in Table 5.
Study characteristics and certainty of evidence for CBD in DRE.

CBD, cannabidiol; DRE, drug-resistant epilepsy; OCEBM, Oxford Centre for Evidence-Based Medicine.
Discussion
Optimal CBD regime
The dosing of CBD varied across studies, with a starting dose of as low as 2.5 mg/kg/day with gradual uptitration, monitoring of clinical response for as short as 8 weeks, to as long as 2 years. The CBD doses were escalated as tolerated, not more than 5 mg/kg/day in every 1–2 weeks, practiced in 10 studies. The maximum dose reported was 50 mg/kg/day, administered in 10 studies. Among these, 12 studies utilized a plant-derived highly purified CBD sesame seed oil-based oral solution known as Epidiolex, with a concentration of 100 mg/mL.
In 2019, Thailand became the first Southeast Asian country to legalize cannabis and cannabinoids for medical purposes. This legalization has led to the common use of CBD, along with THC, for treating various medical conditions, including intractable epilepsy and spasticity in multiple sclerosis patients, as identified by the Thai Ministry of Public Health. 33 However, due to the psychoactive effects of THC, its legally accepted concentration is limited to 0.2%. Therefore, our review exclusively focuses on pure CBD formulations.
The route of administration of CBD across the 16 studies included oral, sublingual, and transdermal routes, with the oral route being the most prevalent. Oral CBD exhibits a half-life of 1–2 days and undergoes extensive first-pass metabolism via an enzyme, which is a member of the cytochrome P450 (CYP) family, CYP3A4, resulting in a bioavailability of approximately 6%. In vitro studies have indicated that CBD is a potent inhibitor of multiple CYP isozymes, including CYP2C and CYP3A. However, the relevance of these in vitro findings at plasma concentrations remains uncertain. The lipophilic properties of CBD facilitate access to intracellular sites, thereby modulating calcium levels in various cell types, including hippocampal neurons. This modulation of calcium homeostasis may underlie CBD’s neuroprotective properties, as CBD targets transient receptor potential channels involved in intracellular calcium modulation. 34
Effects of adjunctive antiseizure therapy with CBD administration
Among the 16 studies reviewed, concurrent ASMs commonly included levetiracetam, clonazepam, carbamazepine, valproic acid, lamotrigine, and clobazam. The concurrent use of clobazam in particular has been reported to result in a variety of side effects. Some of the included studies maintained a consistent clobazam dosage, while others adjusted clobazam doses due to adverse reactions or significant laboratory results related to CBD interactions.27,29 Conversely, some clinicians opted to exclude participants who were using clobazam to control for its potential effects.30,31 The most frequently reported adverse event associated with CBD and clobazam co-administration was somnolence, observed in 6 out of 16 studies. This phenomenon is attributed to increased levels of clobazam and its active metabolite, N-desmethylclobazam, due to their shared metabolic pathway via CYP. 17 While there is evidence suggesting that combined CBD and clobazam treatment may enhance anticonvulsant effects compared to ASMs alone, this relationship appears complex. A lower subthreshold dose of CBD did not promote greater antiseizure effects despite increasing plasma clobazam concentrations, indicating that CBD may possess independent anticonvulsant activity. 35
CBD use on longitudinal EEG changes
Only one study used the identification and quantification of interictal epileptiform discharges (IEDs) to measure the response to CBD therapy. 25 In this EAP, longitudinal EEG data were analyzed, focusing on the number of IEDs per unit of time. These IEDs have been associated with both the frequency and severity of seizures, noting a positive but nonsignificant association between CBD dosage and longitudinal changes in IEDs.36,37
While the precise role of IEDs as an indicator of the effectiveness of antiseizure therapy remains somewhat unclear, it is noteworthy that multiple studies, encompassing both animal and human subjects, have consistently demonstrated a proportionate reduction in IEDs following CBD treatment.38,39 However, there are no reported associations between the frequency of IED and seizure frequency, with or without antiseizure therapy. Still, they reported statistical significance between IED incidence and epilepsy duration (p = 0.04). 40
CBD effects on brain imaging
A couple of studies highlighted an additional area of interest in our investigation pertaining to the brain imaging of participants undergoing CBD therapy. The first of them, by Houston et al., analyzed MRI changes and found significant alterations in white matter integrity, albeit without significant differences after adjustments for various controlling factors. 26 Notably, short-term exposure to CBD did not yield significant changes in gray matter volume or cortical thickness in patients with treatment-resistant epilepsy. 41
Another study investigated the resting-state functional connectivity (rs-FC), revealing substantial alterations across multiple brain regions, encompassing the cerebellum, frontal areas, temporal areas, hippocampus, and amygdala. 23 Furthermore, CBD demonstrated a reduction in activation within brain regions implicated in stimulus conflict resolution and normalization of condition-based functional connectivity in the right superior frontal gyrus. 42 In epilepsy patients, network impairment in the left hippocampus and left thalamus regions was identified, corresponding to changes in rs-FC. 43 This finding underlines the potential utility of investigating rs-FC as a biomarker for treatment response in epilepsy patients. 44
CBD use on cognitive function
We have discussed and learned the significant CBD-induced alteration via neuroimaging. However, the research findings on CBD’s impact on cognitive function vary widely. One of our studies reported no statistically significant association with CBD and changes in cognition. 20 A statistically significant reduction of cognitive functioning associated with frequent or heavy CBD use was reported (95% CI, −0.32 to −0.17; p < 0 .001), while a small positive effect of CBD on working memory, assessed by backward digit span, was noted.45,46 Furthermore, South Korean researchers have suggested that CBD is neurotoxic, proposing that astrocytes may mitigate CBD-induced neurotoxicity by regulating intracellular calcium mechanisms. Calcium imaging showed higher intracellular calcium concentration in neurons treated with CBD than in neurons co-cultured with astrocytes. It is speculated that the astrocytes reduce the neurotoxicity of CBD by regulating intracellular calcium mechanisms and sustaining neuronal viability. 47
Though not discussed in our review, functional outcomes represent a crucial secondary measure of cognitive functioning, encompassing academic and occupational outcomes influenced by various demographic factors. We did not include this in our discussion as the functional outcome is heterogeneous and complex, mounted by multiple acquired factors, which are very demographic-dependent, requiring more specific prospective modeling.48,49
Study limitations
The open-label design prevalent in many studies introduced potential reporting and placebo biases, particularly affecting subjective outcomes such as seizure reduction and quality of life improvements. Additionally, the heterogeneity in outcome reporting and reliance on self-reported data may have contributed to reporting and selection biases. As most studies relied on Epidiolex, the findings may not fully represent the general effectiveness of CBD for DRE across various formulations. Additionally, the concurrent use of ASMs alongside CBD treatment makes it challenging to isolate the effects of CBD alone on DRE or to fully understand its combined impact with ASMs. Consequently, while findings suggest efficacy, the certainty of evidence remains a combination of moderate, low, and very low quality, underscoring the need for further high-quality, multicenter RCTs to better elucidate CBD’s therapeutic potential in this population.
Conclusion
Our review has shown that CBD may reduce seizures as an adjunctive therapy in adult patients with DRE, though pooled results did not reach statistical significance. Given the consistency in the reported positive outcomes observed as seizure reduction across all included studies, the level of evidence on CBD use in adult PWE with DRE is classified as moderate GRADE level and OCEBM level 2. Further prospective multicenter studies are required to explore the use of CBD in adult patients with DRE of various etiologies.
Supplemental Material
sj-docx-1-tan-10.1177_17562864251313914 – Supplemental material for The use of cannabidiol as adjunctive therapy in adult patients with drug-resistant epilepsy: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tan-10.1177_17562864251313914 for The use of cannabidiol as adjunctive therapy in adult patients with drug-resistant epilepsy: a systematic review and meta-analysis by Marjorie Jia Yi Ong, Muhammad Samir Haziq Abd Rahman, Vanessa Lin Lin Lee, Kong Heng Lee, Carmen Jia Yinn Chang, Ching Soong Khoo, Rozita Hod, Hui Jan Tan and Eugen Trinka in Therapeutic Advances in Neurological Disorders
Footnotes
Appendix
Acknowledgements
None.
Correction (April 2025):
Since the original online publication, section “CBD regime and outcomes” has been updated.
Declarations
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
