Abstract
Drug-resistant frontal lobe epilepsy is a challenge in epilepsy surgery because it is the second most common epilepsy type after temporal lobe epilepsy. Additionally, the frontal lobe is the largest brain lobe, making the localization of epileptic foci difficult, thereby preventing necessary surgery and improving seizure control. This review discusses the epidemiology and aetiology of drug-resistant frontal lobe epilepsy, demonstrating that cortical developmental manifestations constitute the most common aetiology of epileptic focus. Additionally, we reviewed the anatomy and symptoms of frontal lobe seizures. The review discusses advances in the neurophysiological study, including magnetoencephalography and neuroimaging modalities, such as quantitative PET scans and 7T MRI, for lesion detection and outcome improvement in frontal lobe epilepsy surgery. Although 7T MRI is unavailable in many epilepsy centres, the imaging modality can increase the rate of lesion detection, particularly when its findings are confirmed using depth electrode implantation, along with the utilization of artificial intelligence and machine learning in interpreting the morphometric analysis of MRI and nuclear imaging.
Introduction
Drug-resistant epilepsy is defined as failure of adequate trials of two tolerated and appropriately chosen and used anti-seizure medication schedules (as monotherapy or in combination) to achieve seizure freedom. 1 The International League Against Epilepsy (ILAE) classifies seizures as generalized, focal or unknown according to aetiology. Focal seizure is anatomically classified according to the lobe in which the seizure originates, and it includes frontal, temporal, parietal, occipital and insular epilepsy. Most patients with epilepsy respond to medical therapy; however, 20%–30% of patients do not respond to antiepileptic drugs. In such cases, surgical management should be considered where the frontal lobe epilepsy is the second most common cause of drug-resistant epilepsy after temporal lobe epilepsy.2–4
Frontal lobe seizure has a different semiology that overlaps with other types of focal epilepsy. In addition, it can be mistaken for a paroxysmal non-epileptic event, and the reverse is not uncommon, where a patient with a nonepileptic event may get diagnosed with drug-resistant epilepsy, which is pseudo-resistance. With increasing awareness among patients and physicians regarding surgical management of drug-resistant epilepsy, which leads to increased patient referrals to epilepsy centres, temporal lobe epilepsy has been recognized as more easily treatable compared to frontal lobe epilepsy because of the characteristic semiology, Electroencephalogram (EEG) findings, or MRI findings. Therefore, the treatment of patients with frontal lobe seizures presents a challenge to physicians.
The present review discusses the anatomy of the frontal lobe with figures illustrating the surface landmarks and epidemiology of frontal lobe epilepsy; notably, obtaining accurate estimates is difficult in frontal lobe epilepsy, although there are recent advancements in neuroimaging and an increased number of epilepsy centres that can confirm a diagnosis based on pathology after surgical resection. In addition, the review discusses the common aetiology, semiology of seizure with the anatomic correlation and mimickers, lateralizing signs, EEG findings in the interictal and ictal phases, depth electrode recording, magnetencephalography, neuroimaging findings (MRI, PET and the 7T MRI), surgical resection outcomes based on the Engel classification, and factors that affect the surgical outcome. Multi-modal assessment of drug-resistant epilepsy, lesion localization with 7T MRI, and Stereoelectroencephalography (SEEG) implantation have significant roles in improving the outcome of epilepsy surgery.
Anatomy of the frontal lobe
The frontal lobe is the largest lobe of the brain and comprises the dorsolateral, mesial and inferior aspects. The dorsolateral aspect consists of three sulci and four gyri, which includes the precentral, superior frontal, middle frontal and inferior frontal gyri. Furthermore, the inferior frontal gyrus comprises three distinct portions: the pars orbitalis, the pars triangularis and the pars opercularis. Anatomically, the precentral gyrus lies anterior to the central sulcus and forms the posterior part of the frontal lobe. In addition, the superior frontal gyrus is separated from the middle frontal gyrus by the superior frontal sulcus, and the middle frontal gyrus is separated from the inferior frontal gyrus by the inferior frontal sulcus5,6 (Figures 1 and 2).

The dorsolateral aspect of the frontal lobe.

Sagittal T1-weighted MRI showed the dorsolateral structure of the frontal lobe.
The medial surface of the frontal lobe contains the corpus callosum, which is separated superiorly from the cingulate gyrus by the callosal sulcus. Superior to the mesial aspect of the frontal lobe is the superior frontal gyrus, which is separated from the cingulate gyrus by the cingulate sulcus. Posterior to the superior frontal gyrus is the paracentral lobule, separated from the superior frontal gyrus by the paracentral sulcus5,7 (Figures 3 and 4).
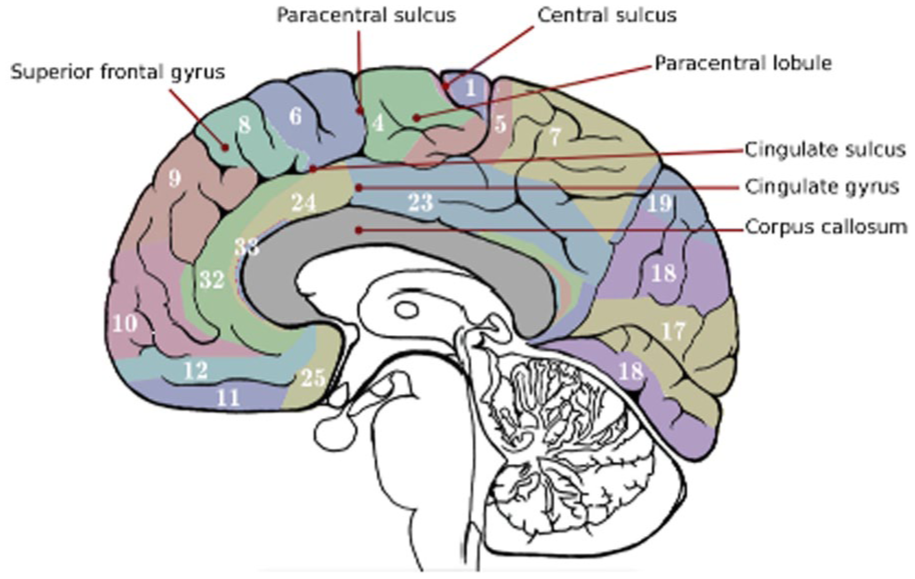
The mesial aspect of the frontal lobe.

Sagittal T1-weighted MRI showed the medial structure of the frontal lobe.
On the inferior surface, the frontal lobe is formed by the gyrus rectus on the medial aspect, which is separated from the four orbital gyri on the lateral aspect by the olfactory sulcus. The four orbital gyri are the anterior, posterior, medial and lateral orbital gyri separated by the H-shaped orbital sulcus5,8 (Figures 5 and 6).

The orbitofrontal aspect of the frontal lobe.

Axial T1-weighted MRI of the brain showing the orbitofrontal structure.
Epidemiology
Seizure affects 2%–5% of the general population, with 30% of the affected individuals developing epilepsy. In adults, focal epilepsy represents 55%–83% of all epilepsy cases, generalized epilepsy represents 6%–32%, and unclassified epilepsy represents 8%–20%. 9 Frontal lobe epilepsy is the second most common type of focal epilepsy, with temporal lobe epilepsy being the most common type. Moreover, frontal lobe epilepsy accounts for ~15%–30% of surgical cases.6,10,11
A study of 68 patients revealed that the most common causes of frontal lobe epilepsy were tumours (35% of cases), followed by dysgenetic lesions (26%), gliosis (21%), vascular malformation (15%) and necrosis (1.5%). 12 In a study from Cleveland Clinic of 70 patients, the most common aetiology was malformations of cortical development with abnormal MRI in 41% patients, followed by malformations of cortical development with normal MRI in 17%, tumour in 19%, vascular malformation in 3%, gliosis and encephalomalacia in 10%, and cryptogenic in 10%. 13 Furthermore, in another study of 23 patients with frontal lobe epilepsy, the most common aetiology was developmental problems in eight (34%) patients, followed by tumour in seven (30%), and other etiologies, such as gliosis in seven (30%) and vascular malformation in two (8%). 14 In tropical and developing countries, infectious aetiology should be considered, as parasitic infections such as cysticercosis, Schistosomiasis, and Hydatid disease can present with frontal lobe seizures.15–17
Pseudo-refractory epilepsy is defined as a seizure inadequately treated due to incorrect diagnosis, use of erroneous and/or low-dose anti-epileptic drugs and poor compliance of patients. In a retrospective study of 449 patients in a tertiary epilepsy centre who were labelled as drug-resistant epilepsy, 66.1% had true refractory epilepsy, 11.9% were misdiagnosed, 10.2% were on inappropriate medicine, 9.3% were on an inadequate dose of anti-seizure drug and 2.5% had poor compliance with anti-seizure medicine. 18 Another prospective study conducted in Denmark involved 41 patients, and 39 patients completed the study; one third of patients developed pseudo-resistant epilepsy due to wrong classification and inadequate adherence to anti-seizure medicine. 19
Semiology of frontal lobe seizure
The frontal lobe is the largest lobe of the brain, with complex connections to the temporal, parietal and insular lobes, which can lead to false localization. Therefore, there is a need to analyze seizure semiology and consider other mimickers carefully. Additionally, some areas of the frontal lobe are silent, and seizures typically only manifest after they spread to other regions. The goal of seizure semiology is to identify the symptomatogenic zone in the brain cortex that produces seizure signs and symptoms or the aura when stimulated. Furthermore, the zone can overlap or be in proximity to the epileptogenic zone (EZ), which is the area of the brain cortex that generates seizures; the complete removal or disconnection of this region resolves the seizures. Additionally, it is crucial to note that seizures can spread throughout the cortex, from one area to an adjacent region or through pathways and connections in the white matter. 20
In a retrospective study of frontal lobe seizures in children, 549 seizures were reviewed from 79 patients, and the mean seizure duration was 25.7 s, and the mean post-ictal confusion was 27 s; 56.5% of seizures were nocturnal. The most common semiology was tonic seizure in 77.2% then versive seizure in 26.7%. Clonic seizures were observed in 17.7% and complex motor seizures in 20.5%, dialeptic seizures were seen in only 3% of all seizures and vocalization in 16%. 21
Symptoms of the precentral gyrus include contralateral tonic posturing, clonic jerks, or cortical myoclonus. Seizures originating from the premotor cortex result in asymmetric tonic posturing (the fencing posture), which is flexion of the ipsilateral arm and extension of the contralateral arm or the Figure 4 posture. In addition, such seizures can lead to complex motor seizures, such as fumbling and picking. The frontal eye field is in the posterior part of the superior frontal gyrus, and the activation of this area leads to versive (an unnatural turn) contralateral gaze and head movement. The frontal operculum is involved in chocking sensation, facial contraction and hypersalivation, and the adjacent posterior inferior frontal gyrus produces expressive aphasia. The prefrontal area can be divided into the dorsolateral (involved in complex automatism) and the ventromedial regions (involved in hyperkinetic motor seizure and psychic aura (fear sensation)).22,23 Table 1 summarizes the common frontal lobe semiology with localization and its mimickers.
Summary of the symptoms of frontal lobe seizure.
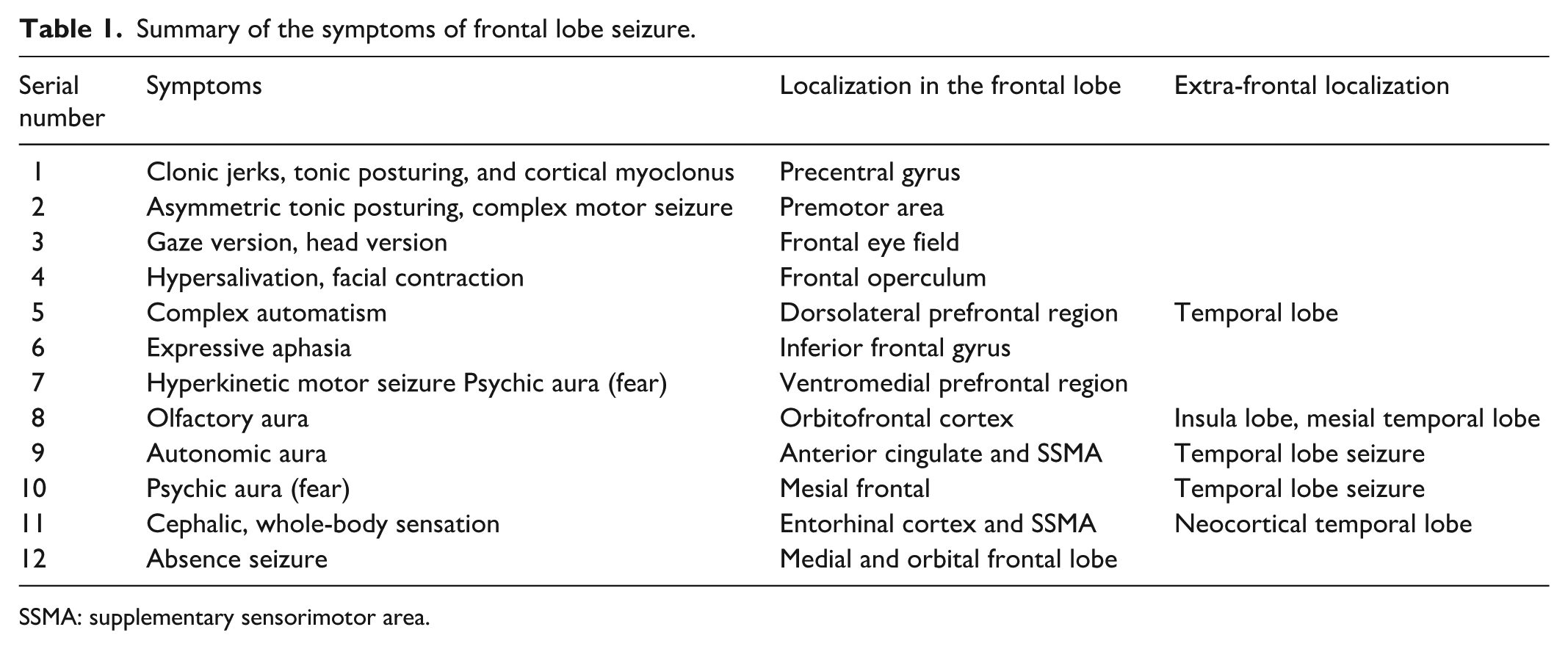
SSMA: supplementary sensorimotor area.
A hypermotor seizure (HMS) arises from the frontal lobe, and it is often mischaracterized as a non-epileptic seizure. Furthermore, HMS is a complex, high-amplitude, violent and unexpected movement in the proximal muscles. HMS is divided into two types: (1) HMS type 1, originating from the ventromedial frontal lobe, is characterized by kicking, boxing or body rocking with automatism, facial expression like fear and (2) HMS type 2, arising dorsal to the mesial premotor cortex and anterior cingulate, is characterized by horizontal movements and trunk rotation. Moreover, HMS can arise from the temporal lobe (mesial, neocortical and pole), insular cortex and parietal lobe. 24 Regarding the lateralization to the contralateral side, the most significant signs are version, unilateral tonic movement, unilateral dystonic posture and unilateral tonic posture. 25
A study involved 54 patients with frontal lobe seizures and classified the clinical semiology into four groups based on the stereotactic EEG findings. The first group was characterized by elementary motor signs (clonic movement, tonic/dystonic contraction and posturing as well as head/eye versive movement) and localize to precentral and premotor gyrus, the second group showed combination of elementary motor signs and non-integrated gestural motor behaviour (complex motor behaviour like hyperkinetic movement, stereotypies and integrated and none-integrated motor behaviour) and localize to premotor and prefrontal regions, the third group showed non-integrated gestural motor behaviour and distal stereotypies. It localized to the anterior lateral and medial prefrontal areas, and the fourth group showed seizures with fearful behaviour, localized to the paralimbic system, which includes the ventromedial prefrontal cortex and the anterior temporal structure. 26
Another study of eight patients with drug-resistant frontal lobe epilepsy was conducted to assess seizure semiology and brain perfusion pattern. A total of 176 seizures were recorded, and the time between the electrographic and seizure onset ranged between 0 and 18 s. Semiology started with staring and automatism in 50% and focal seizures without impaired consciousness in 25%. Seizures continued to upper limb clonus, three patients showed evolution to bilateral tonic clonic seizure, and in one patient, tremor in the upper and lower limbs, ipsilateral to the EZ, was detected before the first minute. Analysis of semiology revealed a frontal epileptogenic network that involves the ipsilateral dorsolateral prefrontal region, primary motor area, basal ganglia and ultimately the entire cerebral cortex. Early injection during Single-Photon Emission Computed Tomography (SPECT) (mean 5.8) helps determine that both the cingulate gyrus and inferior frontal gyrus are part of the estimated EZ. 27
Another study analyzed emotional behaviours in 469 seizures from 42 patients, and the total objective and subjective ictal emotional phenomena were 79%, with objective emotional signs accounting for 64%. Based on the ictal emotional signs, patients were divided into four groups: one group with a cluster of negative behaviour (active threat response) and localized to the posterior orbitofrontal region, anterior cingulate and dorsolateral and/or ventrolateral prefrontal region, the second group (passive fear) localized to the amygdala and mesial temporal structure and posterior orbitofrontal region. 28
Sleep-related hypermotor seizure (SHE), previously known as autosomal dominant nocturnal frontal Lobe epilepsy, needs to be considered in patients with sleep hypermotor seizure with normal MRI, many genes were identified and a recent case series of SHE from a tertiary epilepsy centre present eight cases where six of eight patients had hyperkinetic seizures, two patients had asymmetric dystonic posturing. All seizure arises from NREM sleep, and MRIs were normal in all patients. Genetic studies were conducted for the main known genes, including a single mutation in CHRNA4 and CHRNB2, and three patients had a mutation in DEPDC5. 29
EEG findings in frontal lobe seizure
In a study of patients with tumoural frontal lobe epilepsy, the surface EEG showed normal findings in 13.8% of patients, interictal epileptiform discharge in 10.3%, ipsilateral slowing in 24.1%, and 27.6% was either generalized, bifrontal or false lateralizing. 11
A study of an ictal EEG pattern in frontal lobe epilepsy categorized patients into the mesial frontal lobe and lateral frontal lobe epilepsy groups. In the lateral frontal group, the most common ictal pattern was repetitive epileptiform activity in 36% patients, followed by rhythmic delta activity in 26%, suppression in 14%, rhythmic theta activity in 9%, paroxysmal fast activity in 9%, arrhythmic activity in 3% and rhythmic alpha in 2%; 1% of patients had an obscured ictal onset. Meanwhile, in the mesial frontal group, the most common pattern was paroxysmal fast activity in 33% patients, followed by suppression in 29%, rhythmic delta in 18%, arrhythmic activity in 10%, rhythmic delta in 4% and repetitive epileptiform activity in 4%; the pattern was obscured in 2% of patients. 14 In another prospective cross-sectional study of 175 patients with frontal lobe epilepsy, 461 EEG recordings were analyzed. The most common abnormality was an isolated sharp wave in 97.1%, followed by an isolated spike in 50.2%, and then a polyspike in 6.9%. A seizure was recorded in 51 of the 175 patients, which is 28.9%, and the most common ictal onset was the inferior frontal region. Seizures arising from the prefrontal electrodes did not show any attenuation at the seizure onset. Seizure onset was non-localizable in eight patients and was lateralized to one hemisphere or was undetermined in 18 patients. Ictal onset showed beta activity in 50% of cases with frontocentral seizures, while alpha activity at onset was seen more frequently in seizures from the inferior frontal region. The mean seizure duration was 75 s, with a range of 3 s–15 min. Four main seizure onsets were noted: rhythmic ictal discharge with high beta more than 20 Hz (Figure 7), low amplitude ictal beta activity <20 Hz (Figure 8), diffuse attenuation <10 µV (Figure 9), interictal epileptiform discharge just before seizure onset (Figure 10). 30

Rhythmic ictal discharge with high beta more than 20 Hz.

Low amplitude ictal beta activity <20 Hz.

Diffuse attenuation <10 µV.

Interictal epileptiform discharge just before seizure onset.
Another study looked at drug-resistant frontal lobe epilepsy, where they analyzed 154 patients, and 81 patients fulfilled the criteria. Seizures were only nocturnal in 30%, and those had a favourable outcome post-surgical resection. Interictal discharges were recorded in 93% of patients. Ictal onset was localized in 80% and was obscured by artifact or unlocalizable in 20%. Interestingly, the seizure onset that was maximum at a single electrode had a better outcome. Six ictal pattern was reported; the most common was fast activity in 42% then none-localizable onset followed by slow rhythmic activity in 9.8%, the third pattern was fast activity followed by rhythmic spike, spike and slow wave or polyspike and slow wave in 8.6%, rhythmic spikes, spike and slow wave, polyspoke and slow wave was seen also in 8.6%. The fifth pattern was sharp wave in two patients or spike in four patients (7.4%), and the sixth pattern was slow wave followed by fast activity, which did not have a prognostic correlation. 31
There are no specific EEG patterns when the recording is made using intercranial electrodes; the pattern can be low-voltage, fast and sharp activity, and this has been observed in epilepsy affecting the frontal, temporal, parietal and occipital lobes. Besides, spike and wave activity has been noted in frontal, temporal and occipital lobe epilepsy and delta brush activity has been observed in cortical dysplasia of frontal and parietal lobe epilepsy. Furthermore, the intracranial EEG pattern correlated more with the pathology than with the brain lobe, except for the temporal lobe, where a low-frequency, high-amplitude periodic spike was observed in mesial temporal lobe sclerosis. 32 The frontal lobe is the largest brain lobe, and the implantation will require a high number of contacts to cover the area of the expected EZ. Regarding the implantation of frontal lobe epilepsy, the relationship between the EZ and the spread pathway needs to be determined, and the need for implantation in the contralateral hemisphere should be established. Based on semiology, the frontal lobe into anterior perisylvian, and we must implant the orbitofrontal cortex, anterior insula, precentral opercular cortex and anterior cingulate gyrus and sampling from the temporal lobe also, if we think it is posterior frontal, then sampling should include posterior insula, post central operculum, posterior cingulate gyrus and temporo-occipital gyrus and posterior superior temporal gyrus (Heschl’s gyrus and planum temporale). Implantation in the contralateral hemisphere should be symmetrical. 33
The sensitivity of magnetoencephalography (MEG) was up to 76.9%, with post-surgical outcomes being better in patients with monofocal activity than in those with multifocal activity. 34 A study of 28 patients who had MEG followed by surgical resection, and patients were divided into three groups; group 1 consisted of six patients and had a negative MRI with intraoperative electrocorticography and invasive EEG mapping, group 2 consisted of 11 patients and a negative MRI, and group 3 consisted of 11 patients with a lesion on the MRI. In the first group complete resection of the EZ was achieved with resection of the MEG localization according to the last postoperative MRI in four out of six patients and Engle class 1 was achieved in 50% of patients, the second and third group had complete resection of the MEG localization in 91% and the intraoperative MRI showed incomplete resection of MEG localizer in four patients leading to successful re-resection and Engle class 1 was achieved in 61% of group 2 and 64% in group 3. 35
Stereotactic EEG is becoming more used in invasive evaluation than subdural grids and strips due to less morbidity and ability to sample deep structure in the brain. A study of 71 patients with difficult to localize Drug-resistant epilepsy (DRE), negative MRI represent 63.4%, and the most frequent evaluation was in the frontal lobe about 64%. The SEEG was localized to the EZ in 90.1%, and the frontal lobe resection was the most frequent sub-lobar intervention in 31%. The most common pathology was focal cortical dysplasia (FCD) in 57.7% out of 55 patients and normal MRI was common in FCD patient than other pathology. Sixty-one patients underwent surgical intervention and were followed up for 1 year, with 63.3% achieving an ENGLE class 1 outcome. 36 SEEG plays a major diagnostic role in epilepsy surgery. Still, it is also gaining increasing recognition as a predictive tool for surgical outcomes. A study of 100 patients with follow-up to 1 year, recruited from four comprehensive epilepsy centres, showed that the co-occurrence of gamma spike and preictal spikes is an excellent predictor for surgical outcome. 37 SEEG can have a therapeutic role, as radiofrequency thermocoagulation can be performed in the contact of the EZ. A retrospective study of 44 patients showed that at 24 months 20.5% patients achieved Engle class 1 and accumulative total of 16 patients had surgical resection 38 and in another retrospective study of 30 patients with normal MRI who was followed for 31.07 months, 36.7% achieved ILAE class 1 in the last follow-up and localization of EZ in the limbic system or insula was a good prognostic factor for seizure freedom. 39
Imaging in frontal lobe epilepsy
In a series of 58 patients with frontal lobe epilepsy who were referred for presurgical evaluations, MRI showed a lesion in 55% of the patients. 40 Processing the MRI image and using advance technique along with the machine learning can increase the yield of detecting lesion in MRI, mainly FCD which is the commonest cause of focal drug-resistant epilepsy. In a study of 61 patients with DRE who had type 3 FCD, the MRI imaging was processed with morphometric assessment and was analyzed using artificial intelligence and machine learning that showed that at a threshold of 0.9, the sensitivity was 73.3%, and the specificity was 90%. 41 In another study of 39 patients with FCD proven by pathology, a morphometric analysis was done by a program and showed sensitivity of 64% and specificity of 96% in detecting the FCD, while the visual review of MRI detect the FCD in 43.6%. 42 Another study of 37 patients with FCD showed that 3D magnetic resonance fingerprinting combined with voxel-based morphometry (VBM) of MRI reduce the false positive detection of FCD with sensitivity of 82.1 and specificity of 71.7%. 43 A study of 13 patients with normal MRI showed that using multispectral VBM analysis, the concordance of 46.2% and the concordant/discordant rate was 30.8%. However, in the visual interpretation of VBM, the concordant rate decreases from 15.4% to 46.2%. 44
7T MRI can increase the yield of neuroimaging in drug-resistant epilepsy. In a study of 21 patients (17 adults and four infants), 7T MRI revealed abnormal findings in 29% of patients, and post-surgical histopathology confirmed FCD in these patients. In contrast, four patients with negative 7T MRI results had gliosis on post-surgical histopathology.45,46
Nuclear imaging is playing an increased role in detecting lesion, and in a study of 39 patients with drug-resistant epilepsy, PET scan with an online quantification method improved the sensitivity of detecting hypometabolism by over 50%. 47 In another study of 108 patients with FCD on pathology, the PET/CT (Computed Tomography) detect the FCD in 93.52%, while the MRI detect the FCD only in 20.37%. 48 Ictal SPECT can be used in MRI-negative drug-resistant epilepsy, particularly if extratemporal lobe epilepsy is suspected. It can indicate focal hyperperfusion concordant with the lobe of seizure onset zone on the EEG in 85%, and the injection latency is shorter than 25 s. 49
Neuropsychology of drug-resistant frontal lobe epilepsy
Neuropsychology impairment was reported in 81% of patients with drug-resistant frontal lobe epilepsy in studies done in the Cleveland Clinic, and the assessment included five cognitive domains: language, attention, executive function, processing speed and verbal/visual learning. Four cognitive phenotypes were identified: generalized phenotype (25%) for patients who had impairment in at least four domains, tri-domain phenotype (20%) for patients who had impairment in three domains, domain-specific phenotype (36%) for patients with impairment in one or two domains, and 19% were the intact domain who did not have impairment. The neuropsychological impairment did not have a significant correlation with patient demographic data, seizure location or the epileptogenic focus pathology. 50
Another study examined 71 participants: 34 with drug-resistant frontal lobe epilepsy, 37 with drug-resistant temporal lobe epilepsy and 22 healthy controls. The neuropsychological assessment include intelligence by using the Wechsler Adult Intelligence Test (WAIS), memory by using the extended Rivemaed Behavioural Memory Test (RBMT), attention/psychomotor speed and motor function by using the Trial-Making Test – Part A (TMT-A) and the TMT-B, response inhibition by using the Stroop Neuropsychological Screening Test, concept formation and shifting by using the Wisconsin Card Scoring Test-64 Version and verbal fluency (phonemic and semantic) by using the Verbal Fluency Test.
There was no statistically significant difference among the three groups in terms of age, sex and year of education. Seizures were more frequent in the frontal lobe group compared to the temporal lobe group, and there was no significant difference in the seizure lateralization, epilepsy onset, duration of epilepsy and treatment. The analysis revealed that patients with frontal lobe epilepsy and temporal lobe epilepsy performed significantly worse than the healthy control group in WAIS – digit span, Stroop – interference, phonological fluency, semantic fluency and WCST – number of categories, and RBMT – total score. The frontal lobe epilepsy had a lower score than healthy controls in TMT-B and WCST-perseverative response. Temporal lobe epilepsy patients performed significantly worse in the WCST-number of categories compared to frontal lobe epilepsy patients. 51
Another study of nine patients with drug-resistant frontal lobe epilepsy compared to 24 healthy controls and assessed the episodic and semantic autobiographic memory. The battery used was the Autobiographic Memory Interview, Wechsler Test of Adult Reading, Verbal Pointed Associated subtest, Wechsler Memory Scale, Rey Auditory Verbal Learning Test, Design Memory subset, Rey–Osterreith Complex Figure Test, Symbol Span subset and Controlled Oral Word Association Test. There was no difference in the demographic data for patients with epilepsy and the healthy controls, which included age, sex, full-scale IQ and year of education. The study showed that patients with frontal lobe epilepsy showed worse autobiographic memory compared to healthy controls, and they had a higher level of clinical depression. 52
A retrospective study to analyze the effect of frontal lobe resection in patients with drug-resistant frontal lobe epilepsy by comparing the preoperative neuropsychological evaluation and postoperative assessment included ninety patients. The neuropsychological assessment includes intelligence, attention/working memory, visomotor processing speed, language, executive function and memory. The study revealed that 48% did not experience a meaningful decline in cognitive function after the surgery, while 42% showed a decrease in one or two cognitive domains. The decline was seen in the intelligence, visuomotor processing speed and executive function. There was no relation between the side or site of resection and the cognitive decline. Still, it played a role in the decrease of contralateral manual dexterity after supplementary motor area resection. 53
A prospective study in the pediatric age group evaluated 23 patients with frontal lobe epilepsy before starting the anti-seizure medication. After taking the anti-seizure medication for 1 year, 20 control-matched patients with idiopathic generalized epilepsy and 18 age-matched healthy controls underwent neuropsychological assessment, which included the Wechsler Intelligence Scale for Children – fourth edition (WISC-4) and the Developmental Neuropsychological Assessment – second edition (NEPSY-2). Before starting the anti-seizure medication, the WISC-4 showed significant indifference in all domain in patients with frontal lobe epilepsy compared to the patient with idiopathic generalized epilepsy and healthy control. Also, the NEPSY-2 showed substantial difference between the frontal lobe patient compared to the idiopathic generalized epilepsy and healthy control except for the geometric puzzle subtype which significantly differ only between the frontal lobe group and health control and the word generation sematic category and word generation initial letter subtypes which were significant different only between patient with frontal lobe epilepsy compared to the idiopathic generalized epilepsy. After 1 year, the patients with frontal lobe epilepsy were divided into two groups: one with poor seizure control on polytherapy, which included nine patients and the other group with reasonable seizure control on monotherapy and included fourteen patients. The poor seizure control group showed worse scores on the WISC-2 compared to group 2 in all scores except the processing speed at time zero before starting the antiepileptic, and in all scores at time 1. 54
Surgical treatment and outcomes
According to previous studies, the rate of achieving Engel class 1 after epilepsy surgery in the frontal lobe is 48%–68%, and the mean time for a first seizure after surgery is 33.3 months.11,12,55–57 Many factors affect surgical outcomes in frontal lobe epilepsy, with better outcomes observed in tumours and cortical dysplasia and worse outcomes in cases with gliosis and vascular malformation. 12
A long seizure duration is associated with worse outcomes, and the occurrence of a seizure <2 years is associated with a higher likelihood of seizure freedom. Additionally, a young age at the time of surgery is a favourable prognostic factor. However, the presence of epileptiform discharge on EEG on day 7, 3 months, and 1 year postoperatively is a poor prognostic factor.56,58,59 In addition, the presence of recruiting rhythm and lesion near the eloquent cortex is a poor independent prognostic factor. 58
Furthermore, MRI-negative FCD, a generalized and non-localized ictal pattern on EEG, incomplete lesion resection, seizure in the first postoperative week and an extra-frontal lesion are identified as poor independent prognostic factors. 13
Neuromodulation in frontal lobe epilepsy
In a study of 45 patients with refractory posttraumatic epilepsy who were followed for at least 1 year, 64.4% of patients had more than a 50% reduction in seizure frequency and 15.6% achieved seizure freedom after 24 months. The responder rate increased over time, from 37.4% to 44.4%, 50% and 67.6% at 3-, 12- and 24-month follow-up, respectively. Sixty-six point seven percent patients had lesions on MRI that involved both hemispheres, and 33.3% had lesions that involved only one hemisphere – the negative predictor for outcome after VNS implantation was generalized interictal epileptiform discharge on EEG. The age, sex, duration of seizure, monthly seizure frequency and MRI features did not show a significant difference between the responder and non-responder groups. 60
In a study of 95 pediatric age groups with structurally drug-resistant epilepsy who were followed up on average for 2.6 years (range 1–4.6 years), the rate of seizure freedom was 9.7% in 24 months. Patients were divided into four groups: malformation of cortical development, 26 post-encephalitic lesion, 36 perinatal brain injury 31 and hippocampal sclerosis. 2 The responder rates at 6, 12, 18 and 24 months were 40%, 52.6%, 56% and 59.7%, respectively. The seizure freedom rates at 12, 18 and 24 months were 8.4%, 9.5% and 9.7%, respectively. 61
These studies were not exclusive to frontal lobe epilepsy. Still, the data from studies can be closely matched to drug-resistant epilepsy, and a dedicated prospective study of vagus nerve stimulation in frontal lobe epilepsy can be done in the future.
Another neuromodulation method is responsive neurostimulation (RNS), which allows for direct treatment of tissue, primarily in patients who are not candidates for resection due to the involvement of an eloquent area. A retrospective multicentre study involved 30 patients with neocortical drug-resistant seizure, RNS implanted and the median follow-up was 21.5 months (range 6–52). The median reduction in seizure frequency was 75% and the responder rate for those who had a 50% reduction in seizure frequency was 70%. At the last follow-up, 10% of patients achieved seizure freedom, and 26.7% experienced more than a 95% reduction in seizure frequency. There was no significant difference in seizure reduction between patients who had prior surgical resection and those who did not have prior resection. The frequency of seizure reduction increased over time, as observed in studies of VNS. Among the 30 patients, 13 had only frontal seizures, and nine had seizures arising from the frontal region in addition to other brain regions. 62
Another study of medically resistant epilepsy from eloquent and neocortical regions, which involved 126 patients with an average follow-up of 6 years, showed that the median seizure reduction was 70% in patients with frontal and parietal seizure onset. The treatment was more effective in patients with lesional MRI findings. Similar to the previous study, no significant difference was found between patients who had undergone prior surgery and those who had not undergone surgical resection. 63
Another multicentre retrospective study from eight comprehensive epilepsy centres included 150 patients treated with RNS, the median reduction in seizure was 67% at 1 year, 75% at 2 years and 82% at more than 3 years, and 74% at last follow-up (mean was 2.3 years), 18% of patients had seizure freedom at last follow-up. Forty percent of patients had led implanted in the neocortical frontal lobe. 64
Anterior thalamic deep-brain stimulation is another neuromodulation method used in drug-resistant epilepsy that was approved by the U.S. FDA in 2018. A study from the European multicentre registry, involving 170 patients with follow-up of up to 5 years, showed that patients who completed the study at 5 years had a median seizure frequency reduction from 16 to 7.9/month. However, only 49 patients completed the 5-year follow-up. The effect was more apparent on the focal to bilateral tonic clinic seizure. Regarding the frontal lobe seizure, 15.9% did not benefit, 19.3% improved, 17.4% responders and 28.6% were seizure-free. 65 Another study evaluating the long-term efficacy and safety of thalamic stimulation in drug-resistant epilepsy showed that the median percentage seizure reduction from baseline at 1 year was 41% and at 5 years was 69%. The responder rate (defined as a more than 50% reduction in seizure frequency) was 43% at 1 year and 68% at 5 years. There was no unanticipated adverse event or symptomatic intracranial hemorrhage. 66
Another study compared 12 patients who had stimulation of the anterior thalamic nucleus and 19 patient who had radiofrequency lesion of the anterior thalamic nucleus in patient with drug-resistant epilepsy and were followed for 12 months. Demographic data were similar and also the seizure frequency and duration of disease. The mean seizure reduction was 80.3% in the stimulation group with two none responders and five patients become seizure-free, and 91.3% seizure reduction in the stereotactic lesion group and three patients become seizure-free. 67
Conclusion
Frontal lobe epilepsy is the second most common epilepsy type and accounts for 30% of surgical cases, with the most common aetiology being malformations of cortical development, which is not always detected using 3T MRI, but using advanced technology like 7T MRI, VBM and PET scan, with the quantification method can improve lesion detection. Routine EEG has a low yield in detecting frontal lobe seizures; however, magnetencephalography can detect deep foci away from the scalp, and SEEG has an increasing role in detecting lesions, prognostication, and, in some cases, a therapeutic role through radiofrequency thermal ablation.
Limitation
While this review has its limitations, a significant drawback is that it only includes articles in English, potentially excluding studies from non-English-speaking regions. Other limitations include articles published over 10 years ago, but these are landmark articles that shouldn’t be dismissed. Additionally, the scope of this review – a comprehensive look at drug-resistant epilepsy – is vast and challenging to cover thoroughly. It is a thorough examination of drug-resistant epilepsy – a topic, that is, extensive and difficult to cover comprehensively.
Footnotes
Acknowledgements
Author contributions
Mohammed Alshurem did the conceptualization, reviewing and writing this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Data availability statement
The data are available upon request.
