Abstract
Background:
Parkinson’s disease (PD) is the second most prevalent neurodegenerative disorder globally. Deep brain stimulation (DBS) has become a critical therapeutic option for advanced PD. The efficacy of DBS has been well established for up to 1 or 2 years; however, long-term outcome data for Chinese cohorts are limited, and the duration of the “DBS honeymoon” remains underexplored.
Objective:
This study aimed to evaluate the long-term efficacy (⩾10 years) of subthalamic nucleus (STN)-DBS in patients with PD in Southern China, and to investigate the duration of “DBS honeymoon.”
Design:
Retrospective study.
Methods:
Thirty-one patients who underwent bilateral STN-DBS between 2010 and 2011 were assessed. Motor symptoms were evaluated using the Unified Parkinson’s Disease Rating Scale-Part III (UPDRS-III) in the off- and on-medication states with stimulation. Nonmotor symptoms and the quality of life (QOL) were measured using validated scales. The levodopa equivalent daily dose (LEDD), stimulation parameters, and adverse events were recorded. Genetic testing was performed for seven patients.
Results:
Thirteen patients completed the follow-up at 1, 3, and ⩾10 years. The UPDRS-III (off-state) scores improved by 53.02%, 44.79%, and 22.56% at 1, 3, and ⩾10 years, respectively. Tremor and rigidity showed sustained improvement; sleep remained stable postoperatively. In contrast, emotion, cognition, and QOL improved at 3 years; however, they returned to baseline or declined beyond 10 years. The LEDD reductions were 36.29%, 40.40%, and 29.10% at 1, 3, and ⩾10 years, respectively. Stimulation frequency decreased from 141.70 ± 15.72 Hz at 1 year to 110.00 ± 18.22 Hz ⩾10 years. Additionally, genetic testing identified three mutation carriers and rare complications such as DBS withdrawal syndrome appeared beyond 10 years.
Conclusion:
STN-DBS provided sustained motor improvement, with tremor and rigidity showing the most significant benefits after 10 years. The initial 3 years likely represented a “DBS honeymoon,” with peak improvements in motor and nonmotor symptoms. Genotype may influence the efficacy of DBS, and monitoring rare complications is essential. These findings should be interpreted with caution, given the small sample size and the retrospective design of the study.
Plain language summary
Introduction
Parkinson’s disease (PD) is the second most prevalent neurodegenerative disorder globally. It is characterized by motor symptoms such as bradykinesia, tremor, and rigidity. 1 Deep brain stimulation (DBS) has become a critical therapeutic option for advanced PD, particularly for patients experiencing motor fluctuations.2,3 The efficacy of DBS has been well established for up to 1 or 2 years.4–6 Long-term follow-up studies from international cohorts have reported sustained improvement in motor function and quality of life (QOL) for up to 15 years, representing the longest follow-up duration to date.7,8 In contrast, evidence from Chinese cohorts remains limited, with most studies reporting follow-ups of 5–8 years,9–12 and only one extending to 13 years. However, the primary focus of this study was on motor symptoms, with limited information about nonmotor symptoms and adverse events (AEs). 13 Importantly, long-term studies consistently show that the efficacy of DBS gradually declines over time. 14 In 2009, Simonin et al. 15 introduced the concept of a “DBS honeymoon,” analogous to the “levodopa honeymoon,” describing a period of maximal therapeutic benefit before subsequent decline. The duration and mechanisms underlying this phenomenon remain poorly understood, particularly in Chinese cohorts.
Moreover, despite strict selection criteria, considerable interindividual heterogeneity has been observed in the long-term outcomes of DBS. Current evidence suggests that genetic background may play an important role in an individual’s responsiveness to DBS. 16 It is necessary to predict the clinical outcomes as accurately as possible when informing patients about their suitability for DBS, considering its invasiveness, cost, and possibility of serious AEs. In this context, a better understanding of PD-related genes and their influence on DBS outcomes could enable a more precise and personalized approach to PD treatment. However, many relevant genetic studies are case reports, case series, or retrospective, short-term studies 17 and, therefore, do not allow definitive conclusions regarding the genetic impact on DBS outcomes.
This study aimed to investigate the long-term efficacy of bilateral subthalamic nucleus deep brain stimulation (STN-DBS) in patients with PD from southern China whereas describing genotype-specific long-term responses. Building on the cohort from our previously published 8-year follow-up study,18,19 we conducted a comprehensive follow-up of 13 patients with PD who underwent bilateral STN-DBS at the First Affiliated Hospital of Sun Yat-sen University. Baseline and follow-up data were collected at 1, 3, and ⩾10 years of age, focusing on the clinical outcomes, AEs, and genetic profiles.
Methods
Study design and participants
In this observational study, we retrospectively analyzed 31 patients with PD who underwent bilateral STN-DBS at the First Affiliated Hospital of Sun Yat-sen University between 2010 and 2011. The focus of this analysis was on motor and nonmotor symptoms of patients 10 years after surgery. The inclusion and exclusion criteria were based on previously published studies in our center.18,19 Inclusion criteria included (1) a diagnosis of idiopathic PD according to the British Parkinson’s Disease Society Brain Bank criteria, (2) age 18–75 years, (3) disease duration of 5 years or more, (4) severe levodopa‑induced motor complications despite optimal adjustment of anti‑Parkinsonian medications, (5) at least 30% improvement in motor symptoms assessed by the Unified Parkinson’s Disease Rating Scale Part III (UPDRS III) after a levodopa challenge test, and (6) normal brain magnetic resonance imaging. Exclusion criteria included: (1) the presence of cognitive impairment (score < 26 on Mini‑mental State Examination (MMSE)), (2) severe psychiatric or behavioral diseases, (3) conditions such as severe metabolic/cardiac/respiratory/renal/hepatic diseases, (4) diagnosis of secondary Parkinsonism or multiple system atrophy, or (5) inability to comply with the study protocol. This retrospective analysis corresponds to the ⩾10-year follow-up time point of a long-term postoperative observational study, which was approved by the Medical Ethics Committee of the First Affiliated Hospital, Sun Yat-sen University (Approval No. [2009]116) on November 4, 2009. The original ethical approval covered both the collection of patient data and subsequent retrospective analyses. Written informed consent was obtained from all patients and their family members.
Clinical evaluations
Preoperative assessment: The patients were comprehensively assessed within a week before surgery according to the consensus developed by the MDS in 2006. 20 (1) Motor symptoms: UPDRS-III was used to assess motor symptoms in both the on- and off-medication states; and the Hoehn and Yahr (H–Y) stages were used to assess the disease severity. Details were reported in our previous study.18,19 (2) Activities of daily living (ADL): UPDRS-II was used to assess ADL in both the on- and off-medication states. (3) Nonmotor symptoms: Overall nonmotor symptoms were assessed using UPDRS-I. Specific domains were assessed as follows: (1) Sleep: using the Parkinson’s Disease Sleep Scale Chinese Version (PDSS-CV) and Epworth Sleepiness Scale (ESS); (2) Emotion: using the Hamilton Anxiety Scale (HAMA) and Hamilton Depression Rating Scale (HAMD); (3) Cognition: using the MMSE and the Montreal Cognitive Assessment (MoCA); (4) QOL: using the 39-Item Parkinson’s Disease Questionnaire (PDQ-39); (5) Medication dosage: using the Levodopa equivalent daily dosage (LEDD), referring to the LEDD conversion standard established by the MDS in 2010. 21
Surgical procedure and programming: Electrodes (Model 3389; Medtronic, Minneapolis, MN, USA) were implanted into the STN under local anesthesia, as previously reported. An Implantable pulse generator (IPG; Kinetra; Medtronic) was implanted under local anesthesia. Additionally, the IPG was switched on a month after surgery. The adjustment methods for stimulation parameters were in accordance with those used in our previous study.18,19
Postoperative follow-up: Comprehensive assessments of motor and nonmotor symptoms were performed at 1, 3, and ⩾10 years (i.e., the last available follow-up beyond 10 years) after surgery in on (medication-on/stimulation-on) and off (medication-off/stimulation-on) states using the same scale as that at baseline. The LEDD, stimulation parameters, and AEs were recorded at each time point.
Genetic test and analysis: To further investigate the influence of genetic factors on the long-term efficacy of DBS, we offered genetic testing to all 13 patients who completed follow-up. Seven patients agreed and provided written informed consent, whereas six declined participation. Genomic DNA was extracted from peripheral blood according to the standard operating instructions of the nucleic acid extraction/purification kit (AmCare Genomics Lab, Guangzhou, China). Targeted next-generation sequencing was performed on the AmCareSeq-2000 sequencer (AmCare Genomics Lab) using a capture-based panel comprising 149 PD-associated genes. The 149 genes were curated in 2018 by querying the Human Phenotype Ontology (keyword “Parkinsonism,” HP:0001300; https://hpo.jax.org/) and OMIM (keyword “Parkinson”; http://www.omim.org), and integrating the results from both resources to finalize the panel. Variant interpretation followed the American College of Medical Genetics and Genomics/Association for Molecular Pathology guidelines. 22
Statistical analyses
All statistical analyses were performed using SPSS version 27.0 (IBM Corp., Armonk, NY, USA). Continuous variables were expressed as mean ± standard deviation; however, categorical variables (e.g., gender) were presented as percentages. Data distribution was assessed using the Shapiro–Wilk test. Baseline characteristics were compared across groups (tracked, lost to follow-up, and deceased) using a one-way analysis of variance (ANOVA) for continuous variables and Fisher’s exact test for categorical variables. In the tracked group, the paired-sample Wilcoxon test was used to assess changes in stimulation parameters (voltage and pulse width) between 1 and 3 years postoperatively. Additionally, changes in scores of UPDRS-III, PDQ-39, scales related to nonmotor symptoms (including PDSS-CV, ESS, HAMA, HAMD, etc.), LEDD, and frequency over time were analyzed using repeated measures ANOVA with Geisser–Greenhouse correction. Fisher’s least significant difference method was used for post hoc multiple comparisons. Statistical significance was set at p < 0.05.
Results
Study population
Overall, 31 patients with PD who underwent bilateral STN-DBS at the First Affiliated Hospital of Sun Yat-sen University were enrolled in this study between 2010 and 2011 (patient details are provided in Supplemental Table 1). This cohort had a mean disease duration of 9.97 ± 3.71 years, and an average age at surgery of 59.81 ± 8.19 years. Thirteen patients among the overall 31 completed baseline, 1-year, 3-year, and beyond 10-year follow-ups (Tracked Group). Nine were lost to follow-up (Lost Group), and nine died during the study period (Deceased Group). In the Lost Group, the most common reason for loss to follow-up was relocation (4/9), followed by impaired mobility (3/9), lack of family support (1/9), and battery exhaustion (1/9). In the Deceased Group, the leading cause of death was pneumonia (4/9), followed by heart failure (2/9), septicemia (1/9), cerebrovascular disease (1/9), and bowel cancer (1/9). In the Tracked Group, the mean duration to the last available follow-up was 11.62 ± 1.66 years (range, 10–15 years), with a mean disease duration of 22.15 ± 4.51 years at that time (range, 18–35 years). Detailed follow-up time points and disease durations for each of the 13 patients are provided in Supplemental Table 2. There were no significant differences in the baseline characteristics among the tracked, lost-to-follow-up, and deceased groups (Table 1).
Baseline characteristics of the 31 patients with PD.

Values are presented as mean ± standard deviation, and gender is presented as percentages.
These variables were analyzed using the ordinary one-way ANOVA.
These variables were analyzed using the Fisher’s exact test (two-sided).
ANOVA, analysis of variance; H–Y, Hoehn and Yahr stages; LEDD, levodopa equivalent daily dose; MMSE, Mini-Mental State Examination; MoCA, Montreal Cognitive Assessment; PD, Parkinson’s disease.
Motor outcome
Motor symptoms substantially improved after STN-DBS. In comparison with preoperative off state (47.73 ± 5.98), UPDRS-III scores in off state showed marked improvement at 1 year (22.42 ± 6.77, 53.02%, p < 0.001), 3 years (26.35 ± 6.97, 44.79%, p < 0.001), and beyond 10 years (36.96 ± 7.04, 22.56%, p = 0.001), respectively. Notably, tremors and rigidity showed consistent and significant improvement, exceeding 40% even after 10 years. Additionally, bradykinesia showed improvement across all time points, with a 19% sustained improvement beyond 10 years (p = 0.049). However, the improvement in axial symptoms persisted only for the initial 3 years.
In contrast, when compared with the preoperative on state (15.65 ± 6.42), UPDRS-III scores in the on state showed modest improvement at 1 year (12.00 ± 4.64, 23.32%, p = 0.033), primarily driven by improvement in rigidity (43.30%, p = 0.006). However, scores returned to baseline at 3 years (p = 0.240) and worsened significantly beyond 10 years (28.46 ± 10.35, −81.85%, p < 0.001), predominantly due to deterioration in bradykinesia (6.27 ± 2.98 vs 12.50 ± 6.06, −99.36%, p = 0.004), axial symptoms (2.15 ± 1.64 vs 5.96 ± 3.20, −177.21%, p = 0.002), and tremor (0.35 ± 0.69 vs 1.69 ± 1.25, −382.86%, p = 0.005).
The UPDRS-I score remained stable for the initial 3 years before worsening thereafter. In the off state, the UPDRS-II scores improved for up to 3 years. However, in the on state, the scores revealed an improvement only up to 1 year and returned to baseline at 3 years, followed by deterioration beyond 10 years. The details are presented in Table 2.
UPDRS scores at the baseline and in 1, 3, and beyond 10 years postoperatively.

Values are presented as mean ± standard deviation. The ANOVA for repeated measures showed significant time effects for OFF-UPDRS-II (F = 23.92, p < 0.001), ON-UPDRS-II (F = 12.40, p < 0.001), OFF-UPDRS-III (F = 48.13, p < 0.001), and ON-UPDRS III (F = 9.73, p = 0.003) scores, indicating changes during the follow-up period. Post hoc multiple comparison was performed using the LSD t test. UPDRS with higher scores indicating worse functioning.
p < 0.05 compared to baseline, $$p < 0.01 compared to baseline, $$$p < 0.001 compared to baseline.
p < 0.05 compared to 10 years, **p < 0.01 compared to 10 years, ***p < 0.001 compared to 10 years.
ANOVA, analysis of variance; LSD, least significant difference; UPDRS, Unified Parkinson’s Disease Rating Scale.
Nonmotor symptoms, QOL, and medication
(1) Nonmotor symptoms: compared with baseline, 1. Sleep: PDSS-CV scores remained stable throughout the follow-up periods; however, ESS scores did not significantly differ for up to 3 years but worsened significantly beyond 10 years (p = 0.042; Table 3). 2. Emotion: HAMA and HAMD scores improved at 3 years but returned to baseline after 10 years (Table 3). 3. Cognition: MoCA scores showed the greatest improvement at 3 years (p = 0.047), whereas MMSE scores remained unchanged at 1 and 3 years postoperatively (p = 0.844 and p = 0.739). Beyond 10 years, both MMSE and MoCA scores showed moderate decline (p < 0.001 and p = 0.018, respectively; Table 3).
(2) QOL: The mean scores on the PDQ-39 Synthesis Index (PDQ-39 SI) improved significantly at 1 (p < 0.001) and 3 (p = 0.001) years but returned to baseline beyond 10 years (p = 0.089; Table 3).
(3) Medication: LEDD demonstrated a considerable decline after surgery; the reduction from baseline to 1, 3, and beyond 10 years was 36.29%, 40.40%, and 29.10%, respectively.
Nonmotor scores and QOL at the baseline and in 1, 3, and beyond 10 years postoperatively

Values are presented as mean ± standard deviation. The ANOVA for repeated measures showed significant time effects for MoCA (F = 11.125, p < 0.001), and PDQ-39 SI (F = 19.47, p < 0.001), indicating changes during the followup period. Post hoc multiple comparison was performed using the LSD t test. Higher scores indicate better functioning in PDSS-CV, MMSE, and MoCA, but worse functioning in PDQ-39 SI, ESS, HAMD, and HAMA.
p < 0.05 compared to baseline, $$p < 0.01 compared to baseline, $$$p < 0.001 compared to baseline.
p < 0.05 compared to 10 years, **p < 0.01 compared to 10 years, ***p < 0.001 compared to 10 years.
ANOVA, analysis of variance; ESS, Epworth Sleepiness Scale; HAMA, Hamilton Anxiety Scale; HAMD, Hamilton Depression Scale; LSD, least significant difference; MMSE, Mini-Mental State Examination; MoCA, Montreal Cognitive Assessment; PDQ-39 SI, 39-item Parkinson’s Disease Questionnaire summary index; PDSS-CV, Parkinson’s Disease Sleep Scale Chinese Version; QOL, quality of life.
Programming
From the 3-year follow-up onward, multiple stimulation patterns were employed, including single and double monopolar, and interleaving patterns, particularly beyond 10 years (three patients with single monopolar, six with double monopolar, and four with interleaving patterns). The amplitude significantly increased between 1 and 3 years (18.75%, p = 0.020) and appeared to further increase beyond 10 years. The pulse width remained stable for up to 3 years (p = 0.106) but showed a possible decline beyond 10 years. The frequency remained stable throughout the 3-year mark (p = 0.864) but exhibited a significant reduction beyond 10 years (p < 0.01). Overall, 13 patients underwent IPG replacement, with an average lifespan of 5.69 ± 1.55 years for the non-rechargeable battery. Further details are presented in Table 4.
Stimulation parameters in 1, 3, and beyond 10 years postoperatively.

Amplitude and pulse width between 1 and 3 years were compared using the paired-sample Wilcoxon test; frequency was compared using the ANOVA for repeated measures; –, comparisons of amplitudes and pulse width in the case of different stimulation patterns are not applicable.
Contact 1 refers to the most effective contact; Contact 2 refers to contact with suboptimal efficacy.
indicates p < 0.01 compared to 10 years.
ANOVA, analysis of variance.
AEs and safety
The AEs observed during the 10-year follow-up period were recorded for all patients (n = 31) and grouped into three major categories: surgery-related, DBS device-related, and stimulation/disease-related (Table 5). Surgery-related complications included pouch effusion (6.46%) and poor wound healing (6.46%), both of which improved after surgical intervention.
Incidence of adverse events.

DBS, deep brain stimulation.
The DBS device-related AE included only one case of extension cable fracture, which occurred in patient 21 (patient details are provided in Supplemental Table 1). She maintained good symptom control after DBS and underwent battery replacement 5 years postoperatively. She experienced worsening motor symptoms that were unresponsive to adjustments of stimulation parameters at the 14th year (aged 59 years with a disease duration of 23 years). Increased electrode impedance (13,500 Ω on the right lead and 7368 Ω on the left) was detected, and a brain X-ray confirmed a fracture of the extension cable (Figure 1). Her motor symptoms improved significantly after cable replacement.
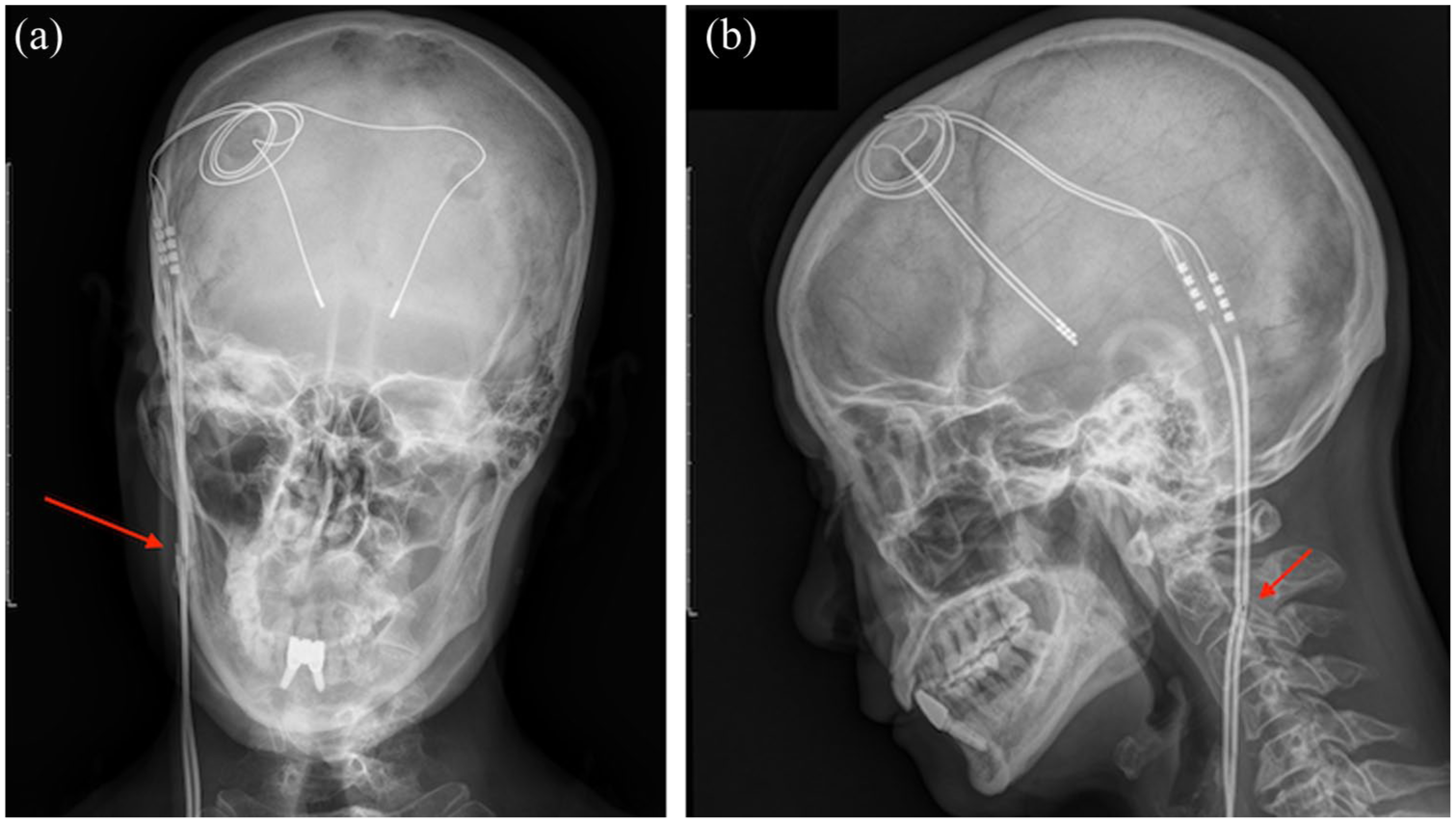
Extension cable fracture in Patient 21, indicated by the red arrows. (a) PA view. (b) Lateral view.
The most common stimulation-related AEs were dyskinesia (29.03%), gait deterioration (25.81%), pain (12.9%), dizziness (9.68%), dysarthria (6.46%), and eyelid apraxia (6.46%). Notably, a 74-year-old male patient (Patient 6) with a rechargeable IPG experienced DBS withdrawal syndrome (DBS-WDS) 12 years postoperatively. He was found unconscious at home and taken to the emergency room. Laboratory tests and brain computed tomography findings were unremarkable. Examination of the DBS device showed that the IPG was switched off, and the battery was exhausted due to insufficient charging. The usual dose of dopaminergic medication (LEDD 150 mg) was ineffective; however, an increased dose (LEDD 570 mg) was administered along with higher stimulation parameters. The patient regained consciousness with gradual improvement in other symptoms, including the akinetic state, dysarthria, and dysphagia. The patient recovered fully 2 weeks after discharge.
Genetic test result
Seven patients who underwent genetic testing were classified into two groups. The MUT+ group (mutation-positive) comprised three patients carrying PD-related variants, including potential risk genes (MGA, CTTNBP2, GIGYF2) and a well-established PD gene (PINK1), all in heterozygosity. And Group MUT− (mutation-negative) included four patients without identified mutations. As shown in Table 6, compared to baseline, within both Group MUT+ and MUT−, the MGA and CTTNBP2 carrier exhibited the best improvements in motor symptoms (49.15% short term, 50.00% long term) and PDQ-39 SI (35.52 vs 12.50, short term; 35.52 vs 34.69, long term). However, it showed the most pronounced deterioration in emotion. In contrast, PINK1 carriers showed moderate motor improvement (44.21%, short term; 7.37%, long term) with the worst outcomes in the PDQ-39 SI (22.03 vs 54.95, long term) and cognition (24.00 vs 14.00, long term). Notably, GIGYF2 carriers exhibited substantial postoperative motor improvement along with favorable nonmotor and QOL outcomes. The details are listed in Table 6.
Demographic and clinical features of patients who had genetic test results.

Improvement of UPDRS-III (off state, %) = (preoperative score − postoperative score)/preoperative score ×100%. Higher scores indicate better functioning in MoCA, but worse functioning in UPDRS-III, PDQ-39 SI, and HAMD.
AD, autosomal-dominant; AR, autosomal-recessive; HAMD, Hamilton Depression Scale; Het, heterozygous; MoCA, Montreal Cognitive Assessment; MUT−, mutation negative; MUT+, mutation positive; PD, Parkinson’s disease; PDQ-39 SI, 39-item Parkinson’s Disease Questionnaire summary index; UPDRS, Unified Parkinson’s Disease Rating Scale.
Discussion
In our study, DBS provided significant and sustained motor improvement, with UPDRS-III scores in the off state improving by 53.02%, 44.79%, and 22.56% at 1, 3, and over 10 years, respectively. Cognition improved at 3 years but declined after 10 years, whereas QOL and emotion returned to baseline after 3 years. LEDD reductions were the most pronounced in the first 3 years, with increasing stimulation amplitude and decreasing frequency after 10 years. Despite being generally consistent with our 8-year outcome, 19 these findings raised new questions and reflections.
Decline in the overall benefit of DBS and “DBS honeymoon”
The efficacy of DBS inevitably declines over time8,13,23–25 despite its dramatic improvements in motor symptoms and QOL over the first few years. Satisfactory symptomatic control and good QOL usually last for 3–5 years postoperatively8,19,24,26 in accordance with the data from longitudinal studies worldwide. This period is referred to as a second honeymoon for PD (i.e., “DBS honeymoon”) as it resembles the levodopa honeymoon, which was first proposed by Simonin et al. 15 in 2009. Only a few studies have investigated the long-term efficacy of DBS until the present day. Therefore, it is unclear why the honeymoon phenomenon is temporary. Patients experienced minor or no motor complications and responded well to adjustment of stimulation parameters during the “DBS honeymoon,” according to our observation. As the disease progressed, Parkinsonism (i.e., bradykinesia, tremors, and rigidity) worsened, axial signs (i.e., gait, balance, speech, and swallowing) evolved, and levodopa sensitivity diminished. This makes disease management much more complex and even contradictory. More powerful electrical stimulation is required to control Parkinsonism. The stimulation parameters were relatively high beyond 10 years of age at our center. Hence, an increase in electrical energy might cause or aggravate AEs to a variable extent, such as dyskinesia, eyelid apraxia, dysarthria, gait disturbances, and hallucinations, which in turn constrain further improvement in Parkinsonism. 27 On the contrary, axial symptoms, which could be either clinical features of PD or side effects of DBS, sometimes benefit from lower parameters; however, usually at the cost of insufficient control of parkinsonism.19,28 Furthermore, levodopa-resistant signs, like postural instability and freezing of gait, were unlikely to improve with DBS. Improvements in some of the initial levodopa-sensitive symptoms were not sustained over the years with DBS, as levodopa sensitivity gradually declined. 29 Taken together, we speculate that the DBS honeymoon is closely associated with disease severity and complexity. The condition of the disease in the early postoperative phase is relatively simple, and it is easier to achieve adequate symptom relief. However, when the situation becomes intractable in the long term, DBS can only provide partial relief.
Discrepancy between improvements in different motor signs
There seems to be a discrepancy between improvements in different motor signs after surgery13,23,24 as indicated by long-term follow-up studies on DBS. In our study, in off state, tremor, rigidity, and bradykinesia sub-scores beyond 10 years improved by 61.63%, 40.63%, and 19.00%, respectively. In comparison with the baseline, the axial sub-score reverted to the preoperative severity level. However, in the on state, the efficacy of DBS on motor symptoms was observed only within the initial 3 years. This was primarily driven by improvements in rigidity and showed significant worsening beyond 10 years, which aligns with previous studies and may be attributed to axial symptoms progression and reduced responsiveness to levodopa.18,19,30 These results show that DBS-induced improvements in tremor and rigidity are greater and last longer compared with bradykinesia and axial symptoms, which is also supported by other long-term studies.13,23,24,31 Reportedly, in off state, beneficial effects of DBS on rigidity and tremor are maintained at 5 years and beyond; nevertheless, benefits for bradykinesia and axial signs start to decline by 5 years.13,23,24,31 Moreover, compared with preoperative on state, additional relief for rigidity is detected over time, in contrast to a gradual deterioration in bradykinesia and axial signs.8,13 There are some possible explanations for these findings. First, the different motor signs of PD may reflect independent mechanisms that progress at varying rates. 32 Recently, study results show that bradykinesia is mainly associated with dysfunction in the cortico-basal ganglia-thalamocortical loop, 33 whereas rigidity is more likely to arise from alterations in brainstem circuits regulating muscle tone and descending projections to the spinal cord. 34 Postural instability and gait disturbance (PIGD) development is probably driven by the degeneration of brainstem cholinergic neurons originating in the pedunculopontine nucleus and striatal dopamine deficiency. 35 Moreover, tremors are not associated with striatal dopamine depletion and are thought to have a distinct pathophysiological mechanism involving the neurodegeneration of dopaminergic neurons in the retrorubral area and abnormal oscillations in the cerebello-thalamocortical loop. 36 Longitudinal, pathological, and neuroimaging studies have shown that tremor-dominant patients with PD have less severe cell loss and neuroalterations, and a better prognosis with slower progression than non-tremor-dominant patients, including those with PIGD and akinetic-rigid subtypes. 37 We hypothesized that the sustained relief of tremors following STN-DBS could be partly attributed to the relatively benign progression of this symptom.
Second, the DBS therapeutic effect on each Parkinsonian feature is mediated by separate neuronal pathways, which are activated differently by chronic electrical stimulation. 38 Bradykinesia and rigidity improvements are associated with the stimulation of tracts connected to the supplementary motor and premotor cortices, respectively. However, tremor improvement is associated with tracts connected to the primary motor cortex and cerebellum, and axial improvement is associated with tracts connected to the supplementary motor cortex and brainstem. 38 Thus, the efficacy of STN-DBS can be influenced by the brain’s structural integrity and the axonal features (diameter, orientation, and myelination) of the fibers within and near the STN. 39 It is speculated that STN-DBS might preferentially interact with the neural networks responsible for rigidity. This may result in stable control of this symptom, whereas circuits unaffected by STN-DBS may engage in the deterioration of bradykinesia, contributing to a decremental surgical response. 32
Changes in DBS parameters
Regarding the stimulation parameters in our cohort, the amplitude gradually increased, the frequency decreased over time, and the pulse width remained relatively stable. The predominant configuration was monopolar stimulation. However, an increase in the use of double monopolar and interleaving patterns 3 years post-surgery was notable, which aligns with other long-term follow-up studies (>10 years).13,40 Changes in programming strategies somewhat reflected disease progression. As mentioned above, amplitude adjustment alone or with frequency or pulse width was effective during the “DBS honeymoon.” Double monopolar or interleaving patterns were considered as the disease became more severe and complex. A lower frequency was adopted when the axial signs became prominent. However, beyond 10 years, only limited benefits were gained by DBS programming, partly owing to the restrictions of programming techniques and the aforementioned AEs related to the spread of the current outside of the main target area.19,28 DBS technology has developed over the past decade, which could be a solution to this study’s challenges. Innovations, including directional electrodes and brain-sensing technology, will enhance programming effect. 41
Genetic influences on DBS outcome
In our study, we identified three out of seven patients as PD-related mutation carriers. The MGA and CTTNBP2 carriers among them showed the greatest motor improvements, whereas the PINK1 carrier showed the least benefit from DBS compared to the GIGYF2 carriers and noncarriers. GIGYF2, MGA, and CTTNBP2 mutations have been suggested as potential PD risk genes. Nonetheless, their pathogenicity remains uncertain,42,43 and their impact on DBS outcomes is largely unexplored; thus, any potential association with DBS efficacy should be regarded as exploratory. In contrast, PINK1 is a well-established pathogenic PD mutation, along with SNCA, PRKN, PLA2G6, LRRK2, and GBA, and accounts for 4%–9% of early-onset PD in Asian populations. 44 Limited data show that PINK1 carriers experience short-term DBS benefits but develop axial symptoms in the long term, becoming unresponsive to medication and neurostimulation, similar to our findings. 45 By contrast, GBA, SNCA, LRRK2, and PRKN mutations have been studied more frequently in DBS-treated patients. SNCA and GBA carriers may experience early benefits from DBS, which are often counteracted by rapid cognitive and neuropsychiatric decline. In contrast, LRRK2 and PRKN carriers show satisfactory motor outcomes, similar to non-mutation carriers, in both short- and long-term follow-ups.16,17 Notably, the PINK1 carrier in our cohort had a heterozygous variant, which, given the autosomal recessive inheritance pattern of PINK1-related PD, may not be pathogenic. In addition, only a small subset of patients underwent genetic testing, representing merely 23% of the total cohort. Consequently, these findings regarding the relationship between genetic variants and DBS outcomes should be interpreted with caution and cannot be readily generalized to the broader PD population.
AEs in the long term
In this study, extension cable fractures for device-related AEs were observed more than 10 years postoperatively. A previous study reported a 7.69% incidence in the long term, primarily due to rotational stress on the lead extension cable system. 46 This result shows that long-term monitoring of DBS hardware integrity is necessary. Stimulation-related AEs in our cohort were predominantly dyskinesia (29.03%), which is consistent with our previous results. 19
DBS-WDS, a life-threatening emergency that typically results from the prolonged cessation of stimulation, should be given special attention, with an incidence of approximately 1.84% in patients with DBS. 47 It is characterized by the sudden exacerbation of Parkinsonism, autonomic dysfunction, and hyperthermia, which are unresponsive to high-dose levodopa. 48 Advanced age, prolonged disease duration, and higher pre-DBS motor scores were found as the potential risk factors for DBS-WDS. 49 Most patients recover with the restart of stimulation; however, those with advanced disease duration (>18 years) and prolonged DBS use (>8 years) may experience unfavorable outcomes, including death. 48 Early intervention, including reimplantation, resumption of stimulation, and family care, is critical for preventing severe DBS-WDS-related outcomes. 47
In this study, both the mortality and dropout rates were 29% (9/31). Pneumonia was the leading cause of death, followed by heart failure, septicemia, cerebrovascular disease, and bowel cancer. Axial symptoms, including dysphagia and postural gait disturbances, which contributed to long-term bedridden states, are considered risk factors for pneumonia and have been associated with increased mortality. 50 In the Lost Group, the most common reason for loss to follow-up was relocation, followed by impaired mobility. This may have introduced selection bias, as patients who completed follow-up might have been in better physical condition, thereby limiting the generalizability of the findings.
This study has several limitations. First, the sample size was small, with only a limited number of patients available for follow-up beyond 10 years due to death, disability, complications, or loss to follow-up. Second, the study was retrospective, conducted at a single center, and employed a non-blinded design. Third, stimulation-off motor assessments were not performed due to patients’ intolerance of this condition, as noted in our previous reports.18,19 Finally, the genetic variants analyzed were of uncertain and non-pathogenic significance, and the unequal and small number of participants carrying specific mutations limited statistical power, so these findings should be regarded as exploratory. Taken together, these limitations emphasize the preliminary nature of our results, which require validation in larger, prospective, multicenter studies with balanced cohorts and extended follow-up.
Conclusion
In conclusion, STN-DBS provided sustained motor improvement for over 10 years in patients with PD, with tremors showing the most significant benefit, followed by rigidity, bradykinesia, and axial symptoms. The first 3 years marked a potential “DBS honeymoon,” with the most pronounced benefits in motor and nonmotor symptoms, and LEDD was reduced the most. Treatment efficacy declined, LEDD increased after 3 years, and a lower stimulation frequency and interleaving patterns were preferred for axial symptoms. Rare complications, including DBS-WDS and lead fractures, require monitoring for cases over 10 years. Genotype may influence the efficacy of DBS.
Supplemental Material
sj-docx-1-tan-10.1177_17562864251388840 – Supplemental material for Long-term efficacy of deep brain stimulation in Parkinson’s disease: over 10-year follow-up and insights into the “DBS honeymoon”
Supplemental material, sj-docx-1-tan-10.1177_17562864251388840 for Long-term efficacy of deep brain stimulation in Parkinson’s disease: over 10-year follow-up and insights into the “DBS honeymoon” by Jianmin Chu, Lei Wu, Lulu Jiang, Jinhua Chen, Jing Gu, Hao Qian, Jie Chen, Yongli Wang, Weixin Chen, Yanmei Liu, Jinlong Liu and Ling Chen in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-2-tan-10.1177_17562864251388840 – Supplemental material for Long-term efficacy of deep brain stimulation in Parkinson’s disease: over 10-year follow-up and insights into the “DBS honeymoon”
Supplemental material, sj-docx-2-tan-10.1177_17562864251388840 for Long-term efficacy of deep brain stimulation in Parkinson’s disease: over 10-year follow-up and insights into the “DBS honeymoon” by Jianmin Chu, Lei Wu, Lulu Jiang, Jinhua Chen, Jing Gu, Hao Qian, Jie Chen, Yongli Wang, Weixin Chen, Yanmei Liu, Jinlong Liu and Ling Chen in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
