Abstract
Background:
Lasmiditan, an oral 5-HT1F receptor agonist, has been recently approved for acute migraine treatment. While its efficacy was confirmed in randomized clinical trials, scarce data is available regarding effectiveness and tolerability in the real-world setting.
Objectives:
To evaluate lasmiditan effectiveness and tolerability in the real-world setting in 16 Italian headache centers.
Design:
LasmiDitan as Acute migRaine Treatment (DART) study is a prospective, multicentric, observational study.
Methods:
We enrolled 58 participants with migraine (84.5% females, age 49.0 (45.2–52.9) years, 24.1% with chronic migraine) reporting 9.4 (7.4–11.3) monthly migraine days. Participants were instructed to treat their migraine attacks with oral lasmiditan 50 or 100 mg. Using an ad hoc electronic diary, participants prospectively collected migraine attack features at baseline and every 30 min after lasmiditan administration, up to 2 h post-dose. The primary outcome was 2-h pain freedom for the first-treated attack after lasmiditan intake. We also collected the occurrence of treatment-emergent adverse events (AE) after administration.
Results:
Overall, participants treated 100 attacks, of which 58 first-treated attacks. Regarding first-treated attacks, 44.8% of subjects rated migraine intensity as severe at lasmiditan intake. Pain freedom 2-h post-dosing was reported in 32.8% (19/58) of individuals and was associated with baseline pain intensity, being higher in subjects treating a mild/moderate attack (p = 0.044). Conversely, it was not influenced by timing of intake (p = 0.375), dosage (p = 0.727), or previous triptan failure (p = 0.351). Regarding all-treated attacks, pain freedom 2-h post-dosing was 37.0% (37/100). At least one AE was reported by 53.4% of participants (31/58), predominantly asthenia, dizziness, somnolence, anxiety or agitation, and paresthesia. Tolerability was rated as good-to-excellent by 51.8% of subjects.
Conclusion:
Our study supports clinical effectiveness of oral lasmiditan 50 and 100 mg for the treatment of acute migraine attacks. Lasmiditan effectiveness was not associated with the previous triptan failure and may therefore represent a valuable therapeutic option in subjects who did not benefit from, or have contraindications to, triptans.
Trail registration:
The study was preregistered on clinicaltrial.gov, NCT05903040 (https://clinicaltrials.gov/study/NCT05903040?cond=migraine&intr=lasmiditan&rank=5).
Introduction
Migraine is a prevalent and disabling neurological disorder mainly affecting individuals in their most productive years of life, often underdiagnosed and undertreated. 1 Until recently, acute treatment of migraine attacks relied on nonsteroidal anti-inflammatory drugs (NSAIDs), acetaminophen, and triptans. 2 Triptans are migraine-specific drugs acting as serotonin 5-HT1B/1D receptors agonists. Despite their disease-specific mechanism of action, triptans do not represent the optimal solution for a large portion of subjects with migraine (30%—40%) due to lack of effectiveness, adverse events (AE), and cardiovascular contraindications.3–6 The OVERCOME study has recently highlighted the persistence of pronounced migraine-related burden and disability despite use of acute medications. 7 Altogether, these observations suggest that proper acute management represents a critical unmet need for people with migraine.
Recently, the efficacy, safety, and tolerability of lasmiditan for migraine acute treatment was confirmed in randomized clinical trials (RCTs).8,9 This drug is now available in our clinical practice, representing an additional option to improve acute migraine care.
The recommended dose of lasmiditan is 100 mg, with the possibility to reduce or increase the dosage based on the effectiveness and tolerability profile, reaching a maximum daily dose of 200 mg. 10 Lasmiditan is a selective serotonin 5HT1F receptor agonist. 10 This receptor is coupled with a Gi/G0 protein and activates a downstream pathway resulting in inhibition of neuropeptides and neurotransmitters release, including calcitonin gene-related peptide (CGRP) and glutamate. 11
The 5HT1F receptor is located in key structures of the trigeminovascular system, both at peripheral and central levels (trigeminal nucleus caudalis and trigeminal ganglion) as well as in other areas that are relevant within migraine circuitry, such as cortex, thalamus, and hypothalamus.10,11 Unlike the 5HT1D/1B receptor, the 5HT1F receptor is not involved in vascular smooth muscle control. 11 This explains its favorable cardiovascular profile, which was also confirmed by both in vivo and in vitro studies.12–14 Lasmiditan therefore appears to be a valuable option in individuals with triptans contraindications for neurological or cardiovascular comorbidities (namely history of stroke, vascular diseases or uncontrolled hypertension), or above 65 years of age.15,16
Three randomized controlled trials and two open label extensions (OLE) have demonstrated lasmiditan effectiveness in controlling pain and migraine associated symptoms at 2 and 24 h from intake.8,9,17–19 Indirect comparisons and network meta-analyses suggest that lasmiditan is associated with a higher prevalence of mild AEs, such as dizziness, paresthesias, nausea and fatigue, compared to other migraine-specific acute treatments.20–22 This might be related to its highly lipophilic nature and the consequent passage across the blood–brain barrier. Due to the reporting of central side effects, some countries, including Italy, mandate patients to refrain from driving and handling heavy machinery for 8 h after lasmiditan intake. 10
Very limited data are available on the performance of lasmiditan in the real-world setting. At state of art, only one study assessed lasmiditan effectiveness and tolerability in real-world practice. 23 The authors enrolled a cohort of 48 Japanese individuals who treated a single migraine attack with lasmiditan 100 mg. 23 They reported a 2-h pain-free rate of 48% with AEs occurring in more than 90% of cases. 23 The FREE project, an American observational study of individuals treated with lasmiditan in the real-world setting, is presently ongoing with the objective to assess the ability of lasmiditan to restore complete functioning in activities of daily living. 24
The primary objective of the multicenter DART study is to assess the effectiveness and tolerability of two dosages of lasmiditan (50 or 100 mg tablets) in the acute treatment of migraine in the real-world setting.
Methods
Study design, patient features, and variables collected
DART is a real-world, prospective, observational, multicentric study, including all consecutive out-patients that treated at least one migraine attack with lasmiditan 50 or 100 mg. In the DART study, participants were instructed to treat up to four migraine attacks. The study was preregistered on clinicaltrial.gov (NCT05903040, link https://clinicaltrials.gov/study/NCT05903040?cond=migraine&intr=lasmiditan&rank=5) and the Italian centers involved are reported in Supplemental Table 1.
The study was approved as part of the Registro Italiano Cefalee (RICe) study, complete details about the RICe study are provided elsewhere. 25 All participants signed a written informed consent before starting treatment with lasmiditan. For data collection, we used the online open database Research Electronic Data Capture (REDCap) and the Empedocle electronic platform (developed for the RICe study). At the time of the study, lasmiditan was not subsidized by the Italian National Health Service.
Inclusion criteria were (i) individuals aged 18 years or older, (ii) diagnosis of migraine without aura, migraine with aura, or chronic migraine (CM) according to ICHD-3; 26 (ii) at least 3 monthly migraine days (MMDs) in the 3 months before enrollment; (iii) good compliance to study procedures; (iv) availability of headache diaries of least 1 month before enrollment.
Exclusion criteria were (i) subjects with any contraindications to lasmiditan; (ii) concomitant diagnoses of medical diseases and/or comorbidities that could interfere with the study according to investigators; (iii) pregnancy and breastfeeding.
Participants were enrolled regardless of the number of previous preventive treatments that proved ineffective (determined as no meaningful improvement in headache frequency after the administration of drugs during at least 3 months at an appropriate dose) or not tolerated, including onabotulinumtoxinA and anti-CGRP monoclonal antibodies (mAbs).
Before prescribing lasmiditan, clinicians performed a thorough anamnestic collection of clinical and demographic features, including migraine diagnosis and duration, accompanying symptoms, duration and severity of the attacks, previous and ongoing preventive treatments, monthly headache days (MHDs), MMDs, number of acute drugs per month, and days with at least one acute drug intake per month. Previous or current use of triptans and self-reported effectiveness were also collected to classify participants in triptan responders and triptan nonresponders. We also assessed the impact of migraine in the previous month (Headache Impact Test, HIT-6), 27 migraine-related disability (MIDAS questionnaire), 28 and the efficacy of current acute therapy (Assessment of Current Therapy Questionnaire, Migraine-ACT). 29 A headache day was defined as any day on which a participant recorded any type of headache, a MMD was defined according to the International Headache Society (IHS) guidelines.30,31
Study participants were instructed to treat up to four migraine attacks with the prescribed dose of lasmiditan, taking the drug at any time from pain onset, regardless of pain severity. In line with our real-world design, neurologists were allowed to prescribe lasmiditan at the dose of 50 or 100 mg, according to their clinical judgment. Of note, Italian regulations suggest a starting dose of 100 mg and the 200 mg formulation is currently not available in Italy.
For each treated attack, participants reported the following data on a dedicated eDiary: timing of headache onset, timing of lasmiditan intake, intensity of pain at lasmiditan intake (Likert scale graded from 0 = none to 3 = severe), disability (Functional Disability Scale (FDS)), accompanying symptoms, most bothersome symptom (MBS) with related intensity. After lasmiditan intake, they reported the same variables indicated above at 30, 60, 90, and 120 (2 h) min (Figure 1). Two hours after lasmiditan intake, participants were allowed to take a rescue medication, including a second lasmiditan dose, up to a maximum dose of 200 mg in 24 h, as per the manufacturer’s guidelines. Only for the first-treated attack, they also recorded headache symptoms in the eDiary 24-h post-dosing and filled in the Migraine-Act questionnaire.

Study design.
We also recorded AEs, collecting type, severity and duration, and subjective tolerability (from very bad to excellent). Participants treating their attacks with triptans, presently or in the past, were also asked to compare the performance of lasmiditan with the triptan they used. All participants were asked to rate their satisfaction with lasmiditan on a scale from 0 = none to 10 = excellent.
Outcomes and analysis
The primary outcome measure of DART study was 2-h pain freedom in the first-treated attack. We also evaluated the occurrence of treatment emergent side effects (TEAEs) over the 24-h period post-dosing.
Secondary outcome measures included (i) 2-h pain freedom across all treated attacks; (ii) 2-h pain relief defined according to IHS guidelines; 30 (iii) ability to function normally 2-h post-dose, evaluated with FDS; (iv) 2-h freedom from the MBS; (v) effectiveness in triptan responders versus nonresponders; (vi) use of rescue medications; (vii) pain freedom at 24 h, defined as the percentage of participants who were pain-free at the 24-h follow-up without the intake of a rescue medication; (viii) sustained pain freedom, defined as the percentage of migraine individuals who were pain-free at 2 and 24 h post-dosing without the intake of any rescue medication; (ix) treatment satisfaction (from 0 to 10); (x) self-reported treatment effectiveness (Migraine-ACT).
Statistical analysis
Due to the scarce data regarding lasmiditan effectiveness in the real-world setting, it was not possible to perform a formal a-priori sample size calculation. Taking into account the study by Ishii et al. (48 treated attacks), 23 our previous experience with the GAINER study (103 treated attacks), 32 and the absence of lasmiditan subsidization, we considered our final sample size (58 participants, and 100 treated attacks) as representative of the population attending our outpatient headache clinics.
For several variables, normality was not confirmed with Kolmogorov–Smirnov test, and statistical analysis was conducted with nonparametric tests. We reported mean (95 confidence interval) for continuous variables and number (percentage) for categorical data. No imputation was performed for missing data. For univariate analyses, we adopted Mann–Whitney or Kruskal–Wallis tests for independent groups comparison, and chi-square test for categorical variables. Cochran Q test was adopted to assess significant modification of pain freedom and prevalence of AEs over time. For all-treated attacks, we performed a multivariate analysis with a logistic regression to assess which variables were independently associated with 2-h pain freedom. The level of significance was set at α < 0.05 with a Bonferroni’s correction for multiple comparisons where appropriate. Statistical analysis was conducted with SPSS software version 21.0 (IBM Corp. SPSS Statistics, Armonk, NY, USA).
Results
Study population
The final study population included 58 participants (84.5% females, age 49.0 (45.2–52.9) years, mean disease duration 33.9 (26.7–41.1) years). Overall, they treated 100 attacks, of which 58 were first-treated attacks. Forty-four (75.9%) participants had episodic migraine (EM), while 14 (24.1%) had CM. Among CM participants, 7 (50.0%) had a concomitant diagnosis of medication overuse headache (MOH). The average number of previously failed preventive drug classes was 3.2 (2.6–3.8), with 81.0% (n = 47) of participants taking a preventive treatment at baseline (stable over the previous 3 months in 48.3% of cases). Thirty-two participants (55.2%) had at least one comorbidity.
At baseline, participants presented 9.4 (7.4–11.3) MMDs and 10.7 (8.5–12.9) MHDs with untreated attacks lasting 28.5 (21.3–35.6) hours. They reported a mean of 9.9 (7.1–12.9) days of acute drugs intake per month and 12.6 (8.3–16.9) doses of acute medications per month. Clinical and demographic features are illustrated in detail in Table 1. Forty-eight participants (82.8%) declared a current or previous use of triptans, with a subjectively reported good/very good effectiveness in 40.0% of cases, and a poor/no effectiveness in 60.0% of cases (Supplemental Table 2).
Clinical and demographic characteristics of study population.

Values in bold are statistically significant. Percentages are expressed on column total.
BoNT-A, botulinumtoxinA; CGRP, calcitonin gene-related peptide; CM, chronic migraine; EM, episodic migraine; mAbs, monoclonal antibodies; MHDs, monthly headache days; MMDs, monthly migraine days; SNRI, serotonin–norepinephrine reuptake inhibitor.
Lasmiditan was prescribed for one or more of the following reasons: lack of effectiveness (67.3%), as a therapeutic alternative (31.0%), or due AEs/lack of tolerability (15.5%) of the usual acute treatment.
First-treated attack: Baseline clinical features of migraine attack treated with lasmiditan
Participants reported lasmiditan intake after 53.9 (30.3–77.5) minutes (min) from headache pain onset; 45 (77.6%) subjects took lasmiditan within 1 h from headache onset. At the time of intake, they rated pain intensity as severe (score 3 on a 0–3 Likert scale) in 44.8% of cases, while the disability was at least moderate/severe in 84.5% of cases. Most participants (94.8%) reported associated symptoms, mainly photophobia (79.3%), phonophobia (70.7%) and nausea (70.7%). The most prevalent MBS was nausea (49.1%), reported as moderate-severe in 80.4% of individuals. Baseline clinical features of the treated migraine attack are detailed in Table 2.
Baseline clinical features of treated migraine attacks.

For the first-treated attack, 41 participants (70.7%) took lasmiditan 100 mg, while the remaining 17 (29.3%) took lasmiditan 50 mg, none took the 200 mg dose.
First-treated attack: 2-h pain freedom
The percentage of participants who were pain free 2-h post-dosing was 32.8% (19/58), 38.6% for EM, and 14.3% for CM (p = 0.113). As reported in Figure 2, the rate of pain freedom after lasmiditan intake gradually increased every 30 min (30 min: 1.7%, 60 min: 17.2%, 90 min: 25.9%, 120 min: 32.8%; p < 0.001) without differences between EM and CM (p > 0.05 at all timepoints).

Two-hour pain freedom in the first and in all treated attacks.
Higher rates of 2-h pain freedom were observed in the subjects who took lasmiditan when baseline pain was mild/moderate when compared to those who took the drug when pain was severe (43.8% vs 19.2%, p = 0.044).
The average timing of intake from pain onset was 25.5 (11.8–39.2) min in individuals who achieved 2-h pain freedom, and 68.1 (33.7–102.5) min for individuals who did not achieve the 2-h pain freedom (p = 0.375). The 2-h pain freedom rate was 29.4% in participants using lasmiditan 50 mg, and 34.1% in individuals using lasmiditan 100 mg (p = 0.727). No clinical/demographic feature was associated with the 2-h pain freedom condition.
First-treated attack: 2-h pain relief, 2-h freedom from the most bothersome symptom, and migraine-related functional disability
Pain relief 2 h post-dosing was achieved by 67.2% of participants (39/58). This outcome was neither influenced by baseline pain severity (p = 0.692) nor by the timing of lasmiditan intake (within 60 min vs more than 60 min; p = 0.862). The mean timing of intake from pain onset was 50.5 (9.1–91.9) min in individuals who achieved 2-h pain relief, and 55.6 (25.6–85.6) min for individuals who did not achieve 2-h pain relief (p = 0.841).
Two-hour freedom from the MBS was reported in 41.4% of cases. Only two participants (10.5%) who achieved 2-h pain freedom still presented nausea as their MBS at the 2-h timepoint. Complete recovery from migraine-related functional disability 2 h after lasmiditan intake was achieved by 24.1% of individuals.
Response at 24 h
Thirteen participants (22.4%) took a rescue medication in the timeframe 2–24 h post-dose. Specifically, 10 (17.2%) took NSAIDs, and one each a combination of analgesic (1.7%), acetaminophen (1.7%), or a second 50 mg lasmiditan dose (1.7%). Pain freedom at 24 h after lasmiditan dose was 56.9% (n = 33), being significantly higher in EM (65.9%) compared to CM (28.6%) (p = 0.028). Eighteen subjects (31.0%) reported sustained pain freedom. Associated symptoms were absent in 38 participants (65.5%).
Detailed comparison between EM and CM subgroups for the first-treated and all-treated attacks are reported in Supplemental Tables 3 to 6.
All treated attacks: Baseline clinical features of migraine attacks treated with lasmiditan
Of the total 100 migraine attacks recorded in the electronic diary, 58 participants treated at least one attack, 28 two attacks, 9 three attacks, and 5 four attacks. Lasmiditan 100 mg was used in 75% of cases, with three participants increasing the dose from 50 mg, and two participants decreasing the dose for subsequent attacks.
The time of intake of lasmiditan was 53.1 (35.6–70.5) min from headache pain onset; in 79 attacks (79.0%) lasmiditan was taken within 1 h from headache onset. At the time of intake, pain intensity was rated severe in 39.0% of cases, while the disability was moderate/severe in 84.0% of cases. Most participants (92.0%) reported associated symptoms, mainly photophobia (79.0%), phonophobia (69.0%), and nausea (72.0%). The most prevalent MBS was nausea (51.0%), reported as moderate-severe in 77.0% of individuals. Baseline clinical features of all-treated migraine attack are detailed in Table 2.
All treated attacks: 2-h pain freedom
Considering all-treated attacks, the percentage of 2-h pain freedom was 37.0% (37/100), 41.3% for EM, and 24.0% for CM (p = 0.283). The rate of pain freedom after lasmiditan intake gradually increased every 30 min, up to 2 h post-dose (30 min: 5.0%, 60 min: 20.0%, 90 min: 30.0%, 120 min: 37.0%; p < 0.001), without differences between EM and CM (p > 0.050 at all-time points).
Pain freedom at 2 h was associated with pain intensity at baseline, with higher rate of pain freedom when treating a mild/moderate attack (45.9%) when compared to severe attacks (23.1%) (p = 0.021). The average timing of lasmiditan intake from pain onset was 40.9 (16.3–65.6) min in individuals who achieved 2-h pain freedom, and 60.3 (36.3–84.2) min in individuals who did not achieve the 2 h pain freedom condition (p = 0.413).
The 2-h pain freedom rate was 28.0% for attacks treated with lasmiditan 50 mg, and 40.0% for those treated with lasmiditan 100 mg (p = 0.282). Considering all-treated attacks, 2-h pain freedom was associated with: i) the usual duration of migraine attacks (18.6 (12.0–25.3) hours in individuals who achieved 2-h pain freedom and 33.6 (26.4-40.8) hours for individuals who did not; p = 0.011), and ii) the presence of phonophobia at baseline (41.7% in individuals who achieved 2-h pain freedom, and 12.5% in individuals who did not; p = 0.045).
A logistic regression model, corrected for gender, lasmiditan dose, timing of lasmiditan intake, migraine diagnosis (episodic or chronic), and presence of phonophobia at baseline, suggested that 2-h pain freedom was associated with i) pain intensity at baseline, with a 2.4 higher probability of being pain-free when treating a mild/moderate migraine attack (p = 0.022), and ii) age, with a slight increase in probability of being pain-free with an older age (p = 0.030). The Hosmer–Lemeshow test showed a proper good-of-fit (p = 0.146). This model explained up to 26.5% (Nagelkerke R2) of variability.
Independently from the number of attacks treated, 25.9% (15/58) of participants achieved 2-h pain freedom in all-treated attacks, while 62.1% (36/58) never achieved 2-h pain freedom. Considering participants who treated at least 2 attacks (n = 28), 25.0% (7/28) achieved 2-h pain freedom in all-treated attacks, while 50.0% (14/28) did not.
All treated attacks: 2-h pain relief, 2-h freedom from the most bothersome symptom, and migraine-related functional disability
Pain relief at 2 h was achieved in 68.0% of attacks (68/100). The outcome was neither influenced by baseline pain severity (p = 0.281) nor by the timing of lasmiditan intake (within 60 min vs more than 60 min; p = 0.797). The mean timing of intake from pain onset was 56.7 (34.3–79.1) min in individuals who achieved 2-h pain relief, and 45.5 (17.3–73.7) min for individuals who did not achieve 2-h pain relief (p = 0.445).
Two-hour freedom from MBS was reported in 46.0% of attacks. Only three participants (8.1%) who achieved 2-h pain freedom still presented nausea as the MBS at the 2-h time point. Complete recovery from migraine-related functional disability 2 h after lasmiditan intake was achieved by 31.0% of individuals. Detailed outcomes are reported in Table 3.
Outcomes assessed at the different time points.

Association with triptan response
Forty-eight participants (82.8%) reported previous use of triptans. Data regarding triptans effectiveness was available for 30 out of 48 subjects (51.7%). All of these 30 subjects used lasmiditan at least once for a total of 50 migraine attacks. Previous triptan response was rated as good/very good by 40% of individuals (12/30) and absent/poor by 60.0% of participants (18/30). Triptan response was not associated with lasmiditan 2-h pain freedom (2-h pain freedom in triptan responders: 16.7% vs triptan non-responders: 27.8%, p = 0.351), 2-h pain relief (p = 0.968) and 2-h freedom from the most bothersome symptom (p = 0.419).
Among the 48 participants who had previously used triptans, the experience with lasmiditan was considered better than triptans in 29.2% of cases, comparable to triptans in 33.3% of cases, and worse than triptans in 37.5%. Reasons for lasmiditan preference over triptans were complete effectiveness (30.0%), higher effectiveness (22.0%), faster action (38.0%), and/ or higher effectiveness on associated symptoms (12.0%; Figure 3).
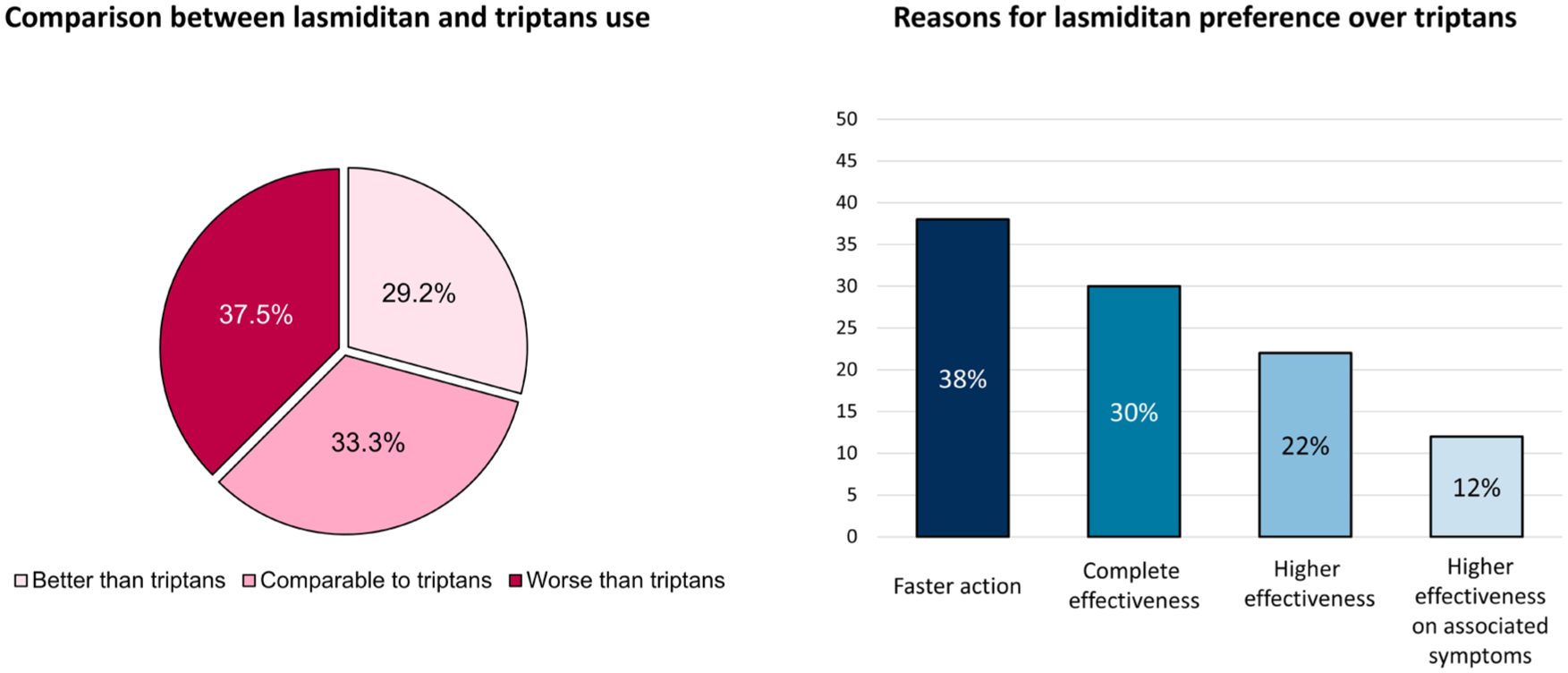
Lasmiditan experience when compared to previously used triptans and reasons for lasmiditan preference over triptans.
Subjective rating of effectiveness
Participants reported a global satisfaction of 6.1 (5.5–6.6) on a 0–10 scale. Migraine-ACT for lasmiditan was higher compared to the usual acute drug (p = 0.001).
Tolerability and AEs
At least one AE was reported by 53.4% of participants (31/58), and in 42.0% of all-treated attacks (42/100). The rate of AEs was 50.0% for the first attack (29/58), 35.7% for the second attack (10/28), 22.2% for the third attack (2/9), and 20.0% for the fourth attack (1/5) (p = 0.220). The rate of AEs was comparable between lasmiditan 50 mg (40.0%) and lasmiditan 100 mg (42.7%) (p = 1.000).
Twelve subjects reported more than one AE. The average number of AEs was 1.6 (1.3–1.9), comparable between lasmiditan 50 mg and 100 mg (p = 0.512). All AEs were mild, and self-limiting, occurring 47.9 (31.2–64.5) min after lasmiditan intake, with a duration of 57.0 (40.6–73.4) min. The most common AEs (>5%) were asthenia (20.0%; 20/100), dizziness (16.0%; 16/100), somnolence (11.0%; 11/100), anxiety or agitation (5.0%; 5/100), and paresthesia (7.0%; 7/100). Dizziness occurred in 24.1% of first-treated attacks (14/58), in 7.1% of second-treated attacks (2/28), and was not reported after the third and fourth treated attacks (p = 0.067). The 2-h pain freedom was not associated with the occurrence of AEs (p = 0.149). The overall lasmiditan tolerability was rated as good/excellent by 51.8% of participants (30/58) in 58.0% of treated attacks (58/100).
Discussion
Our findings can be summarized as follows: i) 2-h pain freedom was observed in 33% of participants in the first-treated attack and in 37% of participants in the overall treated attacks; ii) 2-h pain relief was reported in 67% of participants in the first-treated attack and in 68% of participants in the overall treated attacks; iii) 2-h freedom from the MBS was observed in 41% of participants in the first-treated attack and in 46% of participants in the overall treated attacks; iv) sustained pain freedom was reported by 31% of participants in the first treated attack; v) mild and self-limiting AEs were reported in 42% of all-treated attacks, mainly described as asthenia, dizziness, somnolence, anxiety or agitation, and paresthesia.
The results of our study support the effectiveness of lasmiditan in the real-world setting, with mild AEs reported in 2/5 of cases.
When considering the endpoint of 2-h pain freedom in the first-treated attack, the rate of response was slightly higher in our study when compared to previous RCTs. Indeed, the 2-h pain freedom was achieved by 28.2% (vs 15.3% for placebo) of participants in the SAMURAI trial, and by 25.8% (vs 8.4% for placebo) of participants in the CENTURION study. By contrast, the rate of response was comparable with the SPARTAN study (31.4% vs 21.3% for placebo) and the MONONOFU trial (32.4% vs 16.6% for placebo).8,9,33
We also reported a slightly higher rate of effectiveness for sustained pain freedom at 24 h post-dose when considering the output of a pooled post-hoc analysis of RCTs (16.4%). 34
When taking into account all-treated attacks, comparing our result with the OLE of previous RCTs, we confirm a higher rate of 2-h pain-freedom (GLADIATOR for lasmiditan 100 mg: 29.6%), as well as a higher rate of 2-h pain-relief (GLADIATOR: 56.3%), and 2-h freedom from the MBS (GLADIATOR: 39%). 35
A similar trend for real-life studies compared to RCTs and OLE studies was already highlighted for rimegepant. 32 Some considerations might explain our different findings, mainly the open label nature of our study that cannot exclude a role of a possible placebo response. Furthermore, unlike previous RCTs and OLE studies, lasmiditan intake was not only limited to moderate or severe headache intensity. It is also true that only a minority (5%) of participants took lasmiditan when their pain was mild; thus, this observation cannot completely explain the discrepancy between our study and previous RCTs.
Finally, in our study a higher percentage of participants was on an ongoing preventive treatment (80%) compared to previous RCTs (18.9% and 25.5% for SPARTAN and SAMURAI, respectively).9,34
As far as we know, only one real-life study conducted in Japan assessed the effectiveness and tolerability of a single intake of lasmiditan 100 mg. 23 This study reports a higher percentage of 2-h pain freedom compared to our population (47.9% vs 32.8%). This discrepancy could be explained by the different baseline features of the participants enrolled in the Japanese study, consistent with a lower disabled cohort: i) lower baseline MHDs (8.1 vs 10.7), ii) lower rate of ongoing preventive treatments (58.3% vs 81.0%); iii) exclusion of CM participants. Moreover, in our study around 30% of participants took the 50 mg lasmiditan dose, potentially leading to a final lower rate of 2-h pain freedom.
When considering all treated attacks, 2-h pain freedom was associated with pain intensity at baseline (2.4 times higher when treating a mild/moderate migraine attack), age (slight increase with an older age); by contrast, it was not associated by timing of intake, baseline diagnosis (EM vs CM), or lasmiditan dosage (100 vs 50 mg).
The evidence of a higher rate of pain freedom when treating a mild/moderate attack is consistent with post-hoc analyses of previous RCTs and OLEs. 36 Specifically, in the GLADIATOR study, the higher 2-h pain freedom rate was confirmed in a subpopulation of participants treating migraine attacks of milder intensity. The added value of taking the acute medication when the pain is mild has been demonstrated in some studies on triptans and strongly suggested by the recently published practice recommendations. 16 The early treatment of a migraine attack has the potential to reduce pain burden and time spent with migraine during the day, ultimately reducing disability.
Even though previous indirect comparisons showed a higher effectiveness of lasmiditan 200 mg versus lower dosages, 22 in our study none of the subjects took the 200 mg dose and no differences emerged between the 50 mg and the 100 mg doses.
No differences were found in lasmiditan effectiveness according to migraine diagnosis. To our knowledge, this is the first study evaluating lasmiditan effectiveness in a population of CM participants. The percentage of 2-h pain freedom in CM participants ranged from 14% to 24%. Although these percentages seem lower compared to the EM population, our results support a role of lasmiditan as an acute medication in this particular setting of disabled individuals. However, considering the small number of CM included in our study and the preclinical evidence of increased MOH risk with frequent lasmiditan intake, future studies are needed to validate our findings. 37
Of interest is our finding suggesting the effectiveness of lasmiditan in triptan nonresponders. This is in agreement with a previous study. 38 Despite the results of a recent network meta-analysis describing a higher effectiveness of triptan in a head-to-head indirect comparison, in our cohort, lasmiditan was rated as more effective, or equally effective than triptans in nearly 30% and 33% of cases, respectively. 39 Lasmiditan preference over triptans was mainly due to complete or higher effectiveness, as well as faster action or a higher effectiveness on associated symptoms. These findings support the use of lasmiditan as an alternative strategy in triptan-resistant individuals.
In our cohort, the rate of AEs was high, but showed a decreasing, though not statistically significant, pattern over time (50.0% in the first attack, 35.7% in the second attack, 22.2% in the third attack, and 20.0% in the fourth attack). Previous OLE studies analyzing lasmiditan use in more than one migraine attack described a progressive reduction of AEs occurrence after repeated lasmiditan intake. 35 Unfortunately, it is not possible to completely corroborate this hypothesis with our findings in consideration of the progressive reduction of participants, who have two to three or four attacks treated with lasmiditan. Thus, the observed reduction of AEs may be due to an attrition bias rather than to a true improvement in lasmiditan tolerability with multiple dosing. Compared to previous RCTs, we did not observe dose-related AEs rates. 9 This discrepancy could be explained by the small proportion of individuals who received the 50 mg dose in our study, and the fact that no participant used the 200 mg. In addition, in our observational study, all AEs were self-reported in the diary, and not specifically inquired during an in-person visit as it is routinely done during RCT, which might have led to a lower reporting rate of AEs.
Nonetheless, despite similar AEs characterization, more frequently related to central nervous system symptoms, the prevalence of AEs was higher in our population when compared to longer OLEs. This difference could be explained by the longer follow-up of the OLEs, with the possibility of treating several attacks, and the evidence of a progressive reduction rate after repetitive use. 17 In addition, subjects enrolled in OLEs from parent RCTs already received the investigational drug during the double-blind phase. It is reasonable that subjects with severe AEs, particularly those leading to drug discontinuation, dropped out during the randomized controlled phase of the trials, leading to a more selected population rolling over in the OLE phase.
Some limitations of this study must be acknowledged. We were able to enroll a limited sample size, partly due to the lack of reimbursement criteria with the possibility to take lasmiditan only upon payment. This may have limited the statistical power, at least for secondary analyses. Furthermore, although we adopted a specific diary for data collection, and we performed a thorough patients’ education, we cannot completely rule out a certain degree of recall biases. Moreover, a selection bias cannot be excluded as lasmiditan can only be prescribed by neurologists, thus selecting a population of participants potentially more difficult-to-treat and more prone to medical referral. We must also recognize that we cannot perform a thorough analysis of lasmiditan effectiveness and tolerability in individuals with cardiovascular or psychological comorbidities, as co-existing comorbidities were not properly captured to allow thorough analysis.
Conclusion
Our study supports the clinical effectiveness of lasmiditan 50 and 100 mg for the treatment of acute migraine attacks. Mild and self-limiting AEs were reported in 42% of lasmiditan-treated attacks, with asthenia, dizziness, somnolence, anxiety, and paresthesia being the most reported ones. Notably, lasmiditan effectiveness was not associated with the previous triptan response and was rated by participants as better or comparable to triptans in 29% and 33% of cases, respectively. Lasmiditan may represent a valuable therapeutic option in subjects who failed triptan or have contraindications.
Supplemental Material
sj-docx-1-tan-10.1177_17562864251381886 – Supplemental material for Effectiveness and tolerability of lasmiditan in the acute treatment of migraine: a real-world, prospective, multicentric study (DART study)
Supplemental material, sj-docx-1-tan-10.1177_17562864251381886 for Effectiveness and tolerability of lasmiditan in the acute treatment of migraine: a real-world, prospective, multicentric study (DART study) by Gloria Vaghi, Luigi Francesco Iannone, Michele Corrado, Federico De Santis, Marina Romozzi, Gabriele Sebastianelli, Giorgio Dalla Volta, Marco Bolchini, Andrea Burgalassi, Francesco De Cesaris, Maria Albanese, Nicola Mercuri Biagio, Raffaele Ornello, Simona Sacco, Francesca Pistoia, Gennaro Saporito, Francesco Casillo, Gianluca Avino, Antonio Granato, Antonio Russo, Marcello Silvestro, Catello Vollono, Michele Trimboli, Alberto Doretti, Mariarosaria Valente, Sabina Cevoli, Edoardo Mampreso, Cristina Tassorelli and Roberto De Icco in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-2-tan-10.1177_17562864251381886 – Supplemental material for Effectiveness and tolerability of lasmiditan in the acute treatment of migraine: a real-world, prospective, multicentric study (DART study)
Supplemental material, sj-docx-2-tan-10.1177_17562864251381886 for Effectiveness and tolerability of lasmiditan in the acute treatment of migraine: a real-world, prospective, multicentric study (DART study) by Gloria Vaghi, Luigi Francesco Iannone, Michele Corrado, Federico De Santis, Marina Romozzi, Gabriele Sebastianelli, Giorgio Dalla Volta, Marco Bolchini, Andrea Burgalassi, Francesco De Cesaris, Maria Albanese, Nicola Mercuri Biagio, Raffaele Ornello, Simona Sacco, Francesca Pistoia, Gennaro Saporito, Francesco Casillo, Gianluca Avino, Antonio Granato, Antonio Russo, Marcello Silvestro, Catello Vollono, Michele Trimboli, Alberto Doretti, Mariarosaria Valente, Sabina Cevoli, Edoardo Mampreso, Cristina Tassorelli and Roberto De Icco in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
The “Società Italiana per lo Studio delle Cefalee” (SISC) is acknowledged for the “Registro Italiano delle Cefalee (RICe).”
Declarations
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
