Abstract
Background
The efficacy of triptans as the main acute treatment strategy for migraine headache at the population-wide level needs to be understood to inform clinical decision-making. We summarise key trends in triptan use using more than 25 years of Danish nationwide data.
Methods
We conducted a nationwide register-based cohort study based on all Danish residents with access to public healthcare between 1 January 1994 and 31 October 2019 and summarise informative trends of all purchases of triptans in Denmark in the same period. Complete purchase records of Sumatriptan, Naratriptan, Zolmitriptan, Rizatriptan, Almotriptan, Eletriptan, and Frovatriptan were used.
Findings
Over a 25-year period, triptan use increased from 345 to 945 defined daily doses (DDD) per 1000 inhabitants per year and the yearly prevalence of triptan use increased from 5.17 to 14.57 per 1000 inhabitants. Between 2014 and 2019, 12.3% of the Danish migraine population purchased a triptan. Following their initial purchase, 43% of patients had not repurchased triptans within 5 years. At most, 10% of patients indicating triptan discontinuation tried more than one triptan. The prevalence of triptan overuse, defined as having purchased at least 20 DDDs of triptans per month for 3 consecutive months, increased in parallel with the prevalence of triptan use, prevalent in 56 of every 1000 triptan users every year between 2014 and 2019.
Interpretation
In a cohort with access to free clinical consultations and low medication costs, we observed low rates of triptan adherence, likely due to disappointing efficacy and/or unpleasant side effects rather than economic considerations. Triptan success continues to be hindered by poor implementation of clinical guidelines and high rates of treatment discontinuance.
Introduction
Migraine is a common and frequently disabling neurological disorder characterised by severe and recurring unilateral headaches and accompanying symptoms known as migraine attacks (1). It ranks second of all diseases in terms of years of life lived with disability (2). Migraine has a high lifetime prevalence (3) and an estimated 11.7% (4) of the population suffer from migraine attacks every year. Effective acute treatment for migraine attacks is crucial to alleviate the personal and socio-economic burden imposed by this disorder.
The acute treatment of migraine is predominantly based on potent serotonin 5-HT1B/1D-receptor agonists, collectively termed triptans. They act by stabilising sensory terminals, inhibiting neurotransmission, constricting blood vessels, and inhibiting the release of calcitonin gene-related peptide (CGRP) (5,6). Following their introduction in the early 1990s, triptans were suggested to be extremely effective (7), essentially providing a solution to the problem of acute treatment. Since then, these views have tempered (8), but even so triptans remain the first line of acute treatment when plain analgesics and NSAIDS are not sufficient. Several different triptans are available and they can have different efficacy and/or tolerability profiles. Clinical guidelines recommend trying at least three different triptans before a patient is considered triptan-resistant (9). Patients who frequently use triptans are at risk of medication overuse headache (MOH), a disorder characterised by increased frequency and intensity of migraine attacks (10).
Denmark has a high awareness of headache disorders and the population receives free health care that includes significant reimbursements for their medication expenses. Trends in triptan use in Denmark therefore reflect patients’ treatment need and satisfaction as opposed to availability and/or affordability. Triptans are only available by prescription, which has made it possible to register all triptan purchases since 1994 with linked patient information. We aimed to complete a detailed long-term evaluation of nationwide triptan use, particularly of treatment persistence, discontinuation and repurchase rates, and use the results to draw a number of clinically important conclusions.
Methods and materials
Civil Registration Systems: All Danish residents are issued a unique personal identification number, which is registered in the Civil Registration System (CRS) (11). The CRS continuously monitors vital statistics of all residents in Denmark and contains their current and past relocation history. Personal identification numbers are used as identifiers in national administrative and health registers, allowing identity-secure linkage between public register entities. Using the unique identification number, we linked data on date of birth, date of death/exit, gender from the CRS and the Danish National Prescription Register. Data were retrieved in December 2019.
Prescription Register: The Danish National Prescription Register contains individual-level information on all prescription drug purchases for the entire population of Denmark since 1994 (12). This includes the individual’s personal identification number, product name and manufacturer of the dispensed drug, its Anatomical Therapeutic Chemical (ATC) classification, dose unit, number of dose units in the package and the amount dispensed in terms of defined daily doses (DDDs, https://www.who.int/tools/atc-ddd-toolkit/about-ddd) according to the World Health Organization (WHO). In the case of triptans, one DDD corresponds to a recommended initial dose for the treatment of an acute migraine attack. The dose encoded by the DDD is specific to the triptan formulation, in combination with the chosen route of administration (13).
Data availability: Danish register data used in this study is available for research through Statistics Denmark (14) and the Danish Health Data Authority (15).
Ethical approval: Danish Data Protection Agency (2015-57-0102), section 10 of “The Act on Processing of Personal Data (DK)”, Danish law no 502 as of 23 May 2018.
Patient and public involvement: This research was done without patient involvement. Patients were not invited to comment on the study design and were not consulted to develop patient relevant outcomes or interpret the results. Patients were not invited to contribute to the writing or editing of this document for readability or accuracy.
Study population: The study population consisted of all Danish residents who lived in Denmark between 1 January 1994 and 31 October 2019 and had access to Danish healthcare.
Triptan purchases: We retrieved data on all recorded dispensations of Sumatriptan (ATC: N02CC01), Naratriptan (N02CC02), Zolmitriptan (N02CC03), Rizatriptan (N02CC04), Almotriptan (N02CC05), Eletriptan (N02CC06) and Frovatriptan (N02CC07), for all modes of administration.
Triptan users: In Denmark, triptans are only prescribed to migraine and cluster headache patients, the latter being extremely rare (9). An individual was considered a triptan user if they purchased at least 1 DDD of any triptan during the study period. Information on how many days each month a patient uses triptans was unavailable. The International Classification of Headache Disorders defines triptan overuse as the use of triptans on 10 or more days per month, for 3 consecutive months (1). To approximate this definition and remain conservative, we defined overuse as having purchased ≥ 20 DDD of any triptan, disregarding administration form, per month, for at least 3 consecutive months. This threshold was chosen as some patients may consume more than one dose per day.
Statistical analyses: We summarised triptan use in DDD per 1000 inhabitants per year, DDD per 1000 triptan users per year, prevalence of triptan purchase per 1000 inhabitants per year, and yearly incidence rate of triptan purchase per 1000 individuals. We found this representation to be more intuitive than referring to, for example, population and user rates.
We imputed missing data on triptan purchases in November and December of 2019. The expected number of DDDs purchased and the expected number of incident triptan buyers in that period was assumed to be the same as in November and December of 2018.
We conducted secondary analyses using individuals who purchased triptans for the first time in 1995 or later; that is, the first-time triptan-user (FTTU) cohort (Figure 4). Triptans were first introduced in Denmark in 1992 and, thus, supposed first-time purchasers in 1994 are confounded by individuals who had purchased triptans before the start of the study period. Data on triptan purchases before 1994 are largely unavailable. Follow-up periods (unbroken) for first-time triptan users were defined as the duration between the patient’s first triptan purchase and first event that lead to a loss of consistent access to Danish healthcare (e.g. death, emigration).
Triptan repurchase rates at 1, 2, 3, 4 and 5 years were estimated using subgroups of the FTTU cohort with at least 1, 2, 3, 4 or 5 years of uninterrupted follow up, respectively, since their first triptan purchase. Repurchase rates are the percentage of the relevant individuals that have completed a second purchase within the indicated follow-up interval.
We defined triptan users as triptan-persistent (or just persistent) if they completed at least three triptan purchases within 3 years of their first triptan purchase. The persistence rate is the percentage of individuals that fulfil this criterion. The rate was estimated using a subgroup of the FTTU cohort with at least 3 years of uninterrupted follow-up.
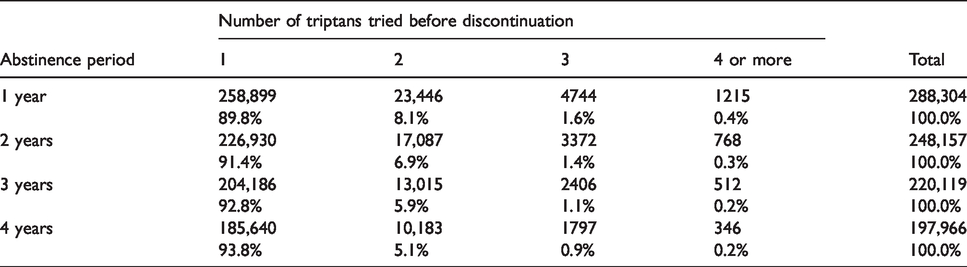
We defined triptan discontinuation to be indicated by a prolonged period of no purchases of triptans. We then assessed how many patients had tried multiple triptans before discontinuing triptan use as per this definition. This analysis was repeated using 1, 2, 3, or 4 or more years of no purchases. This analysis is not based on a strict subset of the FTTU cohort, since individuals with first-time purchases occurring in 1994 were included. Purchase history concerning factors other than the number of different triptans tried were not assessed.
All analyses were performed using the R software, version 3.4.4 (16). Source code is available upon request.
Funding: Lundbeck Foundation, funding grant number R191-2015-844. Candys Foundation, funding grant number CEHEAD. Funding parties did not participate in the conceptualisation, design, collection of data, analysis, decision to publish, or preparation of the manuscript.
Results
Population trends in triptan use
Cohort attributes: We observed 7,435,758 individuals over a combined period of 139.0 million person-years when they were alive and living in Denmark. The median observed time per individual was 24.5 person-years.
Overall, 381,695 individuals purchased triptans at least once. There was a clear difference in overall triptan use prevalence between genders, as 289,024 (75.7%) users were female and 92,671 (24.3%) were male. The five-year prevalence of triptan purchase between 2014 and 2019 was approximately 31 per 1000 inhabitants.
Triptan use in DDDs: The use of triptans increased from 346 DDDs per 1000 inhabitants in 1994 to an estimated 948 DDDs per 1000 inhabitants in 2019. DDDs purchased per 1000 females/year increased from 548 in 1994 to 1489 in 2018, a 172% increase. DDDs purchased per 1000 males/year increased from 137 in 1994 to 347 in 2018, a 153% increase (Supplemental Table 1 and Figure 1).

Timeline of total purchases of triptans in Denmark in DDD per 1000 individuals per year.
After the introduction of all seven triptans, the proportion of DDDs attributed to Sumatriptan decreased to 60.5% in 2006, but thereafter Sumatriptan steadily reclaimed market share. It may be worth noting that patents for Sumatriptan expired in February 2009 and it has been generically available in Denmark since then (Supplemental Table 2).
From 2014 to 2019, no new triptans were introduced and the relative purchase rates of each type of triptan remained constant. Of the total DDD purchased in this period, 74.8% was Sumatriptan, 9.7% Rizatriptan, 8.2% Eletriptan, 5.9% Zolmitriptan, 0.5% Almotriptan, 0.5% Frovatriptan and 0.4% Naratriptan (Supplemental Table 2 and Figure 2).
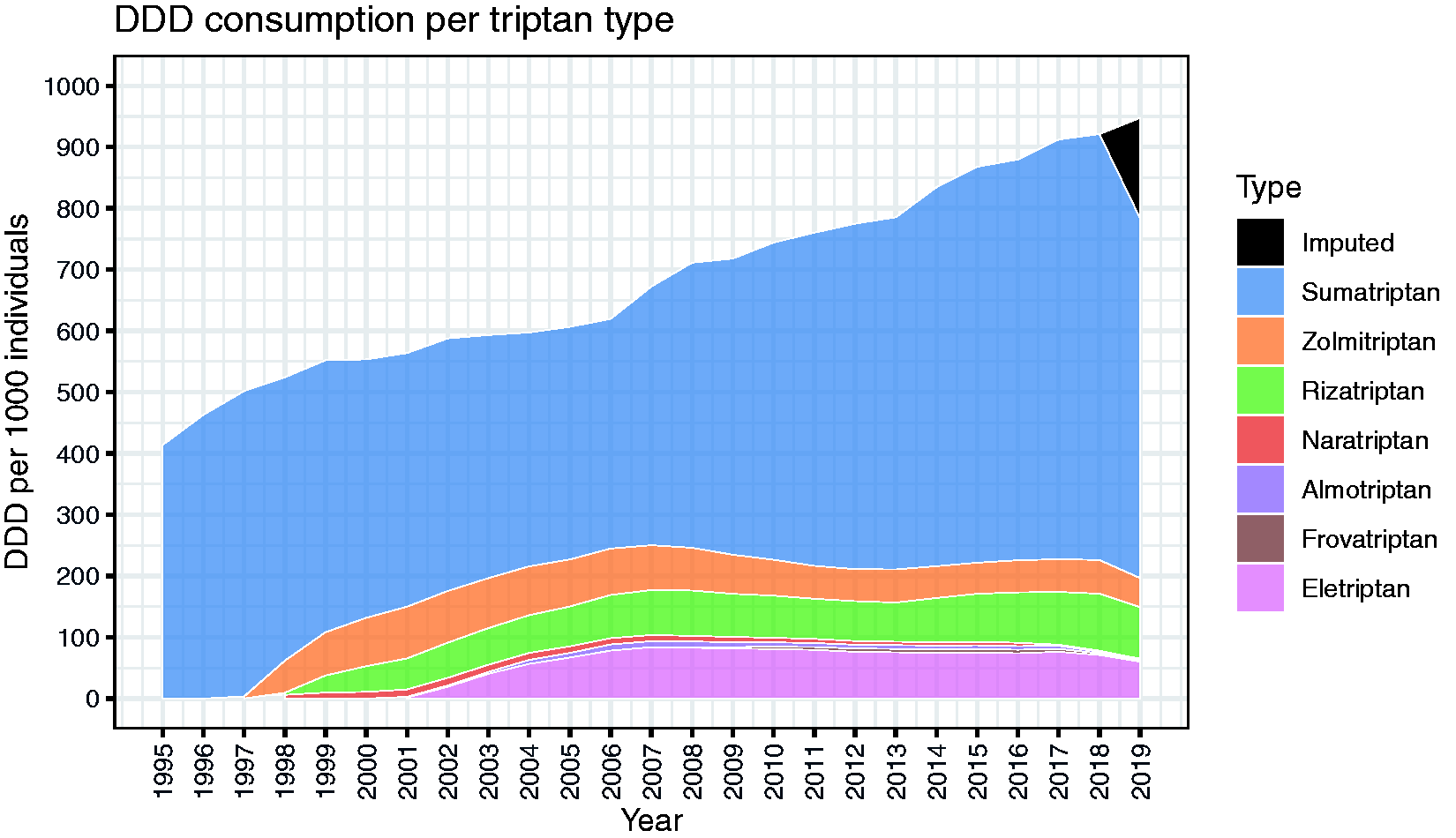
Stacked area graph showing total purchases of triptans stratified by triptan type.
During the study period, 95.2% of all triptans purchased were for oral administration, 2.0% for injection, 1.6% for rectal and 1.2% for nasal administration.
Yearly triptan purchase prevalence: The yearly prevalence of triptan purchase in Denmark increased from 5.2 per 1000 inhabitants in 1994 to 14.5 per 1000 inhabitants in 2019. Approximately one in five triptan buyers every year was male. This ratio was unchanged throughout the study period (Supplemental Table 1).
The purchase prevalence of triptan types other than Sumatriptan reached a combined maximum of 6.5 per 1000 inhabitants in 2006. Subsequently, their prevalence declined, numerically and proportionally, in comparison to Sumatriptan. Between 2014 and 2018, the use of Sumatriptan increased to 10.6 buyers per 1000 inhabitants whereas other triptan types either stagnated or saw a reduction in terms of the number of yearly buyers. Since 1998, the yearly prevalence of purchasing more than one triptan type remained steady at approximately 0.8 buyers per 1000 inhabitants (Supplemental Table 2).
Triptan purchase incidence: The average incidence rate of new triptan buyers from 1995–2019 was 2.58 cases (mean, range 2.26–3.52) per 1000 inhabitants per year. Between 2014 and 2019, the incidence rate remained stable with 2.26–2.48 new triptan users per 1000 inhabitants per year (Supplemental Table 1). The male to female ratio of incident users remained constant at 1:3. Mean age at first purchase was 38.4 years for females and 38.6 years for males (gender data not shown).
Sumatriptan claimed 69.8% of all first-time purchases during the study period (Supplemental Table 2). The number of first-time purchasers claimed by alternative triptans followed a general trend, peaking immediately after their introduction to the market, which then decreased by up to 50% within 1 year. Between 2014 and 2019, Sumatriptan claimed 87.8% of all first-time users (Supplemental Table 2 and Figure 3).

Yearly prevalence of triptan purchase per 1000 individuals.
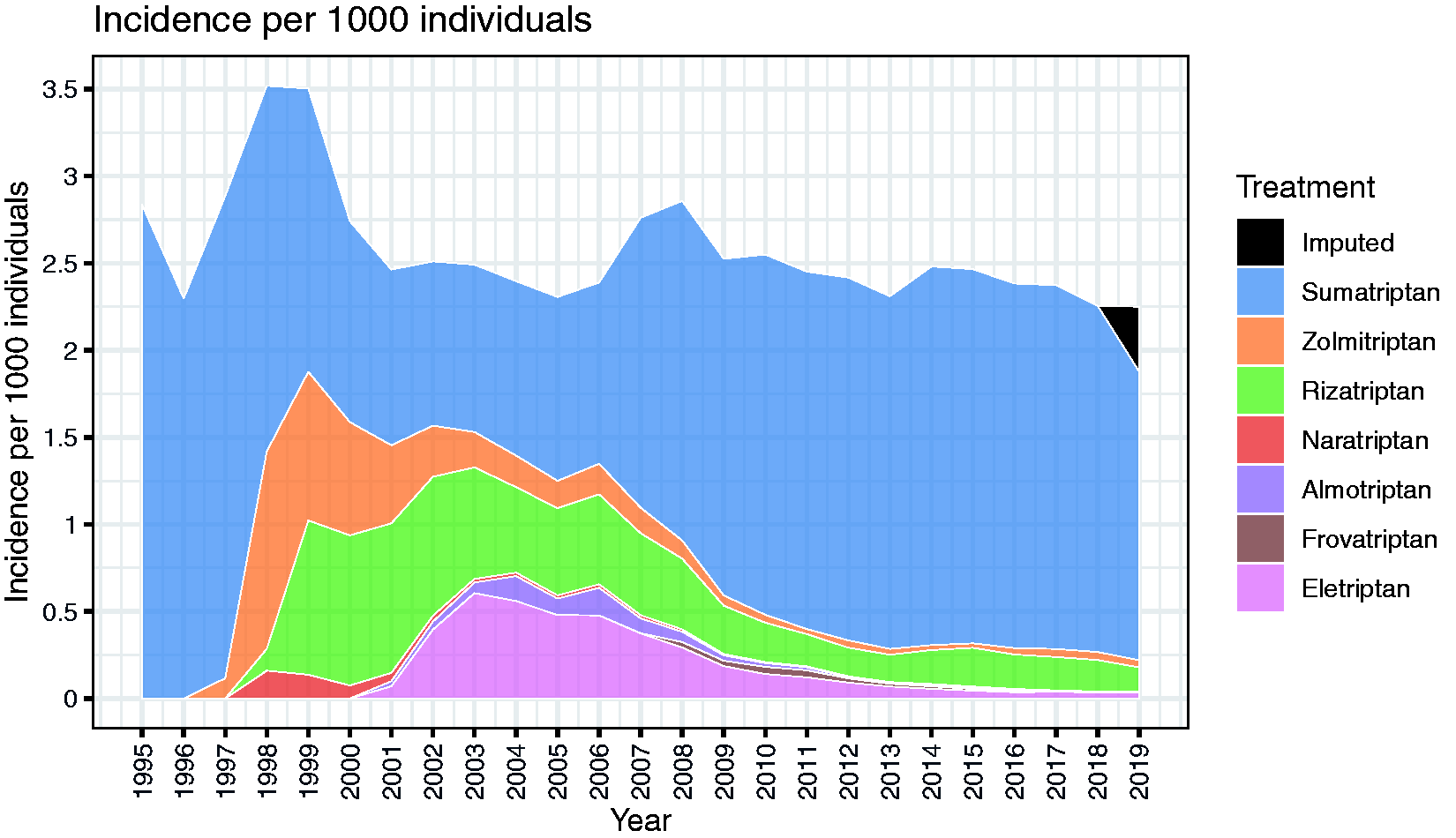
Stacked area graph of historical rates of fist time purchases attributable to each triptan type.
Unmet treatment need
Triptan use within migraine population: Based on estimates of lifetime prevalence of migraine from Danish cohorts (3), we estimate that 12.3% of all migraine sufferers in Denmark purchased a triptan between 2014 and 2019 (Supplemental Table 1.1).
Repurchase rates: First-time buyers represented a steadily decreasing proportion of the total triptan-buyer population every year. For example, in 1998, 19,293 out of 51,489 (37.5%) were first-time buyers. In 2018, it was 12,598 out of 83,002 (15.2%) (Supplemental Table 1).
Repurchase within 1 year was observed in 41.0% of females and 36.0% of males. Repurchase within 2 years was observed in 49.2% of females and 43.5% of males. Failure to repurchase within five years was observed in 41.2% of females and 48.4% of males (Table 3 and Figure 5).

The rate of repurchase within up to 5 years, stratified by gender.
Treatment persistence: Based on 310,530 individuals with at least 3 years of uninterrupted follow-up after their first purchase, we observed a triptan persistence rate of 38.9% among females and 32.2% among males.
Suboptimal use of triptans
According to clinical guidelines, patients should try at least three different triptans before they can be considered triptan resistant. Only 6.2% of patients who discontinued triptan use, based on a 4-year period of no purchase, had tried more than one triptan (Table 4).
Triptan purchase trends between 1994–2018 in Denmark, reported at 6-year intervals.

*DDDs purchased per 1000 inhabitants.
**Prevalence/incidence of triptan purchase per 1000 inhabitants.
Triptan purchase trends between 1994–2018 in Denmark, stratified by triptan type, reported at 6-year intervals.

*DDD purchased per 1000 inhabitants.
**Prevalence/incidence of triptan purchase per 1000 inhabitants.
Proportion of triptan buyers in the FTTU cohort that complete a second purchase within 1–5 years of follow-up.

Rates of trying multiple triptans before triptan discontinuation. Rates for abstinence periods of 1, 2, 3 and 4 or more years are based on all triptan users with at least 1, 2, 3, and 4 or more years, respectively, of unbroken follow-up after their first recorded purchase.
Triptan overuse
The rate of triptan overuse increased during the study period. In the general population, between 1994 and 1999, we observed a yearly rate of approximately 0.53 per 1000 inhabitants, which increased to approximately 0.8 cases per 1000 inhabitants between 2014 and 2019. Between 1994 and 1999, triptan overuse affected approximately 72 out every 1000 triptan buyers every year. This rate then decreased to a minimum of 44.5 per 1000 triptan users in 2007 before rising to approximately 55.7 between 2014 and 2019. Triptan overuse rates were the same between sexes given a prior history of triptan purchase (Table 5 and Supplemental Table 3). In terms of DDDs, 21.8% of all triptan doses were purchased by over-users.
Trends of triptan overuse rates in the overall, male and female populations in Denmark and among yearly triptan buyer subgroups, from 1994 to 2018, reported at 6-year intervals.

*Per 1000 inhabitants.
Discussion
The main results of the present study were that triptan use has seen a slow and steady increase since 1994. Despite this increase, we estimated that only 12.3% of migraine patients in Denmark used a triptan between 2014 and 2019. New triptan users exhibit a low persistence rate and almost 43% had not repurchased triptans within 5 years. In conflict with established guidelines, 90–94% of patients that discontinued triptan-based treatment had tried only one triptan type. Triptan overuse was common and has increased in parallel with triptan use.
In the absence of economic treatment barriers, such as cost of medications and/or consultations, we still noted a significant unmet treatment need in the Danish migraine population. This need was first demonstrated by the small but steady increase in the prevalence of triptan use during the study period, which has not abated. In 2018, the year with the highest rate of triptan purchase (Table 1), 1.5% of the population purchased a triptan. Given an estimated yearly migraine prevalence of 11.7% (4), the prevalence of triptan purchase among those suffering from migraine in 2018 was 12.8%. To account for the possibility that patients can acquire more than a year’s worth of triptans in one purchase, we estimated the 5-year prevalence of triptan purchase (2014–2019) and found it to be only 3.1%. Using estimates for lifetime prevalence of migraine in Denmark, this corresponds to a rate of approximately 12.3% in the migraine population. Hence, 80–90% of Danish migraine patients currently do not use any specific acute migraine medications. Lastly, ergotamine and dihydroergotamine use was extremely rare over the course of the study period and thus had a negligible effect on triptan use statistics.
We found evidence of low adherence to triptan treatment consistent with other studies (17). Following an initial purchase, only 39% of female and 32% of male triptan users purchased a triptan three or more times within 3 years and could therefore be regarded as treatment persistent. Additionally, only 38.6% of first-time triptan buyers had repurchased a triptan within 1 year and only 47.3% within 2 years. Even when given ample time, 42.9% had not repurchased any triptans within 5 years (Figure 5).
The issue of low triptan adherence cannot be explained by successful prophylactic treatment since, based on a Danish nationwide cohort of incident migraine diagnoses between 2005 and 2013, Thomsen et al. show similarly low rates of adherence to prophylactic migraine treatment (18). It is likewise unlikely to be explained by natural remission of migraine symptoms, as observed in, for example, postmenopausal females (19) since, according to Danish guidelines, initial triptan prescriptions are recommended to be only for three to six tablets (9). Our results therefore indicate that the triptans themselves may be to blame, the likely cause being inadequate relief and/or adverse effects that patients deem intolerable. To investigate the latter, register-based approaches are of limited value since triptans generally only cause minor adverse events and their mechanism to induce vasoconstriction of blood vessels, including coronary arteries, rarely leads to reports of serious vascular events (20). Hence, to a large degree, the data relevant to adverse events do not appear in the Danish registers. In order to assess adverse events as predictors of triptan adherence, systematic interviews of discontinuing patients will instead be needed.
Clinical guidelines indicate that a patient should try multiple triptans before they can be considered triptan resistant (21). Specifically, guidelines from the Danish Headache Society state that patients should try at least three different triptans, each for a minimum of three migraine attacks (9). The practical implementation of these guidelines is via trial and error, since there are no clinical features that can predict a patient’s response to a specific triptan (9,22). To estimate adherence to these guidelines, we observed patients after their first triptan purchase (Table 4). Only a small fraction of patients had tried more than one triptan before abandoning this class of medicine. Strict adherence to established guidelines can have the potential to improve patient outcomes, as multiple studies have shown that a triptan can be efficacious even if other triptans are not (7,23,24). It is also known that adverse events can be mitigated by switching to a different triptan (25). Further enquiries into the specific conditions that underlie triptan discontinuation are needed. They should assess to what extent inappropriate skills on behalf of general practitioners, who are responsible for most triptan prescriptions, are to blame and how disappointment in efficacy and/or adverse effects on behalf of patients can reduce their desire to recontact their physician. Future campaigns should aim to inform both the patient and the general practitioner of these issues, as it is clear that these guidelines have not had the intended effect on Danish medical practice.
Compared to other migraine treatments such as ergots and general analgesics, triptan overuse is known to induce a faster onset of MOH (26). Despite increasing awareness of MOH and public awareness campaigns (27), we saw that triptan use carried a constant risk of overuse as evidenced by parallel increases in the prevalence of both use and overuse (Table 5). From 2014 to 2019, we observed a yearly population prevalence of 0.8% of triptan overuse on average. Remarkably, more than one in every five triptan doses sold between 1994 and 2019 was purchased by a patient in an active state of overuse.
The efficacy of drugs is usually determined in randomised, controlled, double-blind clinical trials. In the case of triptans, such trials were often short and based on the treatment of a single attack per patient. Initially, triptans were received with enthusiasm, owing to a documented success rate of 60% in such trials (28). The consensus seemed to be that the problem of treating acute migraine attacks was practically solved. Later, when the definitions of treatment outcomes were scrutinised, it became clear that the initial success criteria were too relaxed. When they were adjusted to more closely reflect patients’ wishes, such as requiring complete relief from headache within 2 h with no recurrence, the success rate of triptans fell to 15–38% (7).
Patient-reported outcomes are gaining acceptance as an important method to judge treatment value and thus to create better personalised care. In a cohort such as ours, with access to free clinical consultations and low medication costs, low rates of persistent use of triptans is likely due to disappointing efficacy and/or unacceptable side effects rather than economic considerations. Triptan success also continues to be hindered by poor implementation of clinical guidelines and the consistent threat of overuse. The approach we employ may be of direct use in numerous settings, such as in studies of childhood cancer survivors, for whom migraines and other headaches are a common but little-appreciated late complication of treatment (29,30).
Conclusion
One seventh of Danish migraine patients currently take a triptan to treat their migraine attacks. Almost half of all patients who receive prescriptions buy a triptan only once within a 5-year period despite migraine being a chronic disorder. Almost no patients try three different triptans, as recommended by clinical guidelines, before abandoning treatment. The overuse of triptans continues unabated despite public information campaigns. We conclude that triptans are far from being ideal drugs for the treatment of acute migraine.
Supplemental Material
sj-xlsx-1-cep-10.1177_0333102421991809 - Supplemental material for Twenty-five years of triptans – a nationwide population study
Supplemental material, sj-xlsx-1-cep-10.1177_0333102421991809 for Twenty-five years of triptans – a nationwide population study by Olafur B Davidsson, Isa A Olofsson, Lisette JA Kogelman, Michael Asger Andersen, Klaus Rostgaard, Henrik Hjalgrim, Jes Olesen and Thomas Folkmann Hansen in Cephalalgia
Supplemental Material
sj-xlsx-2-cep-10.1177_0333102421991809 - Supplemental material for Twenty-five years of triptans – a nationwide population study
Supplemental material, sj-xlsx-2-cep-10.1177_0333102421991809 for Twenty-five years of triptans – a nationwide population study by Olafur B Davidsson, Isa A Olofsson, Lisette JA Kogelman, Michael Asger Andersen, Klaus Rostgaard, Henrik Hjalgrim, Jes Olesen and Thomas Folkmann Hansen in Cephalalgia
Supplemental Material
sj-xlsx-3-cep-10.1177_0333102421991809 - Supplemental material for Twenty-five years of triptans – a nationwide population study
Supplemental material, sj-xlsx-3-cep-10.1177_0333102421991809 for Twenty-five years of triptans – a nationwide population study by Olafur B Davidsson, Isa A Olofsson, Lisette JA Kogelman, Michael Asger Andersen, Klaus Rostgaard, Henrik Hjalgrim, Jes Olesen and Thomas Folkmann Hansen in Cephalalgia
Footnotes
Clinical implications
From 1994 to 2019, the yearly prevalence of triptan use increased from 5.2 to 14.5 per 1000 individuals. The incidence rate of triptan use was relatively constant at 2.58 new users per 1000 individuals per year. In the period from 2014 to 2019, 12.3% of the Danish migraineur population purchased a triptan. Repurchase of triptans within 2 years was observed in 49.2% of females and 43.5% of males. Failure to repurchase within 5 years was observed in 41.2% of females and 48.4% of males. Triptan purchase histories provide clear evidence of non-adherence to clinical guidelines. Triptan overuse rates increased in parallel with triptan use prevalence. Approximately 55.7 of every 1000 triptans users every year between 2014 and 2019 were indicated to have overused triptans.
Acknowledgements
The authors would like to acknowledge the Candys Foundation and Lundbeck Foundation.
Author contributions
Conceptualisation: OD, IO, LK, JO, TH (lead). Data curation: OD (lead), MA, KR. Formal analysis: OD (lead), KR, HH. Investigation: OD (lead), TH. Methodology: OD (lead), LK, KR, HH, JO, TH. Software: OD (lead), MA, KR. Visualisation: OD (lead), MA. Writing-original draft: OD (lead), IO, LK, JO, TH. Writing-review & editing: OD (lead), IO, LK, MA, KR, HH, JO, TH. Supervision: IO, KR, HH, JO (lead). Funding: JO, TH (lead). Administration: HH (lead). Resources: HH (lead).
Data sharing
No additional data are available.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Lundbeck Foundation, funding grant number R191-2015-844. Candys Foundation, funding grant number CEHEAD.
First author declaration
The lead author affirms that the manuscript is an honest, accurate, and transparent account of the study being reported; that no important aspects of the study have been omitted, and that any discrepancy from the study as planned have been explained. He further confirms that he had full access to all the data used in the study and had final responsibility for the decision to submit for publication.
Dissemination
Findings of the study will be disseminated to patient organisations, specialist and non-specialist audience at international conferences and through the National Centre for Headache website.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
