Abstract
Background
A significant proportion of triptan users exhibit an insufficient response or inadequate tolerability to a triptan, and some may develop a contraindication. Lasmiditan, a selective 5-HT1F receptor agonist, may be an option for these individuals. We assessed lasmiditan efficacy in a subgroup of patients in CENTURION (Phase 3 migraine consistency study) who exhibited an insufficient response to triptans, including a subgroup with insufficient response due to efficacy only.
Methods
Patients were randomized to lasmiditan 200 mg for four attacks, lasmiditan 100 mg for four attacks, or placebo for three and lasmiditan 50 mg for one attack. Triptan insufficient responders were pre-defined as patients with insufficient efficacy or tolerability, or who developed a contraindication.
Results
In triptan insufficient responders, lasmiditan was superior to placebo (p < 0.05) for pain freedom beginning at 1 h (both doses); pain relief beginning at 0.5 (200 mg) or 1 h (100 mg); migraine-related disability freedom, much/very much better on the Patient Global Impression of Change, and most bothersome symptom freedom at 2 h; sustained pain freedom; and need for rescue medication. Lasmiditan showed benefit for consistency of effect across attacks for 2-h pain freedom and pain relief. Findings were similar in triptan responders and triptan naïve patients and when the triptan insufficient response definition was based on efficacy only.
Conclusions
Lasmiditan was efficacious across multiple clinically relevant endpoints in the acute treatment of migraine independent of prior response to triptans.
Keywords
Introduction
The goals of acute treatment for migraine include rapid and consistent freedom from pain and other symptoms, restored ability to function, minimal need for repeat dosing or rescue medications, and acceptable tolerability, with minimal or no adverse events (1,2). While triptans have played a crucial role in the management of migraine attacks over the past 30 years, a significant proportion of triptan users exhibit an insufficient response or inadequate tolerability, and some may develop a contraindication.
There is extensive literature on the response rate to triptans. Some 20–40% of individuals with migraine exhibit an insufficient response to triptans (3–6), and 30–60% of patients prescribed a triptan discontinue within one year (7), with lack of efficacy being the greatest predictor of discontinuing (8). Patients with insufficient response to triptans have been reported to have greater migraine-related disability, worse quality of life and impaired work productivity compared to patients who respond to triptans (9). Poorly optimized acute treatment has been associated with progression from episodic migraine to chronic migraine, although whether poorly optimized treatment leads to disease progression or disease progression results in less effective acute treatment is not clear (10).
Not all patients with migraine are able to tolerate triptans. Among those discontinuing triptan therapy, side effects were reported as the reason for discontinuation in 29% of patients (8). Triptans are contraindicated in patients with coronary artery disease, peripheral vascular disease, cerebrovascular disease, and uncontrolled hypertension; a significant proportion of patients with migraine fulfil one or more of these contraindication criteria (11–13).
Recently, lasmiditan, a selective 5-HT1F receptor agonist, has been approved in some countries for the acute treatment of migraine with or without aura. In the single attack Phase 3 SAMURAI and SPARTAN studies (14,15), lasmiditan demonstrated superiority to placebo for pain freedom as well as freedom from migraine-associated most bothersome symptom (MBS) at 2 h post dose. In a post hoc pooled analysis of data from SAMURAI and SPARTAN, patients reported an overall response of either “good”, “poor”, or “none” to their acute migraine medications. Patients reporting a “good” response were classified as good responders, whereas patients reporting a response of “poor” or “none” to their most recent triptan were classified as triptan insufficient responders. Lasmiditan efficacy (assessed by 2-h pain freedom, pain relief, and MBS freedom) was generally similar between patients who reported a good response and those who reported an insufficient response to prior triptan therapy (16). However, overall poor/no response lacks a clear definition and therefore has limitations.
A recent Phase 3 placebo-controlled consistency study of lasmiditan, CENTURION (17), assessed the effect of lasmiditan treatment across four attacks. The first dose findings of CENTURION support those from SAMURAI and SPARTAN. Lasmiditan was superior to placebo for both pain freedom (starting at 1 h post dose) and pain relief (starting at 30 min) and exhibited a sustained effect. Lasmiditan also showed consistency of therapeutic effects.
The aim of this analysis was to assess the efficacy of lasmiditan in a predefined subgroup of patients with an insufficient response to triptans in CENTURION defined through review of guidance documents, literature, and expert input. Additional analyses of pooled data from SAMURAI, SPARTAN, and CENTURION were also performed.
Methods
CENTURION was a modified-parallel, placebo-controlled, double-blind, phase 3 consistency study of patients with migraine, with or without aura, randomized to lasmiditan 200 mg for four attacks, lasmiditan 100 mg for four attacks, or placebo for three attacks and lasmiditan 50 mg for one attack (third or fourth attack). The details of CENTURION have been published (17).
The study was conducted in accordance with the International Conference on Harmonization Good Clinical Practice guidelines and local regulatory requirements. The study protocol was approved by an independent ethics committee or institutional review board at each study site. All patients provided written informed consent for study participation prior to the start of the study.
Triptan insufficient responder subgroups
In CENTURION, a specific, pre-specified definition of triptan insufficient responders was developed based on guidance documents, literature, and expert input from advisory boards as follows:
Pre-specified triptan insufficient responder population
This CENTURION trial subgroup of patients was pre-specified and will be referred to as the triptan insufficient responder (TIR) population. The TIR population was defined based on review of the literature, guidance from the American Headache Society (AHS) and the International Headache Society (IHS), and input from an advisory board of experts in migraine, as patients meeting any of the three criteria:
Did not achieve pain freedom at 2 hours post dose in ≥2 out of 3 attacks with their most recent triptan
Investigators were asked “How well did the subject respond to the medication?” with their recorded response based on patient recall - no response; poor response (pain free at 2 h in ≤1 out of 3 attacks); good response (pain free at 2 h in ≥2 out of 3 attacks). Patients reporting the “no response” or “poor response” to their most recent triptan were considered to be TIR. Published findings support the importance of pain freedom at 2 h post dose in determining acute treatment efficacy; this endpoint aligns with patients’ wishes for rapid onset and complete relief from pain. In a recent position statement, the IHS recommended criteria for defining failure of a triptan, including an inadequate response at 2 h post dose for pain freedom in at least three migraine attacks (18).
2. Acute treatment not optimized - currently taking a triptan and had a score of ≤ 5 based on responses to 4 questions from the migraine Treatment Optimization Questionnaire 6 (mTOQ-6)
The mTOQ is a validated, self-administered questionnaire used to assess the efficacy of current acute migraine treatment (10). In CENTURION, a TIR was defined as having poor or very poor treatment efficacy (score of ≤5) based on responses to four of the mTOQ-6 questions (Figure S1).
Guidance from the AHS (1) emphasizes the preference to assess response of migraine treatments using validated methods, such as the mTOQ. A low mTOQ score (indicating poor response to acute therapy) has been associated with higher Migraine Disability Scores (MIDAS), greater number of headache days/month, higher rates of depression, and more frequent progression from episodic migraine to chronic migraine (10).
3. Most recent triptan discontinued due to lack of efficacy, tolerability issues, or contraindications
Patients currently not taking a triptan discontinued their most recent triptan for one or more of the following reasons – lack of 2-h pain freedom; lack of 2-h pain relief; did not return function or eliminate disability; inconsistent response (pain freedom at 2 h in < 2 out of 3 attacks); migraine recurrence within 24 h; did not relieve associated symptoms (nausea, vomiting, photophobia or phonophobia); did not like route of administration; discontinued due to cardiovascular disease or event; discontinued due to cardiovascular risk factors; discontinued due to contraindication or warning; intolerance to medication. These criteria were based on review of the literature, including recent IHS guidance (19) and findings of Dodick et al. (20,21).
Subgroup of TIR population with two triptan failures
Patients were required to fulfil one or more of the TIR criteria for both of their most recent triptans and an additional triptan (they could fulfil a separate criterion for each triptan).
Subgroup of TIR population defined based on efficacy only
This subgroup, termed TIR efficacy only, excluded patients who only met the contraindications/tolerability issues criterion. That is, to be included in the TIR efficacy only subgroup the patient needed to fulfill at least one of the efficacy-related TIR criteria (did not achieve pain freedom at 2 h in ≥2 out of 3 attacks with their most recent triptan; currently taking a triptan and had a score of ≤5 based on responses to four questions from mTOQ-6; triptan discontinued due to lack of efficacy).
Triptan insufficient responder findings from SAMURAI, SPARTAN, CENTURION
This subgroup was defined as patients whose overall response to their most recent triptan was reported as “Poor” or “None” for an analysis of pooled data from SAMURAI, SPARTAN and CENTURION.
Statistical analysis
The definition of the TIR population was prespecified in the Statistical Analysis Plan for the CENTURION Study. Pain freedom at 2 h post dose for the TIR population was a key secondary endpoint in CENTURION, tested using a graphical multiple comparisons procedure (22) that preserved overall type I error at a one-sided alpha level of 0.025. All additional comparisons were conducted at a two-sided significance level of 0.05.
First attack efficacy analyses were conducted using data from the TIR intent-to-treat (ITT) population, which was defined as all randomized patients who fulfilled the TIR criteria and who used at least one dose of study drug to treat a migraine attack of at least mild pain severity and with any post-dose pain severity assessments at or before 2 h post dose. Analyses were repeated for the modified ITT (mITT) population, which was defined as ITT patients who treated a migraine attack of at least moderate pain severity.
Consistency analyses were conducted using data from the TIR ITT consistency population, which was defined as all patients who fulfilled the TIR criteria and who experienced at least two successes or two failures during an ITT-evaluable attack (i.e. a treated attack of at least mild pain severity with any post dose pain severity assessments ≤2 h post dose). For the control group, only placebo-treated attacks were considered; for the lasmiditan groups, only the first three ITT-evaluable attacks were considered.
Safety and disposition analyses were conducted using data from the TIR safety population, defined as randomized patients fulfilling the TIR criteria and who took at least one dose of study drug. Logistic regression analyses with treatment and region as factors were used for all efficacy analyses. Missing data were handled as follows. A patient was considered a non-responder at a specific time point if they had an evaluable attack but did not provide the necessary data (e.g. pain severity rating, symptom rating) for analysis at the specific time point. A patient was also considered a non-responder if they took any other medication for migraine at or before the specific time point.
Subgroup analyses for those patients meeting TIR criteria versus patients not meeting TIR criteria (TIR vs. Other) were performed for efficacy endpoints. A logistic model with region, treatment, subgroup, and treatment-by-subgroup interaction as explanatory variables was used. The treatment-by-subgroup interaction was tested at the 0.1 significance level.
Additionally, a pre-specified integrated subgroup analysis that included data from Phase 3 SAMURAI, SPARTAN, and CENTURION studies was performed based on patients’ overall response to their most recent triptan (good vs. poor/none and good vs. other) for pain relief and MBS relief at 2 h post dose. Mantel-Haenszel odds ratios and 95% confidence intervals, stratified by study, were calculated. Additionally, the p-value associated with the interaction term from a logistic regression model with terms for study, subgroup, treatment, and subgroup-by-treatment interaction were calculated. A firth logistic regression was used when the logistic regression maximum likelihood did not converge. The treatment-by-subgroup interaction was tested at the 0.1 significance level.
The statistical evaluation was performed using SAS version 9.4 or higher.
Results
Of the 1471 patients in the overall safety population, 633 were considered triptan insufficient responders. Of the triptan insufficient responders, 592 were considered TIR efficacy only (patients who had discontinued their most recent triptan only because of tolerability issues or contraindications were excluded).
For the TIR population, 536 (85%) completed the study. Of the 97 patients who discontinued the study, the most prevalent reasons for discontinuation were an adverse event (n = 36), lack of efficacy (n = 10) and lost to follow up (n = 10).
For the TIR efficacy only population overall, 498 (84%) completed the study. Of the 94 patients who discontinued the study, the most prevalent reasons for discontinuation were an adverse event (n = 33), lack of efficacy (n = 10) and lost to follow up (n = 10).
Baseline characteristics
The triptan experience of patients in the TIR population is shown in Figure 1. The majority of the TIR population had an inadequate response to most recent triptan, an mTOQ score (≤5), or both.

Triptan experience of the CENTURION trial triptan insufficient responder population (n = 633).
Of the 633 TIR patients, 76% had tried one triptan, 17% had tried two triptans, and 7% had tried three or more. Baseline demographics and disease characteristics of the TIR population, as well as the triptan naïve and triptan sufficient responder populations, are shown in Table 1. In the TIR subgroup there was a greater proportion of women, and more patients were from Europe and fewer from Asia. Cardiovascular risk factors were more prevalent in triptan users (triptan sufficient and TIR efficacy patients). The TIR population had a higher MIDAS total score than the triptan sufficient responders (patients who had experienced one or more triptans and did not fulfil the TIR criteria) and naïve populations; this was driven by the TIR efficacy subgroup. Use of preventive treatments was highest in the TIR subgroups.
Baseline characteristics of subgroups defined by triptan experience (CENTURION safety population).

Population definitions: Triptan naïve patients had not previously taken a triptan; triptan sufficient responders (TIR) had previously taken a triptan and did not fulfil the TIR criteria; triptan insufficient responders fulfilled the TIR criteria described in the main text; TIR efficacy only excluded TIRs who discontinued their most recent triptan based only on contraindications/tolerability issues; TIR other comprised patients who discontinued their most recent triptan based only on contraindications/tolerability issues.
MIDAS: Migraine Disability Assessment questionnaire; m: month; SD: standard deviation.
First dose efficacy
In the TIR population, during the first attack both lasmiditan doses showed a statistically-significantly benefit over placebo for pain freedom beginning at 1 h, and for pain relief beginning at 0.5 h for lasmiditan 200 mg and 1 h for lasmiditan 100 mg (p < 0.05 vs. placebo in both cases) (Table 2, Figure 2). Statistically significant differences from placebo were evident in the TIR population for one or both lasmiditan doses for sustained pain freedom at 24 and 48 h (p < 0.05), migraine-related disability freedom at 2 and 24 h, a rating of much/very much better on the Patient Global Impression of Change (PGIC) at 2 h, most bothersome symptom freedom at 2 h, and the need for rescue medication (Table 2). The lasmiditan versus placebo 2-h pain freedom findings were similar for the mITT TIR population (treatment of migraine pain of at least moderate severity) (Table 2).
First dose efficacy findings (CENTURION pre-specified triptan insufficient responder ITT population, unless otherwise specified).

p < 0.05; ‡p < 0.01; *p < 0.001 vs. placebo; ns (based on logistic regression with treatment and region as factors).
Gated secondary endpoint.
CI: confidence interval; h: hour; ITT: intent to treat; MBS: most bothersome symptom; mITT: modified ITT; ns: not significant; OR: odds ratio; PGIC: Patient Global Impression of Change.
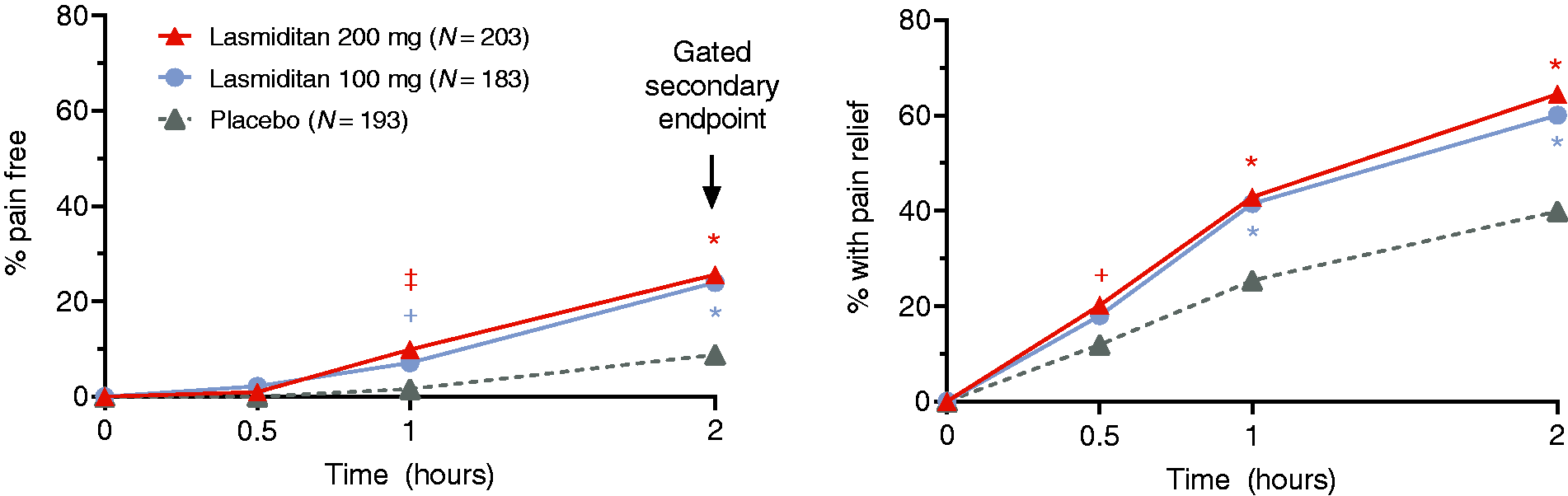
Pain freedom and pain relief during first attack (CENTURION triptan insufficient responder ITT population).
First dose efficacy findings for the TIR patients were similar to those for other CENTURION trial participants (i.e. those not considered TIR) (interaction p-value >0.1 in all cases). Findings for the TIR efficacy only subpopulation (excluding patients who were considered TIR only based on tolerability issues/contraindications) appeared similar to the prespecified TIR population (no formal analysis, Table S1).
For patients who failed two triptans, including their most recent triptan, pain freedom at 2 h was reported for three of 46 (7%) patients in the placebo group compared with seven of 38 (18%) in the lasmiditan 100 mg group (OR [95% CI] vs. placebo, 3.2 [0.8, 12.6]) and 11 of 39 (28%) in the lasmiditan 200 mg group (5.2 [1.4, 19.5]). Pain relief at 2 h was reported for 16 of 46 (35%) for placebo compared with 20 of 38 (53%) for lasmiditan 100 mg (2.0 [0.8, 4.9]) and 27 of 39 (69%) for lasmiditan 200 mg (4.0 [1.6, 10.0]).
In an analysis of pooled data from SAMURAI, SPARTAN and CENTURION, efficacy of lasmiditan, as measured by pain freedom or MBS freedom at 2 h, did not differ based on a patient experience with triptans (poor/none vs. good, poor/none vs. other) (Figure 3).
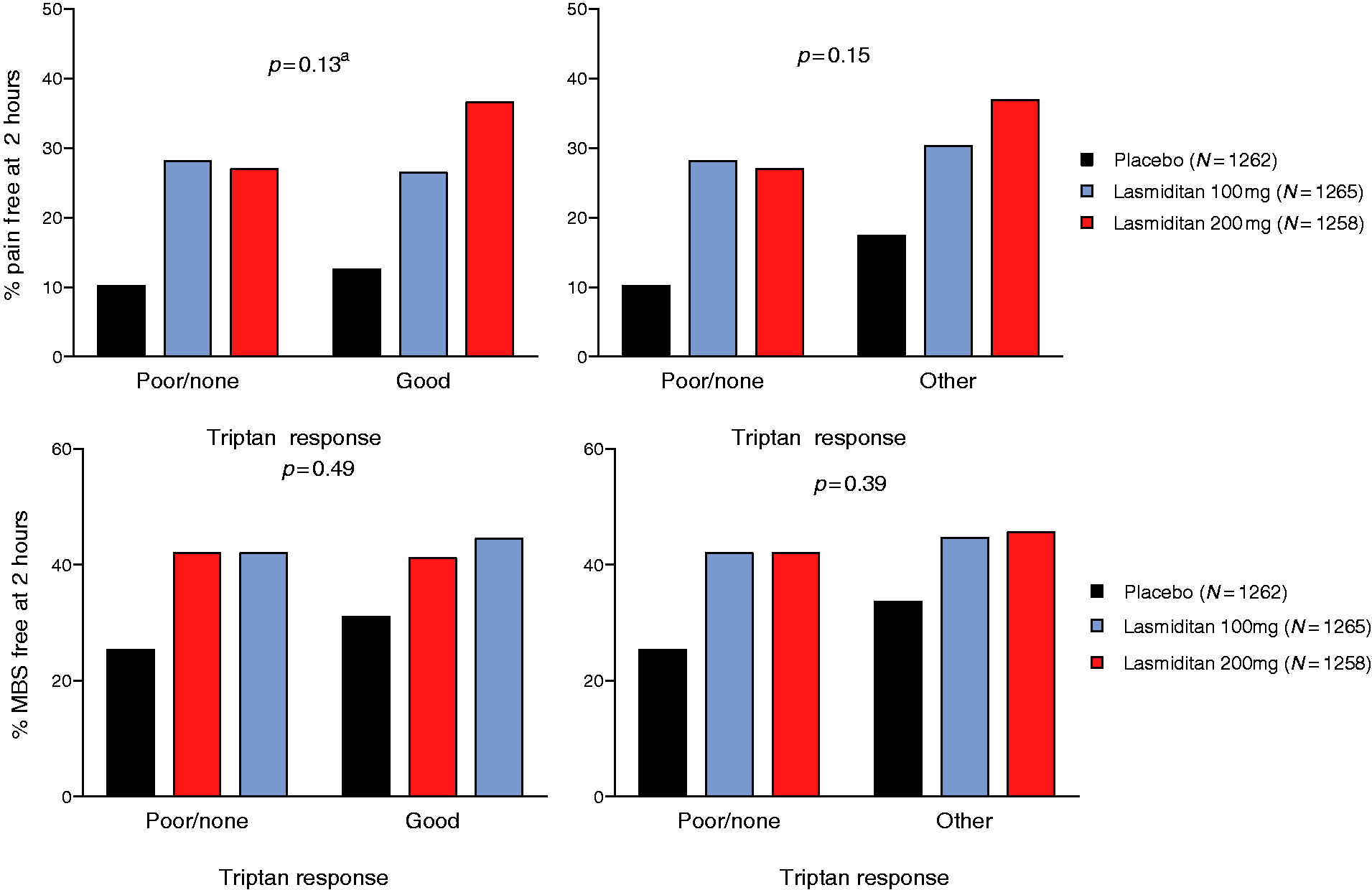
Pain freedom and MBS freedom at 2 h by overall response to triptans (pooled analysis of data from SAMURAI, SPARTAN and CENTURION ITT populations). Triptan response was based on the patient’s overall response to their most recent triptan (Good, Poor or None). Left side figures include only data from patients who had experienced one or more triptans. Right side figures include data from all patients (triptan experienced and triptan naïve). “Other” captures both patients with a good response to their triptan and triptan naïve patients.
Consistency of effect
Intra-patient consistency was assessed as 2-h pain freedom in at least two of three attacks and as 2-h pain relief in at least two or three attacks. In both cases, lasmiditan at either dose was superior to placebo (p < 0.05 in all cases) for the TIR population; for 2-h pain freedom, there appeared to be a dose response (difference from placebo increased with dose) (Figure 4). Findings for the TIR population were similar to those for other patients (interaction p-value >0.1 in both cases). Findings in the TIR efficacy only population were similar to those for the total TIR population (no formal analysis, Table S1).

Intra-patient consistency findings (CENTURION triptan insufficient responder ITT consistency population).
Population consistency was assessed based on percentage of patients who were pain free or had pain relief at 2 h post dose in each attack. Across attacks, the effect of lasmiditan at either dose versus placebo appeared similar (Figure 5).

Population consistency (CENTURION triptan insufficient responder ITT population).
Safety
There were no deaths reported in the TIR safety population; of the 11 serious adverse events, three were considered treatment emergent (one each for placebo, lasmiditan 100 mg, and lasmiditan 200 mg) (Table 3). There were no major cardiovascular events suggestive of vasoconstriction. The most common treatment-emergent adverse events (TEAs) are tabulated (Table 3).
Adverse event findings (pre-specified CENTURION TIR safety population).

Of the serious AEs, three were considered treatment emergent: One after taking placebo (suicidal ideation); one in lasmiditan 100 mg group (asthma); one in lasmiditan 200 mg group (serotonin syndrome [met Hunter’s and Sternbach’s criteria and considered related to study drug by the investigator]).
TEAEs reported in ≥5% of patients in any treatment group.
TEAE: treatment emergent adverse event.
Discussion
In this Phase 3 CENTURION study, 43% of the overall safety population were considered triptan insufficient responders, and the vast majority were considered triptan insufficient responders based on triptan efficacy alone. This is in line with a meta-analysis suggesting that approximately 70% of patients treated with sumatriptan 100 mg did not achieve pain freedom in at least two out of three attacks (3). Additionally, the study inclusion criterion of patients having at least moderate migraine-related disability, as defined by a total MIDAS score of ≥11, may have contributed to a relatively high proportion of triptan-experienced patients being classified as insufficient responders.
A greater proportion of patients in the TIR group (vs. other subgroups) were from Europe and less from Asia, which may reflect different triptan use across geographies. The TIR population had a higher MIDAS total score compared to both triptan-naïve patients and those with a sufficient response to triptan. This was despite the greater use of preventive treatments in the TIR population and suggests that this population had the greatest need for improvement in their migraine therapy.
In CENTURION, lasmiditan was efficacious in the treatment of an acute migraine attack in patients with an insufficient response to triptans; the magnitude of effect was similar to that seen in other trial participants (i.e. those who had a sufficient response to triptans or who were triptan naïve). Excluding patients from the TIR population based on tolerability issues or contraindications (TIR efficacy only population) had little influence on the findings. In an analysis of pooled data from SAMURAI, SPARTAN and CENTURION, the efficacy of lasmiditan did not differ based on a patient experience with triptans (poor/none vs. good, poor/none vs. other).
That lasmiditan has efficacy in patients with an insufficient response to triptans is consistent with its novel approach for acute treatment of migraine. First, lasmiditan has structural and pharmacological dissimilarity to all previous acute migraine treatments. It has a unique pyridinoyl-piperidine chemical scaffold not seen in any established drug (23). Second, the mechanism of action of lasmiditan is novel; lasmiditan is centrally penetrant and selectively targets 5-HT1F receptors on neurons in the central and peripheral trigeminal system to relieve symptoms of migraine attacks by inhibiting pain pathways, including the trigeminal nerve, and without vasoconstriction (24,25).
Safety findings for the TIR population were similar to those for the overall CENTURION population. Common TEAEs in the TIR safety population were similar to those in the overall population (dizziness, paresthesia, fatigue, nausea, vertigo, and somnolence).
For the CENTURION study, the TIR criteria were established based on published literature, guidance from AHS and IHS, and with input from migraine experts. The TIR definition identified patients not experiencing the desired attributes of acute treatment for migraine, known reasons for triptan discontinuation. For example, if a patient stops a drug because it does not work for them, they may be unwilling to try it again at a different dose or earlier during an attack. However, the specific criteria used to define a triptan insufficient responder in this study have not been universally accepted across the migraine field and, to date, a standard definition of this population has not been agreed upon. A potential limitation of our criteria is that in the assessment of treatment efficacy the mTOQ does not isolate the response to triptans in the questioning; instead, it encompasses all treatments that the patient take to treat an acute migraine attack (both prescribed and over-the-counter).
Additionally, patients were not prospectively tested for insufficient response to triptans, there was no requirement for patients to have taken their triptan early in the attack or escalate the dose before they could consider their triptan not efficacious, and the primary definition of TIR did not require patients to fail multiple triptans. The strong therapeutic alliance necessary to carry out such an iterative procedure may not always exist, nor may such an approach be desirable for every patient given the availability of other migraine-specific therapies with an alternative mechanism of action.
Finally, the study did not include a triptan comparator arm, so direct comparative efficacy of lasmiditan versus triptan in patients classified as triptan insufficient responders is not available.
The definition of a triptan non-responder is still debated. In light of the lack of an accepted definition of non-response, we have included a broader patient population in this analysis so as to encompass the entire range of patients who are not satisfied with triptan use. Some experts advocate that the definition of a triptan insufficient responder should consider only lack of efficacy and not discontinuation of triptans due to contraindications or tolerability issues. Accordingly, we conducted a post hoc sensitivity analysis excluding patients with tolerability issues or who had developed a contraindication and found the findings in the TIR efficacy only population were similar to those of the total TIR population.
In conclusion, based on these findings, there is strong evidence that lasmiditan is efficacious in patients who have previously experienced insufficient response to a triptan. Lasmiditan may be a therapeutic option in this subgroup of patients.
Article highlights
Lasmiditan is efficacious in patients who have experienced insufficient response to triptans. The CENTURION trial safety findings for patients with an insufficient response to triptans were similar to those for the overall trial population. Lasmiditan may be a therapeutic option in patients with an insufficient response to triptans.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024211048507 - Supplemental material for Lasmiditan efficacy in the acute treatment of migraine was independent of prior response to triptans: Findings from the CENTURION study
Supplemental material, sj-pdf-1-cep-10.1177_03331024211048507 for Lasmiditan efficacy in the acute treatment of migraine was independent of prior response to triptans: Findings from the CENTURION study by Uwe Reuter, John H Krege, Louise Lombard, Elisa Gomez Valderas, Judith Krikke-Workel, Grazia Dell-Agnello, Sherie A Dowsett and Dawn C Buse in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_03331024211048507 - Supplemental material for Lasmiditan efficacy in the acute treatment of migraine was independent of prior response to triptans: Findings from the CENTURION study
Supplemental material, sj-pdf-2-cep-10.1177_03331024211048507 for Lasmiditan efficacy in the acute treatment of migraine was independent of prior response to triptans: Findings from the CENTURION study by Uwe Reuter, John H Krege, Louise Lombard, Elisa Gomez Valderas, Judith Krikke-Workel, Grazia Dell-Agnello, Sherie A Dowsett and Dawn C Buse in Cephalalgia
Supplemental Material
sj-pdf-3-cep-10.1177_03331024211048507 - Supplemental material for Lasmiditan efficacy in the acute treatment of migraine was independent of prior response to triptans: Findings from the CENTURION study
Supplemental material, sj-pdf-3-cep-10.1177_03331024211048507 for Lasmiditan efficacy in the acute treatment of migraine was independent of prior response to triptans: Findings from the CENTURION study by Uwe Reuter, John H Krege, Louise Lombard, Elisa Gomez Valderas, Judith Krikke-Workel, Grazia Dell-Agnello, Sherie A Dowsett and Dawn C Buse in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: UR has received speaker fees/honorarium for consulting from Abbvie, Amgen, Allergan, Co-Lucid, Eli Lilly and Company, Lundbeck, Medscape, Novartis, StreaMedUp, and TEVA Pharma; he serves as associate editor of the Journal of Headache and Pain and Frontiers in Neurology and as board member of the European Headache Federation. JHK, EVG, JWW, GDA and SAD are full-time employees and minor stockholders at Eli Lilly and Company. LL is a former full-time employee and a minor stockholder at Eli Lilly and Company. DCB has received grant support and honoraria from Alder, Allergan/Abbvie, Amgen, Eli Lilly and Company, Lundbeck and Teva; she is on the editorial board of Current Pain and Headache Reports.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The CENTURION study was sponsored by Eli Lilly and Company.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
