Abstract
Background:
Branch atheromatous disease (BAD) is a subtype of ischemic stroke associated with early neurological deterioration (END) and poor outcomes. Although BAD shares features with large artery atherosclerosis, optimal treatment strategies remain undefined.
Objectives:
To assess the efficacy and safety of early dual antiplatelet therapy (DAPT) and high-intensity statins in reducing END and improving outcomes in BAD.
Design:
A prospective, single-arm study with a historical control group.
Methods:
This study reports the results of the Statin and Dual Antiplatelet Therapy in Preventing Early Neurological Deterioration in Branch Atheromatous Disease trial. Patients with BAD-related ischemic stroke were treated with aspirin, clopidogrel, and high-intensity statins within 24 h of symptom onset. Outcomes were compared with a historical control cohort treated with single antiplatelet therapy and moderate- or low-intensity statins. The primary outcome was the composite of END (defined as an National Institutes of Health Stroke Scale score increase ⩾2 points within 7 days) or recurrent stroke within 30 days. Secondary outcomes included severe END, functional outcomes at 90 days, and safety events.
Results:
A total of 91 patients received intensive therapy and 285 received standard treatment. The primary endpoint occurred less frequently in the intensive group (34.1% vs 48.1%; adjusted risk ratio (aRR), 0.71; 95% confidence interval (CI), 0.52–0.98; p = 0.034). Intensive therapy significantly reduced END at 7 days (34.1% vs 47.0%; aRR, 0.73; 95% CI, 0.54–1.00; p = 0.049) but not recurrent stroke at 30 days (2.2% vs 1.8%; aRR, 1.16; 95% CI, 0.25–5.43). Good outcomes at 90 days (modified Rankin Scale ⩽2) were more common with intensive therapy (73.6% vs 57.2%; aRR, 1.27; 95% CI, 1.09–1.48; p = 0.002). Major bleeding and mortality did not differ between groups.
Conclusion:
Early intensive therapy with DAPT and high-intensity statins significantly reduced END and improved recovery in BAD without compromising safety. Further studies are warranted to validate these findings.
Trial registration:
ClinicalTrials.gov; Identifier: NCT04824911 (https://clinicaltrials.gov/study/NCT04824911).
Plain language summary
Some people who experience a certain type of acute ischemic stroke called branch atheromatous disease (BAD) may get worse within the first few days, even after receiving medical care. This early worsening can lead to more severe disability. In this study, we tested whether starting a combination of dual antiplatelet therapy—aspirin and clopidogrel—along with a high-dose statin (a cholesterol-lowering medication) within 24 hours of stroke onset could help prevent this early decline. We treated 91 patients with this intensive therapy and compared their outcomes to 285 patients from previous years who had received standard treatment. We found that those who received the early intensive therapy were less likely to experience worsening stroke symptoms during the first week and more likely to have better recovery after three months. The treatment did not increase the risk of serious side effects, although mild bleeding occurred slightly more often. These findings suggest that early use of dual antiplatelet therapy and a strong statin may help improve outcomes in people with this specific kind of stroke. More research is needed to confirm these results and guide future treatment recommendations.
Keywords
Introduction
Branch atheromatous disease (BAD) is a unique pathological entity traditionally classified under small-vessel occlusion. It involves the narrowing or occlusion at the origin of large penetrating arteries due to junctional atherosclerotic plaques or microatheromas near the parent artery. 1 This mechanism differs from lipohyalinosis or cerebral amyloid angiopathy, which are commonly implicated in true lacunar infarctions and involve smaller distal perforators. 2 BAD has been increasingly recognized as a significant cause of acute subcortical infarctions, particularly due to its strong association with early neurological deterioration (END) and a higher risk of stroke recurrence, especially progressive motor deficits.3,4
Recent advances in high-resolution magnetic resonance imaging (MRI) have enabled more accurate identification of BAD-related infarcts. 5 Characteristic imaging findings include infarcts in the lenticulostriate territory extending over three or more axial slices, or infarctions of the paramedian pons reaching the ventral surface, without major vessel occlusion.3,6 However, BAD does not fit neatly into existing stroke classification systems such as TOAST, often falling under undetermined etiology due to the lack of significant proximal arterial stenosis and absence of cardioembolic sources. Although BAD is pathologically linked to atherosclerosis, it has long been undertreated or misclassified. Given its atherothrombotic nature, BAD may respond better to strategies targeting both platelet aggregation and plaque stabilization.7,8
Dual antiplatelet therapy (DAPT), typically involving aspirin and clopidogrel, has become a key component in secondary stroke prevention, particularly in patients with minor noncardioembolic strokes.9,10 Furthermore, studies have suggested that DAPT is also effective in reducing the incidence of END.11,12 Similarly, statins play a crucial role in vascular protection beyond lipid lowering. Current clinical guidelines also recommend the early use of statins, as soon as patients are able to tolerate oral medications following an acute stroke event. 13 Achieving low-density lipoprotein cholesterol (LDL-C) levels at or below 70 mg/dL has been linked to a reduced risk of future cardiovascular events when compared to higher LDL-C ranges (90–110 mg/dL), especially among individuals with atherosclerotic disease after ischemic stroke or transient ischemic attack. 14 At present, DAPT combined with high-intensity statin therapy forms the cornerstone of treatment for intracranial atherosclerosis.15,16
Given that both BAD and intracranial atherosclerosis share similar pathological mechanisms with atherosclerosis, the trial aimed to assess the efficacy of early DAPT and high-intensity statin therapy in managing acute small subcortical infarctions caused by BAD. 17
Methods
Study design and population
This article reports the results of the Statin and Dual Antiplatelet Therapy in Preventing Early Neurological Deterioration in Branch Atheromatous Disease (SATBRAD) trial, a prospective, single-center, open-label, single-arm study using a historical control. The SATBRAD trial was designed to assess the efficacy and safety of early DAPT and high-intensity statin treatment in patients with BAD-related acute ischemic stroke. Given that current guidelines recommend DAPT for minor ischemic strokes—and considering that BAD often presents with mild stroke severity—a prospective control group receiving single antiplatelet therapy could raise ethical concerns. 18 Therefore, a historical control design was adopted for this trial.
The study was conducted at Chang Gung Memorial Hospital in Chiayi, Taiwan, from March 2021 through December 2024. 17 Eligible patients met all of the following criteria: (1) a clinical diagnosis of acute ischemic stroke with a National Institutes of Health Stroke Scale (NIHSS) score of 1–8; (2) an ischemic lesion on diffusion-weighted imaging located in the striatocapsular region or brainstem; and (3) imaging features consistent with BAD, defined as a lesion visible on three or more axial MRI slices in the lenticulostriate territory or an infarct extending from the ventral surface of the pons.
Patients with >50% stenosis of the relevant arteries on time-of-flight magnetic resonance angiography were excluded to isolate BAD without significant large-vessel stenosis. Additionally, patients who received intravenous thrombolysis were excluded in both the intervention and historical control groups to minimize confounding effects from reperfusion therapy. Consequently, the study population included only those who were either outside the therapeutic window for thrombolysis or had contraindications to its administration.
Trial intervention
Patients in the intervention group received DAPT (aspirin and clopidogrel) and high-intensity statins within 24 h of stroke onset. DAPT was continued for 21 days, followed by monotherapy with either aspirin or clopidogrel. High-intensity statin therapy consisted of atorvastatin (40–80 mg/day) or rosuvastatin (20–40 mg/day), maintained for 90 days with dose adjustments as needed for tolerability.
Although BAD shares similar atherosclerotic pathology with intracranial atherosclerosis, the patients in our study were excluded if they had >50% stenosis of the relevant parent artery. A prior study showed the safety and efficacy of DAPT for 30 days in patients with NIHSS score up to 15 due to large artery atherosclerosis. 12
Historical control group
The historical control group was selected from previous prospective observational studies conducted between January 2016 and December 2020. These studies focused on evaluating and predicting END or atrial fibrillation.19,20 Patients were included in the historical control group if they met the same inclusion criteria as those in the intervention group, except that they received only single antiplatelet therapy (either aspirin or clopidogrel). Additionally, moderate- or low-intensity statin therapy was administered when total cholesterol levels exceeded 160 mg/dL or LDL-C levels were ⩾100 mg/dL. All clinical data and patient outcomes were prospectively recorded.
Clinical information and outcomes
Patient demographics, including age, gender, and medical history (hypertension, diabetes mellitus, hyperlipidemia, coronary artery disease, atrial fibrillation, smoking status, and prior cerebrovascular disease), were meticulously documented. Vital signs, hematological parameters, biochemical markers, and coagulation profiles were recorded upon hospital admission.
Given that END in lacunar infarction predominantly affects motor function, it was primarily defined as an increase in the NIHSS score by ⩾2 points within 7 days of stroke onset. 21 Additionally, severe END was assessed as an NIHSS increase of ⩾4 points, and both END definitions were also evaluated within the first 3 days. The primary outcome of this study was the proportion of patients who experienced END within 7 days and recurrent stroke within 30 days. Clinical outcomes at admission and at 90 days were evaluated using the modified Rankin Scale (mRS). A good outcome was defined as an mRS score ⩽2, while a favorable outcome was defined as an mRS score ⩽1.
Sample size and power determination
The initial sample size calculation was based on detecting a significant difference in the incidence of END and recurrent stroke between the intervention and control groups. Assuming an END rate of 27% in the historical control group and an anticipated reduction to 15% in the intervention group, a total of 138 participants in the intervention arm and 277 in the control arm were required to achieve 80% power at a two-sided alpha level of 0.05, using a 2:1 historical control-to-intervention ratio. However, due to slower-than-expected patient recruitment—attributable to strict inclusion criteria, the narrow enrollment window, and the impact of clinical and logistical challenges during the study period—the final number of patients enrolled in the intervention group was 91.
Statistical analysis
Statistical analyses were conducted using the Statistical Package for the Social Sciences software (version 27; Chicago, IL, USA). Normality of continuous variable distributions was evaluated using the Kolmogorov–Smirnov test. Depending on distribution, comparisons between two independent groups were performed using Student’s t test for normally distributed continuous variables or the Mann–Whitney U test for nonnormally distributed continuous variables. Categorical variables were compared using the Chi-square test. The 90-day mRS scores were analyzed with an ordinal regression model. The proportional odds assumption was tested and met for the ordinal logistic regression model, supporting the appropriateness of this method for analyzing ordered mRS outcomes. The model was adjusted for age, sex, LDL-C, and baseline NIHSS score.
For binary outcomes such as END and functional outcomes (Table 2), adjusted risk ratios (aRRs) and 95% confidence intervals (CI) were estimated using generalized linear models with a Poisson distribution, log link function, and robust standard errors. The RRs were adjusted for potential confounders, including age, sex, LDL-C, and baseline NIHSS score. For paired observations (within-subject comparisons), appropriate nonparametric or parametric tests were applied. Laboratory data at initial presentation versus follow-up (Table 4) were compared using the Wilcoxon signed-rank test. All tests were two-tailed, and a significance level of p < 0.05 was applied for hypothesis testing.
Results
Study population
A total of 3136 patients were screened for the historical control group and 2439 for the intervention group, as detailed in Figure 1. After applying the inclusion and exclusion criteria, 91 patients received intensive therapy and 285 were included in the historical control group.
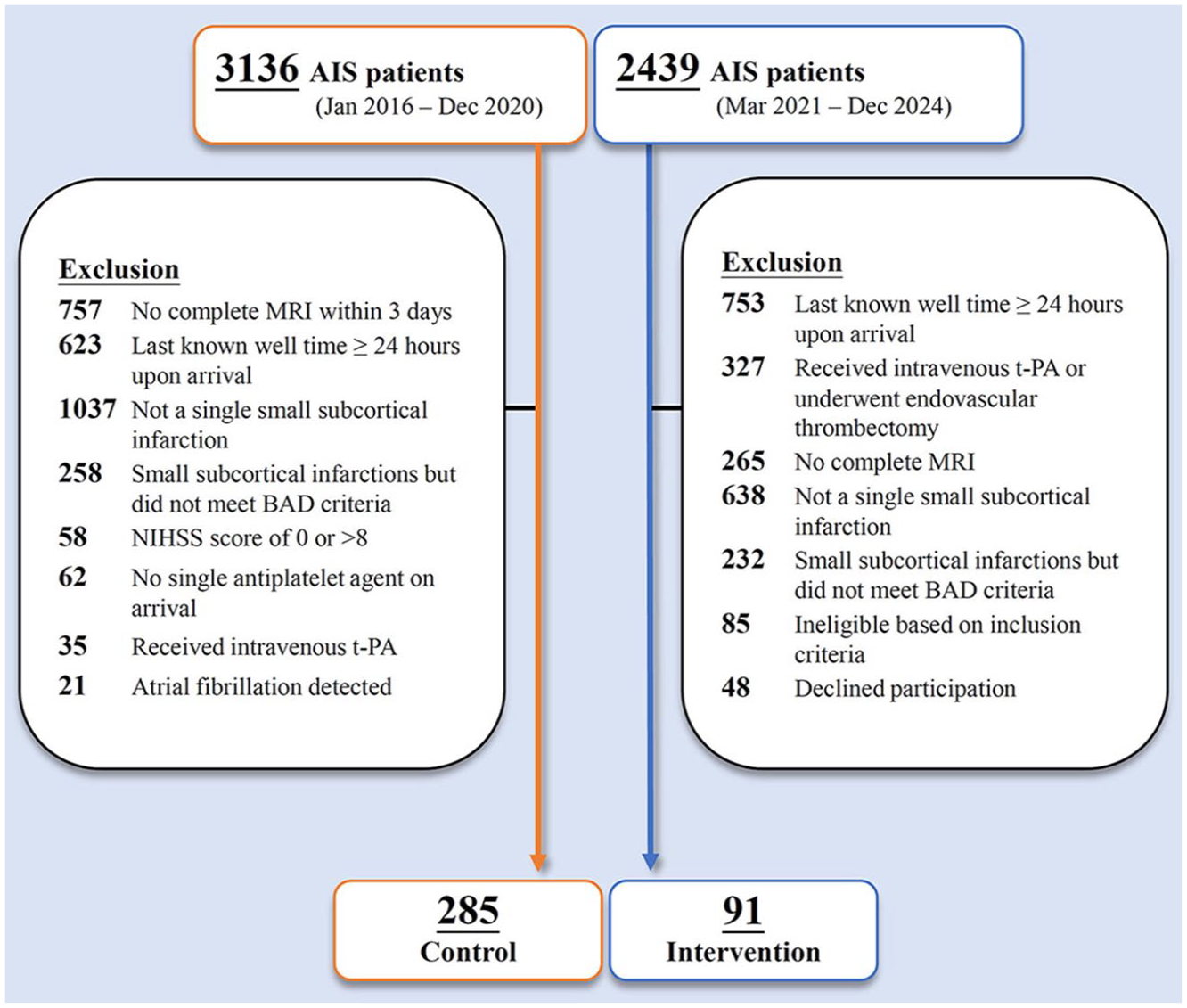
Flow diagram of patient selection for historical control and intensive therapy groups.
Baseline characteristics were largely comparable between groups (Table 1). The median age did not differ significantly (68 vs 70 years, p = 0.448). However, a significantly lower proportion of female patients was observed in the intensive therapy group (27.5% vs 40.7%, p = 0.023). Prevalence of comorbidities such as hypertension, diabetes, hypercholesterolemia, and prior stroke were similar between groups. Coronary artery disease showed a nonsignificant trend toward lower frequency in the intensive group (1.1% vs 6.7%, p = 0.056).
Baseline characteristics of the study groups.

BP, blood pressure; eGFR, estimated glomerular filtration rate; IQR, interquartile range; LDL-C, low-density lipoprotein cholesterol; mRS, modified Rankin Scale; NIHSS, National Institutes of Health Stroke Scale.
No significant differences were observed in baseline NIHSS scores (3 vs 4, p = 0.117) or time from last known well to ED arrival. However, patients in the intensive group had significantly higher LDL-C (125 vs 113 mg/dL, p = 0.001) and total cholesterol levels (196 vs 180 mg/dL, p = 0.002). Diastolic blood pressure was also slightly higher in the intensive group (101 vs 91 mmHg, p = 0.009). The vascular territories of infarction were similar but trended toward a higher proportion of lenticulostriate infarctions in the intensive group (69.2% vs 58.2%, p = 0.062).
Primary and secondary outcomes
The primary composite endpoint of END within 7 days or recurrent stroke within 30 days occurred significantly less frequently in the intensive therapy group (34.1% vs 48.1%; aRR, 0.71; 95% CI, 0.52–0.98; p = 0.034; Table 2). END within 7 days (NIHSS increase ⩾2) was significantly reduced (34.1% vs 47.0%; aRR, 0.73; 95% CI, 0.54–1.00; p = 0.049), as was END within 3 days (31.9% vs 46.0%; aRR, 0.70; 95% CI, 0.50–0.96; p = 0.029). Severe END (NIHSS increase ⩾4) was also lower in the intensive group within both 3 days (14.3% vs 27.0%; aRR, 0.53; 95% CI, 0.31–0.91; p = 0.021) and 7 days (15.4% vs 27.4%; aRR, 0.56; 95% CI, 0.33–0.96; p = 0.034).
Primary and secondary efficacy outcomes.

All effect estimates (RRs and common odds ratio) for intensive therapy were adjusted for age, sex, LDL-C and initial NIHSS score using generalized linear models or ordinal logistic regression, as appropriate. No adjustment was made for multiple comparisons.
Scores on the mRS of functional disability range from 0 (no symptoms) to 6 (death).
COR, common odds ratio; END, early neurological deterioration; IQR, interquartile range; LDL-C, low-density lipoprotein cholesterol; mRS, modified Rankin Scale; NIHSS, National Institutes of Health Stroke Scale; RR, risk ratio.
Recurrent ischemic stroke within 30 days was infrequent and not statistically different between groups (2.2% vs 1.8%, p = 0.850). Similar results were observed at recurrent stroke within 90 days (3.3% vs 2.1%, p = 0.576). Cardiovascular events were uncommon and comparable between groups (3.3% vs 3.9%, p = 0.840). Significantly more patients in the intensive therapy group achieved the LDL-C target of <70 mg/dL at 90 days (67.0% vs 43.2%; aRR, 1.60; 95% CI, 1.31–1.95; p < 0.001).
At 90 days, the median mRS score was lower in the intensive therapy group (1 (IQR 0–3) vs 2 (IQR 1–3), common odds ratio 0.67; 95% CI, 0.42–1.07; p = 0.099). The distribution of mRS scores at 90 days is presented in Figure 2. A shift toward better functional outcomes was observed in the intensive group: mRS 0 (26.4% vs 15.4%), mRS 1 (36.3% vs 31.2%), and fewer patients with mRS 3–6 (26.4% vs 42.8%). Good outcomes (mRS ⩽2) were significantly more frequent (73.6% vs 57.2%; aRR, 1.27; 95% CI, 1.09–1.48; p = 0.002).

Distribution of scores on the mRS at 90 days. Shown are the distribution of scores on the mRS among patients in the intensive treatment group and the historical control group. The mRS score ranges from 0 (no symptoms) to 6 (death).
Safety outcomes
There were no significant differences in 90-day mortality (0% vs 0.7%, p = 1.000) or moderate-to-severe bleeding events (1.1% in both groups; Table 3). Mild bleeding events were more frequent in the intensive therapy group (5.5% vs 1.8%), though not statistically significant (p = 0.054). Liver function abnormalities occurred at similar rates (8.8% vs 7.4%, p = 0.658), supporting a comparable safety profile between the groups.
Safety outcomes.

Liver function abnormalities were defined as elevated transaminases (AST/ALT) exceeding 2× the upper limit of normal.
Bleeding events were categorized as moderate to severe or mild as defined by the GUSTO classification.
ALT, alanine aminotransferase; AST, aspartate aminotransferase; GUSTO, Global Use of Strategies to Open Occluded Arteries.
Laboratory outcomes
Follow-up laboratory data demonstrated that intensive therapy resulted in significant improvements in lipid profiles (Table 4). LDL-C levels decreased markedly from 125 to 60 mg/dL (p < 0.001), and total cholesterol dropped from 196 to 124 mg/dL (p < 0.001; Table 4). HDL-C levels increased modestly (from 41 to 45 mg/dL, p = 0.001), while triglyceride levels declined significantly (from 120 to 83 mg/dL, p < 0.001).
Laboratory data of both groups before and after treatment.
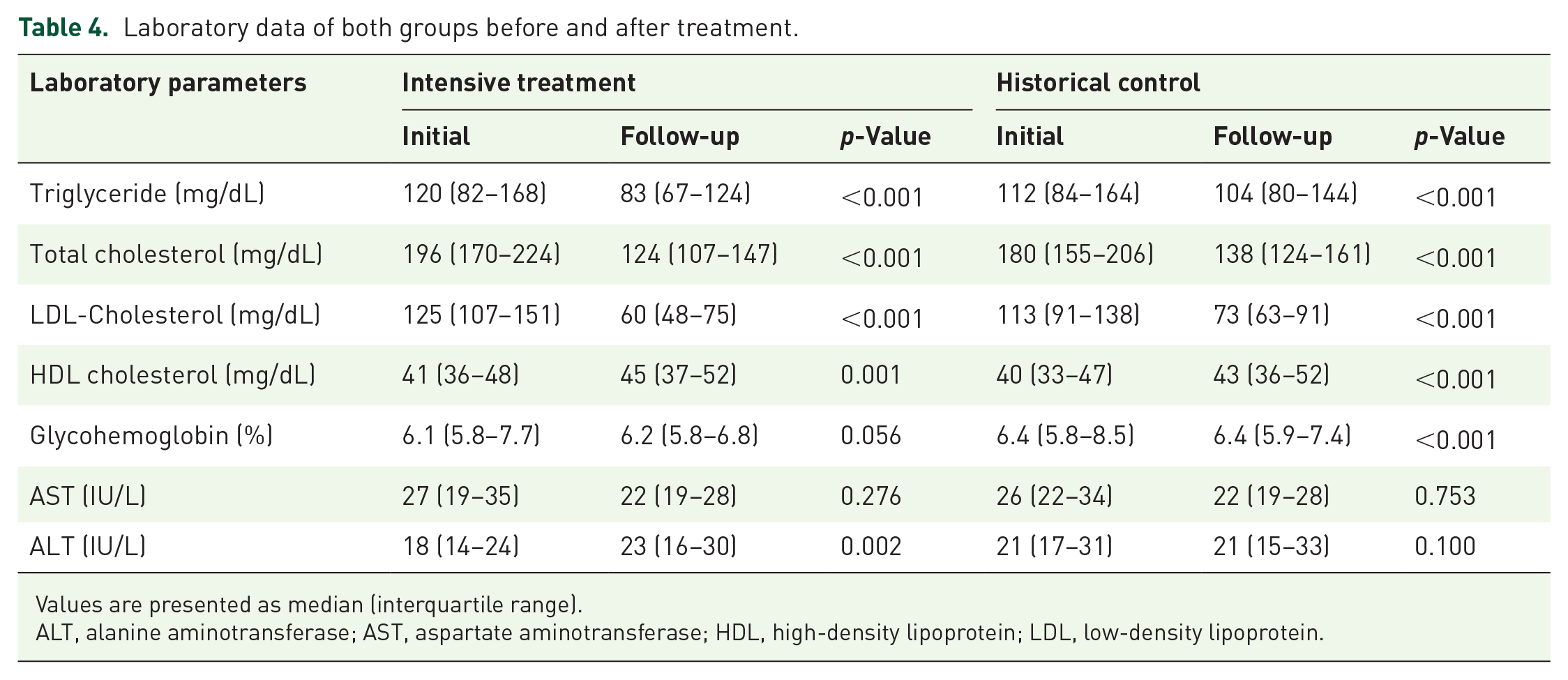
Values are presented as median (interquartile range).
ALT, alanine aminotransferase; AST, aspartate aminotransferase; HDL, high-density lipoprotein; LDL, low-density lipoprotein.
Liver enzyme monitoring showed a slight increase in ALT (from 18 to 23 IU/L, p = 0.002), though values remained within the normal range. AST levels remained stable throughout treatment.
Although baseline LDL-C levels were higher in the intensive therapy group compared to historical controls (125 vs 113 mg/dL, p = 0.001), follow-up measurements revealed significantly lower LDL-C in the intensive group (60 vs 73 mg/dL, p < 0.001).
Discussion
Our study demonstrated that early intensive medical therapy combining DAPT with high-intensity statins significantly reduced the risk of END and improved functional outcomes in patients with acute BAD-related stroke. These findings support the growing consensus that BAD, although historically classified as a small-vessel disease, should be managed more like a large-artery atherosclerosis due to its shared pathophysiological features. 22
The reduction in END can be attributed to the synergistic effects of aspirin and clopidogrel, which inhibit different pathways of platelet activation. This may contribute to reduced thrombus propagation at the site of perforator artery involvement. 23 Although the study included patients with NIHSS scores up to 8—extending beyond the traditional definition of minor stroke—the decision to use DAPT for 21 days was supported by prior evidence, including the Clopidogrel in High-Risk Patients With Acute Non-Disabling Cerebrovascular Events (CHANCE) and Platelet-Oriented Inhibition in New TIA and Minor Ischemic Stroke (POINT) Trials, which demonstrated the efficacy and safety of short-term DAPT in patients with minor stroke (NIHSS ⩽3).9,10,18 Additionally, the recent Antiplatelet Therapy in Acute Mild to Moderate Ischemic Stroke (ATAMIS) trial further extended this evidence to patients with mild-to-moderate stroke severity (NIHSS 4–10), demonstrating benefit and safety of short-term DAPT. 24
Our study uniquely combined intensive DAPT with immediate high-intensity statin therapy, similar to the approach used in the Intensive Statin and Antiplatelet Therapy for Acute High Risk Intracranial or Extracranial Atherosclerosis (INSPIRES) trial.25,26 The INSPIRES trial, a 2 × 2 factorial design, examined both immediate versus delayed statin therapy and DAPT versus single antiplatelet therapy in patients with mild ischemic stroke or high-risk TIA of presumed atherosclerotic origin. While it found that DAPT within 72 h reduced the risk of new stroke at 90 days compared to aspirin alone, immediate statin therapy did not significantly reduce recurrent stroke but was associated with improved functional outcomes. Consistent with these findings, our study also observed improved functional recovery primarily due to the reduction in END from DAPT.
Moreover, our sub-analysis using high-resolution MRI revealed that high-intensity statins significantly reduced contrast-enhanced plaque volume, reinforcing their role in stabilizing atherosclerotic plaques specific to BAD and potentially improving perfusion outcomes.27,28 This imaging-based evidence complements prior findings from the INSPIRES trial, where immediate statin initiation—although not reducing recurrent stroke at 90 days—was associated with better functional outcomes. In our study, the benefit of statins may not have been fully realized within the short timeframe due to the low recurrence rate (2.4%), consistent with previous BAD cohorts (1.9%). 29 Nonetheless, adjusted analyses accounting for baseline LDL-C levels suggest that intensive lipid lowering provided additional vascular benefits in this population.
Our study highlights the importance of early intensive medical therapy in BAD management. By combining DAPT to prevent acute thrombus progression with high-intensity statins to stabilize atherosclerosis, we provide a dual-targeted approach to reducing END and improving long-term functional recovery. This reinforces the notion that BAD should be treated more aggressively, akin to large-artery atherosclerotic strokes. Given the high risk of END in BAD, clinicians should consider initiating DAPT and high-intensity statins as soon as possible in eligible patients. Our findings suggest that this approach may prevent infarct expansion and improve neurological outcomes, particularly in patients with high-risk features such as contrast-enhancing plaques or significant LDL elevation.30,31
In recent years, intravenous tirofiban, a glycoprotein IIb/IIIa inhibitor, have been explored as an alternative strategy to prevent END in high-risk strokes, in which mainly including acute stroke from intracranial stenosis or BAD.32,33 The Safety and Efficacy of Tirofiban for the Prevention of Neurological Deterioration in Acute Ischemic Stroke (TREND) trial demonstrated that tirofiban significantly reduced END rates compared to aspirin alone, suggesting its potential benefit in preventing END. 32 Similarly, the Recurrent Stroke Prevention with Tirofiban in Acute Non-Cardioembolic Cerebral Infarction (RESCUE BT2) trial showed that tirofiban improved functional outcomes at 90 days compared to aspirin, though with a slightly increased risk of intracerebral hemorrhage. 33 These trials suggest that, in selected patients, tirofiban may offer superior early stroke stabilization compared to single antiplatelet therapy.
Despite these promising findings, the role of tirofiban in BAD remains uncertain. Unlike DAPT, which is well established in minor ischemic strokes and BAD-related infarctions, tirofiban is currently reserved for select high-risk patients and requires intravenous administration in a hospital setting. Previous trials demonstrated that DAPT for 21 days reduces stroke recurrence without increasing major bleeding risks, supporting its use as the standard first-line treatment for BAD. 18 Additionally, DAPT has been shown to be superior to single antiplatelet therapy in reducing END within 7 days in patients with mild-to-moderate stroke (NIHSS score 4–10). 24 Given its established safety and effectiveness, DAPT remains a practical and widely accepted option, whereas tirofiban requires further validation in BAD-specific trials. The ongoing STRATEGY trial will clarify whether tirofiban plus aspirin offers superior outcomes compared to aspirin alone in BAD. 34 While tirofiban provides potent and rapid platelet inhibition, its clinical advantage over DAPT remains uncertain. However, given the lack of direct comparisons between DAPT and tirofiban in BAD, clinicians should weigh the risks and benefits carefully, reserving tirofiban for select patients at high risk of END despite standard therapy.
Although our study demonstrates promising results, several limitations warrant consideration. First, the sample size was relatively small, and this was a single-center study, which may limit generalizability. Additionally, the final sample size in the intervention group fell short of the originally calculated target. This shortfall may have reduced the study’s statistical power, particularly for secondary endpoints, and increased the risk of type II error. As such, caution is warranted when interpreting nonsignificant findings, and larger confirmatory trials are needed to validate these results.
Second, the study’s open-label design with a historical control introduces the possibility of selection bias, as treatment allocation was not randomized and the treatment may vary as time passed. The use of a historical control, primarily chosen due to ethical concerns about withholding recommended treatments, may introduce temporal bias related to evolving clinical practices, diagnostic methods, and supportive care improvements. Differences in these practices over the study period could affect comparability between groups and outcomes. Third, given the number of secondary outcomes analyzed, we acknowledge a potential increase in the risk of type I error, which should be considered when interpreting our findings. Lastly, while we observed plaque regression over 6 months in some cases, longer follow-up is necessary to confirm sustained clinical benefits. Future studies should investigate the long-term effects of intensive therapy in BAD and explore optimal treatment durations and intensities.
Conclusion
Our findings support the early initiation of DAPT and high-intensity statins in BAD-related stroke, leading to reduced END and improved functional recovery. These results further support a shift in the clinical approach—treating BAD more like a large-artery atherosclerotic process rather than a small-vessel disease—and highlight that intensive medical therapy plays a crucial role in BAD management. This warrants further large-scale, randomized trials to refine treatment strategies. Future research should also explore the role of other intensive treatment strategies in BAD and determine whether certain high-risk patients may benefit from a more aggressive approaches, such as glycoprotein IIb/IIIa inhibition or anticoagulation. 35
