Abstract
Aims:
To evaluate the effects of treatments with clopidogrel plus aspirin (dual therapy) on early neurological deterioration (END) and outcomes at 6 months in patients with acute large artery atherosclerosis (LAA) stroke.
Methods:
A total of 574 patients with LAA stroke were randomly assigned to receive either dual therapy or aspirin alone (monotherapy). The primary outcome was END. Secondary outcomes included recurrent ischemic stroke (RIS) and outcomes at 6 months.
Results:
The prevalence of END and RIS was lower in patients on dual therapy than in those on monotherapy during the 30 days. At 6 months, dual therapy improved outcomes among older patients and those with symptomatic stenosis in the posterior circulation and basilar artery.
Conclusion:
Clopidogrel plus aspirin is superior to aspirin alone for reducing END and RIS within 30 days and improves outcomes in certain subgroups at 6 months.
Keywords
Introduction
Stroke is a serious health problem worldwide and shows increasing prevalence as the population ages. The risk of recurrent stroke is high immediately after a transient ischemic attack or ischemic stroke, especially during the subsequent hours and days, 1,2 and patients who substantially recover from their initial deficit are at high risk of neurological deterioration as well. 3,4 As a result, clinical guidelines recommend antiplatelet therapy for patients with noncardioembolic stroke. 5
Aspirin is the most commonly used antiplatelet drug worldwide. Compared with placebo, aspirin reduces the relative risk of vascular events by 13% according to 2 meta-analyses 6,7 and stroke risk by 18% in the European Stroke Prevention Study 2. 8 In a large trial, clopidogrel was more effective than aspirin in reducing the combined risk of ischemic stroke, myocardial infarction (MI), or vascular death. 9 However, recent observations have suggested that drug resistance contributes to the failure of therapy in patients taking aspirin or clopidogrel. 10,11 Therefore, intensive clinical research is required to develop more effective interventions. 12
Aspirin and clopidogrel have different modes of action. Aspirin can irreversibly inhibit cyclooxygenase, leading to decreased thromboxane A2, a platelet aggregation activator, while clopidogrel blocks adenosine diphosphate to bind to its receptor on platelets to prevent clot formation. They can synergistically inhibit platelet aggregation, 13,14 and such dual therapy reduces the risk of recurrent ischemic stroke (RIS) events in patients with the acute coronary syndrome. 15,16 Although large-scale trials have not shown a benefit of the combination of clopidogrel and aspirin therapy for secondary prevention of ischemic events after stroke, 17 –19 these trials did not study the early, high-risk period after stroke. Three small pilot trials have shown trends toward a benefit of the combination therapy in patients with acute ischemic stroke. 20 –22 Due to the different pathophysiological bases for stroke subtypes, more data are required before any further conclusion can be drawn. 23,24
Platelet activation, such as platelet aggregation and formation of platelet–leukocyte aggregates, plays a crucial role in the pathophysiology of ischemic stroke through a variety of mechanisms. 25,26 Accumulating evidence shows that platelets contribute to inflammation, 27 and inflammation is important in the progression of atherosclerosis through inflammation-mediated destabilization and rupture of atherosclerotic lesions. 24,28,29 However, it is not known conclusively whether platelet activation is involved in early neurological deterioration (END) and recurrent stroke. We hypothesize that platelet activation may lead to neurological deterioration, recurrent stroke, or both. Furthermore, clopidogrel plus aspirin may be more effective than aspirin alone in reducing the risk of END and recurrent stroke by inhibiting platelet activity more efficiently. Herein, we conducted a randomized study designed to evaluate the effects of combined clopidogrel and aspirin therapy versus aspirin monotherapy on platelet activation, END, and RIS in patients within 30 days of acute large artery atherosclerosis (LAA) stroke and on outcomes at 6 months.
Materials and Methods
Study Populations
The study was reviewed and approved by the Ethics Committee of the People’s Hospital of Deyang City and the Committee of the Third Affiliated Hospital of Wenzhou Medical College. A total of 574 patients who had their first stroke and were admitted within 48 hours of their index stroke to the above-mentioned 2 hospitals were enrolled in the study between August 2009 and December 2011. Informed written consent was obtained from each patient or a family member before enrollment. Detailed medical history and demographic information were obtained using questionnaires. All patients were ≥18 years old with a diagnosis of ischemic stroke by cranial computed tomography (CT) and magnetic resonance (MR) imaging scanning. According to the TOAST classification system, 30 ischemic stroke was divided into five subtypes (LAA, small artery disease, cardioembolism, other determined etiology and undetermined etiology). Only patients with LAA subtype were included for further analysis. A brain CT scan was conducted for all patients before randomization, and a second scan was performed on day 14. Vascular imaging was performed to identify moderate (50%-70%) or greater (>70%) stenosis in the internal carotid, vertebrobasilar, middle cerebral, anterior cerebral, and posterior cerebral arteries by carotid duplex scan, CT angiography, or MR angiography. No patient had a history of carotid endarterectomy or carotid stent therapy, and none underwent carotid endarterectomy or carotid stent therapy during the follow-up period. Patients who presented with or had experienced any of the following conditions were excluded from the study: coma or National Institutes of Health Stroke Scale (NIHSS) score 20 ≥15; any clinically relevant arrhythmia on admission, including atrial fibrillation; any major concurrent illness, including renal failure and malignancies; fever, hypoxia, alterations in consciousness, or any relevant hemodynamic compromise on admission; use of ticlopidine, dipyridamole, other nonsteroidal anti-inflammatory drugs, or other aspirin-containing drugs previously or at the time of the index stroke; administration of heparin or low-molecular-weight heparin within 24 hours before enrollment in the study; any major surgical procedure within 1 week prior to enrollment; malignant paraproteinemias, or a family or personal history of bleeding disorders; platelet count <100 × 109/L or >450 × 109/L; any history of myeloproliferative disorders or of heparin-induced thrombocytopenia; or conditions such as sensitivity to aspirin, asthma, or severe cardiovascular, liver, or renal disease.
Study Design
All enrolled patients were randomly assigned to 1 of the 2 treatment groups, clopidogrel plus aspirin (dual therapy; n = 286) or aspirin alone (monotherapy; n = 288). Randomization into the trial was done through the randomization office of our hospital by means of sealed envelopes or allocation via the Internet.
For patients in the monotherapy group, aspirin was administered daily beginning on the day of admission (200 mg/d for 30 days and 100 mg/d thereafter). Patients in the dual therapy group received 200 mg aspirin and 75 mg clopidogrel daily from the day of admission to day 30 and 75 mg/d clopidogrel thereafter.
Platelet Aggregation
Platelet aggregation was measured by optical platelet aggregometry (OPA). 31 Blood samples (from an antecubital vein) were collected prior to the initial dose of aspirin or clopidogrel and at day 30 of therapy. In brief, whole blood specimens were centrifuged at 200g for 10 minutes to obtain platelet-rich plasma (PRP). Platelet-poor plasma (PPP) was obtained from the remaining specimens by centrifugation at 4000g for 10 minutes. Classical OPA in PRP was performed using a PAP-4D aggregometer (Bio/Data Corporation, Alpha Laboratories Limited, Horsham, Pennsylvania). Platelet aggregation was recorded as changes in light transmission. The recorder was adjusted to ensure that the difference in light transmission between PRP and PPP was 100%. The results of OPA are presented as the amplitude of light transmittance at 5 minutes after addition of the agonists 0.5 mmol/L arachidonic acid (AA) and 10 μmol/L adenosine diphosphate (ADP; Helena Laboratories, Beaumont, Texas).
Platelet–Leukocyte Aggregates
Venous blood was drawn from an antecubital vein prior to administration of the initial dose of aspirin or clopidogrel and again at day 30. It was anticoagulated with 3.8% sodium citrate and processed after 10 minutes of resting time without further manipulation. Platelet–leukocyte aggregates were counted by FC 500 MPC flow cytometry (Beckman Coulter Ltd, Miami, FL, USA) 32 and we used direct fluorescent markers (all commercially available; Coulter Immunotech, Krefeld, Germany). Whole blood was diluted 1:10 with warm hydroxyethyl-piperazineethane-sulfonic acid buffer. Two aliquots of 50 mL were incubated with CD61-phycoerythrin (an activation-independent subunit of the glycoprotein IIb/IIIa complex) to immunologically identify all platelets. Simultaneously, in a 1-step procedure, the sample for measuring platelet activation was stained with anti-CD62P. The other sample was double stained with the pan leukocytic marker CD45 to identify leukocytes. After incubation for 5 minutes, the process was stopped using cold buffer and immediately followed by flow cytometry.
Study Outcomes
Neurological functions of the patients were scored using the NIHSS on admission and on days 3, 7, 14, and discharge. The primary efficacy outcome was END. Early neurological deterioration was defined as an increase of 4 or more points on the NIHSS scale within 10 days after admission but excluding hemorrhagic transformation (HT) of infarct or new infarct in another vascular territory. 33,34 The secondary efficacy outcomes included the RIS and peripheral vascular event (deep venous thrombosis [DVT], pulmonary embolism [PE], or MI) and death during the 30 days of treatment. Recurrent ischemic stroke was defined as a sudden and persistent (>24 hours) deficit occurring after admission, with both clinical and imaging findings of ischemic stroke diagnosed in an independent artery separate from index stroke territory. Death was defined as death due to any cause.
The safety outcomes included hemorrhagic episodes occurring during the first 30 days of treatment. Hemorrhagic episodes were defined as HT of the cerebral infarction, intracerebral hemorrhage (ICH) not associated with cerebral infarction, or extracranial hemorrhage (eg, gastrointestinal bleeding or hematuria). Serious hemorrhage was defined as any symptomatic intracranial hemorrhage or any hemorrhage requiring blood transfusion.
At day 30, or earlier if discharged from the hospital, trained personnel determined the NIHSS and modified Rankin Scale (mRS) scores. At 6 months, mRS scores were assessed by a clinician or a nurse without knowledge of the treatment allocation. Good outcome was defined as an mRS score of 0 to 2, and disability was identified as an mRS score of 3 or greater in survivors. Recurrent ischemic stroke, MI, hemorrhage, DVT, PE, and overall mortality were also documented from day 31 to month 6. All reported efficacy and safety outcomes were confirmed by our central adjudication committee that was unaware of the study group assignments.
Statistical analyses
We determined that a sample of 574 patients would provide 90% power to detect a relative risk reduction of 15% in the percentage of neurological deterioration in the combination therapy group, with a 2-sided type I error of .05, assuming an event rate of 15% in the monotherapy group.
All analyses were performed according to the intent-to-treat principle. SPSS 16.0 statistical software (SPSS, Chicago, Illinois) was used to perform statistical analyses. Categorical variables are presented as frequencies and percentages. For the categorical variables, patient demographics between the groups were compared using chi-square tests, or when expected cell frequencies were small, exact tests were conducted. Continuous variables are presented as mean ± standard deviation and were compared with Student t test between the 2 groups and paired t test between pretreatment and posttreatment of each group. Differences in outcome parameters between the groups were assessed by the odds ratios (ORs) and 95% confidence intervals (CIs). All reported probability values were 2-sided with P < .05 considered significant. All analyses were done by an investigator blinded to both patient identity and the day the recording was taken prior treatment and after treatment.
Results
The trial profile is shown in Figure 1. A total of 574 patients were enrolled, with 286 randomly assigned to the dual therapy group and 288 to the monotherapy group. Four patients were withdrawn (2 in the dual therapy and 2 in the monotherapy group) by the local investigators before baseline assessments because they did not meet the inclusion criteria (they began treatment >48 hours after symptom onset). Thus, 570 patients (284 in the dual therapy group and 286 in the monotherapy group) were assessed for analysis. A total of 4 (0.70%) patients, 2 in the dual therapy group and 2 in the monotherapy group, were lost to follow-up. In all, 9 (3.15%) patients in the monotherapy group and 10 (3.52%) patients in the dual therapy group discontinued the study medication before the end of the study (Figure 1).
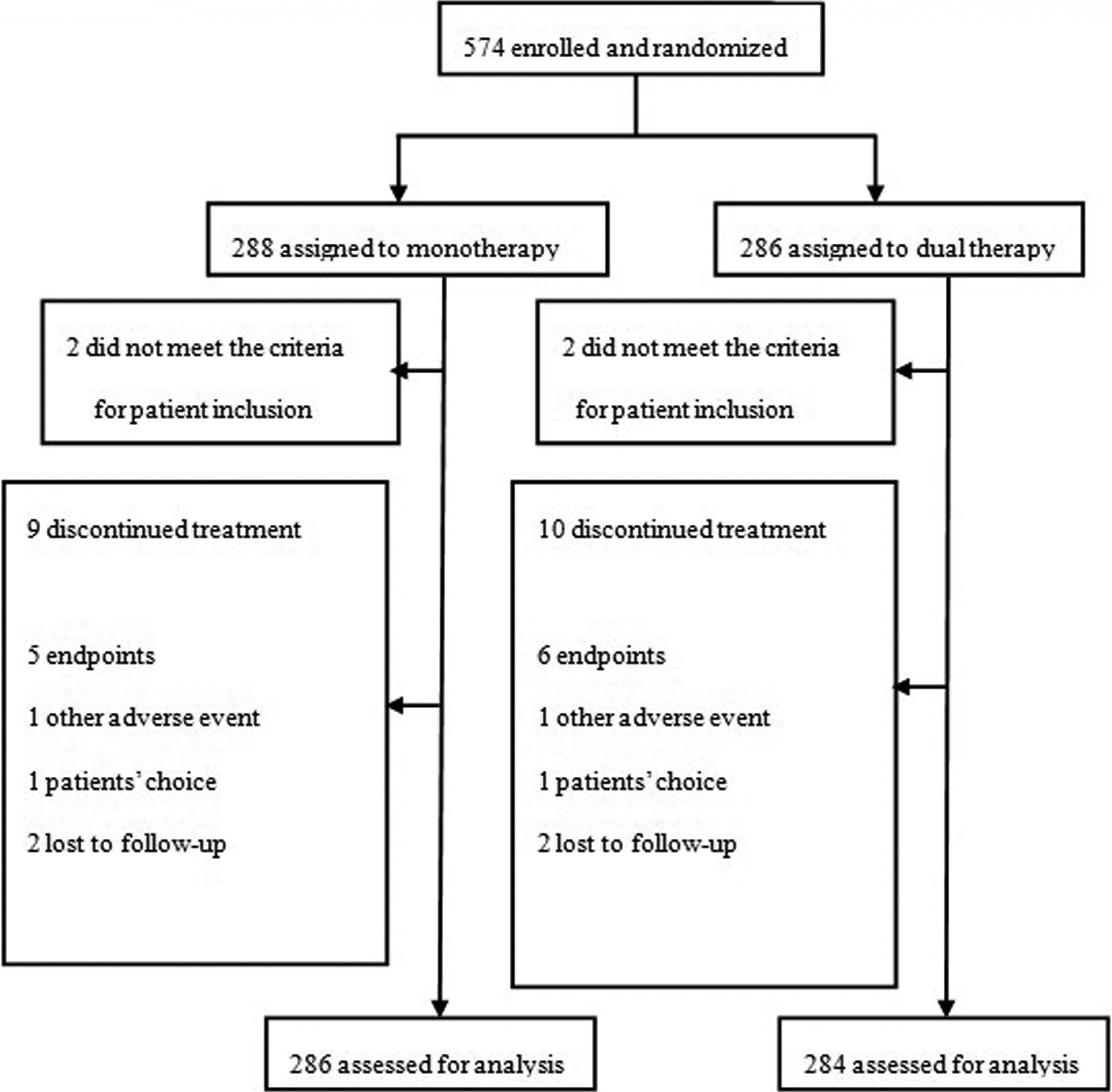
Trial profile.
The clinical characteristics of all patients are summarized in Table 1. The median age was 70 years, and 45.09% of the patients were women. The 2 groups were well balanced regarding baseline characteristics. No significant differences in the clinical characteristic distributions were observed between the 2 groups. The average times from stroke onset to randomization were 36.2 ± 13.3 and 34.4 ± 14.2 hours in the dual therapy group and the monotherapy group, respectively.
Demographics and Baseline Characteristics of Patients in the Dual Therapy (n = 284) and Monotherapy (n = 286) Groups.
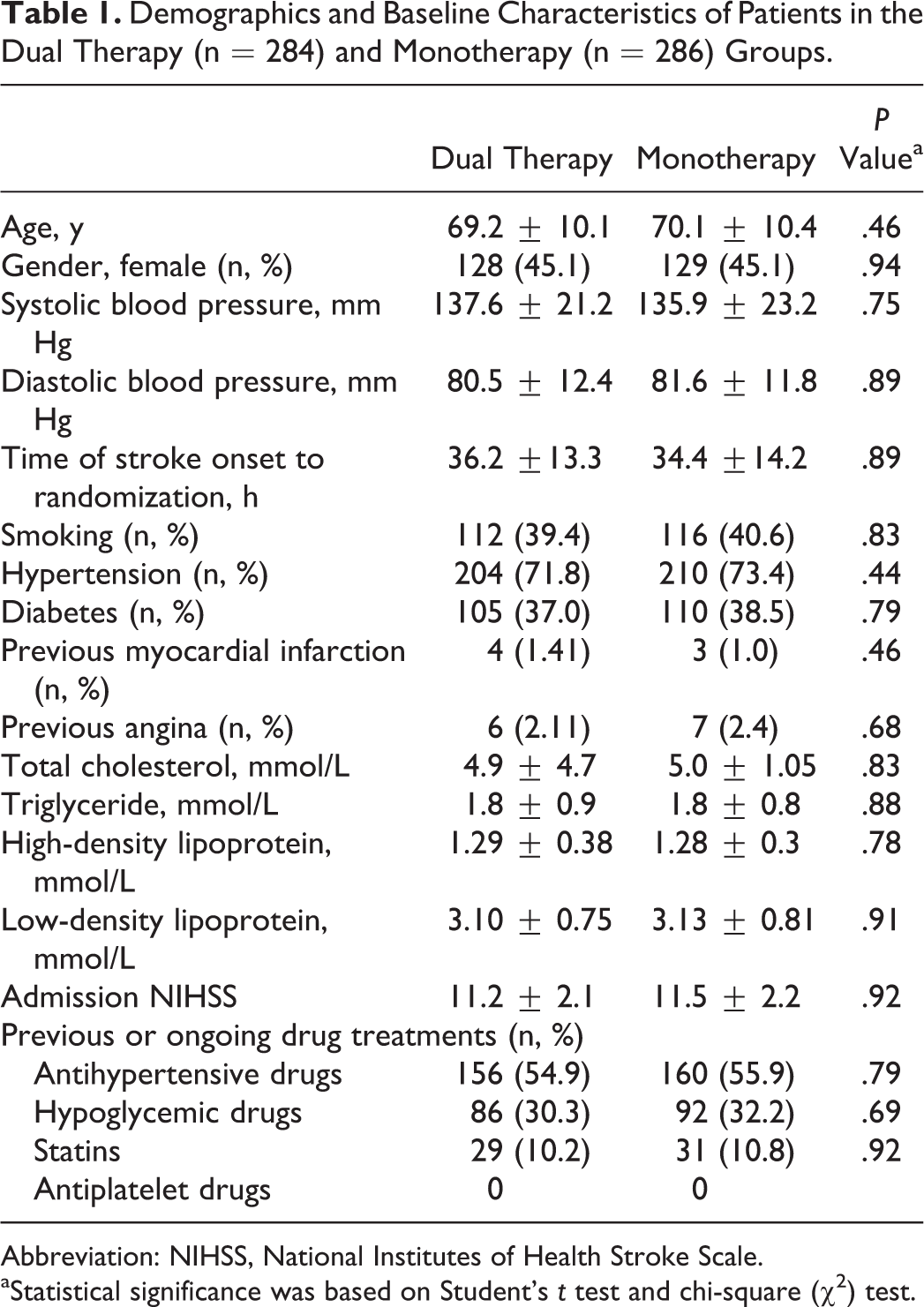
Abbreviation: NIHSS, National Institutes of Health Stroke Scale.
aStatistical significance was based on Student’s t test and chi-square (χ2) test.
The efficacy outcome and safety outcome within 30 days are shown in Table 2. Early neurological deterioration within 10 days posttreatment was observed in 38 (6.7%) patients. The prevalence of END was lower in patients in the dual therapy group than in those in the monotherapy group (3.5% vs 9.8%, OR: 0.34, 95% CI: 0.12-0.86, P < .001). Of the 570 patients, 23 (4%) had RIS within 30 days after admission. Similarly, the prevalence of RIS was lower in patients in the dual therapy group than in those in the monotherapy group (1.7% vs 6.3%, OR: 0.26, 95% CI: 0.08-0.82, P < .001; Table 2). These results suggest that dual therapy with aspirin and clopidogrel can decrease the occurrence of END and RIS more compared to treatment with aspirin alone in patients with LAA.
Outcomes of Patients in the Dual Therapy (n = 284) and Monotherapy (n = 286) Groups Within 30 Days (n, %).a
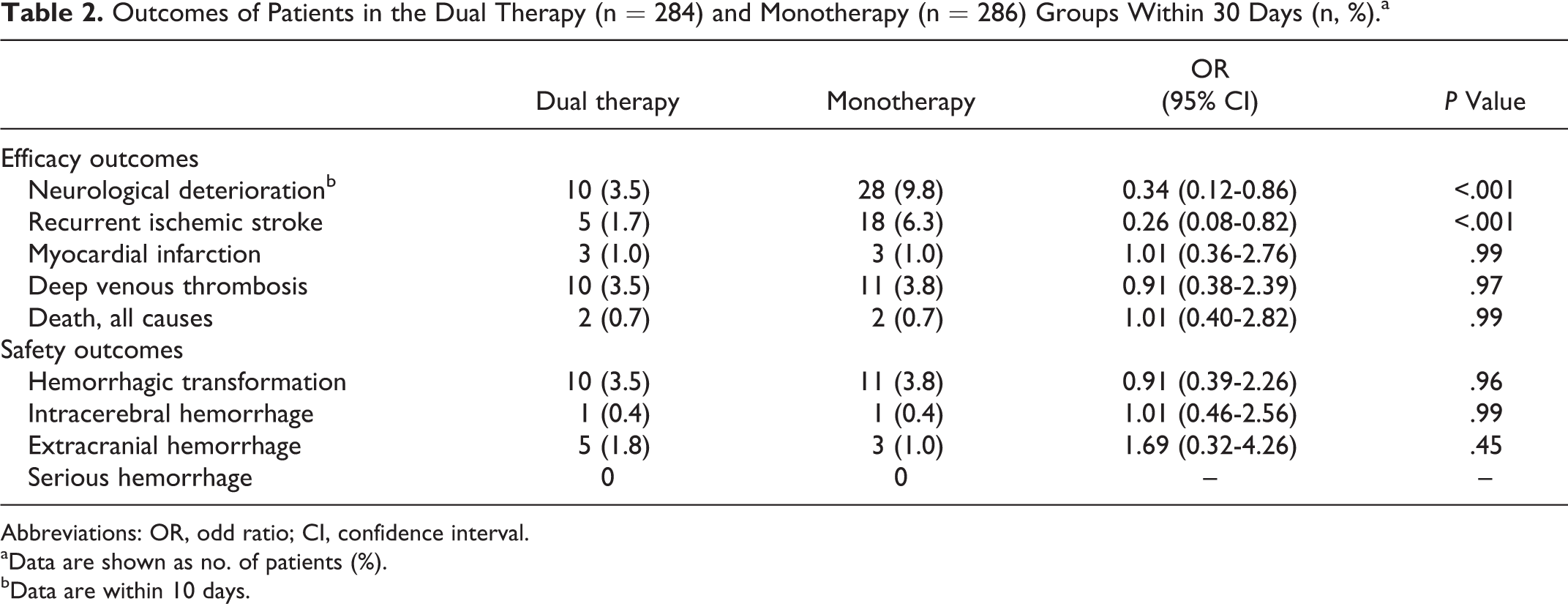
Abbreviations: OR, odd ratio; CI, confidence interval.
aData are shown as no. of patients (%).
bData are within 10 days.
We then tested whether dual therapy with aspirin and clopidogrel would have an effect on platelet aggregation and the formation of platelet–leukocyte aggregates. As shown in Table 3, prior to antiplatelet therapy, there were no significant differences in platelet aggregation and the formation of platelet–leukocyte aggregates between the 2 groups. On day 30 after treatment, platelet aggregation and number of platelet–leukocyte aggregates were significantly lower than on day 1 whether in the dual therapy group or in the monotherapy group. However, platelet aggregation and number of platelet–leukocyte aggregates were more lower in patients given clopidogrel plus aspirin than in patients given aspirin alone (Table 3). Our results suggest that dual therapy with clopidogrel plus aspirin more effectively inhibited platelet aggregation and platelet–leukocyte aggregate formation.
Platelet Aggregation and Platelet–Leukocyte Aggregates of Patients in the Dual Therapy (n = 284) and Monotherapy (n = 286) Groups (%).a
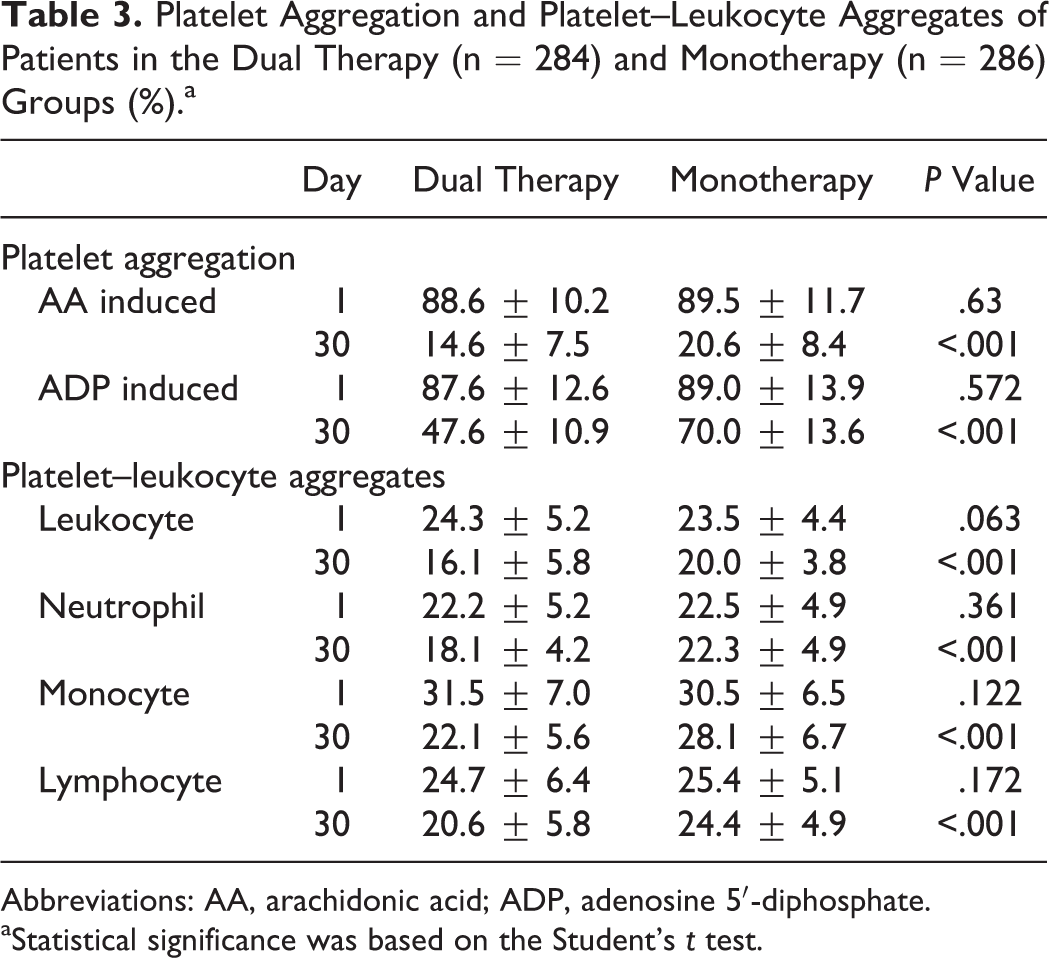
Abbreviations: AA, arachidonic acid; ADP, adenosine 5′-diphosphate.
aStatistical significance was based on the Student’s t test.
We then tested whether platelet activation will contribute to neurological deterioration and recurrent stroke (Table 4). We divided the samples into 2 groups: one with END or RIS and the other one without END or RIS. The data for platelet aggregation and platelet–leukocyte aggregate formation on day 1 were analyzed for our purpose. Compared to patients who did not have subsequent END or RIS within 30 days, the platelet aggregation induced by AA or ADP and platelet–leukocyte aggregate formation at day 1 were higher in patients who did experience these afflictions. Our results indicate that platelet activation plays a role in neurological deterioration and RIS.
Platelet Aggregation and platelet–Leukocyte Aggregates at Day 1 in Patients With (n = 61) and Without (n = 509) Subsequent END or RIS Within 30 Days (%).

Abbreviations: AA, arachidonic acid; ADP, adenosine 5′-diphosphate; END, early neurological deterioration; RIS, recurrent ischemic stroke.
aStatistical significance based on Student’s t test.
As shown in Table 2, no significant difference was observed for the prevalence of MI, DVT, and death from all causes nor was there a significant difference in safety outcome (the frequency of HT, ICH, and extracranial hemorrhage) during 30 days after treatment between dual therapy and monotherapy. We then asked whether a difference exists after 6 months of treatment. As shown in Table 5, there was not any significant difference between aspirin plus clopidogrel dual therapy and aspirin monotherapy in the efficacy outcomes (frequency of RIS, DVT, PE, MI, and all-cause death from day 31 to month 6) and safety outcomes (HT, ICH, and extracranial hemorrhage). There were no serious hemorrhagic events in either groups.
Comparison of Outcome From Day 31 to Month 6 Between the 2 Groups.
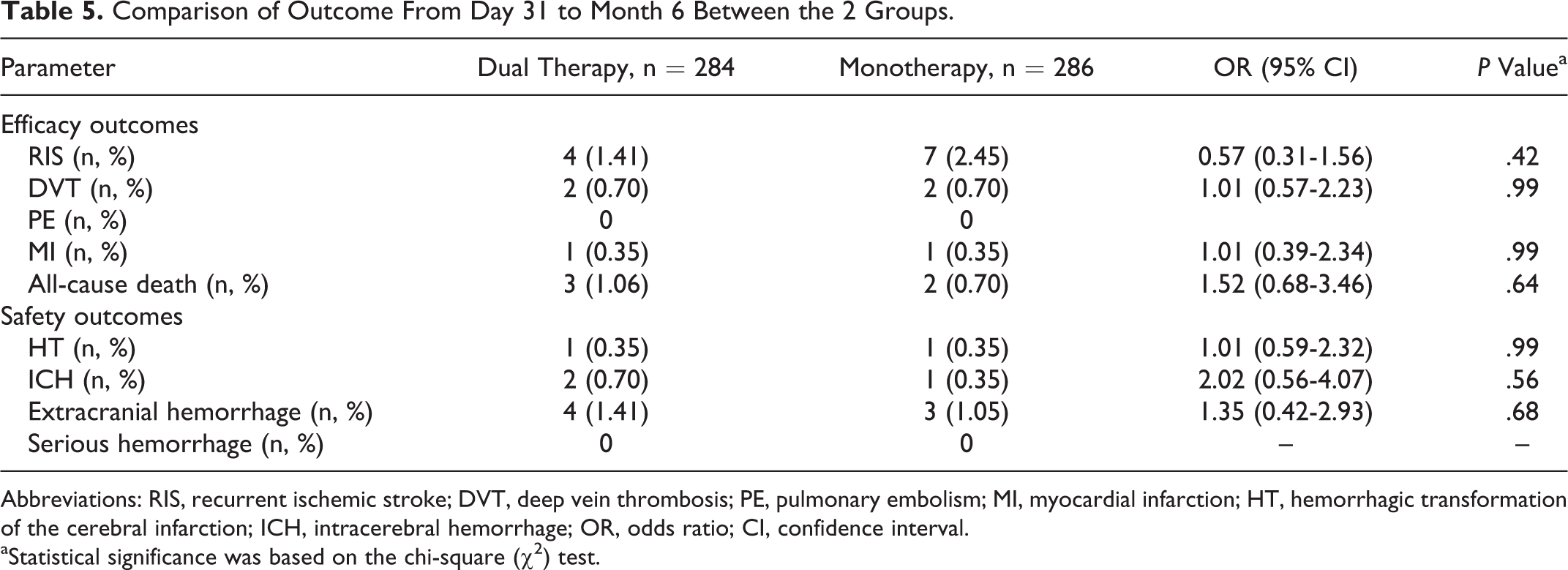
Abbreviations: RIS, recurrent ischemic stroke; DVT, deep vein thrombosis; PE, pulmonary embolism; MI, myocardial infarction; HT, hemorrhagic transformation of the cerebral infarction; ICH, intracerebral hemorrhage; OR, odds ratio; CI, confidence interval.
aStatistical significance was based on the chi-square (χ2) test.
Finally, we evaluated the relationships between treatment efficacy and patient age, sex, NIHSS at baseline, and symptomatic artery (Table 6). In the efficacy outcome analysis at 6 months, there was a significant difference in the frequency of good outcome in patients over the median age of 70 years (64.29% [81 of 126] in the dual therapy group vs 51.16% [66 of 129] in the monotherapy group, P = .038). A significant benefit of dual therapy was also found in patients with symptomatic stenosis in the posterior circulation (frequency of good outcome was 75.0% [51 of 68] in the dual therapy group vs 45.45% [30 of 66] in the monotherapy group, P < .001) and in the basilar artery (77.78% [28 of 36] in the dual therapy group vs 46.88% [15 of 32] in the monotherapy group, P < .001). Our results indicate that there is a beneficial effect of clopidogrel plus aspirin dual therapy in patients older than 70 years and in patients who have stenosis in the posterior circulation or in the basilar artery.
Comparison of mRS in the Dual Therapy Group and Monotherapy Group Within Specified Subgroups at 6 Months.a
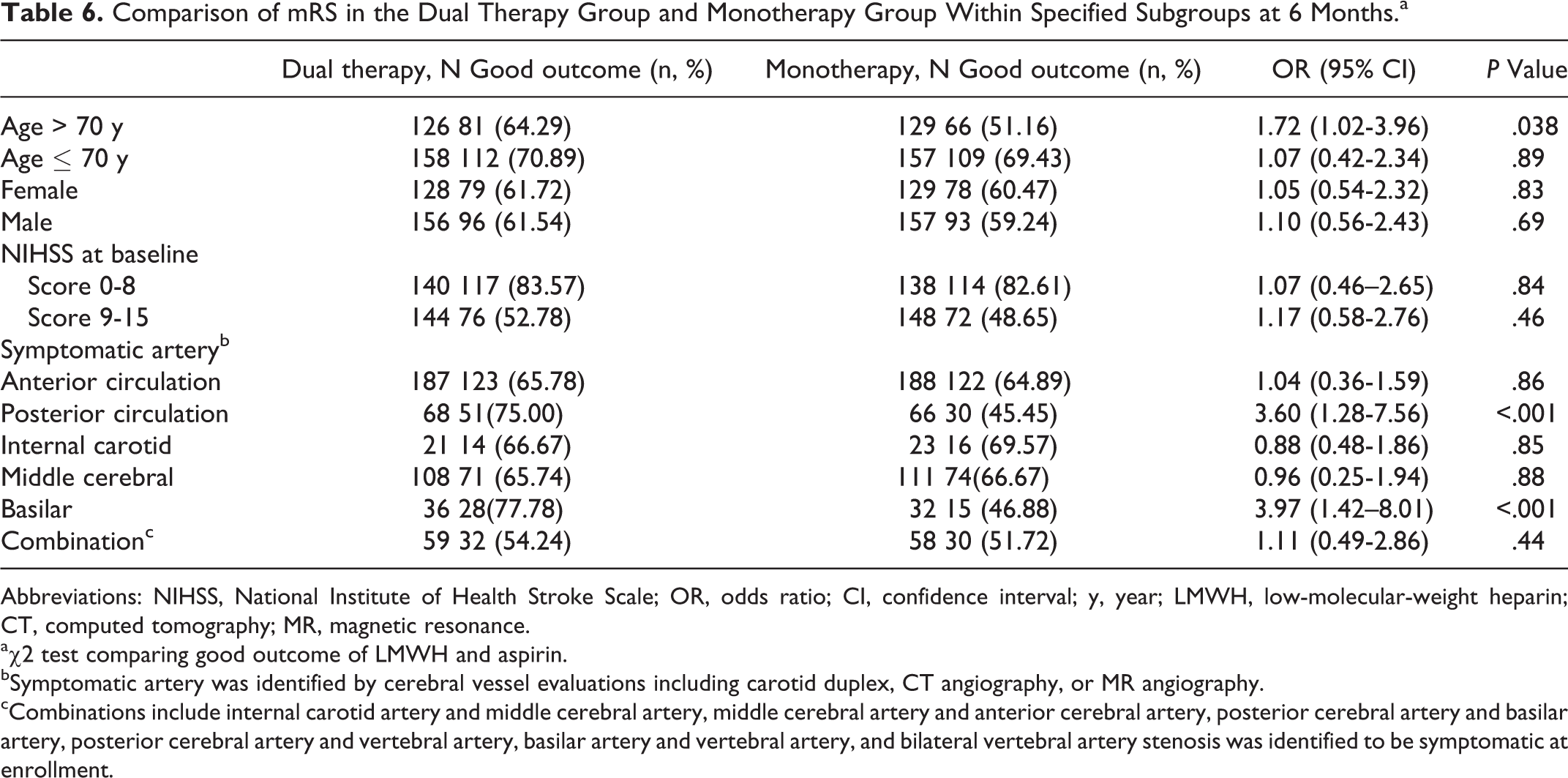
Abbreviations: NIHSS, National Institute of Health Stroke Scale; OR, odds ratio; CI, confidence interval; y, year; LMWH, low-molecular-weight heparin; CT, computed tomography; MR, magnetic resonance.
aχ2 test comparing good outcome of LMWH and aspirin.
bSymptomatic artery was identified by cerebral vessel evaluations including carotid duplex, CT angiography, or MR angiography.
cCombinations include internal carotid artery and middle cerebral artery, middle cerebral artery and anterior cerebral artery, posterior cerebral artery and basilar artery, posterior cerebral artery and vertebral artery, basilar artery and vertebral artery, and bilateral vertebral artery stenosis was identified to be symptomatic at enrollment.
Discussion
Thrombosis and atherosclerosis are major contributors to the development of ischemic stroke. Platelets have a crucial role in triggering arterial thrombosis 35 and in promoting atherogenesis. 36 Platelet activation and platelet–leukocyte aggregates have also been described in patients with stroke. 37,38 However, whether platelet activation and platelet–leukocyte aggregates are involved in the pathogenesis of END and RIS after acute stroke is not known. In the present study, the incidences of AA-induced or ADP-induced platelet aggregation and platelet–leukocyte aggregates on the day of admission were higher in patients who later experienced END or RIS than in patients who did not have these problems. This suggests that high platelet aggregation and platelet–leukocyte aggregates may have a key role in the pathogenesis of END or RIS after acute LAA stroke. Therefore, intensive antiplatelet therapy such as dual antiplatelet therapy may reduce the incidence of END or RIS.
Recurrence results in poor prognosis in patients with ischemic stroke, and antiplatelet therapy has been recommended for the secondary prevention of stroke in patients with a history of noncardioembolic stroke or TIA to reduce nonfatal MI, nonfatal stroke, or vascular death. 39,40 In the present study, we also found that dual antiplatelet therapy was superior to aspirin alone for the prevention of END and RIS. Dual antiplatelet therapy, compared with aspirin alone, was not associated with an increased incidence of hemorrhage. The results of our trial differ from those of other trials of combination therapy with clopidogrel and aspirin after cerebral ischemic events. 17,19,41,42 The possible explanation is that, unlike previous trials, our trial targeted patients after their first acute stroke and who had an NIHSS score less than 15 who may have had a low risk of hemorrhage. Previous trials included patients with more severe strokes than our trial did, and they did not enroll patients in the first hours after an index stroke, during which the risk of END and RIS is particularly high. 43,44 Furthermore, the duration of dual therapy in our trial treatments was 30 days, whereas in previous trials, the treatment period of combination therapy was 18 to 28 months. 17,19 In addition, previous trials included all subtypes of stroke. However, our trial focused on acute LAA stroke. The role of antiplatelet drugs may be different in each subtype of stroke. 40 Several studies have reported that combination therapy with clopidogrel and aspirin is more effective than aspirin alone in reducing microembolic signals and recurrent stroke in patients with predominantly intracranial symptomatic stenosis. 20,21 However, among patients with recent lacunar strokes, the addition of clopidogrel to aspirin therapy did not significantly reduce the risk of recurrent stroke but did significantly increase the risk of bleeding and death. 45
In the present study, platelet aggregation and platelet–leukocyte aggregate formation were not significantly different between the 2 groups at day 1 of treatment. However, at day 30 of treatment, these indicators were lower in patients given dual therapy than in patients who received monotherapy, which indicates that dual therapy can more efficiently inhibit platelet activity. Aspirin and clopidogrel have different biochemical pathways to inhibit platelet adhesiveness. The principal mechanism for the antithrombotic effect of aspirin is inhibition of platelet cyclooxygenase, which decreases the production of thromboxane A2, a potent stimulator of platelet aggregation. The effect of aspirin is more prominent in platelet aggregation induced by AA. The mechanism of clopidogrel is to block ADP to bind to its receptor on platelet to prevent platelet aggregation. 13,14 Thus, decrease in platelet aggregation is more prominent with ADP agonist. This may be a reason that there was no effect of aspirin monotherapy on platelet aggregation induced by ADP (89.0 on day 1 vs70 on day 30), but it had a significant effect on platelet aggregation induced by AA (89.5 on day 1 vs 20.6 on day 30). Therefore, dual therapy with clopidogrel and aspirin may have synergistic effects on platelet aggregation. This may explain why dual therapy is superior to aspirin alone for the prevention of END and RIS.
In the present study, platelet aggregation and platelet–leukocyte aggregate formation were not significantly different between the 2 groups at day 1 of treatment. However, at day 30 of treatment, these indicators were lower in patients given dual therapy than in patients who received monotherapy, which indicates that dual therapy can more efficiently inhibit platelet activity. This may explain why dual therapy is superior to aspirin alone for the prevention of END and RIS.
In our study, there was no significant difference in the frequency of RIS from day 31 to month 6 between the 2 groups. However, for patients over the median age of 70 years, dual therapy was significantly associated with a favorable outcome at 6 months compared with aspirin alone. Age is an independent risk factor for stroke, and the incidence of thrombosis and atherosclerosis increases with increasing age. 46 That may be the reason why dual therapy with clopidogrel and aspirin is more effective than aspirin alone in patients older than 70 years. Age may not only increase the severity of intracranial atherosclerosis but also influence the distribution of occluded vessels. 46 Our results also showed that dual antiplatelet therapy may be more efficacious in patients with posterior circulation stenosis and basilar artery stenosis. The reasons are still unclear. The anterior circulation stroke (ACS) differs from the posterior circulation stroke (PCS) in stroke etiology and stroke mechanisms. Posterior circulation stroke is more often due to atherosclerosis and local branch occlusion, and ACS is more often associated with artery-to-artery embolism and less often associated with local branch occlusion than PCS. 47,48 It may be possible that dual antiplatelet therapies have different efficacies on different involved vessels.
There were no significant differences between the 2 groups with regard to the rates of HT, ICH, or extracranial hemorrhage during the treatment period. No serious hemorrhagic events occurred in the patients overall.
Limitations
Before the proposed drug regime is applied in clinical practice, studies of larger scale are required to validate the advantage of dual antiplatelet therapy in large artery acute ischemic stroke. The results of this 2-center study are from a limited sample size, and therefore, further studies with larger sample sizes and performed across multiple centers are necessary before dual therapy is put into practice. In addition, the beneficial effect of dual therapy on subtypes should be further validated.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
