Abstract
Background:
Branch atheromatous disease (BAD) is a primary cause of early neurological deterioration (END) in penetrating artery occlusion, leading to poor functional outcomes. While it has been proposed to classify BAD under large artery atherosclerosis, uncertainty exists regarding the optimal treatment strategy, including cholesterol-lowering targets.
Objectives:
We aimed to assess the clinical implications and temporal changes of atherosclerotic plaques before and after high-intensity statin treatment.
Design:
This is a high-resolution vessel-wall imaging sub-analysis of the trial of Statin and Dual Antiplatelet Therapy in Preventing Early Neurological Deterioration in Branch Atheromatous Disease (SATBRAD).
Methods:
In this prospective, single-group cohort study, participants in the treatment arm of the SATBRAD trial received early dual antiplatelet therapy and high-intensity statin treatment. The majority of these participants subsequently underwent high-resolution vessel-wall magnetic resonance imaging (MRI). Those with atheromatous plaques in the parent artery continued high-intensity statin treatment for 6 months, followed by a repeat MRI to monitor plaque changes.
Results:
There were 57 patients who underwent vessel-wall imaging and 24 exhibited contrast-enhanced plaques. Patients with contrast-enhanced plaques showed higher rates of END (29.2% vs 6.1%, p = 0.027), perfusion defects (62.5% vs 24.2%, p = 0.004), and lower rates of good outcomes at 3 months (50.0% vs 81.8%, p = 0.011). After adjusting for confounding factors, contrast-enhanced plaque had a negative impact on achieving a good outcome at 3 months (adjusted odds ratio = 0.04; 95% confidence interval = <0.01–0.60). Following high-intensity statin treatment in 36 patients, there was a notable reduction in stenosis (33.7% vs 29.3%, p = 0.005) and contrast-enhanced plaque volume (16.3 vs 11.6 mm3, p = 0.015).
Conclusion:
The study highlighted the association between contrast-enhanced atherosclerotic plaques, END, and poor functional outcomes, with high-intensity treatment leading to plaque volume reduction. These results underscore the shared pathology between BAD and intracranial atherosclerosis, emphasizing the necessity for further research and tailored treatment strategies for BAD.
Trial registration:
ClinicalTrials.gov; Identifier: NCT04824911 (https://clinicaltrials.gov/study/NCT04824911).
Plain language summary
Branch atheromatous disease (BAD) is a major cause of early worsening of stroke symptoms, leading to poor recovery. While some experts believe BAD should be treated like large artery disease, the best treatment approach, including cholesterol-lowering targets, remains unclear. This study aimed to assess how high-intensity statin treatment affects atherosclerotic plaques over time and its impact on patient health. Analyzing detailed vessel images from the SATBRAD trial, where patients received high-intensity statins and magnetic resonance imaging, revealed that 24 out of 57 patients had plaques that showed up clearly with contrast enhancement. These patients were more likely to experience early worsening of stroke symptoms and perfusion compromise and had poorer outcomes. After six months of high-intensity statin treatment, there was a significant reduction in plaque size and vessel narrowing. The study concluded that contrast-enhanced plaques are linked to worse early stroke symptoms and poor recovery, but high-intensity statin treatment can reduce plaque size, suggesting that BAD may share similarities with larger artery disease and highlighting the need for further research and tailored treatments for BAD.
Introduction
Branch atheromatous disease (BAD) has been reported to contribute to small-vessel occlusion in cases of occlusion or stenosis that occur at the origin of large caliber penetrating arteries, due to microatheromas or junctional atherosclerotic plaques. 1 In comparison to other pathologies of lipohyalinosis or cerebral amyloid angiopathy, subcortical infarction from BAD is associated with an increased possibility of early neurological deterioration (END) and recurrent stroke, especially progressive motor deficits.2–4 It has been proposed that BAD should be classified as large artery atherosclerosis rather than small-vessel occlusion.5,6
Dual antiplatelet therapy (DAPT), such as aspirin and clopidogrel, represents the cornerstone of treatment for preventing recurrent strokes in cases of minor acute ischemic stroke.7,8 Several studies have demonstrated the efficacy of DAPT in reducing the risk of END.9,10 Additionally, guidelines recommend the prompt initiation or continuation of statin therapy once oral medications can be safely administered following an acute ischemic stroke. 11 Targeting a low-density lipoprotein cholesterol (LDL-C) level of 70 mg/dL or lower has been associated with a decreased risk of subsequent cardiovascular events compared to levels between 90 and 110 mg/dL, particularly in patients with evidence of atherosclerosis following ischemic stroke or transient ischemic attack. 12 High-intensity statin therapy has shown effectiveness in stabilizing symptomatic intracranial atherosclerotic plaques, as confirmed by high-resolution magnetic resonance imaging (MRI). 13 Plaque morphologies, including irregular surfaces and superior positioning, are predictive of culprit plaques in lenticulostriate artery infarction.
Currently, DAPT and high-intensity statins remain the primary treatments for intracranial atherosclerosis. 14 Given that both BAD and intracranial atherosclerosis share similar pathological mechanisms with atherosclerosis, the Statin and Dual Antiplatelet Therapy in Preventing Early Neurological Deterioration in Branch Atheromatous Disease (SATBRAD) trial aimed to assess the efficacy of early DAPT and high-intensity statin therapy in managing acute small subcortical infarctions caused by BAD. 15 As part of this trial, this study focuses on imaging analysis and aims to explore the clinical implications and temporal evolution of atherosclerotic plaques before and after treatment using high-resolution vessel-wall imaging.
Methods
Study design and population
The SATBRAD trial aimed to compare the efficacy of early intensive treatment for BAD patients initiated within 24 h of stroke onset, using DAPT (aspirin plus clopidogrel) and high-intensity statin treatment, with a historical control group of BAD patients treated with single antiplatelet therapy, without high-intensity statin treatment. The study, conducted at Chang Gung Memorial Hospital in Chiayi, Taiwan since March 2021, is still ongoing, while the sub-analysis focused on vessel-wall imaging has stopped recruiting and is concluding. 15 The study population consisted of patients meeting specific inclusion criteria: (1) have a clinical diagnosis of ischemic stroke with a National Institutes of Health Stroke Scale (NIHSS) score of 1–8; (2) have an ischemic lesion on diffusion-weighted imaging (DWI) located in the striatocapsular territory or brain stem areas, with an axial diameter ⩽20 mm; (3) have BAD, defined by a visible lesion in three or more axial MRI cuts in the lenticulostriate territory or infarcts that extend from the basal surface of the pons. Patients with >50% stenosis of the relevant arteries on time-of-flight (TOF) magnetic resonance angiography (MRA) were excluded.
Trial intervention involves administering dual antiplatelet and high-intensity statin treatment within 24 h of stroke onset. Patients received aspirin and clopidogrel for 21 days, followed by aspirin or clopidogrel alone. High-intensity statin treatment was maintained for 90 days, with adjustments for side effects. All patients were targeted to achieve a blood pressure of 130/80 mmHg. The primary outcome will be the percentage of patients with END within 7 days and recurrent ischemic stroke within 30 days.
This study is a subsequent analysis of vessel-wall imaging for the participants. The patients enrolled in the SATBRAD trial were invited to receive further high-resolution vessel-wall MRIs. Those with atherosclerotic plaques detected in the relevant arteries on initial high-resolution vessel-wall imaging continued high-intensity statin treatment for 6 months and then underwent follow-up MRI.
Clinical information and outcomes
Patient data, age, gender, and medical history such as hypertension, diabetes mellitus, hyperlipidemia, coronary artery disease, atrial fibrillation, and prior cerebrovascular disease, were meticulously documented. Additionally, vital sign measurements, cell counts, blood biochemistry, and coagulation profiles obtained upon admission were diligently recorded. As END in lacunar infarction is mainly associated with motor deficits, END is defined as an NIHSS score ≧4 points within 7 days of stroke onset. 16 Clinical outcomes at admission and at 90 days were evaluated using the modified Rankin scale (mRS). A good outcome is defined as an mRS score ≦2.
MRI protocol
The MRI scans were conducted utilizing a 3 Tesla Siemens Verio MRI system (Siemens Medical System, Erlangen, Germany) equipped with a 32-channel head coil. Various sequences were employed, including axial DWI, axial T1- and T2-weighted images, three-dimensional (3D) TOF angiography, dynamic susceptibility contrast perfusion imaging, and high-resolution vessel-wall imaging for both initial and follow-up assessments. The detailed MRI protocols, excluding vessel-wall imaging, were consistent with those used in our previous study. 17 Black-blood high-resolution vessel imaging using the spatial pre-saturation technique was performed as follows: (1) axial and sagittal T2-weighted images (repetition time (TR)/echo time (TE) = 3200/323 ms, echo train length = 185, slice thickness = 6 mm, flip angle = 180°, matrix = 320 × 266, field-of-view = 149.6 mm, number of excitations = 3.4); (2) axial and sagittal T1 fluid-attenuated inversion recovery, pre-contrast and post-contrast (TR/TE = 700/22 ms, echo train length = 55, slice thickness = 6 mm, flip angle = 180°, matrix = 320 × 320, field-of-view = 190 mm, number of excitations = 3.4). The stroke neurologist (YCH) selected the vessel and site of evaluation based on clinical presentation, infarct site in DWI and 3D TOF MRA.
Image analysis
The location of the cross-section measurement was selected based on the luminal image with TOF MRA, focusing on the area with maximal stenosis. Sagittal images of the middle cerebral artery (MCA) and axial images of the basilar artery, depicting the vertical orientation of blood vessels, were utilized for image analysis.
MIStar (Apollo Medical Imaging Technology, Melbourne, Australia) was utilized to produce perfusion imaging for each patient, generating quantitative cerebral blood flow (CBF), cerebral blood volume, and mean transit time (MTT) maps. The location of the perfusion defect was visually identified using the MTT and CBF maps, with reference to the site of the acute infarction. This approach was employed in our previous study. 17 Patients exhibiting increased MTT or decreased CBF were categorized as having perfusion defects.
For the analysis of vessel-wall imaging, we determined the location for cross-section measurement by identifying the luminal image exhibiting maximal stenosis, using pre-contrast T1 vessel-wall imaging with reference to post-contrast T1 imaging. Additionally, we reconstructed sagittal images oriented vertically to the MCA or axial images oriented vertically to the basilar artery during the image analysis process. Furthermore, we assessed normal vessels adjacent or contralateral to the stenotic area as reference values. To quantify the degree of stenosis, we employed the formula: (1 − lumen area of stenotic lesion/reference lumen area) × 100%. Furthermore, we compared pre-contrast and post-contrast T1 fluid-attenuated inversion recovery images to discern the presence or absence of enhancement and determine the volume of enhancement. The change in stenosis was measured at the same site that exhibited the maximal stenosis in the initial imaging. When analyzing the follow-up imaging, the readers referred to the initial imaging to identify the sites with the greatest stenosis and the locations of enhanced plaques. The plaque location was categorized into three regions: superior, inferior, and lateral. The superior portion was defined as the adjacent area to MCA territory or brainstem, while the inferior portion indicated the corresponding area relative to the superior portion (Figure 1).18,19 Because the plaque in BAD is relatively minor, it can predominantly be identified in one quadrant but may extend to involve two or more quadrants. In such situations, the plaque was classified into the quadrant with the greater plaque burden. The stroke neurologist (YCH) and neuroradiologist (YHT) were responsible for the analysis and the final result depends on the consensus of the two experts.

Illustration of plaque location. (a) The cross-section of the MCA (marked by the red circle) reveals its superior portion, corresponding to the area adjacent to the MCA territory, while the inferior portion indicates the opposite area in relation to the superior portion. (b) In the case of the basilar artery (marked by the red circle), its cross-section illustrates the superior portion, indicating the area adjacent to the brainstem, while the inferior portion denotes the opposite area relative to the superior portion.
The study was approved by the Institutional Review Board of Chang Gung Memorial Hospital (Approval No. 202001386A3) and registered on ClinicalTrials.gov (Identifier: NCT04824911). All participants provided written informed consent before taking part in the study. This manuscript was prepared following the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. A completed STROBE checklist is provided as a Supplemental File.
Statistical analysis
Statistical analyses were conducted using the Statistical Package for the Social Sciences (SPSS) software (version 27, Chicago, IL, USA). The normality of continuous variables was assessed using the Kolmogorov–Smirnov test. Differences between the two groups were examined using the Mann–Whitney U test and Student’s t-test, as appropriate. Categorical data were analyzed using the Chi-square test. Univariable logistic regression models were used to evaluate candidate variables for END and functional outcomes. Subsequently, we constructed a multivariable regression model incorporating all potential predictors through forward stepwise selection with a significance criterion of p < 0.1. Evaluation of changes in vessel stenosis and plaque volumes was performed using the paired-samples t-test. The intraclass correlation coefficient was used to evaluate the inter-rater reliability of two readers. All statistical tests were two-tailed, and a significance level of p < 0.05 was considered indicative of statistical significance.
Results
The patient selection flow chart is depicted in Supplemental Figure S1. The intervention group comprised 82 patients, while the historical control group consisted of 306 patients. Among the 82 patients in the intervention group, 7 were excluded due to renal insufficiency, and an additional 18 refused contrast medium injection, leaving 57 who underwent high-resolution vessel-wall imaging. Thirty-six of these patients diagnosed with atherosclerotic plaque in the parent arteries subsequently underwent 6 months of high-intensity statin treatment, followed by a follow-up MRI. The illustration of patients with acute ischemic stroke and vessel-wall imaging is presented in Figure 2.

Illustration of patients with acute ischemic stroke and vessel-wall imaging. (a) A patient presented with acute infarction in the left pons (arrow) on DWI, exhibiting decreased CBF (arrowhead), and a stenotic basilar artery on TOF angiography (empty arrow). (b) Initial vessel-wall imaging revealed a cross-section of the basilar artery (yellow circle) at the level of the acute infarction, where an atherosclerotic plaque was identified at the superior portion (arrow) with contrast enhancement (red circle, arrowhead). (c) Follow-up vessel-wall imaging at 6 months showed regression of the atherosclerotic plaque (arrow) and contrast-enhanced volume (red circle, arrowhead). (d) A patient presented with acute infarction in the right basal ganglion (arrow) on DWI, displaying decreased CBF (arrowhead), and a normal MCA on TOF angiography (empty arrow). (e) Initial vessel-wall imaging revealed a cross-section of the MCA (yellow circle) at the level of the acute infarction, where an atherosclerotic plaque was identified at the superior portion (arrow) with contrast enhancement (arrowhead). (f) Follow-up vessel-wall imaging at 6 months showed regression of the atherosclerotic plaque (arrow) and contrast-enhanced volume (arrowhead).
Fifty-seven patients in the intervention group underwent high-resolution vessel-wall imaging, with 42 of them diagnosed with atherosclerotic plaques, and 24 of these exhibited contrast-enhanced atherosclerotic plaques. Among patients with or without atherosclerotic plaques (Supplemental Table S1), those with plaques were older (70.1% vs 62.7%, p = 0.005) and had higher LDL-C (141 vs 119 mg/dL, p = 0.011). However, there were no significant differences in stroke factors, initial stroke severity, END rates, or outcomes between the two groups. Patients with contrast-enhanced atherosclerotic plaques had higher systolic blood pressure (183.0 vs 165.4 mmHg, p = 0.013), a higher rate of END (29.2% vs 6.1%, p = 0.027), a higher rate of perfusion defects (62.5% vs 24.2%, p = 0.004), and a lower rate of good outcome at 3 months (50.0% vs 81.8%, p = 0.011) (Table 1). After adjusting for confounding factors using multivariate regression analysis, we found that acute infarction in the brainstem (adjusted odds ratio (aOR) = 7.63; 95% confidence interval (CI) = 1.27–45.7) and visible perfusion defects (aOR = 8.55; 95% CI = 1.40–52.1) were associated with the development of END (Table 2). Additionally, patients with brainstem infarction (aOR = 0.03; 95% CI = <0.01–0.54), plaque located superiorly (compared to inferior location; aOR = 0.01; 95% CI = <0.01–0.38), and contrast-enhanced plaque (aOR = 0.04; 95% CI = <0.01–0.60) had a negative impact on achieving a good outcome at 3 months (Table 3).
Baseline characteristics and outcomes in patients with or without contrast-enhanced plaques.
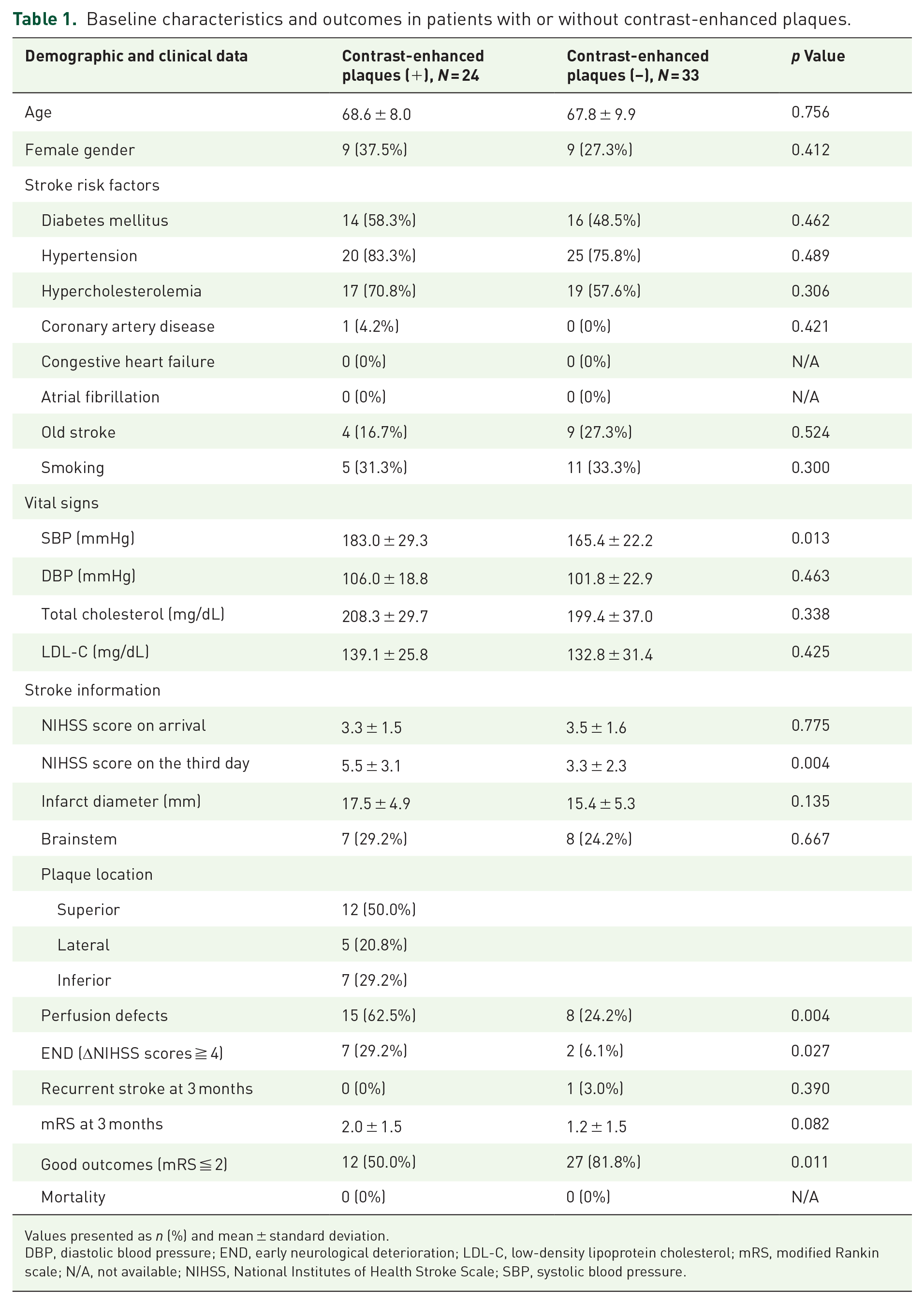
Values presented as n (%) and mean ± standard deviation.
DBP, diastolic blood pressure; END, early neurological deterioration; LDL-C, low-density lipoprotein cholesterol; mRS, modified Rankin scale; N/A, not available; NIHSS, National Institutes of Health Stroke Scale; SBP, systolic blood pressure.
Predictive variables for early neurological deterioration.

With reference to inferior location.
CI, confidence interval; LDL-C, low-density lipoprotein cholesterol; NIHSS, National Institutes of Health Stroke Scale.
Predictive variables for good outcome with mRS ≦ 2 at 3 months.

With reference to inferior location.
CI, confidence interval; LDL-C, low-density lipoprotein cholesterol; mRS, modified Rankin scale; NIHSS, National Institutes of Health Stroke Scale.
In the 42 patients with atherosclerotic plaques, 36 received high-intensity statin treatment for 6 months and underwent follow-up MRI, with 27 having plaques in the MCA and 9 in the basilar artery. Twenty-eight patients took rosuvastatin, and eight took atorvastatin. Perfusion defects were found in 16 (44.4%) patients, and END was observed in 7 (29.2%) patients (Supplemental Table S2). After treatment, there were statistically significant decreases in triglyceride levels (126.3 vs 101.7 mg/dL, p < 0.001), total cholesterol levels (211.7 vs 133.7 mg/dL, p < 0.001), LDL-C (142.5 vs 68.6 mg/dL, p < 0.001), and HbA1c (7.0% vs 6.3%, p = 0.018) (Table 4). The intraclass correlation coefficient for the stenosis measurements between the two readers was 0.783, and for repeated measurements of plaque volume, it was 0.830, indicating good inter-rater and intra-rater reliability. The degree of stenosis (33.7% vs 29.3%, p = 0.005) and the volume of contrast-enhanced plaque (16.3 vs 11.6 mm3, p = 0.015) were significantly decreased.
Laboratory and MRI changes after statin treatment.

Values are presented as n (%) and mean ± standard deviation.
HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; MRI, magnetic resonance imaging.
Discussion
The sub-analysis of the SATBRAD trial, utilizing vessel-wall MRI findings, revealed that contrast-enhanced atherosclerotic plaques were associated with perfusion defects in the perforators, END, and poor functional outcomes. Conversely, the administration of high-intensity statin treatment reduced contrast-enhanced plaque volume and alleviated vessel stenosis. These findings underscore the parallel pathology and clinical relevance shared between BAD and intracranial atherosclerosis, emphasizing the need for additional research and a tailored treatment approach for BAD.
Recent advancements in imaging have elucidated BAD’s association with atherosclerotic plaques obstructing penetrator orifices or proximal penetrators.1,20 Using high-resolution vessel-wall imaging, the junctional plaque at the parent arteries can be visualized and characterized, making it the most well-recognized diagnostic modality for BAD.19,21 While some cases of BAD may stem from microatheroma at the proximal penetrators, studies employing ultrahigh-field TOF angiography or vessel-wall imaging have been utilized to detect the occlusion of lenticulostriate arteries.22,23 However, current methods are still unable to differentiate the occlusion pathology due to microatheroma, lipohyalinosis, or embolism, nor determine the cut point of the length of lenticulostriate arteries for diagnosing BAD. Our study, consistent with previous research, demonstrates that high-resolution vessel-wall imaging of atherosclerotic plaques in the parent arteries holds clinical significance in the diagnosis of BAD.
High-resolution MRI has been instrumental in evaluating the morphological characteristics of atherosclerotic plaques in intracranial atherosclerosis in previous studies. Notably, specific features such as positive remodeling, ulcerative plaques, and contrast enhancement have displayed significant correlations with ischemic stroke.24,25 Contrast-enhanced plaques, indicative of neovascularization, inflammation, and endothelial dysfunction, 26 signify unstable plaques with heightened ischemic stroke risk and downstream hypoperfusion leading to recurrent stroke.27–30 Importantly, our study has unveiled a correlation between contrast-enhanced atherosclerotic plaques, perfusion defects in the perforators, and END, resulting in diminished prospects for functional recovery in BAD. Furthermore, these contrast-enhanced plaques exhibited regression following high-intensity statin therapy, a phenomenon observed in patients with intracranial atherosclerosis. 13 Given that contrast-enhanced plaques signify vulnerable and unstable lesions associated with an increased risk of ischemic stroke events, our findings lend support to the use of high-intensity statins and a targeted reduction in LDL-C levels in treating BAD, mirroring the approach taken for intracranial atherosclerosis. 31
We have also discovered that plaques situated superiorly, as opposed to those in inferior locations, had a detrimental effect on achieving favorable outcomes at 3 months. This observation aligns with prior research indicating that plaque morphologies characterized by irregular surfaces and superior positioning are predictive of culprit plaques in subcortical infarctions resulting from lenticulostriate artery obstruction.19,32,33 It is not unexpected, given that the majority of perforators originate from the superior portion of the MCA and the superior and lateral portions of the basilar artery.34–36 Consequently, plaques positioned superiorly are more likely to block these nearby perforators, leading to ischemic stroke and impaired functional recovery.
In this trial, BAD is defined as a visible lesion in three or more axial MRI slices within the lenticulostriate territory or infarctions extending from the basal pons surface. 1 Although high-resolution vessel-wall MRIs offer direct atherosclerotic plaque detection along parent artery perforators, their detection rate varied from 47% to 74%, as observed in previous studies and our trial.21,37–39 The reasons for this may stem from limitations in spatial resolution, particularly for relatively small junctional atherosclerotic plaques. Additionally, in some cases, microatheromas in proximal penetrating arteries may remain undetectable without the use of advanced sequences.22,23 Thus, prior to the widespread adoption of high-field Tesla MRI with short-duration sequences for vessel-wall imaging, morphological definitions for BAD, such as longitudinal diameter from the proximal MCA or pontine infarction from the ventral surface, offer a straightforward and practical approach in clinical practice.
Several methodological limitations warrant consideration. Firstly, this study was conducted at a single center and involved a relatively small sample size consisting solely of individuals from Taiwan. It is essential to acknowledge that genetic variations may influence patient responses to statin therapy. Secondly, it is important to recognize that vessels in the anterior and posterior circulations possess inherent and distinct histopathological properties. However, due to the limited sample size, we combined these vessels for analysis. Thirdly, while the severity of stenosis and plaque volume was relatively mild compared to intracranial stenosis, we acknowledge the potential of rater-dependent variability. Fourthly, as this was a single-arm study lacking a parallel control group undergoing vessel-wall imaging, it could not determine each factor’s natural progression of atherosclerotic plaques following acute ischemic stroke under standard statin therapy, nor evaluate the influence of blood pressure control or other medications such as antiplatelets, antihypertensives, and hypoglycemic agents. These factors could potentially impact the progression of atherosclerotic plaques, highlighting the need for further investigation in large-scale studies. Lastly, the duration of 6 months may not have been sufficient to adequately assess the morphological changes in atherosclerotic plaques. Moreover, while plaque regression occurred, it is important to note that these preliminary results do not necessarily guarantee clinical benefit. Therefore, further investigation in larger clinical studies is needed to validate these findings comprehensively.
Conclusion
In conclusion, findings from vessel-wall MRI revealed that contrast-enhanced atherosclerotic plaques were associated with perfusion defects in the perforators, END, and poor functional outcomes. Moreover, high-intensity treatment led to a reduction in plaque volume. Further clinical trials are warranted to assess the clinical benefits of high-statin treatment in BAD.
Supplemental Material
sj-docx-1-tan-10.1177_17562864241273902 – Supplemental material for Preliminary results on temporal evolution and clinical implications of atherosclerotic plaque in branch atheromatous disease after statin treatment
Supplemental material, sj-docx-1-tan-10.1177_17562864241273902 for Preliminary results on temporal evolution and clinical implications of atherosclerotic plaque in branch atheromatous disease after statin treatment by Yen-Chu Huang, Yuan-Hsiung Tsai, Leng-Chieh Lin, Hsu-Huei Weng, Jiann-Der Lee and Jen-Tsung Yang in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-2-tan-10.1177_17562864241273902 – Supplemental material for Preliminary results on temporal evolution and clinical implications of atherosclerotic plaque in branch atheromatous disease after statin treatment
Supplemental material, sj-docx-2-tan-10.1177_17562864241273902 for Preliminary results on temporal evolution and clinical implications of atherosclerotic plaque in branch atheromatous disease after statin treatment by Yen-Chu Huang, Yuan-Hsiung Tsai, Leng-Chieh Lin, Hsu-Huei Weng, Jiann-Der Lee and Jen-Tsung Yang in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
