Abstract
Background:
Dual antiplatelet therapy (DAPT) is a cornerstone of secondary prevention in patients with minor ischemic stroke or high-risk transient ischemic attack. The effectiveness and safety of DAPT may differ between patients with posterior (PCI) and anterior circulation infarct (ACI).
Objectives:
We aimed to compare short-term outcomes following DAPT between mild-to-moderate stroke patients with PCI versus ACI.
Design:
Propensity-matched analysis from a prospective real-world multicentric cohort study (READAPT).
Methods:
We included patients with noncardioembolic mild-to-moderate stroke (National Institute of Health Stroke Scale of 0–10) who initiated DAPT within 48 h of symptom onset. Patients were categorized into ACI or PCI based on the infarct(s) location on brain neuroimaging. The primary effectiveness outcome was the 90-day risk of ischemic stroke or other vascular events. The secondary effectiveness outcomes were the 90-day modified Rankin Scale (mRS) score distribution, 24-h early neurological improvement or deterioration, and all-cause mortality. The safety outcomes included the 90-day risk of any bleedings and 24-h hemorrhagic transformation.
Results:
We matched 281 PCI patients with 651 ACI patients. The 90-day risk of ischemic stroke or other vascular events was low and similar between PCI and ACI groups (3.1% vs 2.9%, respectively; hazard ratio 0.98, (95% confidence interval (CI) 0.45–2.14); p = 0.845). Patients with PCI had worse 90-day mRS ordinal distribution compared to those with ACI (odds ratio 1.18 (95% CI 1.01–1.39); p = 0.046). There were no differences in other secondary outcomes. Safety outcomes had low incidence and did not differ between groups (any bleedings: 3.2% vs 2.6%; 24-h hemorrhagic transformation: 1.8% vs 1.2%). We found no differences in the risk of ischemic stroke or other vascular events between patients with PCI and ACI across subgroups defined by sex, age, presumed stroke etiology, stroke severity, prestroke mRS, hypertension, diabetes, acute reperfusion therapies, DAPT loading dose, or presence of symptomatic intracranial stenosis.
Conclusion:
Our findings suggest that effectiveness and safety outcomes after DAPT in patients with mild-to-moderate noncardioembolic ischemic stroke are consistent regardless of infarct location in the anterior or posterior circulation territory. However, patients with PCI may experience worse short-term functional outcome.
Trial registration:
URL: www.clinicaltrials.gov; Unique identifier: NCT05476081.
Keywords
Introduction
While most ischemic strokes occurs in the anterior circulation, approximately 20% involve the posterior circulation, resulting from reduced blood flow to the brainstem, cerebellum, thalamus, and parietooccipital lobes. 1 These two subtypes exhibit some differences in risk factors, etiology, prognosis, and therapeutic approaches.2,3 Anterior circulation infarctions (ACI) are more commonly associated with embolism, 4 whereas posterior circulation infarctions (PCI) are more frequently lacunar. 5 Diagnostically, PCI pose greater challenges due to their nonspecific symptoms, which can lead to delayed recognition and treatment. 6 Moreover, they are associated with a higher risk of stroke recurrences and early neurological deterioration, 7 particularly in cases of large artery stenosis, 8 and often result in worse functional outcomes compared to ACI.9,10
Current international guidelines recommend early short-term dual antiplatelet therapy (DAPT) for all patients with minor ischemic stroke or high-risk transient ischemic attack (TIA).11,12 Although pivotal DAPT trials13–15 included patients regardless of the site of the acute infarct, they did not specifically analyze outcomes based on anterior or posterior circulation involvement. More recently, a secondary analysis from the ticagrelor or clopidogrel with aspirin in high-risk patients with acute nondisabling cerebrovascular events 2 (CHANCE-2) trial found similar 90-day stroke recurrence rates between PCI and ACI patients with minor stroke or TIA who were treated with aspirin plus clopidogrel or ticagrelor. 16 Additionally, a post hoc analysis of the antiplatelet therapy in acute mild-to-moderate ischemic stroke (ATAMIS) trial revealed that, compared to mild-to-moderate stroke patients (baseline National Institute of Health Stroke Scale (NIHSS) of 0–10) with PCI, those with anterior circulation stroke experienced greater reduction in early neurological with DAPT than with aspirin alone. 17 In the real-world settings, the use of DAPT often extends beyond the indications of pivotal trials, potentially offering benefits to patients with greater stroke severity and those who have undergone acute reperfusion therapies.18–20 Therefore, it is important to clarify whether outcomes following DAPT differ across diverse clinical scenarios and specific patient populations, such as those with PCI.
This study aimed to explore differences in short-term outcomes following DAPT in patients with noncardioembolic mild-to-moderate ischemic stroke, comparing those with PCI versus ACI.
Methods
READAPT study
The READAPT study (NCT05476081) is a prospective, observational, multicenter investigation conducted within real-world clinical settings in Italy. Coordinated by the University of L’Aquila and supported by the Italian Stroke Association, it adhered to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for observational research. 21 The study enrolled adults aged 18 and above who had suffered an acute noncardioembolic ischemic stroke or a TIA and were prescribed DAPT for a duration of up to 90 days. The READAPT study was conducted between February 1, 2021, and February 28, 2023, and spanned 64 stroke centers nationwide. Participant recruitment occurred in hospital settings, emergency departments, and open-access neurology clinics. To maximize enrollment, investigators followed structured protocols and issued regular follow-up reminders. Diagnosis of stroke and TIA was established based on standard clinical assessments and the World Health Organization’s criteria. All patients initiating DAPT following an acute noncardioembolic ischemic event were eligible, with no constraints regarding the choice of antiplatelet agents or the timing of DAPT initiation. The decision to prescribe DAPT was left to the discretion of the treating physician, who determined suitability based on individual risk–benefit considerations. Patients were included irrespective of whether they received a loading dose of DAPT (defined as ⩾300 mg for aspirin and clopidogrel or ⩾180 mg for ticagrelor). The exclusion criteria were minimal, disqualifying only patients undergoing prolonged DAPT due to stent-related procedures, and those unable to adhere to study requirements. Participants were enrolled shortly after the qualifying ischemic event and monitored over a follow-up period of 90 ± 10 days through in-person or remote assessments. Methodological details and study definitions have been documented in prior publications.18,19
Study analysis
For this secondary analysis, we selected only patients who experienced mild-to-moderate ischemic strokes (defined by a NIHSS score of 0–10 on admission) and had ⩾1 brain infarct detected on admission or follow-up neuroimaging congruous with their stroke symptoms at onset. Selection was limited to those who started DAPT within 48 h of symptom onset. In contrast, we excluded patients who were prescribed DAPT after carotid endarterectomy or stenting, as well as those who discontinued DAPT prematurely due to the initiation of anticoagulation therapy. These exclusions aimed to reduce potential confounding from procedure-related variables and anticoagulation-associated bleeding risks.
Patients were categorized as having either PCI or ACI based on the acute infarct(s) location. Infarct locations were determined according to the site of hyperintense lesions on the diffusion-weighted imaging (DW) sequences in patients who underwent brain magnetic resonance (MR), or on the site of new hypodense lesion(s) in patients who underwent only brain noncontrast computed tomography (NCCT). Similarly, the number of acute ischemic lesion(s) was assessed using DW sequences on brain MR when available or otherwise using brain NCCT. ACI was defined as the presence of acute infarct(s) in territories perfused by the anterior cerebral artery, middle cerebral artery, or internal carotid artery, while PCI was defined as lesion(s) occurring in brain regions supplied by the posterior cerebral artery, cerebellar arteries, basilar artery, or vertebral artery. Patients with multiple acute infarcts involving both anterior and posterior circulation territories were categorized as PCI only if they presented exclusively with symptoms attributable to the involvement of posterior circulation territories—namely, isolated hemianopsia, ataxia, vertigo, diplopia, typical brainstem syndromes—otherwise, they were categorized as ACI. This symptom-based criterion was adopted to ensure a clinically meaningful classification, in patients where determining the predominant infarct territory based on imaging alone was challenging. Of note, although the presence of lesions in both vascular territories may raise suspicion of a cardioembolic source, we included these patients in the main analysis, as individuals with a confirmed cardioembolic etiology were excluded in accordance with the original READAPT exclusion criteria.18,19 Thus, even if some cases may theoretically reflect a cardioembolic mechanism, their inclusion remains consistent with current guideline recommendations for DAPT in patients without confirmed cardioembolism.11,12 Conversely, isolated watershed infarcts in territories between the anterior and posterior circulation territories were excluded. The location of the acute infarct(s) was adjudicated by local senior neuroradiologists, who were provided with prespecified criteria. Neuroimaging investigations, including the choice of imaging modality, were conducted at the discretion of the treating physician. However, all patients included in the READAPT study underwent at least two brain imaging: one on admission and another after 24 h.
Propensity score matching
We calculated propensity scores for each patient using a multivariable logistic regression model that included prespecified variables potentially acting as confounders when comparing PCI and ACI groups: demographics (age, ethnicity), risk factors (arterial hypertension, diabetes, dyslipidemia, and smoking status), prestroke functional status (modified Rankin Scale, mRS), stroke severity (NIHSS score on admission), symptoms duration (<24 or ⩾24 h), presumed stroke cause, acute stroke treatment (intravenous thrombolysis, endovascular thrombectomy), DAPT characteristics (loading dose, days of DAPT duration), type of brain neuroimaging (brain MRI performed or not performed), and infarcts in the anterior and posterior circulation territories. PCI and ACI patients were matched in a 1:3 ratio, within 0.1 standard deviations of the logit of the propensity score, using greedy nearest neighbor matching. The quality of the matching was assessed by comparing the standardized difference (SD) of means and the ratio of variances between the propensity scores of treatment groups, alongside inspecting graphical distributions of propensity scores and covariates balance in the matched cohort.
Study outcomes
The primary effectiveness outcome was the risk of new ischemic stroke or other vascular events within 90 days, including TIA, myocardial infarction, or death from vascular causes (stroke, systemic hemorrhage, myocardial infarction, congestive heart failure, pulmonary embolism, sudden death, or arrhythmias). Secondary effectiveness outcomes encompassed the 90-day ordinal distribution of the mRS scores and all-cause mortality within the same period, 24-h early neurological improvement and deterioration (⩾2 points decrease or increase in the NIHSS score from baseline, respectively). Safety outcomes included 24-h hemorrhagic transformation, classified according to the Heidelberg bleeding classification system, 22 and the 90-day incidence of any bleeding event, categorized based on the Global Utilization of Streptokinase and Tissue Plasminogen Activator for Occluded Coronary Arteries (GUSTO) classification. 23 Specifically, severe bleeding was defined as fatal hemorrhage, intracranial hemorrhage, or hemorrhage requiring blood or fluid replacement, inotropic support, or surgical intervention. Moderate bleeding referred to events necessitating a transfusion but not posing an immediate life-threatening risk. All other bleeding events were classified as minor. 23
Statistical analysis
Categorical variables were reported as number and percentage, continuous variables as median and interquartile range (IQR). For the primary effectiveness outcomes, the time-to-event 90-day risk of ischemic stroke and other vascular events was compared using Cox regression and the corresponding treatment effect was presented as hazard ratio (HR) and 95% confidence intervals (CIs). Kaplan–Meier hazard functions were used to compare the overall risk of primary effectiveness outcome between patients with PCI and ACI and log-rank test was used to test differences. Furthermore, we conducted a sensitivity analysis excluding patients with multiple infarcts involving both anterior and posterior circulation territories to ensure the robustness of the primary effectiveness outcome in the matched cohorts. For the secondary effectiveness outcomes, the 90-day shift of mRS scores was compared using an ordinal generalized linear model (GLM), and results were presented as odds ratio (OR) with 95% CIs. Furthermore, we calculated risk differences (and 95% CIs) between groups for the rate of 24-h neurological improvement and deterioration. Cox regression was used to compare the 90-day risk of all-cause mortality. For the safety outcomes, we calculated risk differences and 95% CIs between patients with PCI and ACI. All outcomes analyses were performed also in the unmatched cohort. Subgroup analysis of the primary effectiveness outcome in the matched cohorts was performed by calculating risk differences between infarct location in 10 prespecified subgroups (sex (males or females), age (<65 or ⩾65 years), NIHSS score on admission (0–5 or 6–10), presumed stroke cause, pre-stroke mRS (<2 or ⩾2), history of hypertension and diabetes, acute reperfusion therapies, DAPT loading dose, presence of symptomatic intracranial stenosis). Lastly, we used a GLM model with infarct location (PCI vs ACI), subgroup variable, and their interaction term as independent variables to assess the homogeneity in the effect of infarct location by each subgroup variable. The p value was presented for the interaction term. Since this was an exploratory analysis within an observational real-world study, no predetermined sample size was calculated a priori. All statistical analyses were performed using R software, version 4.2 (R Foundation for Statistical Computing, Vienna, Austria). Statistical significance was set at a p value <0.05.
Results
Overall, the READAPT study cohort included 2278 patients. Our study analysis included 1269 patients (median age 70, IQR 60–77), of whom 65.7% were males. The number of patients excluded with reasons are reported in the study flow-chart (Figure 1). A total of 286 patients (22.5%) were classified as PCI, while the remaining 983 patients (77.5%) were classified as ACI. Considering both admission and 24-h follow-up neuroimaging, brain MR was performed in 1019 patients (80.3%), while 250 patients (19.7%) underwent only brain NCCT. All patients underwent brain vessel imaging, using CT angiography (70.5%), MR angiography (38.8%), Doppler ultrasonography (13.4%), or digital subtraction angiography (4.6%).

Study flow-chart.
We matched 281 PCI patients with 651 ACI patients. No missing data were present in the variables used for propensity score calculation or in any baseline variable within both the unmatched and matched cohorts. Balance diagnostics confirmed that the samples were well-matched, with a standardized mean difference of propensity scores between groups at 0.02 (good balance <0.25) 24 and a propensity score variance ratio of 1.02 (good balance range: 0.5–2). 24 Graphical analyses of propensity scores and covariate balance distributions further supported the high quality of the matching (Supplemental Figures 1 and 2). Furthermore, comparisons of baseline characteristics confirmed the strong balance of the matched cohorts, with SDs of propensity scores remaining below 0.20 for all variables, except for a higher prevalence of vertebrobasilar artery stenosis among patients with PCI (30.6% vs 6.3%; SD = 0.660) and a higher frequency of extracranial internal carotid artery stenosis among patients with ACI (17.7% vs 8.6%; SD = 0.273; Table 1).
Baseline characteristics of the unmatched and matched cohorts.

Standardized differences >0.20 are reported in bold.
A standardized difference (of means) <0.20 indicates that groups are well balanced.
Presumed stroke cause was classified according to the TOAST classification system.
Arterial hypertension was defined as a history of blood pressure >140/90 mmHg or the current use of antihypertensive medications.
Dyslipidemia was defined as history of total blood cholesterol levels >220 mg/dL and/or total triglycerides levels >130 mg/dL and/or current used lipid-lowering drugs.
Diabetes mellitus was defined as history of fasting glucose >126 mg/dL or the current use of hypoglycemic medications.
Current smoking was defined as the consumption of ⩾1 cigarette per day over the last year.
Extracranial internal carotid artery stenosis was defined as 50%–99% stenosis or occlusion of the extracranial internal carotid artery, according to the ECST criteria.
Vertebrobasilar artery stenosis was defined as 50%–99% stenosis or occlusion of the vertebral or basilar artery.
Symptomatic intracranial arterial stenosis was defined as 50%–99% stenosis or occlusion of the artery supplying the infarcted territory.
ACI, anterior circulation infarct; CTA, computed tomography angiography; DAPT, dual antiplatelet therapy; ECST, European Carotid Surgery Trial stenosis; IQR, interquartile range; MR, magnetic resonance imaging; MRA, magnetic resonance angiography; mRS, modified Rankin Scale; NIHSS, National Institute of Health Stroke Scale; PCI, posterior circulation infarct; TIA, transient ischemic attack; TOAST, Trial of Org 10172 in the Acute Stroke Treatment; US, ultrasonography.
Primary effectiveness outcome
The rate of 90-day new ischemic stroke or other vascular events was low and similar between patients with PCI and ACI (3.1% vs 2.9%, respectively; HR 0.98 (95% CI 0.45–2.14); p = 0.845; Table 2). Kaplan–Meier hazard functions showed no significant differences in the 90-day risk of ischemic stroke or other vascular events (long-rank test p-value: 0.850; Figure 2). Results of the primary effectiveness outcome analysis in the unmatched cohorts are reported in Supplemental Table 1.
Outcomes comparison in the matched cohort.

Statistically significant p values (<0.05) are reported in bold.
ACI, anterior circulation infarct; CI, confidence interval; mRS, modified Rankin Scale; PCI, posterior circulation infarct.
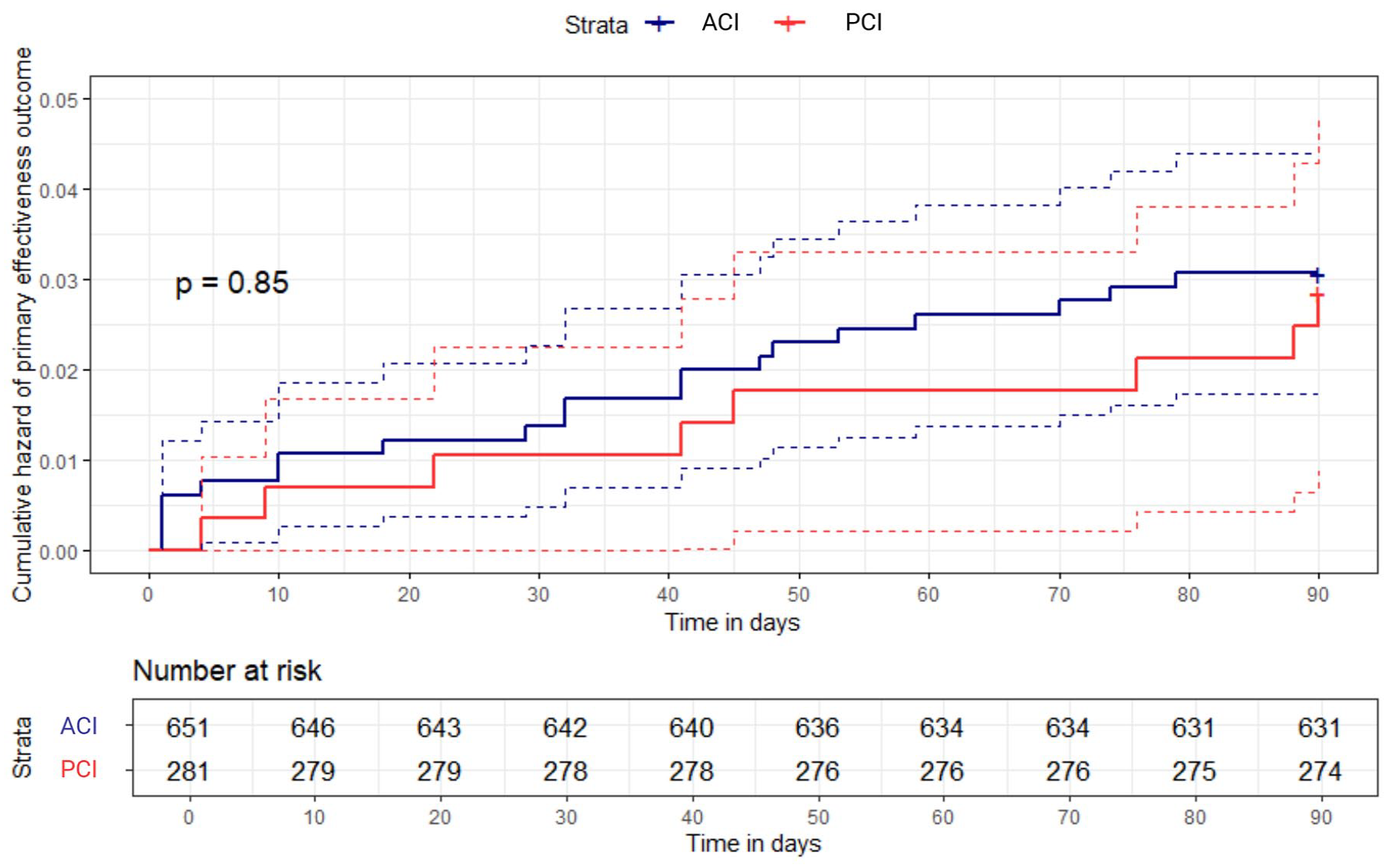
Kaplan–Meier hazard function of 90-day new ischemic stroke or other vascular events. Dashed lines indicate 95% confidence intervals.
Secondary effectiveness outcomes
Patients with PCI had worse 90-day ordinal mRS distribution compared to those with ACI (OR 1.18 (95% CI 1.01–1.39); p = 0.046; Table 2, Figure 3). There were no significant differences in other secondary effectiveness outcomes. Secondary effectiveness outcomes in the unmatched cohorts are compared in Supplemental Table 1.

90-Day modified Rankin Scale scores distribution.
Safety outcomes
The 90-day risk of safety outcomes was low in both patients with PCI and ACI, without significant differences. Specifically, bleeding events occurred in 9 patients (3.2%) with PCI and 17 patients (2.6%) with ACI (risk difference 0.6% (95% CI −1.8% to 3.0%); p = 0.628). Bleeding events were predominantly minor in both groups, accounting for 88.8% of total bleedings in patients with PCI and 70.6% in those with PCI. Moderate-to-severe bleedings occurred in only one patient (0.4%) with PCI and five patients (0.8%) with ACI. There was no significant difference in bleeding severity between groups (p = 0.485). The risk of 24-h hemorrhagic transformation was similarly low in both patients with PCI and ACI (1.8% vs 1.2%, risk difference 0.6% (95% CI −1.2% to 2.3%); p = 0.540; Table 2). Safety outcomes in the unmatched cohorts are compared in Supplemental Table 1.
Subgroup analysis for the primary effectiveness outcome
Results of subgroup analysis for the primary effectiveness outcome in the matched cohorts are shown in Supplemental Table 2. There was no significant difference in the 90-day risk of new ischemic stroke or other vascular events between patients with PCI and ACI across any prespecified subgroup. Interaction analysis revealed no heterogeneity (Supplemental Table 2).
Sensitivity analysis for the primary effectiveness outcome
Our sensitivity analysis of the primary effectiveness outcome, performed after excluding patients with multiple acute lesions involving both the anterior and posterior circulation territories, confirmed that the 90-day risk of new ischemic stroke or other vascular events did not differ between patients with PCI and ACI (3.0% vs 2.9%, respectively; HR 1.02, (95% CI 0.44–2.35); p = 0.968). Among patients with multiple infarcts involving both circulation territories, no events were observed in those classified as PCI, while two events occurred in those classified as ACI (0% vs 7.1%, respectively), with no statistically significant difference (p = 0.366).
Discussion
This propensity-matched analysis from the READAPT study demonstrated that patients with noncardioembolic mild-to-moderate ischemic stroke involving the posterior or anterior circulation territory had a similarly low incidence of new ischemic events at 90 days. Notably, this finding remained consistent across prespecified subgroups supporting its applicability to diverse patient populations, including those with moderate stroke severity and those who underwent acute reperfusion therapies. Additionally, no significant differences were observed in the risk of 24-h early neurological deterioration. However, patients with PCI experienced worse 90-day functional outcome compared to those with ACI. The 90-day risk of bleeding complications following DAPT was low and did not differ based on the vascular territory affected by the index stroke. Overall, these findings further support DAPT as an effective and safe option for secondary stroke prevention in patients with mild-to-moderate stroke, regardless of stroke location.
Within our cohort, the incidence of new ischemic strokes or other vascular events at 90 days was similarly low in both PCI and ACI patients (2.9% vs 3.1%). This contrasts with a previous study suggesting a higher risk of stroke recurrence in minor stroke or TIA patients with PCI compared to those with ACI. 8 However, the study included patients with milder symptoms, and not all received DAPT, as it predated its routine use. 8 Additionally, the time from symptom onset to treatment was not specified, introducing potential inconsistencies. 8 A recent secondary analysis CHANCE-2 trial found no difference in the risk of 90-day stroke recurrence between PCI and ACI patients with minor stroke or TIA treated with aspirin plus clopidogrel or ticagrelor, 16 aligning with our findings. However, the rate of new ischemic events was consistently higher compared to our population both in patients with PCI (8.3%) and ACI (7.4%). 16 The observed effectiveness of DAPT in reducing the risk of new ischemic stroke in our cohort may appear greater than that reported in the CHANCE-2 trial. 16 This discrepancy could be attributed to differences in baseline risk profiles, such as the predominance of Asian patients (>95% in the CHANCE-2 trial), 16 who are known to have a higher risk of vascular recurrences compared to European patients. 25 Additionally, optimal control of comorbidities may have further contributed to the reduced incidence of new vascular events in our population, as more than 80% of patients were discharged on concomitant therapy targeting vascular risk factors. Furthermore, the relatively low prevalence of vertebrobasilar stenosis among PCI patients in our population (30.6%) may have influenced the results, as this condition is known to be associated with a higher risk of recurrent ischemic events. 8
For the secondary effectiveness outcomes, we observed a worse distribution of 90-day mRS scores among PCI versus ACI patients, consistent with previous studies.9,10 Patients with PCI often experience strokes involving critical brainstem structures, the cerebellum, or the thalamus, which can lead to disabling symptoms such as ataxia, dysphagia, dysarthria, or ocular movement abnormalities. These deficits, even in the absence of significant motor impairment, may contribute to a worse functional outcome at 90 days despite similar ischemic event rates. 26 Notably, while patients with PCI had significantly worse 90-day mRS scores than those with ACI in the unmatched cohort (Supplemental Table 1), this difference was attenuated after matching, remaining marginally significant (p = 0.046). Thus, we cannot rule out that this residual difference was due to unmeasured confounders. Conversely, the observed rate of early neurological deterioration was similarly low in both patients with PCI and ACI (4.6% vs 4.0%). This finding aligns with prior evidence suggesting that early DAPT may be effective in preventing thrombus propagation in atheromatous branches during the acute phase, even in patients with PCI, which are more prone to early deterioration (reported rates ranging from 10.3% to 28.6%)7,27 due to limited collateral circulation, increased susceptibility to branch atheromatous disease, and greater hemodynamic vulnerability of posterior circulation penetrating arteries in respect to those supplying anterior brain territories. On the other hand, a post hoc analysis of the ATAMIS trial suggested that DAPT may be more effective in reducing the risk of early neurological deterioration in patients with mild-to-moderate posterior circulation stroke compared to those with anterior circulation involvement. 17 However, early neurological deterioration was assessed over a longer time window (up to 7 days), and there were differences in the distribution of stroke causes, including a higher proportion of patients with undetermined etiology (>50%). 17
For the safety outcomes, the overall incidence of 90-day bleeding events and 24-h hemorrhagic transformation was low across our study population, consistent with findings from DAPT landmark trials.13–15 Notably, the majority of bleeding events (>70%) were mild in severity in both PCI and ACI patients. This result indicates a consistent and favorable safety profile for DAPT across different infarct locations, with no evidence of varying intracerebral bleeding risk between PCI and ACI patients.
Lastly, we observed no significant differences in the risk of new ischemic events between PCI and ACI patients across subgroups stratified by sex, age, presumed stroke etiology, stroke severity, prestroke mRS, hypertension, diabetes, acute reperfusion therapies, DAPT loading dose, or the presence of symptomatic intracranial stenosis. This finding suggests that patients with noncardioembolic mild-to-moderate stroke may derive similar benefits from early DAPT, regardless of whether the infarct affects brain areas supplied by the anterior or posterior circulation, across key clinical and demographic subgroups. In particular, the absence of a significant risk difference between PCI and ACI patients with mild (NIHSS 0–5) or moderate (NIHSS 6–10) stroke severity reinforces previous evidence supporting DAPT as an effective treatment option also for moderate stroke patients,20,28 which were excluded from pivotal DAPT trials,13–15 suggesting that its effectiveness might not be influenced by the affected vascular territory, even in this specific patient population.
Strengths and limitations
The main strengths of this study lie in the adoption of rigorous procedures to ensure data quality, reinforced by regular quality checks of the READAPT electronic database, and the implementation of propensity score matching to control for confounders. However, we cannot rule out residual confounding. Although all patients underwent at least one brain neuroimaging at admission and one after 24 h, a proportion of patients (19.7%) underwent only brain NCCT. Therefore, we cannot rule out that some brain infarcts, particularly small lesions or those in the posterior circulation, have gone undetected due to the use of NCCT alone, which has lower sensitivity compared to MR. This may have led to patient exclusion, misclassification, or underestimation of the number of acute infarcts. Specifically, if any lesions were missed, it is possible that some patients actually had multiple infarcts potentially indicative of an underlying cardioembolic source. Nevertheless, brain MR was performed at admission or after 24 h in the majority of the patients who were excluded due to negative neuroimaging (84.6%). Also, to account for this potential confounder, we matched patients with PCI and ACI based on the type of brain neuroimaging performed. Similarly, patients with multiple acute infarcts involving both circulations were categorized according to a symptom-based approach. Nonetheless, we cannot completely exclude the possibility that some of these patients had mixed symptomatology that was not fully captured. However, this potential source of misclassification was accounted for in the matching process, and these cases represented only a small proportion (4.8%) of the total study population. Moreover, we performed a sensitivity analysis by excluding these patients, yielding consistent results with the overall study analysis. Another limitation was the use of the NIHSS score, which is known to underestimate the severity of posterior circulation strokes. 29 This may have led to an imbalance between groups that was not fully addressed by propensity score matching. Since stroke severity is a strong predictor of long-term functional outcomes, 30 any mismatch in actual severity between groups could have influenced the comparison of 90-day mRS scores. However, given that we observed a worse 90-day mRS score distribution in patients with PCI versus ACI, this limitation likely impacted the magnitude of this finding rather than its overall direction. Additionally, outcomes adjudication was performed by local investigators who were not blinded to the infarct location, which may have biased the assessment of some outcomes, such as the 90-day mRS score. Notably, the READAPT study included a short-term follow-up of 90 days, in line with pivotal DAPT trials13–15; however, this follow-up period may have been too short to detect differences in outcomes following DAPT between PCI and ACI patients. Longer-term follow-up studies are needed to confirm whether the absence of a significant difference in the risk of new ischemic stroke and major bleeding events after DAPT persists beyond the 90-day follow-up period. Furthermore, patients who discontinued DAPT early due to the need for anticoagulation were excluded from this analysis. Although this group represented only 1.9% of the initial READAPT population (Figure 1), their 90-day outcomes were not recorded, preventing an assessment of the impact of transitioning from DAPT to anticoagulation on outcomes. Lastly, our cohort was predominantly composed of non-Hispanic White patients, which may significantly limit the generalizability of our findings to other ethnic groups.
Conclusion
In conclusion, our findings suggest that effectiveness and safety outcomes following DAPT in patients with mild-to-moderate noncardioembolic ischemic stroke remain consistent across infarct locations in the posterior or anterior circulation territories. This supports the broad applicability of DAPT as a secondary prevention strategy in both stroke subtypes. However, patients with PCI may experience worse short-term functional outcomes after DAPT compared to those with ACI, potentially reflecting the inherently poorer functional prognosis associated with this stroke subtype. This underscores the importance of tailored rehabilitation strategies to enhance recovery in PCI patients.
Supplemental Material
sj-docx-1-tan-10.1177_17562864251351100 – Supplemental material for Real-world outcomes following dual antiplatelet therapy in mild-to-moderate ischemic stroke with anterior versus posterior circulation infarct: a READAPT study propensity matched analysis
Supplemental material, sj-docx-1-tan-10.1177_17562864251351100 for Real-world outcomes following dual antiplatelet therapy in mild-to-moderate ischemic stroke with anterior versus posterior circulation infarct: a READAPT study propensity matched analysis by Federico De Santis, Raffaele Ornello, Eleonora De Matteis, Lucio D’Anna, Michele Romoli, Tiziana Tassinari, Valentina Saia, Silvia Cenciarelli, Chiara Bedetti, Chiara Padiglioni, Bruno Censori, Valentina Puglisi, Luisa Vinciguerra, Maria Guarino, Valentina Barone, Marialuisa Zedde, Ilaria Grisendi, Marina Diomedi, Maria Rosaria Bagnato, Marco Petruzzellis, Domenico Maria Mezzapesa, Vincenzo Inchingolo, Manuel Cappellari, Cecilia Zivelonghi, Paolo Candelaresi, Vincenzo Andreone, Giuseppe Rinaldi, Alessandra Bavaro, Anna Cavallini, Maria Grazia Piscaglia, Valeria Terruso, Marina Mannino, Alessandro Pezzini, Giovanni Frisullo, Francesco Muscia, Maurizio Paciaroni, Maria Giulia Mosconi, Andrea Zini, Ruggiero Leone, Carmela Palmieri, Letizia Maria Cupini, Michela Marcon, Rossana Tassi, Enzo Sanzaro, Giuli Papiri, Giovanna Viticchi, Daniele Orsucci, Anne Falcou, Simone Beretta, Roberto Tarletti, Patrizia Nencini, Eugenia Rota, Federica Nicoletta Sepe, Delfina Ferrandi, Luigi Caputi, Gino Volpi, Salvatore La Spada, Mario Beccia, Claudia Rinaldi, Vincenzo Mastrangelo, Francesco Di Blasio, Paolo Invernizzi, Giuseppe Pelliccioni, Maria Vittoria De Angelis, Laura Bonanni, Giampietro Ruzza, Emanuele Alessandro Caggia, Monia Russo, Agnese Tonon, Maria Cristina Acciarri, Sabrina Anticoli, Cinzia Roberti, Gaspare Scaglione, Francesca Pistoia, Alberto Fortini, Antonella De Boni, Alessandra Sanna, Alberto Chiti, Leonardo Barbarini, Marcella Caggiula, Maela Masato, Massimo Del Sette, Francesco Passarelli, Maria Roberta Bongioanni, Manuela De Michele, Stefano Ricci, Simona Sacco and Matteo Foschi in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-2-tan-10.1177_17562864251351100 – Supplemental material for Real-world outcomes following dual antiplatelet therapy in mild-to-moderate ischemic stroke with anterior versus posterior circulation infarct: a READAPT study propensity matched analysis
Supplemental material, sj-docx-2-tan-10.1177_17562864251351100 for Real-world outcomes following dual antiplatelet therapy in mild-to-moderate ischemic stroke with anterior versus posterior circulation infarct: a READAPT study propensity matched analysis by Federico De Santis, Raffaele Ornello, Eleonora De Matteis, Lucio D’Anna, Michele Romoli, Tiziana Tassinari, Valentina Saia, Silvia Cenciarelli, Chiara Bedetti, Chiara Padiglioni, Bruno Censori, Valentina Puglisi, Luisa Vinciguerra, Maria Guarino, Valentina Barone, Marialuisa Zedde, Ilaria Grisendi, Marina Diomedi, Maria Rosaria Bagnato, Marco Petruzzellis, Domenico Maria Mezzapesa, Vincenzo Inchingolo, Manuel Cappellari, Cecilia Zivelonghi, Paolo Candelaresi, Vincenzo Andreone, Giuseppe Rinaldi, Alessandra Bavaro, Anna Cavallini, Maria Grazia Piscaglia, Valeria Terruso, Marina Mannino, Alessandro Pezzini, Giovanni Frisullo, Francesco Muscia, Maurizio Paciaroni, Maria Giulia Mosconi, Andrea Zini, Ruggiero Leone, Carmela Palmieri, Letizia Maria Cupini, Michela Marcon, Rossana Tassi, Enzo Sanzaro, Giuli Papiri, Giovanna Viticchi, Daniele Orsucci, Anne Falcou, Simone Beretta, Roberto Tarletti, Patrizia Nencini, Eugenia Rota, Federica Nicoletta Sepe, Delfina Ferrandi, Luigi Caputi, Gino Volpi, Salvatore La Spada, Mario Beccia, Claudia Rinaldi, Vincenzo Mastrangelo, Francesco Di Blasio, Paolo Invernizzi, Giuseppe Pelliccioni, Maria Vittoria De Angelis, Laura Bonanni, Giampietro Ruzza, Emanuele Alessandro Caggia, Monia Russo, Agnese Tonon, Maria Cristina Acciarri, Sabrina Anticoli, Cinzia Roberti, Gaspare Scaglione, Francesca Pistoia, Alberto Fortini, Antonella De Boni, Alessandra Sanna, Alberto Chiti, Leonardo Barbarini, Marcella Caggiula, Maela Masato, Massimo Del Sette, Francesco Passarelli, Maria Roberta Bongioanni, Manuela De Michele, Stefano Ricci, Simona Sacco and Matteo Foschi in Therapeutic Advances in Neurological Disorders
Footnotes
Appendix
READAPT study group names list.
| City | Site | Collaborator(s) name(s) |
|---|---|---|
| Alessandria | Stroke Unit, Department of Neurology, SS. Biagio e Arrigo | Martina Valente |
| Ancona | Experimental Medicine and Clinical Department, Marche Polytechnic University | Silvia Paolucci |
| Ancona | Department of Neurology, INRCA | Leonardo Biscetti Valentina Cameriere Eleonora Ponente |
| Bari | Department of Neurology and Stroke Unit—“F. Puca,” AOU Consorziale Policlinico | Martina Caccamo Debora Galotto |
| Barletta | Department of Neurology and Stroke Unit, “M. R. Dimiccoli” General Hospital, Barletta, ASL BT, Italy | Sergio Altomare Maurizio Giorelli |
| Bologna | IRCCS Istituto delle Scienze Neurologiche di Bologna, Department of Neurology and Stroke Center, Maggiore Hospital | Federica Naldi Chiara Bassi |
| Cesena | Department of Neurology and Stroke Unit—Bufalini Hospital | Claudia Faini |
| Chieti | Clinics of Neurology, Stroke Unit, Ospedale SS Annunziata | Paola Ajdinaj Anna di Giovanni |
| Città di Castello | Department of Neurology, Città di Castello Hospital | Elisa Sacchini |
| Crema | Department of Cardiocerebrovascular Diseases, Neurology-Stroke Unit—ASST Ospedale Maggiore di Crema | Antonio Zito |
| Cremona | Department of Neurology, ASST Cremona Hospital | Alessia Giossi |
| Fermo | Department of Neurology, Fermo Hospital | Patrizio Cardinali Stefania Maria Angelocola |
| Florence | Stroke Unit, Careggi University Hospital | Laura Tudisco |
| Florence | Internal Medicine, San Giovanni di Dio Hospital | Maraia Cristina Baruffi |
| Genoa | Neuroscience, IRCCS Ospedale Policlinico San Martino | Alessandro Canessa Davide Sassos |
| L’Aquila | Department of Biotechnological and Applied Clinical Sciences, University of L’Aquila—San Salvatore Hospital | Maria Grazia Vittorini |
| Legnano | Department of Neurology, ASST-Ovest Milanese | Serena Gallo Cassarino Gloria Valcamonica |
| Lecce | Department of Neurology, Vito Fazzi Hospital | Annalisa Rizzo |
| Lucca | Unit of Neurology, San Luca Hospital | Marco Vista |
| Mirano-Venice | Department of Neurology, ULSS 3 Serenissima | Elisabetta Menegazzo |
| Monza | Department of Neurology, Fondazione IRCCS San Gerardo dei Tintori Monza | Susanna Diamanti Carlo Ferrarese |
| Naples | Department of Neurology and Stroke Unit, AORN Antonio Cardarelli | Antonio De Mase Emanuele Spina |
| Novara | SCDU Neurologia—Stroke Unit, Azienda Ospedaliero-Universitaria “Maggiore della Carità” | Thomas Fleetwood |
| Novi Ligure | Department of Neurology, San Giacomo Hospital | Gian Luca Bruzzone |
| Parma | Department of Medicine and Surgery, University of Parma; Stroke Care Program, Department of Emergency, Parma University Hospital | Antonio Genovese |
| Pescara | Stroke Unit, “S.Spirito” Hospital | Daniela Monaco Pierluigi Tocco |
| Pistoia | Department of Neurology, San Jacopo Hospital | Chiara Menichetti Federica Letteri Serena Nannucci |
| Reggio Emilia | Department of Neurology, AUSL—IRCCS di Reggio Emilia | Rosario Pascarella Federica Assenza |
| Rimini | Neurology Unit, “Infermi” Hospital, AUSL Romagna | Enrico Maria Lotti Giorgia Bernabè |
| Rome | Department of Neurology, Sant’Andrea Hospital | Filomena Di Lisi |
| Rome | Department of Systems Medicine, Tor Vergata University Hospital | Ilaria Maestrini |
| Rome | Department of Neurology and Stroke Unit, S. Eugenio Hospital | Novella Bonaffini Maria Chiara Ricciardi |
| Rome | Stroke Unit, Azienda Ospedaliera San Camillo Forlanini | Chiara De Fino |
| Rome | Stroke Unit, Emergency Department, Policlinico Umberto I Hospital, Rome, Italy | Danilo Toni Nicoletta Giuseppa Caracciolo |
| Rome | Neuroscienze, Organi di Senso e Torace, Fondazione Policlinico Universitario Agostino Gemelli | Irene Scala Simone Bellavia |
| San Benedetto del Tronto | UOC Neurologia, Ospedale “Madonna del Soccorso” | Cristina Paci Emanuele Puca |
| San Giovanni Rotondo | Department of Neurology, Casa Sollievo della Sofferenza | Giovanni Matteo Fratta |
| Siena | Urgency and Emergency Department, Azienda Ospedaliera Universitaria Senese | Ivo Giuseppe De Franco Maurizio Acampa |
| Siracusa | Department of Neurology, Umberto I Hospital | Roberto Cappellani Eleonora Sgarlata |
| Verona | Department of Neuroscience, Azienda Ospedaliera Universitaria Integrata Verona | Mara Zenorini |
| Vicenza | Department of Neuroscience, San Bortolo Hospital | Cristina De Luca Francesco Perini |
Acknowledgements
None.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
