Abstract
Background:
Elevated baseline systolic blood pressure (SBP) was associated with poor outcomes following dual antiplatelet therapy (DAPT) in patients with non-cardioembolic minor ischemic stroke (MIS) or high-risk transient ischemic attack (TIA) in clinical trials.
Objectives:
We aimed to assess the impact of admission SBP on the short-term outcomes after DAPT in patients with non-cardioembolic MIS or high-risk TIA.
Methods:
We performed an inverse probability weighted (IPW) analysis from a prospective multicentric real-world study (READAPT) including patients with non-cardioembolic MIS (National Institute of Health Stroke Scale of 0–5) or high-risk TIA (ABCD2 ⩾4) who initiated DAPT within 48 h of symptom onset. The primary effectiveness outcome was the 90-day risk of new ischemic stroke or other vascular events. The secondary effectiveness outcomes were the 90-day modified Rankin Scale score ordinal shift, vascular and all-cause mortality, 24-h early neurological improvement or deterioration. The safety outcomes included the 90-day risk of moderate-to-severe and any bleedings, symptomatic intracranial hemorrhage, and 24-h hemorrhagic transformation. We used Cox proportional hazards regression with restricted cubic splines to model the continuous relationship between SBP and the hazard ratio (HR) of new vascular events. We selected SBP = 124 mm Hg as cut-off point for the IPW weighting. Outcomes were compared using Cox and generalized logistic regression analyses, adjusted for residual confounders.
Results:
From 2278 patients in the READAPT cohort, we included 1291 MIS or high-risk TIAs (mean age 70.6 ± 11.4 years; 65.8% males). After IPW, patients with admission SBP ⩾124 mm Hg versus <124 mm Hg had a significantly higher risk of 90-day ischemic stroke or other vascular events (adjusted HR: 2.14 (95% CI 1.07%–4.98%); p = 0.033) and of 24-h early neurological deterioration (adjusted risk difference: 1.91% (95% CI 0.60%–3.41%); p = 0.006). The overall risk of safety outcomes was low, although patients with SBP ⩾124 mm Hg on admission showed higher rates of 90-day moderate-to-severe and any bleeding events (adjusted risk difference: 1.24% (95% CI 0.38%–2.14%); p = 0.004 and 6.18% (95% CI 4.19%–8.16%); p < 0.001; respectively), as well as of 24-h hemorrhagic transformation (adjusted risk difference: 1.57% (95% CI 0.60%–2.55%); p = 0.001). Subgroup analysis showed a significant interaction between admission SBP, sex, and time to DAPT start in predicting 90-day new vascular events (p for interaction <0.001 and 0.007, respectively).
Conclusion:
In patients with non-cardioembolic MIS or high-risk TIA, higher levels of admission SBP may be associated with an increased risk of new vascular events, early neurological deterioration, and bleeding after DAPT use. Future studies should further investigate if optimizing blood pressure management may further improve prognosis.
Introduction
Dual antiplatelet therapy (DAPT) has become the standard of care in the secondary prevention of new ischemic events in patients who have experienced non-cardioembolic minor ischemic stroke (MIS) or high-risk transient ischemic attack (TIA).1–3 Previous studies showed that admission systolic blood pressure (SBP) may influence the choice of initial antiplatelet therapy, as well as the efficacy of DAPT in preventing ischemic stroke recurrences.4–6 In particular, a post-hoc analysis of CHANCE reported that DAPT was associated with a lower risk of new vascular events in patients with a baseline SBP greater than or equal to 140 mm Hg. 5 Similarly, a secondary analysis of the Platelet-Oriented Inhibition in New TIA and Minor Ischemic Stroke (POINT) showed that having a baseline SBP <140 mm Hg was associated with a significant reduction (64%) in the 90-day risk of ischemic stroke recurrences in patients treated with DAPT compared with aspirin alone, whereas patients with baseline SBP ⩾140 mm Hg did not show any benefit for DAPT over aspirin monotherapy. 6 These findings may indicate a synergistic effect between controlled BP in the acute and subacute phases of stroke and the antithrombotic actions of DAPT within the first-week post-event. Additionally, while trials on blood pressure reduction shortly after ischemic stroke have generally shown neutral to negative outcomes,7,8 they did not rule out the potential benefit of moderate BP control (targeting SBP <140 mm Hg). This approach, when combined with DAPT, may reduce early stroke recurrence by simultaneously mitigating multiple vascular risk factors. These observations highlight the need to clarify the relationship between SBP levels during the acute and subacute phases of the index event and DAPT outcomes, especially in real-world settings, where DAPT is often used beyond the indications of clinical trials, including patients with higher pre-stroke disability, a higher burden of vascular risk factors, and those treated with reperfusion therapies.9–11 Hence, we conducted a post-hoc analysis of the Real-Life Study on Short-Term Dual Antiplatelet Treatment in Patients With Ischemic Stroke or Transient Ischemic Attack (READAPT) study, to assess the impact of admission SBP on short-term outcomes following DAPT in a real-world population of patients with non-cardioembolic MIS or high-risk TIA.
Methods
READAPT study
The READAPT study (NCT05476081) is a prospective, observational, multicenter study conducted in a real-world setting. The study was coordinated by the University of L’Aquila and supported by the Italian Stroke Association – Associazione Italiana Ictus. It adheres to the guidelines outlined by the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE). 12
The READAPT study recruited all individuals aged 18 years or older, regardless of hospitalization, who presented with acute non-cardioembolic ischemic stroke or TIA and received a short course of DAPT lasting up to 90 days, irrespective of stroke severity or timing of symptom onset. Recruitment occurred across 64 Italian stroke centers from February 1, 2021 to February 28, 2023. The study enrolled patients consecutively admitted to hospitals, seen in emergency departments, or evaluated in open access outpatient clinics specializing in urgent neurological assessments. To promote the comprehensive inclusion of eligible patients on DAPT, investigators received detailed instructions and periodic reminders. The initial stroke diagnosis was confirmed by the investigators per routine practice and World Health Organization criteria. Eligibility was limited to those with an acute non-cardioembolic stroke or TIA. No further eligibility restrictions, including specific antiplatelet regimens or timing between symptom onset and DAPT initiation, were imposed. Enrollment was independent of whether a loading DAPT dose was given (⩾300 mg for aspirin and clopidogrel or ⩾180 mg for ticagrelor). Exclusion criteria included participation in interventional randomized controlled trials at stroke onset, continuous DAPT for stenting procedures, and conditions likely to interfere with adherence to study protocols. Patients were enrolled shortly following the qualifying event and followed up for 90 ± 10 days, with follow-up visits conducted by investigators in person or remotely. Detailed descriptions of the study procedures and definitions used in READAPT have been previously published.9–11
Study analysis
This post-hoc exploratory analysis of the READAPT cohort includes patients with MIS (defined as a National Institute of Health Stroke Scale (NIHSS) score of 0–5 on admission) or high-risk TIA (symptoms lasting less than 24 h and an ABCD2 score of ⩾4), who initiated DAPT within 48 h of symptom onset. Patients who had not available SBP on admission, and those who underwent urgent carotid endarterectomy or stenting during the follow-up period were excluded, as were those who discontinued DAPT early due to initiation of anticoagulation therapy. The latter exclusion was made to avoid confounding effects of anticoagulation-related bleeding on DAPT safety evaluations. SBP was measured by local investigators upon admission as part of the routine clinical protocol, either in the emergency department for hospitalized patients or in the outpatient clinic, prior to the neurological evaluation and any therapeutic intervention. For this analysis, we included the first SBP measurement recorded within 2 h of patient admission to ensure consistency in data collection. Given the multicenter nature of the study, blood pressure was measured using standard clinical sphygmomanometers or automated devices available at each site, following institutional protocols. While variations in measurement techniques across centers may exist, all participating investigators were trained in standardized BP measurement practices to minimize inter-center variability. Additionally, SBP was consistently recorded with the patient in a supine position. If no SBP measurement was documented within this period, the data were considered unavailable, and the patient was excluded from the analysis.
Outcomes
The primary effectiveness outcome was the risk of ischemic stroke or other vascular events (TIA, myocardial infarction, vascular death) within 90 days. Secondary effectiveness outcomes included a 90-day modified Rankin Scale (mRS) score shift, early neurological improvement or deterioration (defined as ⩾2 points decrease or increase in NIHSS from baseline, respectively) within 24 h, 90-day vascular mortality (death from stroke, systemic bleeding, myocardial infarction, heart failure, pulmonary embolism, sudden death, or arrhythmia), and all-cause mortality at 90 days. Safety outcomes included moderate-to-severe bleeding events, any bleeding event, symptomatic intracranial hemorrhage (sICH), and hemorrhagic transformation within 24 h. Bleeding severity was categorized according to the Global Use of Strategies to Open Occluded Coronary Arteries (GUSTO) classification criteria. 13 Specifically, we classified the clinical impact of bleeding events as moderate-to-severe if they involved fatal hemorrhage or hemorrhage that, regardless of hemodynamic compromise, required blood or fluid replacement, inotropic support, or surgical intervention. Conversely, bleeding events that did not meet these criteria were classified as minor. 13 sICH was defined as any intracranial hemorrhage resulting in a worsening of four or more points on the NIHSS. 14 Outcomes were adjudicated by local investigators via electronic follow-up case-report forms.
Statistical analysis
Relationship between admission SBP and 90-day risk of new ischemic stroke or TIA
To analyze the relationship between SBP and the 90-day risk of new ischemic events, we first conducted a logistic regression model treating SBP as a continuous variable. Since linear models assume a constant effect of SBP across its entire range, we further applied restricted cubic splines (RCS) to assess potential non-linearity. Specifically, Cox proportional hazards regression with RCS was employed to model the continuous relationship between SBP and the hazard of new vascular events. Knots for the splines were set at the 10th, 50th, and 90th percentiles of SBP. The median SBP was used as the reference, and hazard ratios (HRs) for varying SBP levels were estimated relative to this reference. Log HRs were computed and exponentiated to yield HRs, with 95% confidence intervals (CIs) calculated using the standard errors of the log HRs. CIs were derived by adjusting each log HR by ±1.96 times the standard error, then exponentiating the bounds. To identify a clinical cut-off for SBP, the HR curve was examined to locate values where the HR approximated 1, indicating no significant increase or decrease in risk. The SBP level closest to HR = 1 was selected as a potential cut-off point for stratification.
Inverse probability weighting
To balance patients based on baseline characteristics, we used inverse probability weighting (IPW) to create a pseudo-weighted sample between patients with admission SBP above and below the cut-off value determined through RCS analysis. We first estimated propensity scores using logistic regression, modeling the probability of being in the group with admission SBP above the cut-off based on a pre-specified set of baseline covariates: age, gender, smoking status, diabetes, hypercholesterolemia, prior use of antihypertensive medication, baseline NIHSS, modified Rankin Scale mRS, intravenous thrombolysis, endovascular therapy, and DAPT loading dose. From the logistic regression model, we calculated propensity scores for each patient and used these scores to assign inverse probability weights. The covariate balance between groups was evaluated by calculating the standardized mean difference. Additionally, we examined the distribution of propensity scores to verify overlap between groups, and we applied stabilized weights to mitigate the influence of extreme weights and enhance model stability.
Outcomes analysis in the weighted population
For the primary effectiveness outcomes, the time-to-event 90-day risk of ischemic stroke and other vascular events was compared using Cox regression, and the corresponding treatment effect was presented as HR and 95% CIs. Kaplan–Meier hazard functions were used to compare the overall risk of ischemic stroke and other vascular events between admission SBP groups and the log-rank test was used to test differences. For the secondary effectiveness outcomes, the 90-day shift of mRS scores was compared using an ordinal generalized linear model (GLM) and results were presented as odds ratio with 95% CIs. Furthermore, we calculated risk differences between groups for the rate of 24-h neurological improvement and 24-h neurological deterioration, while Cox regression analysis was used to compare the risk of 90-day vascular death and all-cause mortality. For all the safety outcomes, we also calculated risk differences with 95% CIs between the SBP groups. All the analyses were adjusted for residual confounders (i.e., variables that showed a SD >0.10 in the weighted population). Specifically, we obtained adjusted risk differences by employing multivariate GLM with binomial distribution and identity link function.
Subgroup analysis for the primary effectiveness outcome in the weighted population
Subgroup analysis of the primary effectiveness outcome in the weighted population was performed in 12 prespecified subgroups: age (⩽75 years or >75 years), sex (males or females), presence of arterial hypertension or diabetes, presumed stroke cause (large artery atherosclerosis, small artery occlusion, undetermined), symptoms duration (⩾24 h or <24 h), intravenous thrombolysis, endovascular thrombectomy, DAPT loading dose and time to DAPT start (<24 h or 24–48 h). We used a GLM model with admission SBP, subgroup variable, and their interaction term as independent variables to determine the homogeneity in the effect of SBP by each subgroup variable. Each model was adjusted for age and sex, when applicable. The p value was presented for the interaction term. Lastly, marginal effects analysis was employed to visualize and exanimate predicted probabilities of 90-day new vascular events across the range of SBP within each subgroup.
All statistical analyses were performed using R software (R Core Team), version 4.2.2. Statistical significance was set at a p value <0.05.
Results
From 2278 patients in the READAPT cohort, a total of 1291 patients (mean age 70.6 ± 11.4 years; 70.9% minor ischemic strokes and 29.1% high-risk TIAs) were included in the analysis, of whom 849 (65.8%) were males. The number of excluded patients with reasons is reported in the study flowchart. Figure 1 note, admission SBP was not available in only 128 patients (5.6%).
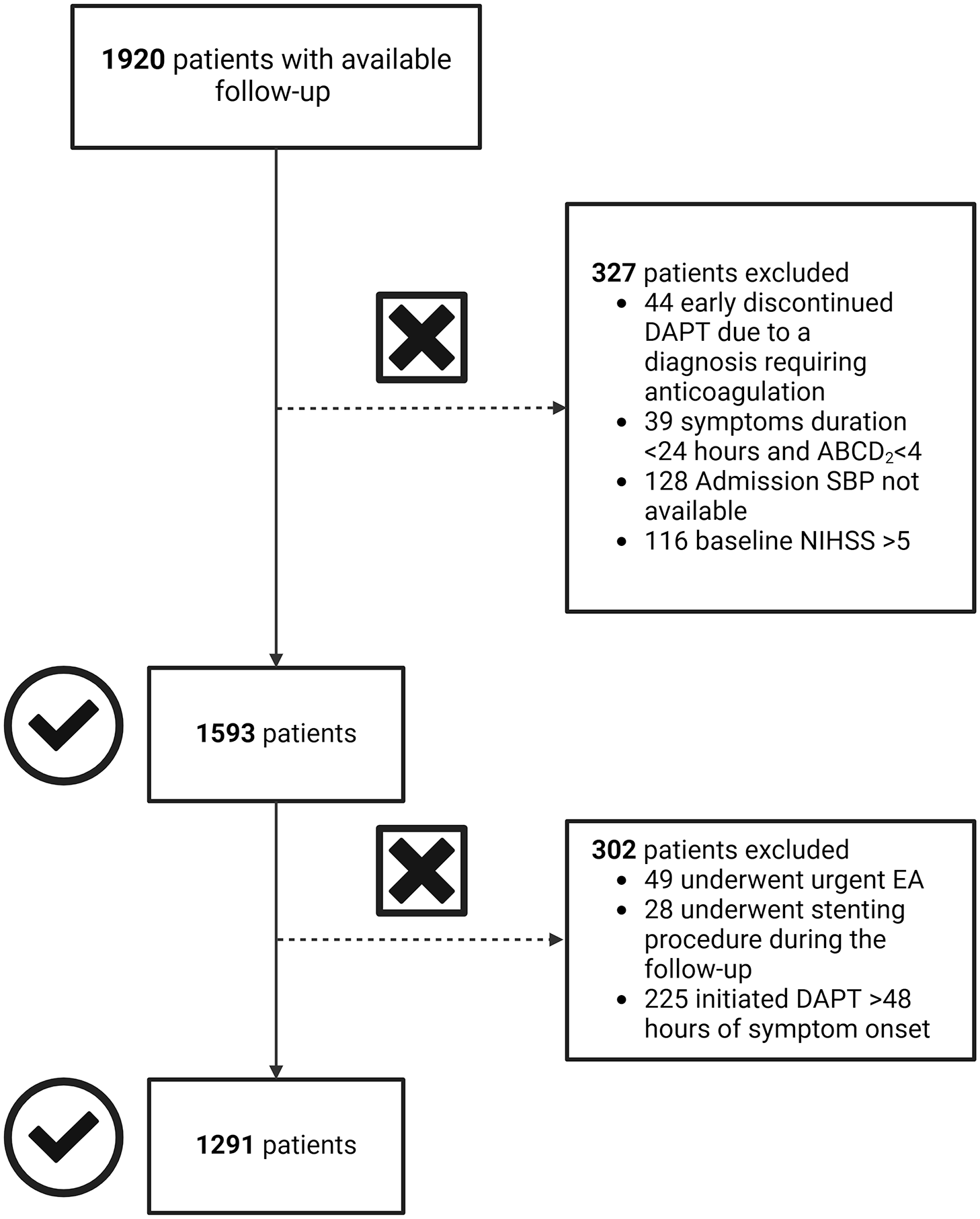
Study flowchart.
Relationship between admission SBP and 90-day risk of new ischemic stroke or TIA
The logistic regression model evaluating the association between admission SBP as a continuous variable and the 90-day risk of new ischemic stroke or TIA revealed that each 1 mmHg increase in SBP was associated with a HR of 1.01 (95% CI 1.001–1.02, p = 0.037). The Cox proportional hazards regression with RCS revealed a non-linear association between admission SBP and the hazard of 90-day new vascular events. We identified an admission SBP cut-off of 124 mm Hg as the first point where the HR approximated 1 (Figure 2). A second point was identified at extreme SBP values (>200 mm Hg), marked by a sharp increase in the risk of the primary effectiveness outcome. We selected the first point (SBP = 124 mm Hg) as clinical cut-off for this analysis, as it was closer to the SBP range of neutral risk, offering a more reliable threshold for identifying patients transitioning out of high-risk states. We identified 1162 patients (90.0%) with baseline SBP ⩾124 mm Hg, while the remaining 129 patients (10.0%) had an SBP <124 mm Hg at admission.

HR for admission SBP predicting 90-day risk of new ischemic stroke or other vascular events.
Inverse probability weighting
Weighted and unweighted results for baseline characteristics are presented in Table 1. Overall, good balance was obtained for most of the baseline variables which showed a SD of the propensity scores <0.10. Measures of balance diagnosis indicated that the samples were adequately weighted, with a standardized difference of the propensity scores means between groups of 0.04 (good balance <0.25), ratio of variances of propensity scores 0.74 (good balance between 0.5 and 2). 15 Graphics of propensity scores and covariates balance distributions confirmed a good overall quality of the weighting (Figures S1 and S2).
Baseline characteristics.

SMDs >0.10 in the weighted population are reported in bold, indicating suboptimal weighting (except for baseline mean blood pressure values).
SMD <0.10 indicates that groups are well balanced.
Only patients with symptoms duration <24 h.
Presumed stroke cause was classified according to the Trial of Org 10172 in the Acute Stroke Treatment (TOAST) classification system.
Arterial hypertension was defined as a history of blood pressure >140/90 mm Hg or the current use of antihypertensive medications.
Dyslipidemia was defined as history of total blood cholesterol levels >220 mg/dL and/or total triglycerides levels >130 mg/dL and/or currently used lipid-lowering drugs.
Diabetes mellitus was defined as history of fasting glucose >126 mg/dL or the current use of hypoglycemic medications.
Current smoking was defined as the consumption of ⩾1 cigarette per day over the last year.
DAPT, dual antiplatelet therapy; IQR, interquartile range; mRS, modified Rankin Scale score; NIHSS, National Institute of Health Stroke Scale score; SBP, systolic blood pressure; SD, standard deviation; SMD, standardized means difference; TIA, transient ischemic attack.
Primary effectiveness outcome
The rate of 90-day new ischemic stroke or other vascular events in the weighted population was significantly lower in the group with admission SBP <124 mm Hg than in those with SBP ⩾124 mm Hg (aHR 2.14 (95% CI 1.07–4.98); p = 0.033) (Table 2). Kaplan–Meier hazard function confirmed that the overall 90-day risk of ischemic stroke and other vascular events was lower in patients with admission SBP <124 mm Hg (log-rank test p value: 0.034) (Figure 3).
Outcomes comparison in the weighted population.

Statistically significant p values (<0.05) are reported in bold.
Adjusted for baseline variables with SMD >0.10 (arterial hypertension, previous TIA/ischemic stroke, ongoing clopidogrel or lipid-lowering drugs at the index minor ischemic stroke/TIA, antihypertensive drugs on discharge, type of antiplatelet prescribed after DAPT).
DAPT, dual antiplatelet therapy; mRS, modified Rankin Scale; SBP, systolic blood pressure.

Kaplan–Meier cumulative function of 90-day new ischemic stroke or other vascular events in the weighted population.
Secondary effectiveness outcomes
The 90-day ordinal mRS distribution was similar between admission SBP groups (aOR 0.91 (95% CI 0.81–1.02); p = 0.127). Conversely, patients with admission SBP ⩾124 mm Hg showed higher rates of 24-h early neurological deterioration compared to those with admission SBP <124 mm Hg (adjusted risk difference: 1.91% (0.60%–3.41%); p = 0.006). The 90-day risk of other secondary effectiveness outcomes did not differ between groups (Table 2).
Safety outcomes
We found a low rate of 90-day safety outcomes in both patients with admission SBP ⩾124 mm Hg and <124 mm Hg (Table 2). However, patients with admission SBP ⩾124 mm Hg showed a significantly higher risk of 90-day moderate-to-severe (adjusted risk difference: 1.24% (0.38%–2.14%); p = 0.004) and any (adjusted risk difference: 6.18% (4.19%–8.16%); p < 0.001) bleeding events, as well as of 24-h hemorrhagic transformation (adjusted risk difference: 1.57% (0.60%–2.55%); p = 0.001) (Table 2).
Subgroup analysis for the primary effectiveness outcome in the weighted population
Admission SBP was associated with the incremental probability of 90-day risk of ischemic stroke and other vascular events regardless of the age group (p = 0.344), presence of diabetes (p = 0.279), history of hypertension (p = 0.118), symptoms duration (p = 0.239), presumed stroke cause (large artery atherosclerosis p = 0.394, small artery occlusion p = 0.123, undetermined cause p = 0.084), intravenous thrombolysis (p = 0.278) or endovascular thrombectomy (p > 0.999), DAPT loading dose (p = 0.087). Yet a significant interaction between admission SBP and sex was found in predicting 90-day risk of ischemic stroke and other vascular events (p for interaction <0.001) and with time to start DAPT (p for interaction = 0.007) (Figure 4).

Margin plots illustrating the relationship between admission SBP and the probability of 90-day ischemic stroke or new vascular events, analyzed by sex (a) and time to DAPT start (b).
Discussion
Our study suggests that baseline SBP impacts short-term outcomes in patients with non-cardioembolic MIS or high-risk TIA receiving DAPT. Specifically, in this exploratory analysis, patients with higher SBP levels on admission had significantly higher risk of 90-day new ischemic stroke or other vascular events, 24-h early neurological deterioration, and 90-day bleeding events compared to those presenting with lower SBP values. Furthermore, we identified female sex and delayed DAPT initiation after the index event as potential factors that may interact with admission SBP in increasing the risk of ischemic recurrences within 90 days.
In our study, we documented a non-linear relationship between SBP at admission and the risk of ischemic vascular events within 90 days. In our weighted cohorts, the risk of 90-day new ischemic stroke and other vascular events was significantly higher in patients with admission SBP ⩾124 mm Hg, compared to those with SBP <124 mm Hg. Notably, patients with SBP <124 mm Hg on admission still represented a high-risk population, as they were largely receiving antihypertensive treatment (70.3%) at the time of the index stroke. Although patients with MIS or TIA and low admission SBP are less commonly encountered in clinical practice, our findings align with current guidelines recommending SBP targets of 120–130 mmHg for patients at high cardiovascular risk. 16 This may further reinforce the importance of achieving and maintaining stringent BP control in patients undergoing DAPT for secondary stroke prevention. Previous post-hoc analyses from landmark clinical trials have explored the impact of baseline SBP on 90-day outcomes in patients with non-cardioembolic MIS or high-risk TIA.5,6 In particular, secondary analyses of the CHANCE and POINT trials revealed that patients receiving DAPT with an admission SBP of 140 mm Hg or higher experienced significantly higher stroke event rates compared to those with lower values (8.7% vs 6.7% in the CHANCE and 6.5% vs 5.2% in the POINT post-hoc analyses).5,6 Notably, in these studies, the threshold of 140 mm Hg was prespecified. Conversely, in our study, the cut-off of 124 mm Hg was determined using a restricted cubic spline analysis, suggesting that the risk associated with elevated SBP may emerge at lower thresholds than previously recognized in clinical trials. This nuanced finding may be attributable to the higher burden of comorbidities often present in real-world patient populations. Additionally, differences in patient characteristics, such as older age at the time of the index stroke or TIA, varying ethnic backgrounds, and greater pre-stroke systemic impairment, may have further amplified the detrimental effects of elevated SBP on the risk of recurrent ischemic events within our population.
For the secondary outcomes, we found that patients with admission SBP ⩾124 mm Hg had a significantly higher risk of 24-h early neurological deterioration compared to those with baseline SBP <124 mm Hg. DAPT has been shown to reduce the occurrence of early neurological deterioration primarily due to its strong antithrombotic effects, which can improve cerebral blood flow. 17 In particular, a recent post-hoc analysis of the Antiplatelet versus R-tPA for Acute Mild Ischemic Stroke (ARAMIS) trial showed more treatment effect of DAPT on the risk of 24-h early neurological deterioration in MIS patients with SBP ⩾140 mm Hg (adjusted risk difference: −5.2%, 95% CI −8.2% to −2.3%), while no effect was found in the subgroup with admission SBP <140 mm Hg. 18 Changes in blood pressure during acute ischemic stroke often reflect impaired cerebral autoregulation, caused by direct damage to modulatory centers.19,20 For example, an increase in cerebral perfusion pressure beyond 30% can signify overall autoregulatory dysfunction. 20 This impaired autoregulation has been linked to neurological deterioration by disrupting cerebral blood flow and perfusion pressure. 21 Consequently, the level of SBP at admission may influence the effectiveness of DAPT in preventing early neurological deterioration by modulating its impact on cerebral blood flow and perfusion.
For the safety outcomes, we observed a low rate of 90-day bleeding events within our cohort, indicating a favorable overall safety profile of short-term DAPT irrespective of admission SBP. However, we identified a significant association between elevated SBP and an increased risk of bleeding complications, including moderate-to-severe bleeding and hemorrhagic transformation. While the role of prior hypertension as a risk factor for hemorrhagic transformation remains a subject of debate, 22 elevated SBP on admission has been implicated in exacerbating this risk through mechanisms such as vascular remodeling, impaired autoregulation, and inflammatory responses, all of which can compromise the integrity of the blood-brain barrier.23–25 In contrast with our findings, prior secondary analysis of landmark trials showed similar rates of bleeding events between patients receiving DAPT or aspirin, regardless of their baseline SBP values.5,6 This divergence may be attributed to several factors, including the more rigorous BP monitoring and proactive management typically implemented in controlled trials compared to real-world clinical practice. Additionally, the greater variability in DAPT dosing and concurrent chronic therapies, especially antihypertensive medications, observed in real-world settings compared to clinical trials may further exacerbate the risk of bleeding events in patients with elevated baseline SBP.
Our subgroup analysis adjusted by age identified a significant interaction between admission SBP and female sex, suggesting an increased risk of 90-day ischemic recurrences in women with elevated baseline SBP values. This finding might underscore a complex and multifactorial relationship between sex, SBP, and DAPT outcomes, which may not have been fully captured in previous trials. Women generally exhibit a higher prevalence of hypertension and small vessel disease, both of which can increase vascular vulnerability and contribute to worse outcomes.26,27 Moreover, sex-related differences in platelet reactivity and the pharmacokinetics of antiplatelet drugs may influence the effectiveness of DAPT, potentially altering the balance between ischemic protection and bleeding risk, particularly in patients with uncontrolled hypertension. 28 These factors suggest that sex-specific considerations may be important in optimizing blood pressure and antiplatelet management strategies.
Moreover, we observed a significant interaction between admission SBP and a prolonged time to DAPT start (12–48 h) after adjusting for age and sex, increasing the risk of 90-day new ischemic stroke or other vascular events. Major trials have consistently demonstrated that prompt initiation of DAPT (within 12–24 h) significantly reduces the risk of early ischemic stroke recurrence compared to aspirin alone (adjusted risk difference from −1.5% in the POINT trial to −3.5% in the CHANCE trial).1–3 Although a significant reduction in 90-day ischemic recurrences has been shown in patients starting DAPT versus aspirin alone up to 72 h after the index cerebrovascular event (adjusted risk difference of −1.9% in the Intensive Statin and Antiplatelet Therapy for Acute High-Risk Intracranial or Extracranial Atherosclerosis (INSPIRES) trial), 28 our observation may suggest that the synergistic effect of controlled SBP and the antithrombotic actions of DAPT may diminish as the time to initiation extends beyond the optimal window. Elevated SBP levels may enhance the risk of short-term recurrent events by accelerating irreversible vascular and hemodynamic disruptions in the absence of immediate antithrombotic intervention, 29 potentially driving early ischemic recurrences.
Lastly, while chronic hypertension and diabetes are well-established risk factors for stroke recurrence, 30 their lack of significant interaction with admission SBP in our subgroup analysis for the primary effectiveness analysis may be attributed to factors such as the homogeneity of our high-risk cohort, potential treatment effects, or the influence of unmeasured variables, including BP variability over time, adherence to and intensification of antihypertensive therapy, lifestyle and behavioral factors (e.g., smoking, diet, physical activity), and genetic variability affecting BP regulation and platelet reactivity. Further investigation in future studies incorporating longitudinal BP monitoring, treatment adherence assessments, and additional clinical and biomarker data are warranted to elucidate the relationship between SBP levels and stroke recurrence risk after DAPT in these subgroups of patients with elevated cardiovascular risk.
Strengths and limitations
The primary strength of this study lies in the rigorous procedures implemented to enhance data accuracy and quality, supported by regular quality checks of the READAPT electronic database. However, our findings should be interpreted in lights of certain limitations. First, although we applied rigorous IPW weighting and adjusted the outcome comparisons for unbalanced covariates, residual confounding cannot be completely ruled out due to the observational nature of the READAPT study. Specifically, while SBP was measured in all patients upon admission and before initiating DAPT, we did not record the exact time between the onset of the index stroke/TIA and the SBP measurement, which may affect its association with subsequent adverse outcomes. Of note, within our population, only 129 patients had baseline SBP ⩽124 mm Hg versus 1162 above. Although we used IPW to address the imbalance in group sizes, ensuring that the smaller group contributed proportionately to the analysis, this approach can pose challenges related to the statistical stability of the weights and generalizability of the results. To mitigate these potential biases, we applied stabilized weights. Second, we could not evaluate whether antihypertensive treatment effectively controlled elevated blood pressure or explore blood pressure variability within the first 24 h, a critical period closely linked to the risk of adverse events, particularly early neurological deterioration. Similarly, we did not record information on the specific types of antihypertensive medications used during the acute or subacute stroke phase, which may have influenced SBP differently based on their distinct pharmacological properties. While our analysis focused on admission SBP, it is important to acknowledge that antihypertensive medication use during the study period may have influenced both SBP trajectories and clinical outcomes, potentially acting as a confounder. However, given the observational nature of our study, we were unable to account for dynamic BP changes over time or medication adherence post-admission. Future studies with longitudinal BP monitoring and detailed treatment data are needed to better understand the interplay between antihypertensive therapy, SBP control, and vascular outcomes in this high cardiovascular risk population. Additionally, we were unable to investigate whether elevated baseline blood pressure might have helped maintain cerebral perfusion in vulnerable brain regions, as no pre-stroke blood pressure data was available. Third, our findings lack detailed information on cerebral autoregulation, such as measurements of cerebral blood flow or perfusion in the acute phase following stroke, which could clarify the association between baseline SBP and the efficacy of DAPT in improving neurological function. Fourth, although all patients underwent brain imaging (non-contrast computed tomography or magnetic resonance imaging) upon admission, we did not collect detailed neuroimaging findings or results from brain vessel examinations. As a result, the stroke diagnosis and presumed cause relied solely on assessments by local investigators. Fifth, we were unable to measure blood levels of antiplatelets, limiting our ability to assess patient adherence to DAPT, determine the exact onset of its effects, or confirm ongoing antiplatelet activity in patients already on prior antiplatelet therapy. Sixth, patients who discontinued DAPT early due to a need for anticoagulation were excluded from this analysis. Although this subset represented only 1.9% of the initial READAPT population (Figure 1), we did not record their 90-day outcomes. Finally, our cohort showed a significant imbalance toward non-Hispanic white participants, which may limit the generalizability of our findings to other ethnic groups. As this was an exploratory analysis within a real-world prospective cohort, no a priori sample size calculation was performed. Therefore, the absence of pre-specified power calculations may have limited the ability to detect smaller effect sizes, particularly in subgroup analyses.
Conclusion
In conclusion, our findings indicate that admission SBP could play a significant role in influencing short-term outcomes following DAPT in patients with non-cardioembolic MIS or high-risk TIA. Higher levels of admission SBP appear to be linked with an increased likelihood of adverse events, including new vascular events, early neurological deterioration, and a heightened risk of bleeding complications. Notably, our analysis also suggests that certain factors, such as female sex and a prolonged interval between the index cerebrovascular event and the initiation of DAPT, may amplify the impact of high admission SBP on the risk of ischemic recurrences. Our observations underscore the importance of considering admission SBP as a potential modifiable risk factor in this patient population. Future research should investigate the underlying pathophysiological mechanisms driving these associations and explore targeted interventions to optimize blood pressure management. These efforts may have the potential to ultimately improve patient outcomes in those receiving DAPT after non-cardioembolic MIS or high-risk TIA.
Supplemental Material
sj-docx-1-tan-10.1177_17562864251332720 – Supplemental material for Admission systolic blood pressure and short-term outcomes after dual antiplatelet therapy in patients with minor ischemic stroke or transient ischemic attack
Supplemental material, sj-docx-1-tan-10.1177_17562864251332720 for Admission systolic blood pressure and short-term outcomes after dual antiplatelet therapy in patients with minor ischemic stroke or transient ischemic attack by Federico De Santis, Eleonora De Matteis, Lucio D’Anna, Michele Romoli, Tiziana Tassinari, Valentina Saia, Silvia Cenciarelli, Chiara Bedetti, Chiara Padiglioni, Bruno Censori, Valentina Puglisi, Luisa Vinciguerra, Maria Guarino, Valentina Barone, Marialuisa Zedde, Ilaria Grisendi, Marina Diomedi, Maria Rosaria Bagnato, Marco Petruzzellis, Domenico Maria Mezzapesa, Vincenzo Inchingolo, Manuel Cappellari, Cecilia Zivelonghi, Paolo Candelaresi, Vincenzo Andreone, Giuseppe Rinaldi, Alessandra Bavaro, Anna Cavallini, Stefan Moraru, Maria Grazia Piscaglia, Valeria Terruso, Marina Mannino, Alessandro Pezzini, Giovanni Frisullo, Francesco Muscia, Maurizio Paciaroni, Maria Giulia Mosconi, Andrea Zini, Ruggiero Leone, Carmela Palmieri, Letizia Maria Cupini, Michela Marcon, Rossana Tassi, Enzo Sanzaro, Giulio Papiri, Giovanna Viticchi, Daniele Orsucci, Anne Falcou, Simone Beretta, Roberto Tarletti, Patrizia Nencini, Eugenia Rota, Federica Nicoletta Sepe, Delfina Ferrandi, Luigi Caputi, Gino Volpi, Salvatore La Spada, Mario Beccia, Claudia Rinaldi, Vincenzo Mastrangelo, Francesco Di Blasio, Paolo Invernizzi, Giuseppe Pelliccioni, Maria Vittoria De Angelis, Laura Bonanni, Giampietro Ruzza, Emanuele Alessandro Caggia, Monia Russo, Agnese Tonon, Maria Cristina Acciarri, Chiara Di Fino, Cinzia Roberti, Giovanni Manobianca, Gaspare Scaglione, Francesca Pistoia, Alberto Fortini, Antonella De Boni, Alessandra Sanna, Alberto Chiti, Marcella Caggiula, Maela Masato, Massimo Del Sette, Francesco Passarelli, Maria Roberta Bongioanni, Manuela De Michele, Stefano Ricci, Raffaele Ornello, Simona Sacco and Matteo Foschi in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
