Abstract
Introduction:
Patients with ischemic stroke or transient ischemic attack (TIA) and cancer face unique risks of recurrent ischemic events and bleeding. It is unclear whether this increased risk is present even in patients with minor ischemic stroke or transient ischemic attack (TIA) receiving dual antiplatelet therapy (DAPT). This study aimed to evaluate the impact of cancer on the short-term outcomes after DAPT in patients with non-cardioembolic minor ischemic stroke or high-risk TIA.
Patients and methods:
This was a secondary analysis of the prospective multicentric READAPT study (NCT05476081), including patients with non-cardioembolic minor ischemic stroke (NIHSS ⩽ 5) or high-risk TIA (ABCD2 ⩾4) who initiated DAPT within 48 h of symptom onset. The primary effectiveness outcome was the 90-day risk of new ischemic stroke or other vascular events (TIA, myocardial infarction, death due to vascular causes). Secondary outcomes included 90-day mRS score distribution and all-cause mortality. The primary safety outcome was the 90-day risk of any bleeding, with secondary safety outcomes including 24-h hemorrhagic transformation. We used Inverse Probability Weighting to compare outcomes between patients with and without cancer.
Results:
From 2278 patients in the READAPT study cohort, we included 1561 patients (mean age 70.3 ± 11.7 years; 65.4% males), of whom 206 (13.2%) had cancer, categorized as either active (27.7%) or in remission (72.3%). After weighting, overall cancer patients had a higher risk of 90-day new ischemic stroke or other vascular events (weighted HR 1.78, 95% CI 1.20–2.63, p = 0.004) and worse 90-day mRS score distribution (OR 1.24, 95% CI 1.10–1.41, p < 0.001) compared to patients without cancer. The 90-day risk of bleeding did not differ between cancer and no-cancer groups overall. When analyzing cancer subgroups, patients with active cancer had significantly higher risk of 90-day ischemic stroke or other vascular (weighted HR 2.75, 95% CI 1.70–4.45, p < 0.001) and any bleeding (weighted HR 2.51, 95% CI 1.27–4.97, p = 0.008) events compared to no-cancer patients. In contrast, patients with cancer in remission had comparable risks to those without cancer. Furthermore, hematological malignancies were associated with a substantially higher risk of 90-day new ischemic stroke or other vascular events compared to solid tumors (weighted HR 8.15, 95% CI 5.06–13.14, p < 0.001).
Conclusions:
Patients with minor ischemic stroke or high-risk TIA and active cancer have increased risk of ischemic and bleeding events after DAPT. Conversely, patients with cancer in remission have similar outcomes compared to those with no cancer.

Keywords
Introduction
Early short-term dual antiplatelet therapy (DAPT) has become a cornerstone in the secondary prevention of new ischemic events in patients who have experienced non-cardioembolic minor ischemic stroke or high-risk transient ischemic attack (TIA).1,2 The combination of aspirin and a P2Y12 inhibitor has been shown to significantly reduce the risk of early vascular recurrences in the general population, particularly within the critical first 21 to 90 days following the index event.3–5 Real-world data indicate that the risk of outcome events in patients with minor ischemic stroke and high-risk TIA receiving DAPT is low. 6 To enhance preventive strategies, it is crucial to identify populations with a higher residual risk. In this context, patients with cancer remain an underexplored group.
Cancer has been associated with both risk of ischemic stroke recurrence and bleeding complications.7,8 Indeed, cancer-associated hypercoagulability, driven by tumor-mediated prothrombotic factors, systemic inflammation, and endothelial dysfunction, significantly increases the risk of ischemic events. This hypercoagulable state may attenuate the efficacy of antiplatelet therapy, thereby complicating efforts to prevent recurrent stroke in this population.7,8 Concurrently, both the presence of cancer and its associated treatments heighten the risk of bleeding,7,8 a concern that is further exacerbated by the use of DAPT. Furthermore, the heterogeneous nature of cancer, characterized by variations in tumor type, stage, and treatment regimens, adds significant complexity to the management of DAPT in this patient population. Hematological malignancies, for example, are associated with a substantially higher thrombotic risk compared to solid tumors, 9 while patients with advanced-stage cancer face a markedly increased risk of bleeding complications. 10
In this study, we aimed to evaluate the impact of cancer on the short-term effectiveness and safety outcomes after DAPT in a real-world cohort of patients with non-cardioembolic minor ischemic stroke or high-risk TIA.
Methods
READAPT study
The READAPT (NCT05476081) is a prospective, observational, multicenter study carried out in real-world clinical settings across Italy. The study was coordinated by the University of L’Aquila and supported by the Italian Stroke Association. It followed the reporting principles of the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for observational research. 11 Participants included adults aged 18 and older who experienced an acute non-cardioembolic ischemic stroke or transient ischemic attack (TIA) and were prescribed dual antiplatelet therapy (DAPT) for up to 90 days. The READAPT study was conducted between February 1, 2021, and February 28, 2023, and spanned 64 stroke centers throughout Italy. Recruitment took place in hospitals, emergency departments, and open-access neurology clinics, aiming for broad representation of the eligible population. Investigators ensured comprehensive inclusion by providing detailed protocols and regular follow-up reminders. Stroke and TIA diagnoses were confirmed according to routine clinical practices and World Health Organization criteria. Eligibility criteria focused solely on patients with acute non-cardioembolic stroke or TIA initiating DAPT, without restrictions regarding the type of antiplatelet agents or the timing of DAPT initiation. The decision to treat the patient with DAPT, and consequently their inclusion in the READAPT study, was made by the treating physician on an individual basis. Physicians were free to make this decision based on their clinical judgment, considering the risk-benefit assessment for each patient. Patients were included whether or not they received a loading dose of DAPT (⩾300 mg for aspirin and clopidogrel or ⩾180 mg for ticagrelor). Exclusion criteria encompassed only patients enrolled in interventional trials at the time of stroke onset, those receiving continuous DAPT for stent-related procedures, and individuals unable to comply with the study protocol. Patients were enrolled soon after the qualifying event and monitored for a follow-up period of 90 ± 10 days through in-person or remote visits. Comprehensive details of the study methodology and definitions used have been published previously.12,13
Study analysis
For the purposes of this exploratory pre-specified analysis, we selected only patients who underwent DAPT according to recommendations from available guidelines.1,2 Specifically, we included patients presenting with minor ischemic strokes (defined by a National Institute of Health Stroke Scale [NIHSS] score of 0 to 5 at admission) or high-risk TIAs, characterized by symptom resolution within 24 h and an ABCD2 score of 4 or higher and no evidence of new ischemic lesion(s) at neuroimaging. Inclusion was limited to those who began dual antiplatelet therapy (DAPT) within 48 h of symptom onset. Conversely, we excluded patients prescribed DAPT following carotid endarterectomy or stenting, as well as those who discontinued DAPT early due to the initiation of anticoagulation therapy. These exclusions were made to minimize potential confounding from procedure-related factors or anticoagulation-associated bleeding risks, which could impact the safety assessment of DAPT.
Patients with cancer where stratified into two groups: those with active cancer and those with cancer in remission. We defined active cancer as (1) a diagnosis of cancer that occurred within 6 months of the index event or during hospitalization, (2) cancer treatment with radiotherapy, chemotherapy or surgery or a combination of them within 6 months of the index event, (3) a previous history of malignancy and a diagnosis of recurrence or metastasis within 6 months of the index event. Cancer in remission was defined as a previous history of malignancy in the absence of active cancer criteria. The site of cancer (non-active or active) was classified as belonging to one of the following categories: gastrointestinal, lung, genitourinary, breast, hematological, skin, other. Definition of cancer was assessed locally by the treating physician and additional investigations were performed as needed at physician’s discretion. Non-systematic assessment was performed to exclude potential occult malignancies.
Study outcomes
The primary effectiveness outcome was the risk of new ischemic stroke or other vascular events (TIA, myocardial infarction, death due to vascular causes – i.e. death caused by stroke, systemic hemorrhage, myocardial infarction, congestive heart failure, pulmonary embolism, sudden death, or arrhythmias) within 90 days. Secondary effectiveness outcomes included the 90-day modified Rankin Scale (mRS) score ordinal distribution and 90-day all-cause mortality. The primary safety outcome was the 90-day risk of any bleeding event, as defined by any category of bleeding severity according to the Global Utilization of Streptokinase and Tissue Plasminogen Activator for Occluded Coronary Arteries (GUSTO) classification. 14 Accordingly, we classified bleeding events as severe if they involved fatal hemorrhage, intracranial hemorrhage, or required blood or fluid replacement, inotropic support, or surgical intervention. Moderate bleeding was defined as requiring a transfusion but not posing a life-threatening risk. Bleeding events that did not meet these criteria were classified as mild. 14 The secondary safety outcome was 24-h hemorrhagic transformation, as defined by any category of the Heidelberg bleeding classification system. 15
Statistical analysis
• Inverse probability weighting
To achieve balanced baseline characteristics between patients with and without cancer, we applied Inverse Probability Weighting to create a pseudo-weighted cohort. Propensity scores were generated using a logistic regression model incorporating pre-defined baseline variables: age, sex, ethnicity, smoking status, arterial hypertension, diabetes, hypercholesterolemia, baseline NIHSS and mRS, presumed stroke/TIA cause, symptoms duration, intravenous thrombolysis, endovascular thrombectomy, DAPT duration and loading dose.
Propensity scores were then used to assign inverse probability weights to each patient. To assess balance between groups, standardized mean differences were calculated for each covariate. We also checked the overlap of propensity score distributions between groups to ensure sufficient comparability and applied stabilized weights to reduce the impact of extreme values and improve the reliability of the model.
• Outcomes analysis in the weighted cohorts
The primary effectiveness outcome was assessed using Cox regression, with hazard ratios (HRs) and 95% confidence intervals (CIs) reported for comparisons between patients with and without cancer. The 90-day shift in the distribution of modified Rankin Scale (mRS) scores was analyzed using an ordinal generalized linear model and presented as odds ratio (OR) with corresponding 95% CIs. Additionally, HRs with 95% CIs were calculated for the 90-day risk of all-cause mortality. The primary safety outcome was similarly evaluated using Cox regression to compare risk between the cancer and no-cancer groups. Risk difference (95% CIs) was calculated for 24-h hemorrhagic transformation. Weighted Kaplan-Meier cumulative function was used to visualize the overall risk for both primary effectiveness and safety outcomes, with the log-rank test applied to determine the statistical significance of differences between groups. Competing risk analysis was also employed to estimate the 90-day cumulative incidence of primary effectiveness and safety outcomes at 90 days, with death as a competing event which prevents the occurrence of those outcomes. Gray test was used to assess the equality of cumulative incidence functions for primary effectiveness and safety outcomes and all-cause death between the cancer and no-cancer groups. Furthermore, the 90-day risks of primary effectiveness and safety outcomes were compared among patients with active cancer, cancer in remission, and those without cancer using Cox regression models and Kaplan-Meier cumulative hazard curves. To assess the 90-day distribution of mRS scores between patients with active and non-active cancer, we employed an ordinal generalized linear model. Lastly, we compared the 90-day risks of primary effectiveness and safety outcomes between patients with solid (gastrointestinal, genitourinary, breast, skin and other) versus hematological cancers using the same statistical approach. As the outcomes of the present analysis were exploratory and no prior assumptions were made, a sample size was not prespecified.
All statistical analyses were conducted using R software (version 4.2.2). Statistical significance was set at a p-value of <0.05.
Results
Of the 2278 patients in the READAPT study cohort, 1561 were included in the analysis (mean age: 70.3 ± 11.7 years, 65.4% males). Among these, 71.6% had minor ischemic stroke, while 28.4% experienced high-risk TIA. The number of excluded patients with reasons is reported in the study flow-chart (Figure 1).

Study flow-chart.
Within our sample, 206 patients (13.2%) had cancer. Specifically, 149 patients (72.3%) had cancer in remission while 57 (27.7%) had active cancer. Regarding the cancer site, 26 patients (12.6%) had gastrointestinal cancer, 9 patients (4.4%) had lung cancer, 43 patients (20.9%) had genitourinary cancer, 39 patients (18.9%) had breast cancer, 21 patients (10.2%) had hematological cancer, 13 patients (6.3%) had skin cancer, and 55 patients (26.7%) had cancer at other sites. Among the 57 patients with active cancer, 7 patients (12.3%) had gastrointestinal cancer, 3 patients (5.3%) had lung cancer, 11 patients (19.3%) had genitourinary cancer, 9 patients (15.8%) had breast cancer, 12 patients (21.1%) had hematological cancer, 4 patients (7.0%) had skin cancer, and 11 patients (19.3%) had cancer at other sites (Table 1).
Baseline characteristics.
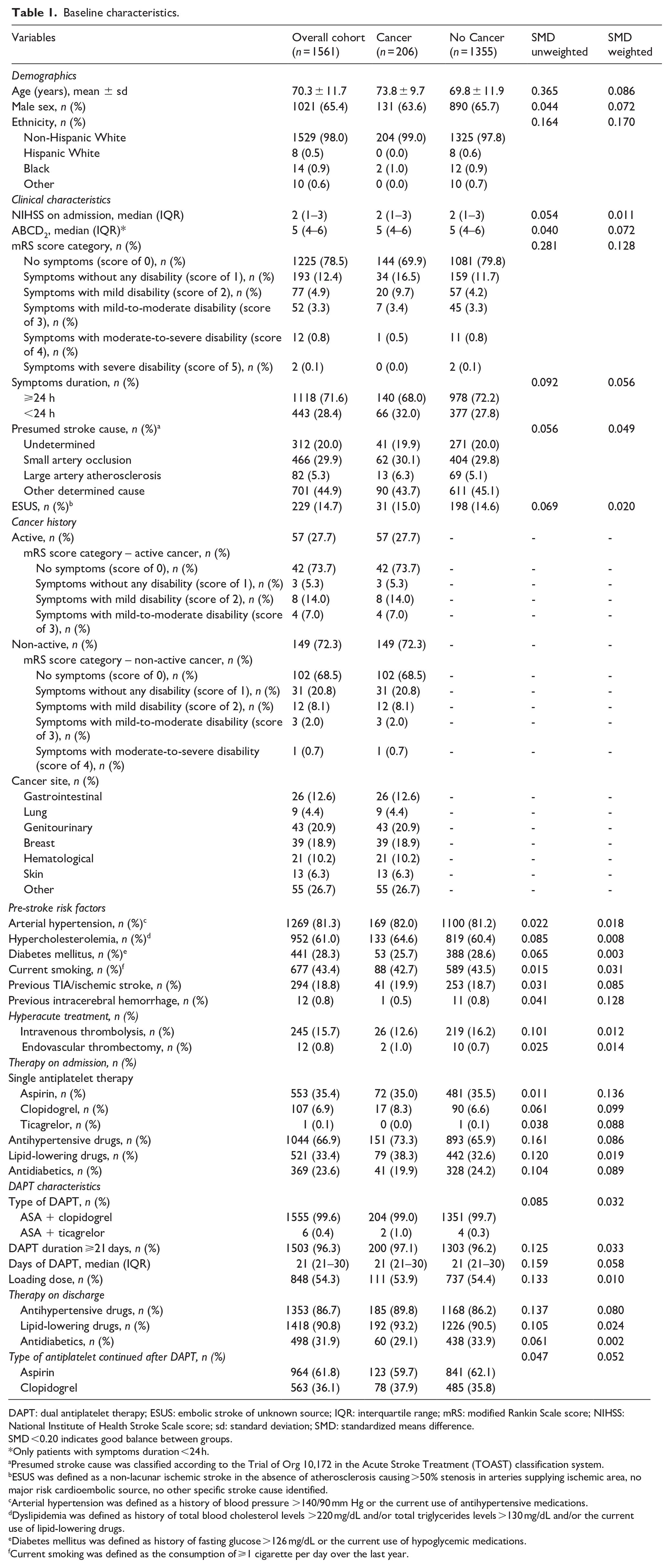
DAPT: dual antiplatelet therapy; ESUS: embolic stroke of unknown source; IQR: interquartile range; mRS: modified Rankin Scale score; NIHSS: National Institute of Health Stroke Scale score; sd: standard deviation; SMD: standardized means difference.
SMD <0.20 indicates good balance between groups.
Only patients with symptoms duration <24 h.
Presumed stroke cause was classified according to the Trial of Org 10,172 in the Acute Stroke Treatment (TOAST) classification system.
ESUS was defined as a non-lacunar ischemic stroke in the absence of atherosclerosis causing >50% stenosis in arteries supplying ischemic area, no major risk cardioembolic source, no other specific stroke cause identified.
Arterial hypertension was defined as a history of blood pressure >140/90 mm Hg or the current use of antihypertensive medications.
Dyslipidemia was defined as history of total blood cholesterol levels >220 mg/dL and/or total triglycerides levels >130 mg/dL and/or the current use of lipid-lowering drugs.
Diabetes mellitus was defined as history of fasting glucose >126 mg/dL or the current use of hypoglycemic medications.
Current smoking was defined as the consumption of ⩾1 cigarette per day over the last year.
Inverse probability weighting
Weighted and unweighted baseline characteristics are summarized in Table 1. No missing data were recorded in the variables used to calculate the propensity scores, nor in any baseline variables within the weighted and unweighted cohorts. Overall, a satisfactory balance was achieved for all baseline variables which showed a standardized difference (SD) of propensity scores below 0.20. Balance diagnostics indicated that the samples were well-weighted, with a mean standardized difference in propensity scores between groups of 0.03 (acceptable balance < 0.25) 16 and a variance ratio of propensity scores of 1.15 (acceptable range: 0.5–2). 16 Graphical representations of propensity scores and covariate balance distributions further confirmed the overall quality of the weighting process (Supplemental Figures S1 and S2).
Primary effectiveness outcome
Within the weighted cohorts, patients with concomitant cancer showed a higher risk of 90-day new ischemic stroke or other vascular events compared to those without cancer (5.8% vs 3.0%; weighted HR 1.78 [95% CI 1.20–2.63]; p = 0.004; Table 2). Weighted Kaplan-Meier cumulative function showed that the overall incidence of new ischemic stroke and other vascular events was significantly higher among patients with concomitant cancer versus no cancer (long-rank p = 0.004; Figure 2(a)). Competing risk analysis confirmed the higher 90-day cumulative incidence of primary effectiveness outcome among patients with cancer (Supplemental Figure S3). Gray test showed that cumulative incidence curves were statistically different for primary effectiveness outcome (p = 0.011) but not for all-cause death (p = 0.117).
Outcomes comparison.

DAPT: dual antiplatelet therapy; mRS: modified Rankin Scale.
Statistically significant p values (<0.05) are reported in bold.
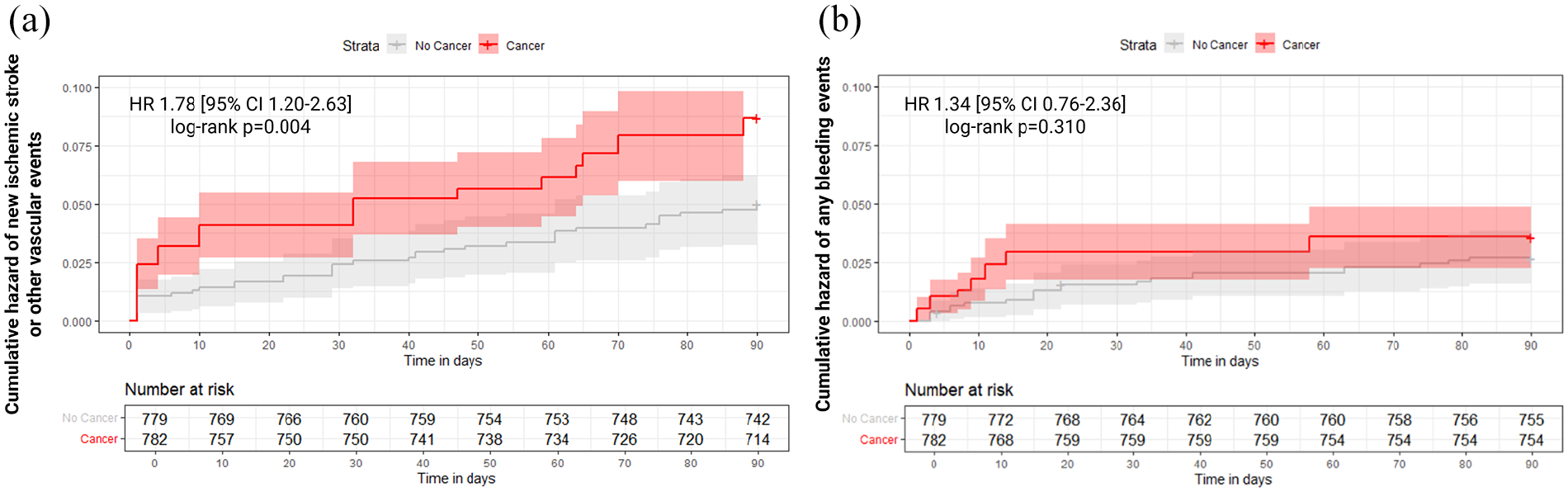
Weighted Kaplan-Meier cumulative function of 90-day (a) primary effectiveness and (b) safety outcomes by cancer status. CI: confidence intervals; HR: hazard ratio. Shaded areas indicate 95% confidence intervals. The estimates and the number at risk shown below the curves reflect the weighted pseudo-population.
Secondary effectiveness outcomes
Patients with concomitant cancer had less favorable 90-day mRS score distribution compared to those without cancer (weighted OR 1.24 [95% CI 1.10–1.41]; p < 0.001; Table 2, Figure 3). Conversely, there was no significant difference between groups in terms of 90-day all-cause mortality (2.4% vs 0.7%; weighted HR 1.74 [95% CI 0.51–5.96; p = 0.375; Table 2).
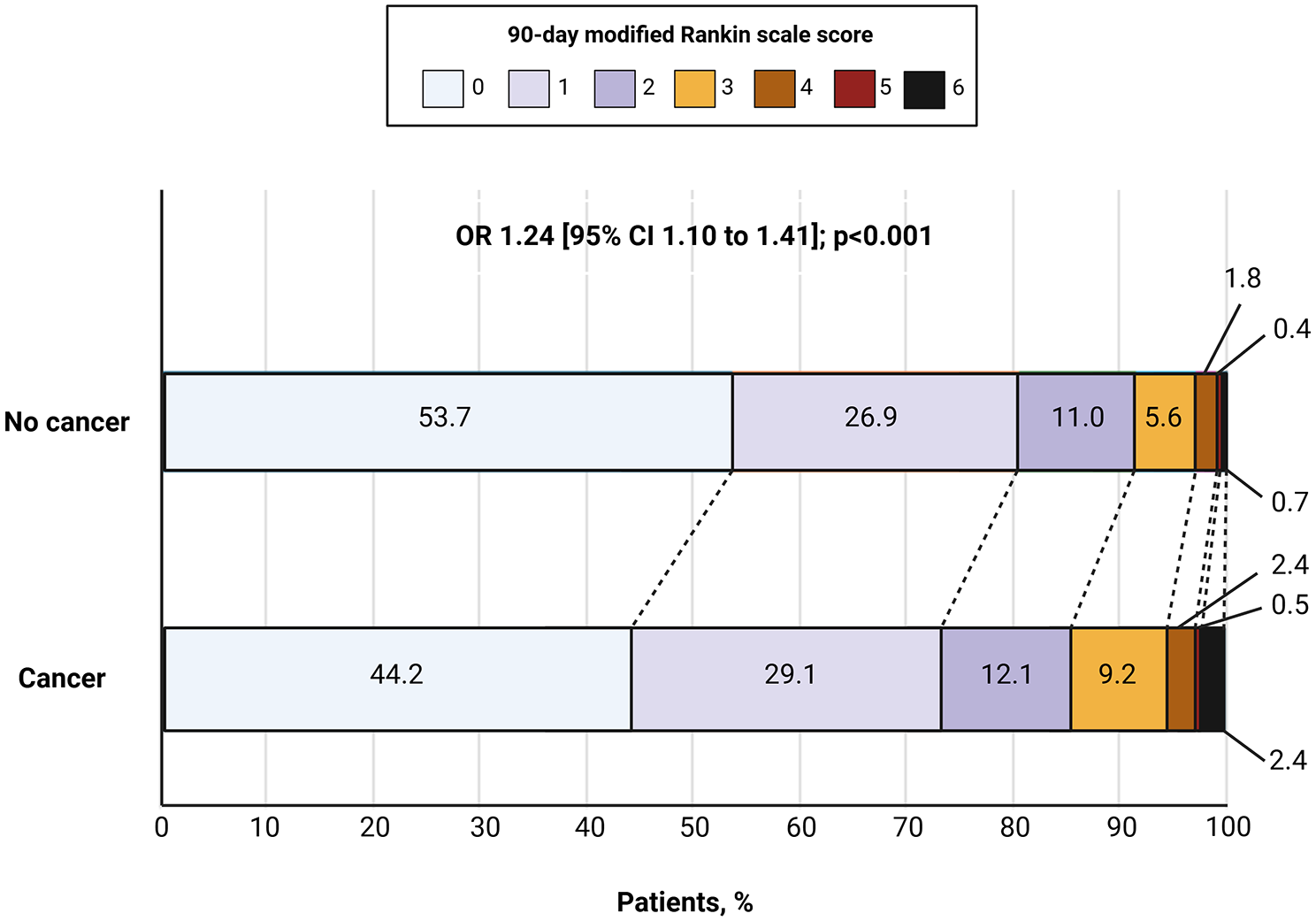
90-day modified Rankin Scale (mRS) Score ordinal distribution by cancer status in the weighted cohorts. CI: confidence interval; OR: odds ratio (cancer vs no cancer).
Primary safety outcome
The risk of 90-day bleeding events was similar between patients with and without concomitant cancer (3.4% vs 2.7%; weighted HR 1.34 [95% CI 0.76–2.36]; p = 0.310; Table 2). Weighted Kaplan-Meier cumulative function (long-rank p = 0.300) and competing risk analysis (Gray test p = 0.787 for primary safety outcome and p = 0.118 for all-cause death) confirmed the similar incidence of bleedings (Figure 2(b), Supplemental Figure S4). The majority of bleeding events observed in both patients with and without cancer were mild-to-moderate (85.7% vs 83.7%, respectively), while severe bleedings occurred in a single patient with cancer (14.3%) and in five patients without cancer (16.3%). There was no significant difference in bleeding severity between groups (p = 0.898).
Secondary safety outcome
We observed no significant difference in the risk of 24-h hemorrhagic transformation between patients with and without cancer (0.5% vs 1.0%; weighted risk difference −0.5% [95% CI −0.2% to 0.4%]; p = 0.244; Table 2).
Active cancer and cancer in remission versus no cancer
Patients with active cancer showed a significantly higher risk of 90-day ischemic stroke or other vascular events and any bleeding events compared to those without cancer (7.0% vs 2.7%, weighted HR 2.75 [95% CI 1.70–4.45]; p < 0.001 and 7.0% vs 2.7%, weighted HR 2.51 [95% CI 1.27–4.97]; p = 0.008, respectively), while those with cancer in remission had similar risks of both outcomes to their counterparts without cancer (5.4% vs 2.7%, weighted HR 1.26 [95% CI 0.80–1.99; p = 0.311 and 2.0% vs 2.7%, weighted HR 0.55 [95% CI 0.24–1.24; p = 0.184, respectively; Supplemental Table S1 and Figure S5). The 90-day mRS score distribution was similar between patients with active cancer and those with cancer in remission (OR 1.14 [95% CI 0.92–1.41; p = 0.230).
Hematological versus solid cancer
Patients with hematological malignancies had a higher risk of 90-day new ischemic stroke or other vascular events compared to those with solid tumors (23.8% vs 3.8%, weighted HR 8.15 [95% CI 5.06–13.14; p < 0.001), while the 90-day risk of any bleeding events was similar between the two groups (4.8% vs 3.2%, weighted HR 1.27 [95% CI 0.44–3.66]; p = 0.657; Supplemental Table S2 and Figure S6). Of note, the proportion of patients with active cancer was significantly higher in patients with hematological malignancies compared to solid tumors (12/21, 57.1% vs 45/185, 24.3%; p = 0.029).
Discussion
This secondary analysis of the READAPT study evaluated the impact of cancer on the 90-day effectiveness and safety outcomes after DAPT in patients with non-cardioembolic minor ischemic stroke or high-risk TIA. Our findings indicate that patients with cancer have a worse 90-day functional outcome and a significantly higher risk of new ischemic events after DAPT compared to those without cancer. Notably, patients with active malignancy had not only higher risk of 90-day ischemic stroke or other vascular events but also an increased risk of bleedings. Patients with cancer in remission had similar risks to non-cancer patients. Additionally, hematological malignancies were associated with a higher risk of ischemic stroke recurrence compared to solid tumors. These findings highlight the need to further investigate better secondary prevention strategies in active cancer patients, given their increased risk of both ischemic and hemorrhagic events.
Cancer patients with ischemic stroke or TIA present a clinical challenge due to the heightened risks of both thrombosis and bleeding.7,8 Malignancy-driven prothrombotic states, influenced by tumor-related factors and cancer therapies, contribute to ischemic recurrences. Endothelial injury promotes the release of pro-thrombotic mediators, such as von Willebrand factor, enhancing platelet aggregation. 17 Histopathological studies of thrombi in cancer-associated stroke further highlight the role of platelets and thrombin in stroke pathogenesis.18,19 Existing research has primarily focused on comparing anticoagulation with antiplatelet therapy for stroke prevention in cancer patients.20–22 A retrospective study of 263 stroke patients with active cancer found similar recurrent thromboembolism rates between antiplatelet and anticoagulant therapy. 20 Likewise, post-hoc analyses of the Atrial Cardiopathy and Antithrombotic Drugs in Prevention After Cryptogenic Stroke (ARCADIA) and New Approach Rivaroxaban Inhibition of Factor Xa in a Global Trial versus Aspirin to Prevent Embolism in Embolic Stroke of Undetermined Source (NAVIGATE ESUS) showed comparable ischemic stroke recurrence rates between apixaban or rivaroxaban and aspirin in ESUS patients with cancer.21,22
Our findings suggests that the pro-thrombotic state associated with cancer may impair DAPT’s effectiveness in preventing recurrent ischemic cerebrovascular events, leading to worse short-term functional outcome. However, our study cannot fully determine whether these recurrences are primarily driven by cancer itself or conventional cardiovascular risk factors. Notably, ESUS prevalence was comparable between cancer and non-cancer patients in our cohort (15.0% vs 14.6%), suggesting that ischemic risk persists across stroke etiologies.
Regarding bleeding risk, cancer patients face a major bleeding incidence of up to 20% annually, further heightened in those with stroke or brain pathologies. 23 Major bleeding is a key predictor of mortality, particularly in older individuals, those with multiple comorbidities, and patients with gastrointestinal/genitourinary cancers or metastatic disease - common features of cancer-related stroke. 23 In our cohort, active cancer patients had a significantly higher 90-day bleeding risk than non-cancer patients, though most events were mild to moderate. While our analysis does not provide definitive guidance on optimal antithrombotic strategies, it underscores the importance of individualized bleeding risk assessment to inform safe secondary prevention therapy.
Our findings also highlight the elevated ischemic stroke risk in patients with hematological malignancies compared to solid tumors. While all cancer patients are at increased thromboembolic risk, this risk is particularly high in hematological malignancies such as multiple myeloma, leukemia, and lymphoma.24,25 The mechanisms vary based on comorbidities, cancer type, and treatment. For example, acute promyelocytic leukemia is frequently associated with disseminated intravascular coagulation, 26 while therapies like asparaginase, tyrosine kinase inhibitors, and immunomodulatory agents increase thrombotic risk through mechanisms that remain incompletely understood.27–29 Although we did not collect detailed data on cancer stage and treatment, the higher prevalence of active cancer among patients with hematological malignancies compared to those with solid tumors within our cohort (57.1% vs 24.3%) may have contributed to their elevated risk of ischemic recurrences, potentially also driven by concurrent exposure to prothrombotic chemotherapies. These patients are also prone to bleeding due to thrombocytopenia. 9 Interestingly, in our study, the 90-day bleeding risk did not differ between hematological and solid tumor patients treated with DAPT. This may reflect rigorous patient selection and close monitoring, mitigating bleeding risk. However, our study’s short follow-up could underestimate cumulative bleeding risk. Additionally, hematological malignancies comprised only 10.2% of our cancer cohort, and we lacked detailed data on thrombocytopenia, limiting subgroup analysis. Therefore, these findings require confirmation in larger prospective cohorts.
Strengths and limitations
The main strength of this study lies in the rigorous procedures implemented to enhance data accuracy and quality, as ensured by regular quality checks of the READAPT electronic database. However, our findings should be interpreted in lights of some relevant limitations. First, despite the use of rigorous inverse probability weighting and adjustment for covariates, residual confounding cannot be fully excluded due to the observational design of the READAPT study. Importantly, we lacked data on cancer stage, laboratory findings, and concurrent treatments, which may have influenced DAPT outcomes. This is particularly relevant for safety assessments, as advanced-stage cancer and chemotherapy might increase the risk of thrombocytopenia and bleeding, especially in patients with active or hematologic malignancies. Furthermore, certain chemotherapeutic agents, particularly those used to treat hematological malignancies, may heighten the risk of ischemic events, as previously discussed. Second, although the vast majority (>95%) of patients in our cohort received DAPT for at least 21 days (Table 1), we did not capture the proportion of cancer patients who discontinued the antiplatelet agent prescribed after DAPT during the follow-up period, either due to anticancer treatment or surgical procedures, which may have impacted on the rate of short-term ischemic recurrences. Similarly, temporary DAPT adjustments due to cytopenia or other cancer treatment-related complications were not accounted for. Third, we were unable to evaluate the impact of frailty or cachexia, both of which may significantly affect clinical outcomes in this high-risk population. Fourth, we cannot rule out the potential presence of occult cancers in the no-cancer group. While this limitation may reduce the strength of the observed associations, it is unlikely to affect their overall direction. Fifth, although all patients underwent brain imaging (non-contrast computed tomography or magnetic resonance imaging) upon admission, we did not collect detailed neuroimaging findings or results from brain vessel examinations. As a result, the stroke diagnosis and presumed cause relied solely on assessments by local investigators. Similarly, the decision to treat patients with DAPT was made by treating physicians on a case-by-case basis, considering the individual risk of bleeding or thromboembolic events. Therefore, we cannot exclude the possibility of selection bias, as some cancer patients at higher risk of bleedings may not have been treated with DAPT and were consequently not included in the READAPT study. Also, investigators were not blinded to cancer status, which may have introduced detection bias, especially for subjective outcomes such as the mRS score. Sixth, patients who discontinued DAPT early due to a need for anticoagulation were excluded from this analysis. Although this subset represented only <2% of the initial READAPT population (Figure 1) and no patient with active cancer or cancer in remission transitioned to anticoagulation, we did not record their 90-day outcomes. Lastly, our cohort showed a significant imbalance toward non-Hispanic white participants, which may limit the generalizability of our findings to other ethnic groups.
In conclusion, our results indicate an increased risk of ischemic and bleeding events in patients with minor ischemic stroke or high-risk TIA and active cancer treated with early short-term DAPT, compared to their counterparts without cancer. In contrast, patients with cancer in remission had similar outcomes compared to patients without cancer. The risk of new ischemic stroke and other vascular events is particularly evident in patients with hematological malignancies. Given the high thromboembolic risk, there is the need of further studies to investigate additional strategies. Future research should explore nuanced, cancer-specific strategies for secondary stroke prevention, including optimizing DAPT intensity or duration, evaluating alternative antithrombotic approaches such as anticoagulation, or combining antiplatelet agents with therapies targeting cancer-associated thromboinflammatory pathways. Additionally, integrating baseline coagulation profiles and cancer-specific risk factors – such as tumor type, stage, and treatment status – into stroke prevention protocols may enable more personalized and effective risk reduction strategies.
Supplemental Material
sj-pdf-1-eso-10.1177_23969873251333282 – Supplemental material for Effectiveness and safety of dual antiplatelet therapy in patients with minor ischemic stroke or transient ischemic attack and cancer: A secondary analysis of the READAPT study
Supplemental material, sj-pdf-1-eso-10.1177_23969873251333282 for Effectiveness and safety of dual antiplatelet therapy in patients with minor ischemic stroke or transient ischemic attack and cancer: A secondary analysis of the READAPT study by Matteo Foschi, Federico De Santis, Lucio D’Anna, Michele Romoli, Tiziana Tassinari, Valentina Saia, Silvia Cenciarelli, Chiara Bedetti, Chiara Padiglioni, Bruno Censori, Valentina Puglisi, Luisa Vinciguerra, Maria Guarino, Valentina Barone, Marialuisa Zedde, Ilaria Grisendi, Marina Diomedi, Maria Rosaria Bagnato, Marco Petruzzellis, Domenico Maria Mezzapesa, Vincenzo Inchingolo, Manuel Cappellari, Cecilia Zivelonghi, Paolo Candelaresi, Vincenzo Andreone, Giuseppe Rinaldi, Alessandra Bavaro, Anna Maria Cavallini, Maria Grazia Piscaglia, Valeria Terruso, Marina Mannino, Alessandro Pezzini, Giovanni Frisullo, Francesco Muscia, Maurizio Paciaroni, Maria Giulia Mosconi, Andrea Zini, Ruggiero Leone, Carmela Palmieri, Letizia Maria Cupini, Michela Marcon, Rossana Tassi, Enzo Sanzaro, Giulio Papiri, Giovanna Viticchi, Daniele Orsucci, Anne Falcou, Susanna Diamanti, Roberto Tarletti, Patrizia Nencini, Eugenia Rota, Federica Nicoletta Sepe, Delfina Ferrandi, Luigi Caputi, Gino Volpi, Salvatore Laspada, Mario Beccia, Claudia Rinaldi, Vincenzo Mastrangelo, Francesco Di Blasio, Paolo Invernizzi, Giuseppe Pelliccioni, Maria Vittoria De Angelis, Laura Bonanni, Giampietro Ruzza, Emanuele Alessandro Caggia, Monia Russo, Agnese Tonon, Maria Cristina Acciarri, Sabrina Anticoli, Cinzia Roberti, Giovanni Manobianca, Gaspare Scaglione, Francesca Pistoia, Alberto Fortini, Antonella De Boni, Alessandra Sanna, Alberto Chiti, Marcella Caggiula, Maela Masato, Massimo Del Sette, Francesco Passarelli, Maria Roberta Bongioanni, Manuela De Michele, Stefano Ricci, Eleonora De Matteis, Raffaele Ornello and Simona Sacco in European Stroke Journal
Footnotes
Acknowledgements
None.
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Piscaglia reports grants from Sanofi Genzyme; grants from Roche Health Solutions Inc.; grants from Novartis Pharma; grants from Biogen; and grants from Merck Company Foundation. Dr Paciaroni reports compensation from Boehringer Ingelheim for other services; compensation from PFIZER CANADA INC for other services; compensation from Bristol-Myers Squibb for other services; compensation from iRhythm Technologies for other services; compensation from SANOFI-AVENTIS U.S. LLC for other services; and compensation from Daiichi Sankyo Europe GmbH for other services. Dr Zini reports compensation from Bayer Healthcare for other services; compensation from Boehringer Ingelheim for consultant services; compensation from Alexion Pharmaceuticals for consultant services; and compensation from CSL Behring for consultant services. Dr Ornello reports grants from Novartis; compensation from Teva Pharmaceutical Industries for other services; compensation from AbbVie for data and safety monitoring services; compensation from Teva Pharmaceutical Industries for other services; compensation from Eli Lilly and Company for other services; compensation from Novartis for other services; compensation from H. Lundbeck AS for other services; compensation from Eli LIlly for data and safety monitoring services; grants from Pfizer; grants from Allergan; travel support from Teva Pharmaceutical Industries; and compensation from Teva Pharmaceutical Industries for consultant services. Prof Sacco reports compensation from Novartis for other services; compensation from Novo Nordisk for consultant services; compensation from Boehringer Ingelheim for consultant services; compensation from Teva Pharmaceutical Industries for consultant services; compensation from Allergan for consultant services; employment by Università degli Studi dell’Aquila; compensation from Novartis for consultant services; compensation from Allergan for consultant services; compensation from PFIZER CANADA INC for consultant services; compensation from Abbott Canada for consultant services; compensation from H. Lundbeck A S for consultant services; compensation from AstraZeneca for consultant services; and compensation from Eli Lilly and Company for consultant services. The other authors report no conflicts.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Consent to participate
All participants gave written informed consent to be enrolled in the study according to the Declaration of Helsinki.
Ethical considerations
The READAPT study received approval from the Internal Review Board of the University of L’Aquila in February 2021 (code 03/2021).
Guarantor
Professor Simona Sacco, MD, serves as the guarantor for this manuscript. As such, she accepts full responsibility for the integrity of the work, including the data and the accuracy of its analysis, and ensures that all aspects of the manuscript adhere to the highest standards of scientific rigor and ethical conduct.
Contributorship
Prof Sacco, Dr Ornello and Dr De Matteis designed and coordinated the study. Dr Foschi and Dr D’Anna wrote and edited the manuscript. Dr Foschi performed data analysis. Prof Sacco, Dr Ornello, Dr D’Anna, Dr Foschi, Dr De Matteis, and Dr De Santis contributed to the interpretation of data. All authors contributed to the acquisition of data and critically read and revised the manuscript before submission.
ORCID iDs
Data availability
The complete dataset used for this study will be shared upon request from any qualified researcher to the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
