Abstract
Background:
Time elapsed from stroke onset and baseline infarct volume is influential on endovascular thrombectomy (EVT) outcomes.
Objectives:
This study aimed to explore the utility of early infarct growth rate (EIGR) measured by apparent diffusion coefficient (ADC) in predicting symptomatic intracranial hemorrhage (sICH) of ischemic stroke patients after EVT.
Methods:
We retrospectively analyzed patients from the prospectively maintained stroke registry admitted between January 2019 and March 2023, presenting with large vessel occlusive stroke in the anterior circulation. EIGR was defined as ischemic core volume on magnetic resonance perfusion imaging (ADC ⩽620 × 10−6 mm2/s) divided by the time from stroke onset to imaging. sICH was diagnosed according to the Heidelberg Bleeding Classification within 72 h after the procedure.
Results:
A total of 315 patients met the inclusion criteria. We observed sICH in 36 (11.4%) patients. After adjusting for the potential confounders, increased EIGR was confirmed to be independently associated with a higher risk of sICH (adjusted odds ratio, 1.033; 95% confidence interval (CI), 1.018–1.048; p = 0.001). Similar results were also confirmed when EIGR was analyzed as a categorical variable. Using a logistic regression model with restricted cubic splines, we found a linear correlation between EIGR and sICH risk (p = 0.001 for linearity). Furthermore, adding EIGR to a model containing conventional risk factors significantly improved risk reclassification for sICH (category-free net reclassification index, 0.393; 95% CI, 0.227–0.560; p = 0.001; integrated discrimination improvement, 0.245; 95% CI, 0.146–0.343; p = 0.001).
Conclusion:
Increased EIGR may predict the sICH in ischemic stroke patients who receiving EVT.
Introduction
Endovascular thrombectomy (EVT) is the standard of care for ischemic stroke patients with large vessel occlusion in the anterior circulation because it has been robustly correlated with both reperfusion rate increase and significant functional improvement.1–3 However, despite its efficacy, EVT is more likely to induce subsequent symptomatic intracranial hemorrhage (sICH) than best medical therapy. 4 The incidence of sICH was reported to be up to 14.0% of an Asian population, 5 which might reduce the benefit–risk ratio of the EVT treatment.6–8 Therefore, it is of great vital to identify the factors associated with sICH development to alleviate the subsequent sICH and improve functional outcomes.
Following large vessel occlusion, growth of the irreversibly injured ischemic brain tissue (ischemic core) occurs at different speeds across individuals, with some patients achieving large infarcts within a very short period while others can have small stroke sizes despite longer times. 9 The early infarct growth rate (EIGR) captures both initial infarct volume based on perfusion imaging and time from stroke onset to imaging acquisition (defined as ischemic core volume on baseline perfusion imaging divided by onset-to-imaging time), 10 which might represent the stroke evolution. Recent studies have evaluated the prognostic implications of EIGR in large vessel occlusive stroke patients.10–12 Data from the International Stroke Perfusion Imaging Registry (INSPIRE study) showed that the EIGR was a major modifier of the therapeutic effect of EVT. 11 Also, in a prospective multicenter cohort study of imaging selection for EVT (SELECT study), the EIGR was confirmed to be strongly correlated with both collateral status and clinical outcomes after EVT. 10 However, the relationship between EIGR and sICH risk has not been clarified to date. Moreover, most previous studies were limited by infarct volumes measured on computed tomography perfusion imaging, which is less precise than magnetic resonance imaging (MRI).
In this study, we aimed to investigate whether EIGR calculating from apparent diffusion coefficient (ADC) on magnetic resonance perfusion correlated with the sICH in large vessel occlusive stroke patients after EVT.
Materials and methods
Study design and patients’ selection
This was a retrospective analysis conducted on a prospectively collected cohort of consecutive ischemic stroke patients treated with EVT at Nanjing First Hospital. For this study, we included consecutive EVT-treated patients from January 2019 to May 2023, fulfilling the following criteria: (1) older than 18 years; (2) acute anterior circulation large vessel occlusion (defined as occlusion of the internal carotid artery, M1 or M2 segments of the middle cerebral artery); (3) available good-quality pretreatment MRI perfusion-weighted imaging. Patients were excluded from the study if the following criteria were met: (1) treated with devices other than a stent-like retriever and aspiration system; (2) unable to perform the follow-up NCCT; (3) diagnosed with a concomitant aneurysm, arteriovenous malformation, moyamoya disease, or hematological system diseases.
Clinical assessment
Demographic characteristics and clinical variables were collected by investigators blinded to the outcomes of interest. In particular, we obtained data on age, sex, medical history, stroke severity, Alberta Stroke Program Early CT Score (ASPECTS), stroke etiology, intravenous fibrinolysis with recombinant tissue plasminogen activator (rt-PA) before EVT, occlusive vessel, collateral status, vessel recanalization status, and laboratory data. Stroke severity was measured by the National Institutes of Health Stroke Scale (NIHSS). 13 The Trial of Org10172 in Acute Stroke Treatment (TOAST) classification was used to classify stroke types. 14 The pretreatment collateral status was evaluated using the DSA-based American Society of Interventional and Therapeutic Neuroradiology/Society of Interventional Radiology collateral flow grading system, with grades 0–1 indicating poor collateral status. 15 Successful reperfusion was defined as modified thrombolysis in cerebral infarction grade of 2b or 3 on angiographic images. 16
Calculation of infarct growth rate
All patients have performed the MRI examination (including diffusion-weighted imaging and perfusion-weighted imaging) before EVT treatment. The EIGR was calculated as follows: initial ischemic core volume/time from stroke onset to imaging acquisition. 10 Ischemic core volume was determined by ADC ⩽620 × 10−6 mm2/s on diffusion-weighted imaging using automated software. For “wake-up” stroke patients, the last moment the patient was known to be well was used to determine stroke onset time.
Outcome measures
The follow-up NCCT was usually performed 24–72 h after EVT. In this study, sICH was confirmed according to the Heidelberg Bleeding Classification,5,6,17 and defined as the primary outcome measure. The radiological images were reviewed in consensus readings by two trained neuroradiologists who blinded to the clinical data. Functional outcomes assessed by the 90-day modified Rankin Scale (mRS) score were defined as the secondary outcome measure.
Statistical analysis
Data are summarized as number (%), mean standard deviation, or median (interquartile range (IQR)). Comparisons of variables between the groups with or without sICH were conducted using Pearson’s Chi-square test or Fisher’s exact method for categorical variables, and Student’s t-test or the Mann–Whitney U test for continuous variables, as appropriate.
Multivariable regression logistical models were used to evaluate the independent effect of EIGR on sICH. Model 1 was adjusted for age and sex. Model 2 was adjusted for model 1 adding with variables with p value <0.1 in the univariate analysis, including pretreatment ASPECTS, poor collateral status, and baseline glucose levels. Sensitivity analysis was also conducted using subgroups consisting of patients with exact time of stroke onset to test the robustness of our findings. An odds ratio (OR) with a 95% confidence interval (CI) was calculated. The pattern and magnitude of the correlation between EIGR and sICH using a logistic regression model with restricted cubic splines with three knots (at 5th, 50th, and 95th percentiles) adjusted for covariates included in model 2. 18 Furthermore, the net reclassification index (NRI) and integrated discrimination improvement (IDI) were performed to evaluate the predictive value of adding the EIGR into model 1 and model 2, separately. A two-sided p value < 0.05 was considered statistically significant. All statistical analyses were performed using R software (version 4.3.1; R Foundation, Vienna, Austria).
Results
Patients population
Between January 2019 and March 2023, 315 eligible patients (63.7% male, mean age 68.7 ± 11.8 years) who received thrombectomy were included in this analysis.
Among these patients, median NIHSS was 13.0 (IQR, 8.0–16.0), median ASPECTS was 9.0 (IQR, 8.0–9.0), median ischemic core volume was 19.0 mL (IQR, 6.0–56.4), and median EIGR was 4.2 mL/h (IQR, 1.1–15.3). Furthermore, 132 (41.9%) patients received intravenous thrombolysis before the EVT procedure, 160 patients (50.8%) showed poor collateral status at baseline, and 276 patients (87.6%) obtained successful reperfusion.
Patient characteristics stratified by tertile of EIGR
Figure 1 represents calculating EIGR for three illustrative cases. Overall, these patients had a median EIGR of 4.2 mL/h (IQR, 1.1–15.3) (Supplemental Figure 1). The cut-off of EIGR tertile was as follows: <1.8 mL/h (first tertile), 1.8–10.5 mL/h (second tertile), and >10.5 mL/h. The patient characteristics stratified by tertile of EIGR were demonstrated in Supplemental Table 1. As compared with patients with first tertile EIGR, those with third tertile of EIGR had higher NIHSS (median, 14.0 vs 10.0; p = 0.001), higher baseline blood glucose levels (mean, 7.6 vs 6.6 mmol/L; p = 0.016), lower pretreatment ASPECTS (median, 8.0 vs 9.0; p = 0.001), shorter time from onset to MRI acquisition (median, 2.3 vs 7.3 h; p = 0.001), larger core volume on ADC (median, 64.0 vs 4.0 mL; p = 0.001), more proximal clot location (internal carotid artery, 41.0% vs 28.6%; p = 0.006), higher t-PA rates (52.4% vs 31.4%; p = 0.009), and higher prevalence of poor collateral status (61.0% vs 47.6%; p = 0.033) and sICH (24.8% vs 3.8%; p = 0.001). Age, sex, and vascular risk factors distribution were similar among the subgroups based on the tertile of EIGR.

Representative images of three illustrative cases and calculating EIGR. (a) A 63-year-old patient with an ischemic lesion in the right ICA territory; sICH was not found in follow-up NCCT. ADC ⩽620 × 10−6 mm2/s = 54 mL, time from onset to imaging = 9.50 h, EIGR = 54/9.50 = 5.7 mL/h. (b) A 57-year-old patient with an ischemic lesion in the right ICA territory; sICH was observed in follow-up NCCT. ADC ⩽620 × 10−6 mm2/s = 23 mL, time from onset to imaging = 2.25 h, EIGR = 23/2.25 = 10.2 mL/h. (c) An 88-year-old patient with an ischemic lesion in the left ICA territory; sICH was observed in follow-up NCCT. ADC ⩽620 × 10−6 mm2/s = 110 mL, time from onset to imaging = 2.22 h, EIGR = 110/2.22 = 48.9 mL/h.
The effect of EIGR on sICH
Within 72 h after EVT, 36 patients (11.4%) developed sICH. Table 1 showed the comparison between patients with and without sICH. There were significantly higher values in the following variables in the sICH than in the no-sICH group: baseline blood glucose levels (mean, 8.2 vs 7.3 mmol/L; p = 0.009), ischemic core volume measured by ADC (median, 67.0 vs 17.0 mL; p = 0.048), and EIGR (median, 17.7 vs 3.5 mL/h; p = 0.001). Moreover, compared with the patients without sICH, those with sICH had a higher prevalence of poor collateral status rate (83.3% vs 46.6%; p = 0.007) and lower baseline ASPECTS (median, 8.5 vs 9.0; p = 0.036).
Baseline characteristics of ischemic stroke patients with and without sICH after EVT.

ADC, Apparent diffusion coefficient; ASPECTS, the Alberta Stroke Program Early Computed Tomography Score; CE, cardio-embolism; EIGR, Early infarct growth rate; EVT, Endovascular thrombectomy; Hs-CRP, Hyper-sensitive C-reactive protein; IVT, Intravenous thrombolysis; LAA, large-artery atherosclerosis; MRI, Magnetic resonance imaging; NIHSS, National institute of health stroke scale; sICH, Symptomatic intracranial hemorrhage; TOAST, Trial of Org10172 in Acute Stroke Treatment.
In the multivariable regression analysis, EIGR independently and positively correlated with sICH (OR, 1.033; 95% CI, 1.018–1.048; p = 0.028; Table 2, Model 2) after adjusting for demographic characteristics, pretreatment ASPECTS, poor collateral status, and baseline glucose levels. The OR of sICH increased by 17.6% for each 5 mL/h increase in EIGR. The observed relationship remained significant when EIGR was analyzed as a categorical variable (highest tertile vs lowest tertile, OR, 7.451; 95% CI, 2.391–23.200; p = 0.001; Table 2, Model 2). The sensitivity analysis also confirmed the significant association between the EIGR and risk of sICH (OR, 1.028; 95% CI, 1.011–1.045; p = 0.001) after being adjusted for the same confounders in Model 2.
Multivariate regression analysis for the association between EIGR and risk of sICH.

Model 1: adjusted for age and sex.
Model 2: adjusted for demographic characteristics, pretreatment ASPECTS, poor collateral status, and baseline glucose levels.
ASPECTS, Alberta Stroke Program Early CT Score; CI, confidence interval; EIGR, early infarct growth rate; OR, odds ratio; sICH, symptomatic intracranial hemorrhage.
In addition, multiple-adjusted spline regression model further confirmed the dose–response correlation between EIGR and sICH (p = 0.225 for nonlinearity; p = 0.001 for linearity; Figure 2). Adding EIGR to a model containing conventional risk factors significantly improved risk reclassification for sICH (category-free NRI, 0.393; 95% CI, 0.227–0.560; p = 0.001; IDI, 0.245; 95% CI, 0.146–0.343; p = 0.001; Table 3).

The correlation between EIGR levels and risk of sICH was assessed using restricted cubic spline regression with three knots (at 5th, 55th, and 95th percentiles). The odds ratio was controlled for the same variables as model 2 in Table 2.
Reclassification statistics (95% CI) for sICH after adding EIGR levels.
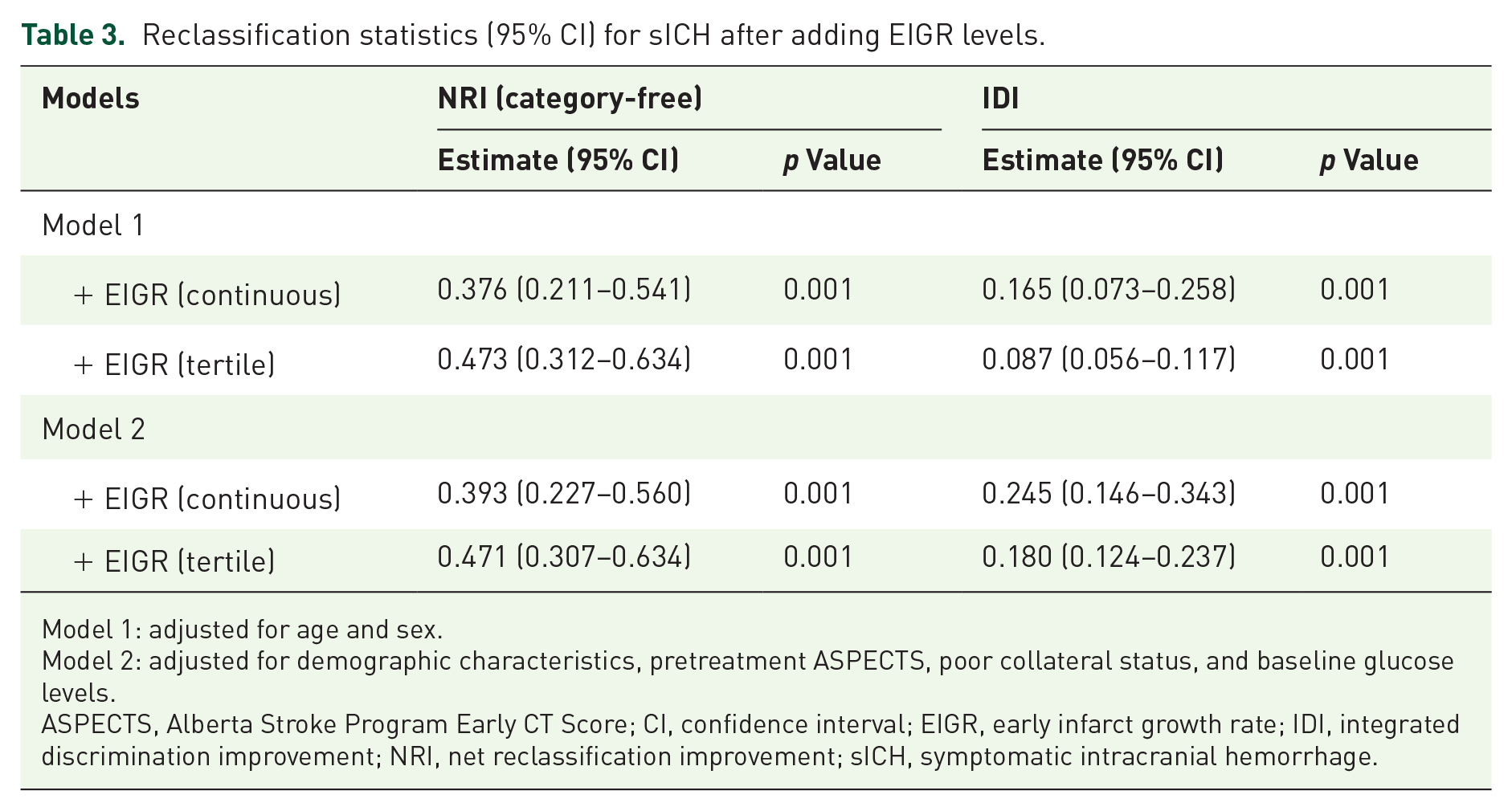
Model 1: adjusted for age and sex.
Model 2: adjusted for demographic characteristics, pretreatment ASPECTS, poor collateral status, and baseline glucose levels.
ASPECTS, Alberta Stroke Program Early CT Score; CI, confidence interval; EIGR, early infarct growth rate; IDI, integrated discrimination improvement; NRI, net reclassification improvement; sICH, symptomatic intracranial hemorrhage.
EIGR as independent determinants of clinical outcomes
The distribution of the mRS score at 90 days according to the tertile of EIGR was demonstrated in Figure 3. There was a significant difference across the EIGR tertile in the overall distribution of mRS scores with multivariable ordinal regression controlling for the same confounders in model 2 (adjusted OR, 1.605; 95% CI, 1.250–2.063; p = 0.001). When an mRS score of 0–2 was defined as a favorable neurological outcome, patients with higher EIGR decreased the likelihood of achieving good functional outcome (highest tertile (36.2%) vs lowest tertile (57.1%); p = 0.006).

Distribution of modified Rankin Scale score at 90 days stratified by the EIGR tertile.
Discussion
This study confirmed a significantly increased risk of sICH after EVT in patients with high EIGR, even after adjusting for several potential confounders. Similar results were observed using subgroups consisting of patients with exact time of stroke onset. Adding EIGR to conventional risk factors improved risk prediction for sICH. Furthermore, EIGR was independently associated with lower odds of achieving favorable outcome 90 days after stroke. Our findings suggested that EIGR could provide important predictive information for clinical outcome in ischemic stroke patients who receiving EVT treatment.
Previous studies have reported the large variability of EIGR within 24 h after symptoms onset.10,11,19,20 The median EIGR in our study was 4.3 mL/h, which is higher than reported in the DEFUSE-2 (3 mL/h). 20 Actually, 10% of patients in the DEFUSE-2 study had distal middle cerebral artery (MCA) occlusion and were very slow progressors. Such patients were excluded from this study, and this may explain part of the difference observed. We found that higher levels of baseline blood glucose were associated with EIGR, which was similar to prior literature. 10 Hyperglycemia may damage the blood–brain barrier via several potential biological mechanisms, such as inducing inflammatory response, oxidative stress, and excitatory chemokines, and directly attacking the neurovascular unit, 21 all of which may exacerbate the infarct progression. Furthermore, in line with previous studies,10,22 our study demonstrated that collateral status was another determinant of EIGR. Relatively poorer collaterals may decrease the perfusion to brain tissue within the oligemic regions, which could worsen the injury of cerebral ischemia. 23 Interestingly, our data indicated that increased EIGR was correlated with a shorter time from onset to imaging, and a higher baseline NIHSS score and rate of IVT treatment. One possible explanation lies in that patients with higher EIGR exhibit severe neurological deficits after stroke onset, which encourages them to be transferred to the stroke center quickly and received the standard treatment. Notably, a recently published work also highlighted the shortened time from onset to imaging in patients with faster early infarct growth, both in the CT-cohort and MR-cohort. 11
The rate of sICH was 11.4% and higher compared with landmark randomized controlled trials such as REVASCAT Trial (4.9%), 24 DAWN (5.6%), 25 and DEFUSE-3 (7.0%), 26 but in the range of ACTUAL registry (13.8%). 5 The broadened inclusion criteria, expansion of clinical indications, and less favorable treatment conditions were more prevalent in real-world clinical practices than in the randomized controlled trials. These factors may contribute to the increased sICH rate in our study. We found that EIGR was significantly correlated with worse functional outcome at 90 days, as seen in two prior studies reporting this association using CT-perfusion imaging,10,27 and two using MRI.11,28 More importantly, our data demonstrated that patients with an increased EIGR had a higher likelihood of sICH after EVT, even after adjustment for confounders. Every 5 mL/h augment in EIGR increased the odds of sICH by 17.6%. However, in the secondary analysis of SELECT (Optimizing Patient Selection for Endovascular Treatment in Acute Ischemic Stroke), Sarraj et al. utilized the CT-perfusion to measure EIGR and reported that fast progressors of infarct volume had an increased trend of sICH (as compared to slow progressors, 11% vs 5%), but the difference did not reach a statistically significant. 10 While there are no clinical trials for any medical treatment to minimize the sICH risk, a more precise individualized risk stratification for these complications could help target treatments and evaluate the need for intensive medical care. The subgroup of patients with a high risk of sICH might also benefit from specific preventive treatment, such as blood pressure management, hypoglycemic treatment, and antioxidative stress therapy. Our results confirmed that EIGR could contribute significantly toward such an individualized risk stratification for sICH in large vessel occlusive stroke patients who receiving EVT.
Our study found that baseline ASPECTS, poor collateral status, and baseline blood glucose levels were significant factors of sICH, which is similar to previous studies.5–8 Hyperglycemia may damage the blood vessel integrity via several biological mechanisms, such as inducing the release of cytokine release, producing oxidative stress, and leading to edema, both of these pathophysiological processes may exacerbate the development of sICH. 29 Infarct volume on initial imaging has been reported to be an important variable that induced parenchymal hematoma after endovascular stroke treatment.6,7 There have been several studies reported on the correlation between time from onset to imaging and reperfusion hemorrhage; however, the results are controversial.7,30–32 Some studies found that increasing time from onset to imaging was positively associated with hemorrhage.7,30,31 In contrast, some evidence showed that time from onset to baseline imaging did not influence the risk of hemorrhagic transformation, 32 which is in line with our data. A previous study also found that time interacted significantly with stroke progression and functional independence. 10 We therefore assumed that it is important to add the time from onset to imaging when assessing the impact of initial stroke size on clinical outcome in patients who undergoing reperfusion therapy. EIGR combinates these two variables, which represent the evolution of infarct volume over time in patients with a persistent large vessel occlusion. Taken altogether, these data suggest the importance of EIGR in future clinical trials evaluating who may benefit from EVT treatment.
The strengths of our study included using ADC to measure infarct volume, and recruiting a Chinese population of large vessel occlusive stroke patients in the anterior circulation, all of which makes this group appropriate for examining influences of EIGR on the presence of sICH. However, this study also has certain limitations. First, the retrospective design has selection bias, which may result in our study sample not being representative of the general population. Second, several confounding variables of sICH, including pretreatment ASPECTS, collateral status, and baseline glucose levels, were further controlled by multivariable regression models. However, residual confounding factors probably persist. Third, the time of ADC acquisition was calculated based on the last-known well time; therefore, some of the calculations may not be accurate for “wake-up” stroke patients. Finally, because the concept of linear infarct volume growth was accepted before using diffusion-weighted imaging, 28 we only calculated infarct size based on a single imaging at the time of presentation. Future studies using serial imaging on MR-perfusion are warranted for infarct growth, which might provide more information on the association of time with stroke development.
In conclusion, we demonstrated the potential of EIGR in predicting the sICH in patients with ischemic stroke undergoing EVT. Future randomized trials are warranted to clarify the safety and efficacy of EVT in patients with faster core growth.
Supplemental Material
sj-doc-1-tan-10.1177_17562864241306561 – Supplemental material for Early infarct growth rate is associated with symptomatic intracranial hemorrhage after endovascular thrombectomy
Supplemental material, sj-doc-1-tan-10.1177_17562864241306561 for Early infarct growth rate is associated with symptomatic intracranial hemorrhage after endovascular thrombectomy by Wei Wang, Zhihang Huang, Shuaiyu Chen, Yan E, Jingwen Qi, Yi Xie, Mouxiao Su, Yingdong Zhang, Teng Jiang and Xiaohao Zhang in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
