Abstract
Recent randomized controlled clinical trials (RCTs) have revolutionized acute ischemic stroke care by extending the use of intravenous thrombolysis and endovascular reperfusion therapies in time windows that have been originally considered futile or even unsafe. Both systemic and endovascular reperfusion therapies have been shown to improve outcome in patients with wake-up strokes or symptom onset beyond 4.5 h for intravenous thrombolysis and beyond 6 h for endovascular treatment; however, they require advanced neuroimaging to select stroke patients safely. Experts have proposed simpler imaging algorithms but high-quality data on safety and efficacy are currently missing. RCTs used diverse imaging and clinical inclusion criteria for patient selection during the dawn of this novel stroke treatment paradigm. After taking into consideration the dismal prognosis of nonrecanalized ischemic stroke patients and the substantial clinical benefit of reperfusion therapies in selected late presenters, we propose rescue reperfusion therapies for acute ischemic stroke patients not fulfilling all clinical and imaging inclusion criteria as an option in a subgroup of patients with clinical and radiological profiles suggesting low risk for complications, notably hemorrhagic transformation as well as local or remote parenchymal hemorrhage. Incorporating new data to treatment algorithms may seem perplexing to stroke physicians, since treatment and imaging capabilities of each stroke center may dictate diverse treatment pathways. This narrative review will summarize current data that will assist clinicians in the selection of those late presenters that will most likely benefit from acute reperfusion therapies. Different treatment algorithms are provided according to available neuroimaging and endovascular treatment capabilities.
Keywords
Introduction
Stroke is a devastating disease that may lead to permanent disability and death. 1 Acute ischemic stroke care has recently been revolutionized with the advent of acute reperfusion therapies. When acute cerebral ischemia occurs due to the occlusion of an intracranial artery, the part of the brain that is irreversibly lost (ischemic core) is surrounded by brain parenchyma that can be salvaged (penumbra) if prompt recanalization takes place. 2 Early recanalization is associated with a more than fourfold increase in the odds of favorable functional outcome (FFO) and a 76% decrease in mortality. 3 By monitoring recanalization with real-time transcranial Doppler in a prospective international multicenter study, 4 we have shown that a 30-min delay in recanalization leads to a 16% reduction in the likelihood of FFO in patients with acute ischemic stroke (AIS) with large vessel occlusions (LVOs) in the intracranial circulation. 5
There are currently two available reperfusion therapies that have been shown to improve outcome in AIS patients: intravenous (IV) thrombolysis (IVT) and endovascular therapy (EVT). Reduction in disability has been well established with both treatments in multiple RCTs up to 4.5 h for IVT and 6 h for EVT using standard neuroimaging: computed tomography (CT) for IVT and CT plus CT angiography (CTA) for EVT.6,7 EVT provides survival benefits in addition to functional independence in AIS patients with LVOs. The number needed to treat (NNT) to reduce disability by at least one level on the modified Rankin scale (mRS) for one patient is 2.6, NNT to achieve functional independence (mRS 0–2) is 5 and there appears to exist a significant 17% relative risk reduction in 3-month mortality corresponding to an NNT of 31.7,8
Recent RCTs have extended the therapeutic time window for both IVT and EVT in AIS but also increased treatment algorithm complexity. Different reported imaging and clinical selection algorithms may appear complex to stroke physicians and this may in turn hamper wide implementation of AIS treatment protocols in real-world everyday clinical practice. As the final part of translational medicine cycle, communication of scientific knowledge in a comprehensive manner to all important stakeholders in stroke care is required to achieve widespread progress and improve stroke patient results. 9 The present narrative review will summarize current knowledge that may assist clinicians in the selection of those late presenters that will most likely benefit from acute reperfusion therapies. Different treatment algorithms will be outlined according to available neuroimaging and endovascular treatment capabilities, while the discussion will be limited to LVOs in the anterior circulation.
IVT after 4.5 h from last seen well
The IVT treatment paradigm has been rather straightforward until recently. Systemic thrombolysis using alteplase (0.9 mg/kg, maximum dose 90 mg over 60 min with initial 10% of dose given as bolus over 1 min) improves clinical outcome in eligible patients when administered within 4.5 h of stroke onset (American Heart Association/American Stroke Association (AHA/ASA), Class I recommendation; Level A evidence up to 3 h and Level B-randomized from 3 to 4.5 h). 10 Pretreatment brain imaging with noncontrast CT is adequate for most patients, mainly to exclude intracranial hemorrhage (ICH) and, to a lesser extent, reveal early ischemic changes (AHA/ASA Class IB non-randomized recommendation). Routine use of advanced neuroimaging within the 4.5-h window is thought to either provide no benefit [magnetic resonance imaging (MRI)] or even harm patients (multimodal CT and MRI) by increasing onset-to-treatment time without providing valuable information that has significant impact on treatment decisions for the average AIS patient. IVT should be used in all eligible patients even if the patient is also eligible for EVT (bridging therapy, AHA/ASA Class IA recommendation). 11 A major contribution to facilitate decision-making in patients when there is uncertainty about eligibility for IVT is the most recent publication of the updated European Stroke Organisation (ESO) IVT guidelines. 12 ESO guidelines assess the overall quality of evidence as high, moderate or low, and provide strength of recommendation either strong or weak; some of these recommendations will be presented throughout the paper and in the illustrative treatment algorithms.
Using standard neuroimaging, IVT effectiveness is extremely time dependent. A meta-analysis of ATLANTIS (A and B), ECASS (I, II and III), NINDS (parts 1 and 2) and EPITHET trials has shown that in the first 90 min after symptom onset, IVT is associated with an increase in the adjusted odds of 3-month FFO (defined as mRS scores of 0–1) of 155%. 13 This benefit rapidly falls to 64% for the 91–180-min time window and to 34% for the 181–270-min time window. Efficacy analyses show that benefit ceases to be statistically significant after 270 min. Besides loss of efficacy, there is a concerning increase in the odds of mortality of 49% in AIS patients treated after 270 min of symptom onset. Since time restriction is a major cause of nontreatment with IVT, 14 a lot of effort has been undertaken to select AIS patients that could be safely thrombolysed in extended time windows or with unknown time of symptom onset but this has been a fruitless effort until recently.
The WAKE-UP study
The WAKE-UP study was an RCT that examined the safety and efficacy of IVT in patients with wake-up stroke (WUS). 15 AIS patients with unknown symptom onset time or patients that woke up with symptoms, presenting within 4.5 h from awakening with positive diffusion-weighted imaging (DWI) MRI of the brain and absence of signal in fluid-attenuated inversion recovery (FLAIR) imaging [DWI(+)/FLAIR(−)] were randomized to either IVT with alteplase 0.9 mg/kg or placebo. It should be noted that DWI/FLAIR mismatch is not a territorial mismatch; as soon as any DWI lesion becomes FLAIR positive, there is no mismatch and patients are excluded from treatment. AIS patients treated with IVT had higher rates of 3-month FFO compared to placebo (53% versus 42%) despite a greater risk for parenchymal hematoma 2 (PH2). The rate of symptomatic ICH (sICH) was 2% in thrombolysed patients, compared to 0.4% in the placebo group. Notably, the rate of sICH following IVT treatment in WUS was lower than the rates reported in other RCTs of IVT for AIS. LVO patients that were candidates for EVT were excluded from the WAKE-UP study but still a 30% of included patients harbored an LVO, producing a rather heterogeneous study sample without clear selection criteria for IVT versus EVT.
It should be clarified that the WAKE-UP trial protocol does not aim to extend IVT time window; on the contrary, it uses advanced neuroimaging (MRI) aiming to identify WUS patients in the 0–4.5-h time window from last seen well (LSW). However, interobserver agreement for acute ischemic lesion visibility on FLAIR imaging is only moderate (κ between 0.46–0.65). 16 DWI/FLAIR mismatch has been shown to predict stroke onset <4.5 h with a sensitivity of 78% and positive predictive value of 83% [87% for middle cerebral artery (MCA)]. 17 In the seminal observational PRE-FLAIR study of AIS patients with known time of symptom onset, it has been shown that among 271 DWI(+)/FLAIR(+) patients, almost half (125) had symptom onset <4.5 h. 18 Another important consideration is that for every six DWI(+)/FLAIR(−) patients, one would had symptom onset >4.5 h (41/245 patients). A Chinese study appears to confirm the inaccuracy of the DWI/FLAIR dissociation to identify symptom onset <4.5 h. 19 Among 601 patients treated with IVT after MRI showing positive DWI but negative T2-weighted or FLAIR imaging, 274 patients were treated 4.5–12 h from LSW; 76 were WUS, leaving 198 (1 in 3) patients with confirmed symptom onset after 4.5 h.
Another major drawback of this approach is that it requires emergent MRI for AIS, limiting application to high-resource countries and stroke centers and to those AIS patients that do not have contraindications for brain MRI (e.g. agitation, pacemaker). The WAKE-UP study group has recently published a promising report that found CT-based quantitative net water uptake comparable to MRI DWI/FLAIR dissociation. 20 The AHA/ASA 2019 updated guidelines for the early management of AIS provide a IIa recommendation for IVT in WUS according to the WAKE-UP protocol. 10 ESO IVT guidelines advocate thrombolytic treatment for patients with AIS on awakening and DWI/FLAIR dissociation for whom mechanical thrombectomy is either not indicated or not planned (quality of evidence, high; strength of recommendation, strong). 12
The EXTEND trial
After numerous neutral RCTs, the Extending the Time for Thrombolysis in Emergency Neurological Deficits (EXTEND) trial was the first phase III randomized, placebo-controlled study to show benefit of IVT for AIS beyond 4.5 h. 21 Advanced neuroimaging was used for patient selection with CT perfusion imaging or perfusion-diffusion MRI, and images were processed by the Rapid Processing of Perfusion and Diffusion (RAPID) automated software platform (iSchemaView; https://ischemaview.com/home). Patients were included if they fulfilled three criteria:
Ischemic core <70 ml, measured on CT perfusion as brain volume with cerebral blood flow <30% of normal brain regions (rCBF) or on MRI as apparent diffusion coefficient (ADC) <620 μm²/s.
Critically hypoperfused brain volume >10 ml from ischemic core volume, measured on CT or magnetic resonance (MR) perfusion as delayed arrival of an injected tracer agent (time to maximum of the residue function, Tmax > 6 s).
Perfusion lesion–ischemic core mismatch ratio >1.2.
IVT was associated with an absolute increase of 6% in the rate of 3-month FFO despite a statistically nonsignificant increase in sICH rate (6% with alteplase versus 1% with placebo) that, importantly, did not affect mortality rates at 3 months. It should be kept in mind however, that the rate of sICH in the EXTEND trial was among the highest reported in RCTs of IVT for AIS. In addition, the treatment benefit reached significance only after adjustment for age and baseline National Institutes of Health Stroke Scale (NIHSS) score, but these adjustments have been predefined in the study protocol.
Two characteristics of the study should be highlighted. First, patient recruitment took place before the publication of the positive EVT trials, permitting the inclusion of patients with LVOs who would be unethical to randomize to placebo treatment in current everyday clinical practice. Second, 70% of randomized patients had an underlying LVO [internal carotid artery (ICA), M1 or M2 branch of MCA]. This is an important point as IVT is a highly effective recanalization treatment for short thrombi resulting in MCA occlusions, especially for the M2 segment. 22 Future IVT studies, by excluding patients with AIS caused by an M2 occlusion because of expanding EVT eligibility, will have difficulty showing positive results, as much of the benefit shown in previous IVT trials may be attributed to high rates of successful recanalization of M2 occlusions.23,24 An example of averted EVT after IVT in a patient presenting with NIHSS score of 20 due to an underlying M2 MCA occlusion is presented in Figure 1. A second important point is that despite a high-volume cut-off at 70 ml of infarcted tissue, most patients had an extremely low ischemic core volume (<4 ml), raising doubts on the validity of the positive results for AIS patients with larger ischemic cores. Clinicians should be cautious in performing IVT 4.5–9 h after symptom onset when ischemic core volume approaches the 70 ml limit. The updated ESO guidelines for IVT in AIS advocate IVT for patients with ischemic stroke of 4.5–9 h duration (known onset time) and with CT or MR perfusion core/penumbra mismatch, and for whom mechanical thrombectomy is either not indicated or not planned (quality of evidence, low; strength of recommendation, strong). 12 ESO guidelines also suggest that the EXTEND protocol may be used for WUS (quality of evidence, moderate; strength of recommendation, strong). However, it should be noted that since the EXTEND trial was not focused on WUS and treatment effect became evident only after (predefined) adjustment, the quality of evidence lack behind those of the WAKE-UP trial (moderate versus high, respectively).

A 79-year-old woman with a history of mesenteric ischemia 10 years ago and hypertension, is presenting 75 min after abrupt onset of complete aphasia and right hemiplegia (NIHSS 20). Noncontrast brain CT showed a dot sign in the right insula, suggestive of right M2 occlusion (arrow, a); it was missed on initial assessment. Due to diagnostic uncertainty of the attending physician, she subsequently had a brain MRI, which showed early diffusion restriction on the left hemisphere (ADC maps b, DWI c1 and c2). On gradient echo images (d1, d2) the thrombus was documented (arrows) as well as slow flow on adjacent leptomeningeal arteries (face-mask artifacts are seen on the anterior part). The patient had no contraindication to thrombolytic recanalization treatment and received 3 ml (15 mg) tenecteplase as bolus intravenously (0.25 mg/kg) 135 min after symptom onset. A DSA revealed a persistent M4 occlusion that was considered of minor importance (arrow, e) and EVT was averted. The patient improved (NIHSS 2 at 24 h). A brain MRI showed reversal of most of the initial DWI lesion; only some diffuse, small, subcortical acute ischemic lesions of the left hemisphere were detected (f). DWI reversal is a rare finding post-reperfusion; persistent decrease in final infarct volume compared to baseline DWI has been described in only 3% of treated patients. 25 Of note, the patient showed DWI lesions on the left cerebellar hemisphere (g2) that were not present in the initial MRI (g1). It is possible that these lesions were present but not depicted in the baseline DWI brain MRI. The sensitivity of DWI for acute ischemic lesions in the posterior fossa is inferior to that in the cerebral hemispheres and may be missed in the hyperacute phase. 26 Another probable cause would have been embolization to new territory caused by DSA 27 or, less likely, caused by IVT (as we have described previously). 28 Minutes after IVT the patient developed an asymptomatic acute right orbital hematoma (h). She was discharged on antiplatelets with right facial paralysis and mRS score of 1, 3 days after admission.
The concept that IVT is safe when there is little or no established brain ischemia, irrespective of time, is biologically plausible but was hard to prove without the proper imaging techniques. Now, for the first time, RCTs provide data to support it. A most recent single-center retrospective study took a step further in establishing the role of IVT in unknown time of symptom onset or extended time windows using advanced neuroimaging. Macha et al. 29 in Erlangen, Germany, performed IVT in 184 patients with evidence of salvageable brain tissue at risk on CT or MR perfusion imaging (mismatch between hypoperfusion versus infarcted core, mismatch quotient > 1.4). Patients had either unknown or >4.5 h duration of symptoms, without upper time limit; 36% of patients received EVT in addition to systemic thrombolysis. The authors reported that IVT and bridging therapy in the unknown or extended time window appeared safe in AIS patients with restricted or no established ischemic core on CT perfusion imaging or MRI ADC, while the use of CT perfusion led to faster door-to-needle times. Treatment with IVT appeared safe: sICH according to safe implementation of treatments in stroke (SITS)-MOST criteria was reported in only 1.6% of treated patients. It is important to point out the low volume of ischemic core on baseline imaging: median volume was 0 ml (interquartile range 0–4.5 ml on CT perfusion and 0–12.5 ml on MRI ADC). Given this low volume of ischemic core in most treated patients, implementation of the Erlangen protocol in clinical practice, as off-label treatment, should incorporate the additional restrictive criterion of 0–5 ml of ischemic core, until more data are available. Another advantage of IVT protocols using perfusion imaging in the extended time window (>4.5 h) may be related to the fact that advanced neuroimaging may reliably discriminate stroke mimics (seizure, migraine) from AIS patients and avert systemic thrombolysis. 30
Symptomatic hemorrhagic transformation of an ischemic brain infarct is the most feared complication of recanalization therapies. In the original NINDS t-PA trial, increasing NIHSS scores and hypodensity or mass effect on baseline CT were independently associated with increased ICH risk. 31 Several predictive instruments for estimating sICH risk after thrombolytic therapy have been developed in recent years (SEDAN, HAT, SITS SICH, MSS, TURN, SPAN-100, GRASPS).32–38 These scores were mainly used for research purposes and none prevailed or was incorporated into clinical guidelines. Most scores report an increase in hemorrhagic risk with increasing age, blood glucose, NIHSS and baseline CT hypodensities (Table 1). A recently published nomogram incorporates data on chronic disease (hypertension, diabetes mellitus, atrial fibrillation), cerebral small vascular disease burden score (calculated from four distinct imaging markers on MRI), NIHSS and onset-to-treatment time for thrombolysis; despite its reported superiority in predicting hemorrhagic transformation post-IVT compared to the aforementioned clinical scores, its complexity precludes clinical use in the acute setting. 39
Scores for predicting post-IVT sICH in early time windows.

Asp, aspirin; Clop, clopidogrel; CT, computed tomography; Incr., increasing; IVT, intravenous thrombolysis; MCA, middle cerebral artery; mRS, modified Rankin scale; NIHSS, National Institutes of Health Stroke Scale; OTT, onset-to-treatment time; p, point on relevant scoring scale; PLT, platelet count; SBP, systolic blood pressure; sICH, symptomatic intracranial hemorrhage.
Current guidelines recommend that the extent of early ischemic changes on perfusion imaging (or any other reported hemorrhagic risk factor) should not be used as a criterion to withhold IVT <4.5 h from LSW for patients who otherwise qualify for treatment. 8 Despite the limited clinical utility of scores for predicting post-IVT sICH in early time windows, and although they may be based on standard neuroimaging, they describe risk factors for hemorrhagic transformation that may aid the clinician stratify the low-risk patient that could potentially be treated with the lowest possible risk for bleeding in the off-label clinical scenarios discussed in the current review. All these scales agree on the association of patient age and stroke severity with the risk of hemorrhagic transformation. Recent research would also preclude IVT for the mildest of strokes that present with nondisabling symptoms. 40 As a conclusion, a low-risk patient for sICH after off-label IVT (as a standalone therapy or as part of bridging therapy as discussed later) should fulfill the following criteria: age <80 years and mild yet disabling stroke severity with limited hypodensities/ischemic core on imaging. Definitive answers may be provided from the ongoing TIMELESS study that is currently recruiting AIS patients in the 4.5–24 h time window (ClinicalTrials.gov identifier: NCT03785678). Patients with ICA or MCA occlusion and favorable penumbral imaging detected on either CT or MRI will be randomized to either tenecteplase 0.25 mg/kg (max 25 mg) or placebo; estimated study completion time is April 2022.
Expert opinion: IVT for AIS > 9 h from LSW may be safe in low-risk patients with mismatch quotient >1.4 and low ischemic core volume (<5 ml on validated or automated RAPID CTP). MR perfusion does not seem to offer any advantage over CT perfusion and may delay treatment.
Patient selection for IVT > 4.5 h using noncontrast brain CT has not yet been proven safe. Thrombolysis in Stroke With Unknown Onset Based on Non-Contrast Computerized Tomography (TRUST CT) is a recently published retrospective multicenter stroke registry that compared 117 AIS patients with unknown time of stroke onset treated with IVT based on an initial Alberta Stroke Program Early Computerized Tomography score (ASPECTS) of ⩾7, with 112 propensity score-matched, nontreated AIS patients. sICH rates were similar in both groups (four versus one, respectively). FFO at 3 months was significantly more common in the IVT (33.3%) than the control (20.5%) group (adjusted OR, 1.94; p = 0.05). 41 However, imbalances remained between study groups despite propensity matching, notably differences in the LSW to presentation times and symptom discovery to presentation times, both being more prolonged in the control group, resulting in delays in the initiation of medical management. Though promising, more data are required before declaring safety of IVT in prolonged time windows based solely on noncontrast CT. ESO guidelines advise against IVT based on noncontrast CT in the 4.5–9-h time window (quality of evidence, moderate; strength of recommendation, strong). 12
Metanalyses
After publication of the positive results of the EXTEND trial, an individual patient data meta-analysis included two additional RCTs that tested IVT after 4.5 h. 42 The European Cooperative Acute Stroke Study-4/Extending the time for thrombolysis in emergency neurological deficits (ECASS-4/EXTEND) trial randomized patients to alteplase after perfusion MRI. 43 Investigators used the EXTEND clinical eligibility criteria but preferred visual assessment of MR perfusion diffusion imaging. The Echoplanar Imaging Thrombolytic Evaluation Trial (EPITHET) also used perfusion MRI in the extended thrombolysis time window in the 3–6-h time window; 44 only patients treated within the 4.5–6-h window were included. Both studies reported neutral results. The meta-analysis showed that IVT increases the odds of achieving FFO at 3 months, despite an increase in sICH (5%) but not in mortality; 61% of patients had LVO on angiographic imaging and would be eligible for EVT according to current international recommendations. As expected, median volume of infarct core was low (8 ml) and median perfusion mismatch volume was extensive (47 ml). As part of the meta-analysis, the investigators performed central adjudication of perfusion mismatch using the automated RAPID software in 405 out of 411 patients who had available raw imaging data. Applying automated processing excluded around 24% of patients, mostly patients with small perfusion lesions. As expected, this analysis implied that the benefit of IVT is driven by patients meeting perfusion mismatch criteria. FFO was 36% with alteplase versus 26% with placebo, with an OR higher than the primary analysis (2.1 versus 1.8), which also reached statistical significance. On the contrary, excellent outcomes were no different among patients without mismatch (treatment group fared worse numerically), whereas when comparing mRS 5–6, a startling difference of 25% with alteplase versus 8% with placebo was noted. Despite being underpowered, this analysis further consolidates the role of perfusion mismatch for proper patient selection for IVT in extended time windows.
A second meta-analysis of individual patient data from IVT RCTs for stroke of unknown time of onset with perfusion-diffusion MRI, perfusion CT, or MRI with DWI-FLAIR mismatch was recently published. This meta-analysis was mostly based in the WAKE-UP study (contributing 60% of total patients). Patients from the THAWS trial were included, a neutral Japanese trial that used the WAKE-UP imaging protocol for patient selection and was prematurely halted after the positive results of the WAKE-UP trial. 45 THAWS tested the controversial 0.6 mg/kg dose of alteplase for IVT, inserting significant heterogeneity in the meta-analysis, but a sensitivity analysis excluding this trial did not alter the results. The authors also included patients with unknown time-onset stroke from the EXTEND and ECASS-4 trials; both used the entirely different concept of penumbral imaging for patient selection, as already discussed. From a total of 843 individuals, 85% had available MRI scans and 95% among them proved to have a DWI/FLAIR mismatch; 15% had CT perfusion imaging that estimates surviving brain parenchyma rather than predicting stroke onset <4.5 h. This heterogeneity is concerning and the results of a meta-analysis that combine two very different strategies for patient selection should be read with caution. In brief, this meta-analysis also showed a net benefit for all functional outcomes across the entire range of the mRS scores despite an increased risk of sICH (3%). The study also reported a statistically significant increase in deaths at 3 months: 27 (6%) patients died in the alteplase group and 14 (3%) patients died among controls (adjusted OR 2.06; p = 0.04). Of the 27 deaths in the alteplase group, 7 were attributable to sICH, 4 to recurrent or progressive stroke, 2 were of unknown cause, and the remaining 14 deaths were of non-neurological cause and unrelated to treatment or index stroke. In the control group, all 14 deaths were of non-neurological cause and unrelated to treatment or index stroke. A most recent post hoc analysis of the THAWS study has shown positive results in treated patients that had >6.4 ml of DWI lesions on MRI; the subgroup was small (n = 42, 26 patients treated with IVT) and no sICH has been recorded. 46
Our group has also performed a systematic review and meta-analysis of RCTs using advanced neuroimaging with either CT or MRI to identify candidates with substantial penumbral or DWI/FLAIR mismatch who could benefit from treatment with alteplase. 47 Four RCTs (859 total patients) were identified: the EXTEND, WAKE-UP, ECASS-IV trials and a small (n = 12) study by Michel et al. 48 In unadjusted analyses, IVT was associated with a higher likelihood of 3-month FFO, complete recanalization and sICH (3% with alteplase versus 0.5% with placebo), with no significant difference in the odds of all-cause mortality at 3 months. In the analyses adjusted for age and baseline stroke severity, IVT was associated with a higher probability of 3-month FFO and sICH with no significant difference in the odds of all-cause mortality.
In addition, our collaborative group has shown that, in our everyday clinical practice experience, 10% of AIS patients fulfill WUS criteria or present in the 4.5–9-h time window; after excluding all LVO patients who underwent EVT, only 1.3% of AIS patients are eligible for IVT according to EXTEND neuroimaging and clinical eligibility criteria (number needed to screen, 77). 49 A further 10% of EXTEND noneligible patients fulfill the WAKE-UP study criteria and can be treated with IVT. Thus, the combination of CTP and brain MRI for WUS or in the 4.5–9-h time window can increase IVT use by 1.9% (1.3% due to perfusion imaging using the EXTEND trial criteria and 0.6% due to brain MRI using the WAKE-UP study criteria) among consecutive AIS patients (number needed to screen, 53). The two neuroimaging strategies were compared in a most recent analysis from the WAKE-UP study group. 50 In a post hoc analysis of the WAKE-UP study, a subgroup of patients that had RAPID perfusion imaging besides standard MRI scans were examined for persistent penumbra at the time of imaging. DWI-FLAIR mismatch was more prevalent than PWI-DWI mismatch (48% versus 26%; p < 0.0001), prevalence of PWI-DWI mismatch was similar in patients with (27%) or without (24%) DWI-FLAIR mismatch. In the small subgroup of 208 randomized patients with available PWI imaging, PWI-DWI mismatch status did not modify the treatment response (p for interaction = 0.73). 51 The authors concluded that using MRI may be more inclusive than CT perfusion for stroke of unknown onset; however, we should be careful not to confound stroke of unknown time of onset with WUS, and keep in mind that >90% of patients in the WAKE-UP trial were wake-up patients, the remaining <10% being aphasic or patients of truly unknown time of onset. Shifting the screened stroke population from WUS to unknown time of onset strokes, would most likely render futile any attempt to show that DWI-FLAIR mismatch is more inclusive than perfusion imaging, since most patients >4.5 h of symptom onset would be excluded. The important finding of the aforementioned post hoc analysis is that for WUS patients, DWI-FLAIR mismatch may be more inclusive, since lacunar strokes can be missed with CT perfusion but captured with DWI and eventually be treated (Figure 2). 52

A 90-year-old man woke up with right hemiplegia, dysarthria and NIHSS score of 8 points at 15 h after LSW. CT, CTA and RAPID CT perfusion were negative for early ischemic changes, LVOs or brain oligemia and the patient received no treatment. The patient had a brain MRI the following day that showed a small acute ischemic lesion involving the left corticospinal tract.
Since the WAKE-UP protocol is MR-based, whereas the EXTEND protocol uses either CT perfusion or MR perfusion, the clinician currently faces the dilemma of whether to choose CT or MRI for WUS. One might consider using a cut-off NIHSS score <6 as a surrogate for non-LVO stroke and opt for MRI in these patients. However, this solution is imperfect since the dilemma is not LVO versus non-LVO but lacunar (captured only by MRI) versus nonlacunar. Peripheral arterial occlusions do not necessitate MRI, since they would be detected by CT perfusion, especially if the CT scanner provides full brain coverage during perfusion imaging. The only single study that incorporates all necessary information to follow either treatment protocol is MR perfusion and it should be preferred for WUS patients where available.
Expert opinion: there are currently two equally valid neuroimaging strategies for selection of appropriate candidates for IVT in WUS, based on DWI-FLAIR mismatch or perfusion imaging. Choice of protocol primarily depends on local resources. MR perfusion provides all necessary data to satisfy both WAKE-UP and EXTEND trial protocols and is therefore the most inclusive single imaging study for selection of WUS for treatment with IVT.
Endovascular treatment in the anterior circulation before and after 6 h from LSW
EVT within 6 h after symptom onset
LVO strokes represent up to one-third of AIS but are accountable for three-fifths of dependency and more than nine-tenths of mortality after AIS. 53 IVT is associated with low odds of recanalization in extensive thrombi that are common in LVO patients. 54 Pretreatment with systemic thrombolysis in LVO patients eligible for mechanical thrombectomy results in successful reperfusion in only 1 of 10 cases, negating the need for additional endovascular reperfusion. Tandem occlusions appear to be the least responsive to IVT pretreatment. 55
EVT provides a solution to this problem, as major brain arteries are readily accessible with intravascular catheters. After years of neutral RCTs, numerous recent clinical trials have recently provided solid evidence that EVT significantly improves clinical outcomes in AIS. Previous failures may have led investigators to adopt stringent selection protocols to prove benefit in the subgroup of patients that would gain the most out of endovascular reperfusion therapies. The appropriate selection of candidates for EVT was a crucial component of the success of recent RCTs (the other being advances in technical components of the catheters used for EVT). 56 EVT using modern stent retrievers and aspiration devices is associated with increased likelihood of complete recanalization [risk ratio (RR), 2.22; p < 0.00001] and 3-month functional independence (mRS scores of 0–2; RR, 1.72; p < 0.00001) that corresponds to an absolute increase in the rates of complete recanalization of 44% (NNT = 2) and of functional independence of 16% (NNT = 6). It is also a very safe intervention that does not appear to increase sICH risk compared to best medical management (4.6% versus 4.3%). 57 The neuroimaging modalities that are sufficient for patient selection in the early time window of 6 h include noncontrast CT and CTA or brain MRI and MR angiography (MRA). CT or MRI may exclude alternative diagnoses and assess the extent of the infarct; noninvasive vascular imaging (CTA or MRA) may reveal a causative LVO occlusion of the anterior circulation, involving either the ICA or the MCA segment 1 (M1) (level of evidence IA). 10
AHA/ASA guidelines not only support the use of plain neuroimaging modalities but they also advise against using advanced neuroimaging in most cases during the 6-h time window (Class IB recommendation). There are two reasons for this recommendation. First, time is brain and imaging protocols should not be time consuming; any delay should be justified for providing clinically relevant information that significantly affects treatment decisions; there are no randomized data currently to indicate such a necessity in AIS patients with LVOs in the early time window. We have shown in a meta-analysis of EVT RCTs that advanced neuroimaging selection appears to be associated with more pronounced improved outcomes in EVT-treated patients compared to LVO patients receiving best medical management, 58 the caveat being that it may also disqualify patients with suboptimal perfusion profiles from a potentially beneficial treatment. This has been aptly described ‘denominator fallacy’ by Goyal. 59 Indeed, the Highly Effective Reperfusion Evaluated in Multiple Endovascular Stroke Trials (HERMES) collaboration meta-analysis showed that estimated ischemic core volume was independently associated with functional independence but did not modify the treatment benefit of EVT. 60 The EXTEND-IA trial, one of the RCTs included in the HERMES meta-analysis, used stringent perfusion criteria leading in hyper selection of patients and reported the highest absolute benefit from EVT but necessarily included only a small minority of LVO patients; many among them would be eligible for EVT in other RCTs with more relaxed imaging inclusion criteria. 61 Second, obtaining additional neuroimaging data may complicate patient selection, by providing information that is difficult to put into context (e.g. by revealing a large ischemic core in early time windows). Such patients may be excluded from treatment out of fear of treatment futility or increased hemorrhagic risk, but it has been shown that CT perfusion may overestimate infarct core in very early time windows (<1 h). This phenomenon has been called ‘ghost infarct core’ and it has been measured more than 10 ml in 16% of LVO patients in a retrospective study with a mean symptom onset to CTP time of 107 min (range, 50–227 min). 62 In these patients, mean difference in the final infarct core volume was −27 ml and it would have not probably influenced treatment decision in most of them, but the range was quite extensive, from −14 to −48 ml, suggesting that it might have been relevant in some of them. 63 Notably, even DWI lesions may regress, especially if they are small, 64 although rarely even larger ones may regress after successful recanalization, as presented in Figure 1. Data from the MR CLEAN trial provide further support to this recommendation. Being the first positive trial (leading to premature termination of subsequent trials since clinical equipoise has been lost), MR CLEAN required only CT and CTA with no collateral or perfusion assessment. 65 Benefit from treatment was preserved even for patients with extensive early ischemic signs on CT. 66 In a subgroup of patients with CT perfusion (who did not alter treatment decisions) no effect of perfusion patterns to treatment benefit was detected, even though perfusion imaging had prognostic value. 67
EVT after 6 h from symptom onset
In the ‘first wave’ of positive EVT RCTs, only a minority of patients was randomized after 6 h from symptom onset in the Endovascular Treatment for Small Core and Anterior Circulation Proximal Occlusion with Emphasis on Minimizing CT to Recanalization Times (ESCAPE) 68 and in the Randomized Trial of Revascularization with Solitaire FR Device versus Best Medical Therapy in the Treatment of Acute Stroke Due to Anterior Circulation Large Vessel Occlusion Presenting within Eight Hours of Symptom Onset (REVASCAT) trials. 69 Specifically, 19% of patients in the ESCAPE trial (59 out of 315) were randomized between 5.5 h (likely to have groin puncture after 6 h from symptom onset) and 12 h. 70 There was no evidence of heterogeneity of EVT treatment effect before and after 5.5 h from symptom onset (p = 0.134, likelihood ratio test). The absolute risk difference favoring intervention was 19.3% for functional independence at 90 days, and the shift analysis without increase in sICH. The ESCAPE trial used multiphase CTA and excluded patients with bad collaterals from treatment. 71 REVASCAT also randomized 20 patients between 6 and 8 h from LSW. Although in subgroup analysis there seemed to be no difference of treatment effect before and after 4.5 h, the number of patients in the 6–8 h time window was too low to allow for statistical analysis. Since extending the therapeutic window from 6 to 24 h may result to an increase up to 27% in patients receiving EVT, the importance of recent late time window RCTs becomes evident. 72 It should be noted that the HERMES meta-analysis showed that benefit of EVT using standard neuroimaging remains statistically significant up to 7 h and 18 min from LSW. This temporal cut-off has been adopted by the 2019 joint ESO and European Society for Minimally Invasive Neurological Therapy (ESMINT) EVT Guidelines. 73
The DWI or CTP Assessment with Clinical Mismatch in the Triage of Wake-Up and Late Presenting Strokes Undergoing Neurointervention with Trevo (DAWN) study proved the efficacy of EVT up to 24 h after symptom onset. 74 The DAWN study’s design was unique in implementing a clinical–radiological dissociation strategy; MRI or CT perfusion with the use of RAPID software was only used to estimate infarct core. Instead of searching for radiological mismatch in perfusion imaging (salvageable tissue), patients with low ischemic core volume and severe neurological deficit (NIHSS ⩾ 10 or 20 depending on age and ischemic core) were randomized to EVT or conservative treatment. Those >80 years of age were selected with strict ischemic core criteria as it is known that elderly patients have dismal prognosis even with moderate sized brain infarction. 75 Results were highly in favor of EVT (3-month functional independence: 49% in the EVT group versus 13% in the best medical management group) and analogous to the positive results of early time window EVT trials. This counterintuitive result was named ‘late window paradox’ and can be explained by the fact that in extended time windows and at the time the DAWN trial was recruiting patients, there was no alternative recanalization therapy (IVT). 76 The other probable explanation is that advanced neuroimaging helps clinicians select only ‘slow progressors’, patients with adequate collaterals that keep extensive areas of ischemic brain parenchyma alive for extended time periods. According to crude calculations, 90% of LVO patients would have sizable viable brain parenchyma in the first hour after symptom onset and would therefore benefit from effective recanalization therapy, 70% would still retain salvageable tissue 2–4 h after onset, whereas, in extended time windows (6–9 h) a mere 10% might still gain benefit from treatment. 77
The Endovascular Therapy Following Imaging Evaluation for Ischemic Stroke (DEFUSE) 3 trial randomized LVO patients 6–16 h after LSW. Perfusion imaging with CT or MR was used to select patients with ischemic core less than 70 ml and favorable perfusion profile. Once again, there was a significant clinical benefit from EVT (3-month functional independence: 45% in the EVT group versus 17% in the best medical management group) and mortality was reduced at the limits of statistical significance (14% versus 26%, p = 0.05). 78 Treatment benefit persisted for all examined subgroups including WUS patients. Following these extremely important positive results, AHA/ASA Guidelines strongly recommend EVT using CTP or DWI according to the protocols of the DAWN and DEFUSE 3 trials in the extended time window beyond 6 h (Class IA for 6–16 h based on two RCTs and Class IIaB for 16–24 h based on one RCT). 10
Since DAWN and DEFUSE 3 used different selection strategies, one may wonder which path to choose, assuming that all the required neuroimaging tools are readily available. DEFUSE 3 has used less-stringent selection criteria including lower NIHSS scores (⩾6 points versus ⩾10 points in DAWN) and larger ischemic core (⩽70 ml versus 51 ml in DAWN). A subgroup analysis of DEFUSE 3 documented that 40% of included patients did not fulfill DAWN criteria; in this subgroup of patients EVT retained a strong therapeutic effect. 78 The reverse may also be true; a single-center study indicated that one in three DAWN-eligible patients may be ineligible for DEFUSE 3 79 and it should be noted that the DAWN trial protocol has two extra advantages. First, it remains the only RCT to show benefit of EVT in the 16–24-h time window. Second, due to a simpler imaging protocol, perfusion imaging is not necessary if standard MRI (with DWI, FLAIR, hemosiderin and time-of-flight sequences) is used, since infarct volume in DWI can be easily calculated with a variety of software tools. It is obvious that stroke center imaging capabilities will determine which path to follow; ideally, any of the DAWN or DEFUSE 3 protocols for 6–16 h and the DAWN protocol for 16–24 h from LSW. After the initial positive results, relaxing selection criteria for EVT in extended time windows became a matter of heated research since recanalization even for larger infarct volumes may still benefit LVO stroke patients. 80 Ongoing RCTs may soon provide an answer to this question. Notably, the cost-effectiveness of EVT in extended time windows has also been shown in many subgroups. 81 As a final note, despite the lesser importance time from LSW takes with penumbral imaging, it remains a significant predictor of outcome when similar groups of patients treated with EVT in early versus late time windows are compared. 82
Adherence to selective trial-defined criteria in extended time windows may provide unprecedent NNTs for acute stroke treatment but comes with a price, hyper-selectivity. A retrospective single-center study found that 70% of LVO patients presenting in the 6–24-h time window would be ineligible for DAWN or DEFUSE 3 treatment; 26% of them received EVT and 30% among them achieved functional independence at 3 months. 83 Of course, these patients fared worse than trial-eligible patients in this single-center registry since they had more severe strokes and larger ischemic cores at baseline, but still fared better than the control arms of DAWN (3-month functional independence rate, 13%) and DEFUSE 3 (3-month functional independence rate, 17%). Patients with large ischemic cores, limited penumbra, pretreatment disability and advanced age (>80 years) tended to have worse outcomes.
Expert opinion: EVT may be offered as an off-label therapeutic option in selected AIS patients presenting 6–24 h after LSW without fulfilling DAWN or DEFUSE 3 criteria, especially when they are younger than 80, have no pre-existent disability and have favorable penumbral imaging, since the natural history of LVO in late time windows is dismal without recanalization.
An observational study from three American centers that used the DAWN eligibility criteria for EVT after 24 h and up to 6 days has also been published. Procedural, efficacy, and safety outcomes in 21 patients were comparable with the DAWN treatment arm, suggesting that DAWN criteria may be used beyond 24 h from LSW. 84 An illustrative case of a patient that fulfilled DAWN criteria and has been treated with EVT 4 days after symptom onset can be seen in Figure 3. Another interesting retrospective single-center study from Korea has been recently published. 85 Among 150 AIS LVO patients presenting after 16 h and up to 10 days from LSW, 24 (16%) were treated with EVT after visual inspection of collateral circulation (85%) or perfusion images (74%) for moderate to good collaterals or core–penumbra mismatch, respectively. In propensity score-matched analyses, EVT was associated with better odds of 3-month functional independence and 3-month functional improvement (mRS score shift, despite a statistically nonsignificant increase in PH2, 13% versus 3%). These preliminary results may be promising but, considering the high rate of PH2 and the subjective evaluation of perfusion maps, cannot support such a treatment strategy outside RCTs. Visual inspection of perfusion maps substantially misclassifies patients compared to automated analysis, not because of interobserver variability as one may assume, but due to the inherent inability of the human examiner to correctly assess volumetric data. 86

A previously healthy 51-year-old man presents with mild aphasia and partial right hemianopia 30 min since LSW (NIHSS 4). He also admits unusual headache for the last 2 days. CT was normal (ASPECTS 10), RAPID CTA showed a possible dissection (left panel a, arrow) and complete occlusion of left ICA and reduced artery density on the left parietooccipital area (right panel a). CT perfusion showed a large area of hypoperfusion on the same area with no ischemic core (total mismatch) (b). HIR was low (0.2) and CBV index (relative CBV in Tmax > 6 s) was high (0.7), both suggestive of good collaterals. IVT with alteplase was initiated 1 h after LSW. Transcranial Doppler showed frequent (approximately 1/min) HITS (c, arrow). We did not proceed with EVT due to low NIHSS score and occlusive dissection of the left ICA. RAPID MR perfusion 4 h later showed an infarction and persistent penumbra (d). Fat saturation MR sequences confirmed the ICA dissection (e, arrow). The patient remained stable for 3 days, when he abruptly developed right hemiparesis and deterioration of aphasia (NIHSS 11) that receded after induced hypertension with noradrenaline. CTA showed a distal left M1 occlusion (f, arrow). RAPID CT perfusion revealed a moderate infarction (ischemic core 29 ml, roughly corresponding to hypodensities on noncontrast CT) and an extensive penumbra (130 ml) involving most of the superficial left MCA distribution. The patient continued to have fluctuations of its neurological examination and EVT was decided since he fulfilled clinical (NIHSS > 10 and age < 80) and radiological (ischemic core ⩽ 30 ml) DAWN criteria, in an off-label 60-h time window. Mechanical thrombectomy of left M1 and stenting of the dissected left ICA with two stents in a telescopic manner were successfully performed. Post-treatment MRI showed scattered areas of infarction of the left hemisphere with no hemorrhagic transformation and the patient had a partial recovery at his discharge, 3 days later (NIHSS 5).
Similar to off-label IVT, off-label EVT should only be considered in AIS patients with low risk for complications. Pre-procedural, procedural and post-procedural risk factors for sICH have been described post-EVT in early time windows. 87 Antiplatelet therapy, high C-reactive protein, elevated mean arterial blood pressure, hyperglycemia and low ASPECTS were found to be independent predictive factors of sICH after EVT. 88 In the Endovascular Treatment for Ischemic Stroke (ETIS) registry, increasing age and ASPECTS, smoking, general anesthesia, angiographic poor collaterals and embolization in a new territory independently predicted parenchymal hemorrhage. 89 There have been several reports of neuroimaging markers predicting symptomatic hemorrhagic transformation after recanalization therapies: 90 leucoaraiosis,91,92 high Tmax (>14 s), 93 low ADC (<550), 94 permeability measures, 95 low cerebral blood volume (CBV).96,97 A patient treated with EVT 11 h after symptom onset with very low CBV on baseline perfusion imaging that developed sICH 10 h after successful recanalization is presented in Figure 4. Hence, a simple and readily available validated tool to predict hemorrhagic transformation in early or late time windows is needed. A first attempt has been the thrombolysis in cerebral ischemia (TICI)–ASPECTS–glucose (TAG) score: hyperglycemia, low ASPECTS and moderate or no recanalization were independently associated with increased rates of sICH in a retrospective analysis of a prospective single-center stroke database. 98 TAG score has been validated in the Blood Pressure After Endovascular Treatment (BEST) registry: 99 the TAG score was associated with an almost 50% per-point likelihood increase of post-MT sICH and a moderate total prognostic capability (area under the curve, 0.69). 100 About one in two patients achieved recanalization after 6 h and patients with favorable penumbral imaging were included up to 24 h from LSW; however, there was no separate report of the validity of the TAG score in late time windows. Still, the score may be used after 6 h from LSW. In Asian populations, having possibly higher sICH risk after EVT, 101 presumably because of increased intracranial atherosclerosis prevalence, 102 the ASPECTS, Baseline Glucose, Poor Collateral Circulation, Passes With Retriever, and Onset-to-Groin Puncture Time (ASIAN) score has been developed based on data derived from the Endovascular Treatment for Acute Anterior Circulation Ischemic Stroke Registry (ACTUAL) in China. 103 Only one in four patients had groin puncture more than 6 h from LSW, limiting the ASIAN score applicability in late time windows. 104 Finally, a nomogram has been derived from the Italian Registry of Endovascular Stroke Treatment in Acute Stroke (IER) and, once again, time to treatment was independently associated with sICH risk, along with collateral status, nonsuccessful recanalization, advanced age and higher baseline NIHSS; however, most patients were treated early and only one in four patients had groin puncture >280 min from LSW. 105 Peri-procedural risk factors associated with increased sICH risk are beyond the scope of this review. In conclusion, comprehensive patient selection for off-label EVT in extended time windows dictates a multidisciplinary approach, involving neurologists, neuroradiologists and neurointerventionalists.
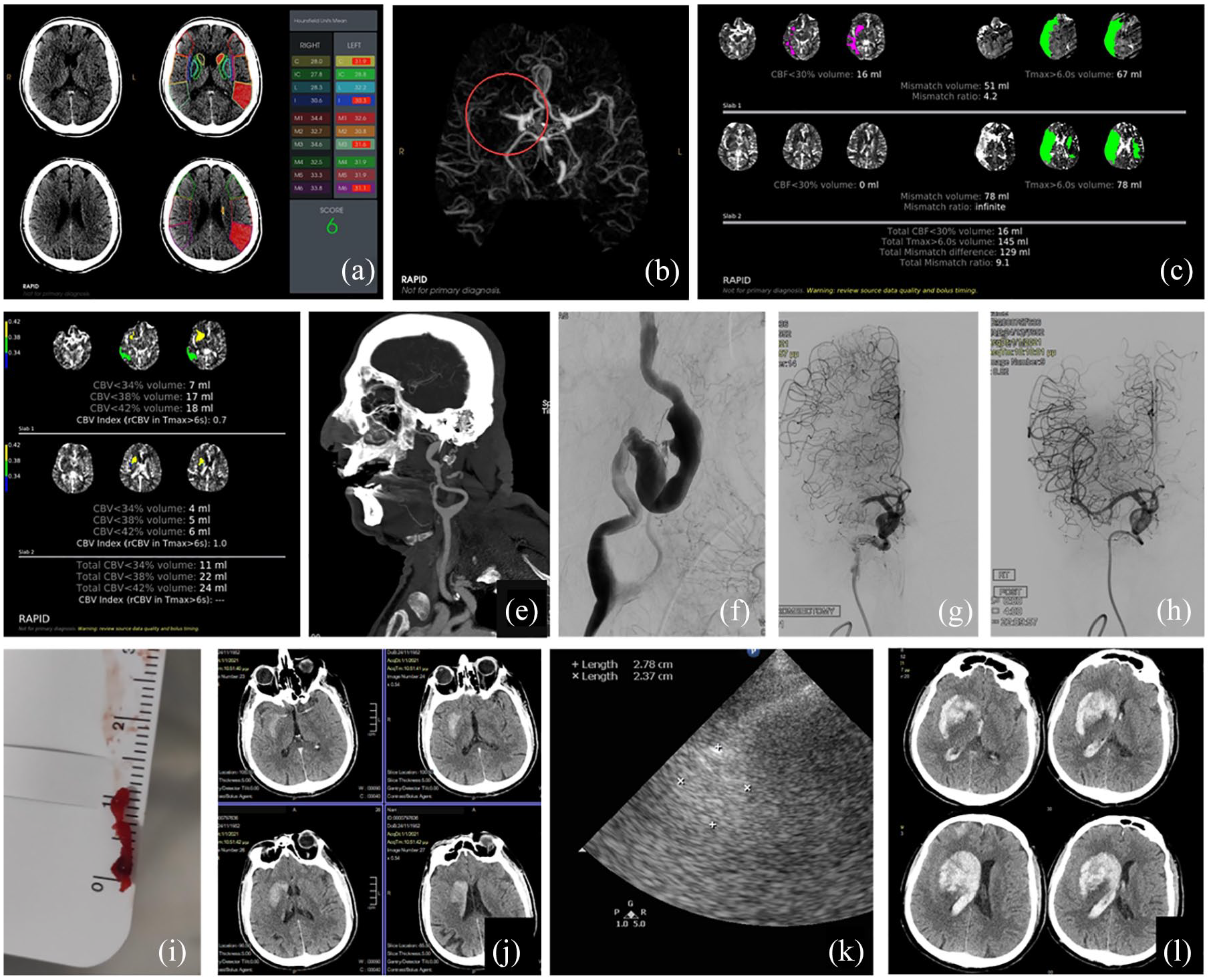
A 68-year-old man with a history of atrial fibrillation and hypertension, treated with 15 mg rivaroxaban once daily the day before symptom onset, presented with left hemiplegia, left hemineglect and left sensory deficits (NIHSS score 19 points) 10 h after LSW. CT showed hypodensity of the right lentiform nucleus (ASPECTS 6) whereas automated ASPECTS erroneously estimated a score of 6 on the left (normal) brain hemisphere (a). Intracranial CTA showed a right M1 occlusion (b). RAPID CT perfusion showed an estimated ischemic core volume of 16 ml and Tmax > 6 s of 145 ml, slightly overestimated as it comprised also areas of the left (normal) brain hemisphere, due to patient movement (c). EVT was decided according to the DEFUSE 3 criteria. Despite the fact that CBV maps are not part of the inclusion imaging criteria of DAWN or DEFUSE 3 studies, it is of note that the patient showed a restricted area of 11 ml involving the right lenticular nucleus with very low CBV (<34%, d). Extracranial CTA showed a giant partially thrombosed aneurysm of the right ICA (e), that was confirmed with DSA (f). The patient had good collaterals on DSA and full recanalization with one pass was achieved with a Solitaire stentriever and Penumbra thromboaspiration device (Solumbra technique) without the use of a balloon-guided catheter due to the cervical ICA aneurysm (h). A 12-mm thrombus was retrieved (i). Post-EVT noncontrast CT showed luxury perfusion and contrast uptake in the left basal ganglia (j). Despite aggressive BP management (SBP target < 140 mmHg) the patient suddenly developed stupor 10 h later (NIHSS 21). Emergent transcranial Doppler at the bedside showed preserved right M1 patency, but a 2.8 × 2.4 cm hyperechogenic lesion was shown with transcranial sonography (k). Noncontrast brain CT confirmed a massive parenchymal hematoma type 2 with intraventricular extension (l). The patient was intubated and an external ventricular drain was placed. He remained intubated 1 month after treatment.
Expert opinion: EVT may be offered as an off-label therapeutic option in selected AIS patients presenting 1–6 days after LSW if they fulfill DAWN criteria and carry a low risk of reperfusion hemorrhage; a multidisciplinary team should carefully take treatment decisions.
Perfusion imaging in AIS: the role of post-processing software
Perfusion software uses four-dimensional data (volume over time) to produce cerebral hemodynamic maps that reflect the probability of infarction in case of nonrecanalization. An extensive presentation of perfusion imaging in stroke is beyond the scope of this narrative review.106,107 There are reasons to explain why brain perfusion imaging has not received prime time in AIS until recently. In spite of its long history in brain research and brain tumor imaging, acute hemodynamic disturbances caused by LVOs and the need for reliable image post-processing in the acute phase of stroke, rendered a fast and accurate depiction of the ischemic core and penumbra challenging. All positive studies presented in Table 1 used RAPID software imaging for acute CT perfusion and MR perfusion for all or most of their patients. Being fully automated, it obviates the need for a neuroradiologist on site, a luxury for many stroke centers during weekends and after hours. Besides allowing for advanced neuroimaging around the clock for all recruiting centers in multicenter studies, central and automated imaging processing gave unprecedent internal validity to AIS RCTs, permitting reliable comparison of treatment effects regardless of the infrastructure of each center. Last, RAPID software has shown superior accuracy to other commercially available software packages in peer-reviewed articles. We have shown that reported differences in imaging interpretation are important and appear to limit the use of the DAWN and DEFUSE 3 criteria on core volumes derived from other programs, as they might substantially overestimate the ischemic core volume and lead to the inappropriate disqualification of patients who are eligible for MT. 108 It is clear that the generic term ‘perfusion imaging’ cannot be applied to different software that provide different results for the same patient. Many studies of perfusion imaging in AIS may use software that is suboptimal to the one used in positive published RCTs. 109 However, there is always room for improvement and research is ongoing; a correction for presumable core overestimation in white matter may further improve RAPID CTP accuracy. 110 The parameter of time from onset may also prove to play a role in alternative processing software.109,111 Current single-value thresholds to predict tissue outcome may oversimplify ischemic tissue fate; increased complexity generated by accumulating data may be solved by deep-learning algorithms in the foreseeable future. 112
Advanced neuroimaging for the selection of patients with distal intracranial occlusions for EVT
All RCTs and most observational studies on EVT mentioned in this narrative review have recruited AIS patients with anterior circulation LVOs involving mostly ICA and/or MCA M1 occlusions. Progress in neurointerventional material and techniques continuously expands treatment frontiers to include more-distal occlusions. A recently published comprehensive review on EVT for distal, medium vessel occlusions (DMVOs) proposed the term LVO to be reserved for ICA, MCA M1 segment, basilar and vertebral artery, all vessels with diameters usually > 2mm, whereas DMVOs refer to more-distal arterial segments of diameter 0.75–2 mm. 113 Detection of DMVOs with CTA in the acute phase may be challenging even for experienced neuroradiologists; CTP has been shown to increase the yield of CTA for DMVO detection by revealing brain ischemia and directing attention of the examiner to a small part of the arterial tree.114,115 Despite the paucity of evidence for the role of EVT for DMVOs in extended time windows, the possibility for treatment should be considered, especially for cases with severe clinical deficit as these occlusions are associated with a high risk of long-term disability. 116
There are many barriers to extrapolating data from LVOs to DMVOs: extreme variation in documentation, definitions, quality of data, ASPECTS not helpful in M2, core sizes are much smaller, mismatch calculation may need other perfusion parameters (Tmax > 6 s may be too long for distal occlusions), collateral filling is faster and collateral scores may have to be adapted accordingly, and mRS endpoints may be different. 117 DMVOs are an exciting frontier in AIS recanalization therapies and advanced neuroimaging for proper patient selection is an area of intense research but, on the time of writing of this narrative review, no recommendations can be safely made for DMVO treatment in extended time windows.
Effective EVT in advanced time windows without advanced neuroimaging: fact or fiction?
Every hour of delay in successful recanalization is a significant factor related to long-term independence with an estimated 1.5% decreased probability of FFO per hour delay of reperfusion. Late presenters have lower rates of successful and excellent reperfusion, higher complication rates and may need increased number of passes during EVT. 118 There is no high-level evidence from RCTs proving efficacy of EVT in extended time windows without advanced neuroimaging. Real-world data from the North American Solitaire Stent Retriever Acute Stroke (NASA) and the Trevo Stent Retriever Acute Stroke (TRACK) registries suggest EVT is equally safe and effective before and after 6 h. 119
The arguments in favor of standard neuroimaging for extended time windows are the following: EVT is both safe and effective; initial studies have shown efficacy but had too restrictive inclusion criteria; relaxing these criteria will increase benefit for more stroke patients, not only those with the most favorable imaging profile. Opinion leaders in EVT around the globe have favored the standard neuroimaging approach. Many of them have co-authored the recent manifesto that challenges the ischemic core concept in AIS imaging. 120 They describe three shortcomings related to the current advanced imaging paradigm:
CT and MRI cannot reliably determine tissue viability, their use is therefore not justified for patient selection.
There is more to stroke than imaging; clinical factors should play a role in decision-making.
Discrepancies between core volume and clinical outcome are not unusual.
The first two alleged problems are general principles that every stroke physician would support but are slightly irrelevant since RCTs have proven that ‘imperfect’ advanced neuroimaging works well enough for patient selection and the same RCTs used clinical in addition to neuroimaging criteria; the only way to improve acute stroke care is through new RCTs comparing novel clinical or imaging strategies to the ones already approved. We should keep in mind that before 2015 there were three negative RCTs for EVT: SYNTHESIS Expansion Trial, Mechanical Retrieval and Recanalization of Stroke Clots Using Embolectomy (MR RESCUE), and Interventional Management of Stroke (IMS) III.121–123 Although there were numerous reasons for their negative results,124,125 it might be worth mentioning that important lessons were learned on the role of the imaging selection of patients was also key for the success of the later trials.126,127 Current imaging selection criteria are thus the product of many trials and errors and are supported by positive RCTs; they can therefore not be changed in the absence of equally high-quality data.
The third argument is more challenging to tackle. It is true that many patients with extensive infarctions of the brain may well have a favorable prognosis. Restricting brain volume criteria would deprive these patients from a meaningful benefit from an intervention. A meta-analysis of seven RCTs in the early time window confirmed that final infarct volume is a strong predictor of long-term outcome but also that there are quite a few patients with large-volume strokes (even >100 ml) that fare well (mRS scores of 0–3), whereas others with moderate-volume infarctions (<50 ml) have grave prognosis (mRS scores of 4–6). 128 It is obvious that volumetric infarct description completely omits the eloquence of brain tissue from prognostication. A young patient with a massive right MCA infarction of 100 ml will most probably have a different outcome depending whether the infarct extends by <10 ml towards the internal capsule (Figure 2); when eloquent brain tissue is at stake, effective recanalization and salvage of a small brain area might make the difference by saving the patient from decades of disability, but we still do not have at our disposal the tools to add quality to quantity. 129 What is more important though, incorporating qualitative data will obviously need much more, not less, advanced neuroimaging. Preliminary results with mRS-weighted brain maps published by MR CLEAN trial investigators have shown improved outcome prediction130,131 and further research is underway to provide a ‘stroke brain atlas’, 132 despite considerable complexity arising not only from the difficult task of delineating the most incapacitating brain lesions, but also from the importance of their interaction in determining final clinical outcome. 133
Leaving aside tissue-at-risk eloquence, the debate whether we should allow EVT for larger infarct volumes on the grounds of excellent EVT benefit–risk ratio in most subgroups of LVO AIS patients is an important one. EVT did not show benefit in stroke patients with very low ASPECTS (0–2) in the HERMES meta-analysis. 7 Benefit was significant for ASPECTS 3–5, but patients with such extended infarcts were excluded from late time window trials: the DAWN trial excluded patients with estimated ischemic core volumes >50 ml and there were only 18 such patients in the DEFUSE 3 trial.74,78 Even in earlier windows, a pooled analysis from the Optimizing Patient Selection for Endovascular Treatment in Acute Ischemic Stroke (SELECT) and Trevo registries demonstrated marked variability in outcomes in patients with ASPECTS of 3–5 with differing baseline ischemic core volumes. 134 A multicenter, retrospective study conducted by the Jeunes en Neuroradiologie Interventionnelle Research Collaborative (JENI-RC), analyzed data from 172 patients with large ischemic cores (mean volume 102 ml); core perfusion mismatch ratio [defined as the volume of critically hypoperfused tissue (Tmax > 6 s) divided by the core volume] >1.72 increased the odds of 3-month functional independence. 135 Ongoing trials are investigating EVT in patients with large ischemic cores: Thrombectomy for Emergent Salvage of Large Anterior Circulation Ischemic Stroke (TESLA; ClinicalTrials.gov identifier: NCT03805308); Efficacy and Safety of Thrombectomy in Stroke With Extended Lesion and Extended Time Window (TENSION; ClinicalTrials.gov identifier: NCT03094715); A Randomized Controlled Trial to Optimize Patient’s Selection for Endovascular Treatment in Acute Ischemic Stroke (SELECT 2; ClinicalTrials.gov identifier: NCT03876457); Large Stroke Therapy Evaluation (LASTE; ClinicalTrials.gov identifier: NCT03811769); and Mechanical Retrieval and Recanalization of Stroke Clots Using Embolectomy (RESCUE-Japan LIMIT; ClinicalTrials.gov identifier: NCT03702413).
On the other hand, standard neuroimaging for all stroke patients, before or after 6 h after symptom onset, has several benefits. It is a cheaper and faster approach, increasing the percentage of eligible LVO patients and, probably even more importantly, the number of stroke centers that can provide EVT in extended time windows. A most recent study reported that stroke centers that routinely use CTP for EVT patient selection tended to treat fewer LVO patients without any difference in outcomes or complications compared to stroke centers that usually selected patients with no perfusion imaging. 136 The under-treatment bias with routine CTP seemed to be maintained in the late time window patients. However, the question of safety is even more pressing than that of benefit. The SELECT prospective study, studied CT and CT perfusion images from AIS patients mostly in early time windows (<8 h) who were treated either with EVT or conservative treatment at the treating physician’s discretion. Patients with favorable CT but unfavorable CT perfusion profiles had lower functional independence and high rates of symptomatic ICH, neurological worsening, and mortality. 137 A recent commentary of the SELECT2 Investigators meticulously delineates the uncertainty behind simple neuroimaging for patients with larger cores and is relevant for the debate for extended time windows. 138 Using the Alberta Stroke Program Early CT score (ASPECTS) to estimate established infarction is a strategy appearing simple but in practice is prone to errors; even after ASPECTS training, one in three readings from neuroradiologists resulted in misclassification (ASPECTS 0–2 versus 3–5 versus 6–8) in 20 illustrative cases. 139 Automated ASPECT evaluation is far from perfect, as our illustrative case shows [Figure 4(a)].
Supposing that EVT is safe in late time windows, even a marginal benefit from recanalization therapies may prove to be significant. It is of interest that in many RCTs, EVT did not increase rates of hemorrhagic complications, notably sICH. sICH (3.8% versus 3.5%, p = 0.90) and parenchymal hematoma type 2 (5.6% versus 4.8%, p = 0.52) did not differ between the EVT and control groups in the HERMES meta-analysis. However, real-world data from a large French registry (ETIS) raise the rate of PH2 after EVT at 11%; however, no sICH rates were reported. 89 Benefit of EVT was preserved in the HERMES meta-analysis, even for infarcts affecting more than 33% of the MCA territory or with ASPECTS <6, but the risk of sICH was higher in the EVT group than in the control group. 140 In extended time windows, neither DAWN nor DEFUSE 3 trials showed any difference in sICH or PH2 between study arms; however, both studies used advanced neuroimaging and selected patients with favorable penumbral pattern, notably small volume ischemic core lesions (Table 2). Late presenters in the ESCAPE trial had higher rates of asymptomatic intracerebral hemorrhage with EVT (48.5% versus 11.5%, p = 0.004) but there was no sICH recorded in either the interventional or the conservative treatment arms. An observational single-center study included 63 anterior circulation LVO stroke patients, either WUS or presenting 6–24 h after symptom onset; patients were eligible for EVT if ASPECTS ⩾7, whereas no collateral assessment was performed. sICH rates were only 3%, similar to the 4% sICH rates in patients treated <6 h from symptom onset in the same center. 141 An Irish retrospective multicenter study included 25 patients (WUS or symptom onset >12 h) that were treated with EVT provided they presented good or moderate collateral status on multiphase CTA according to the ESCAPE trial criteria. 142 No cases of sICH were reported, an important finding for one of the rare studies of EVT in extended time windows based solely on multiphase CTA for patient selection. A retrospective analysis of 32 patients that were treated with EVT >6 h from LSW using DEFUSE 3 MRI criteria but had previously had CTA (on a primary stroke center or on site), showed that for patients with good CT ASPECTS (8 or higher), and good CTA collateral score, DWI findings were rarely discordant. Of course, all included patients had good MR perfusion profiles, meaning that no conclusion can be inferred regarding the diagnostic accuracy of CTA. However, a key complementary finding was that DWI offered the greatest specificity for excluding ineligible patients, thus, when facing a questionable CT/CTA-based imaging result, DWI might be the most specific test to make treatment decisions. 143
Advanced neuroimaging parameters used in positive RCTs (DAWN, DEFUSE 3, EXTEND, WAKE-UP), post hoc RCT analysis (THAWS substudy) and case-control study (Seoul) of AIS recanalization treatment in extended time windows or WUS; all studies provide ischemic core volume on presentation. Cohort studies without control arms that showed safety of IVT in Erlangen and safety and efficacy of EVT comparable to the results of the DAWN study (Pittsburg) are also presented. EVT studies involve exclusively anterior circulation LVO. Please note that despite allowing for moderate-volume stroke treatment, all studies actually treated mostly patients with small or no infarction cores.
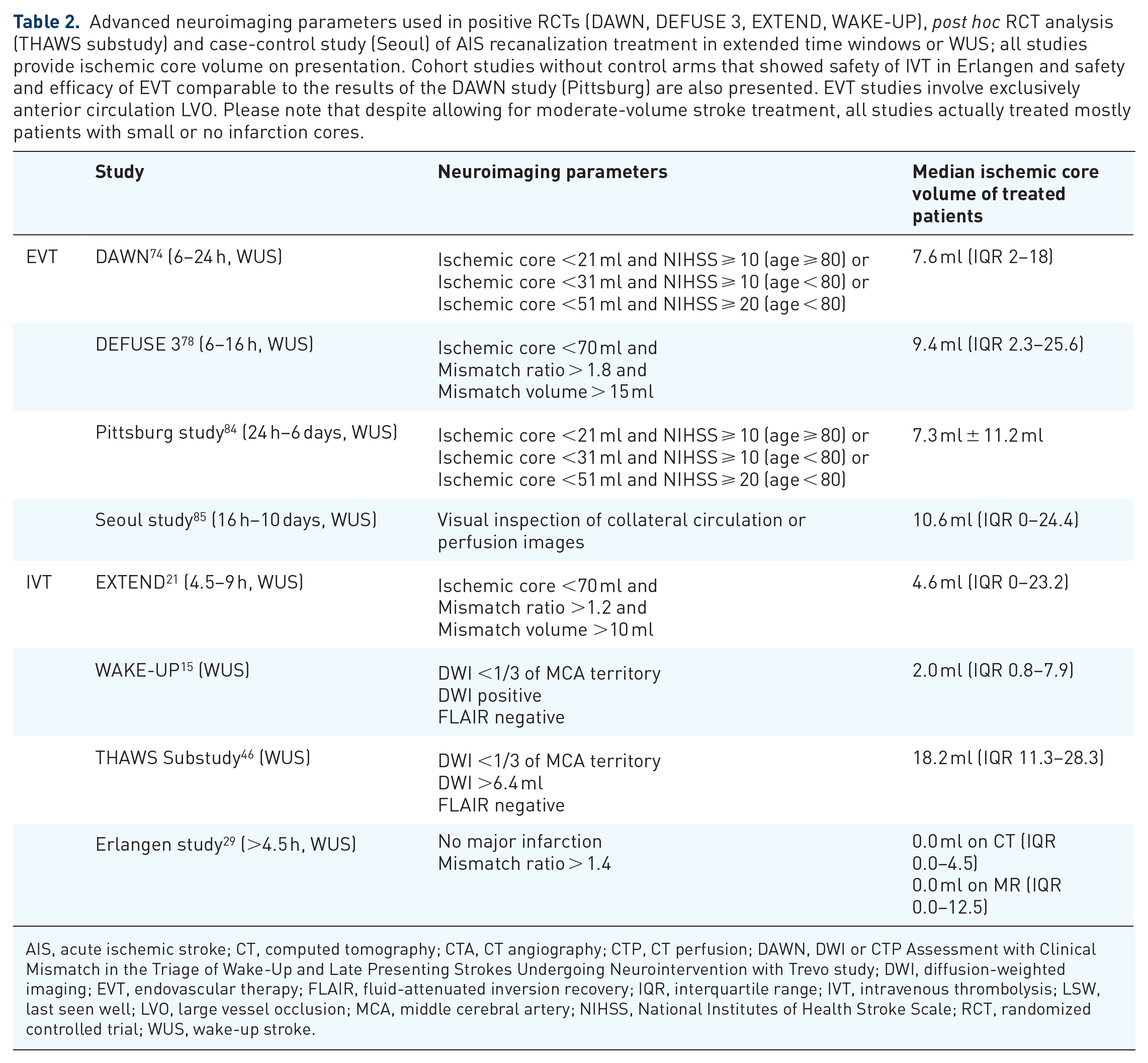
AIS, acute ischemic stroke; CT, computed tomography; CTA, CT angiography; CTP, CT perfusion; DAWN, DWI or CTP Assessment with Clinical Mismatch in the Triage of Wake-Up and Late Presenting Strokes Undergoing Neurointervention with Trevo study; DWI, diffusion-weighted imaging; EVT, endovascular therapy; FLAIR, fluid-attenuated inversion recovery; IQR, interquartile range; IVT, intravenous thrombolysis; LSW, last seen well; LVO, large vessel occlusion; MCA, middle cerebral artery; NIHSS, National Institutes of Health Stroke Scale; RCT, randomized controlled trial; WUS, wake-up stroke.
A recent paper from the ESCAPE study group analyzed imaging and clinical data of 35 patients treated with EVT > 6 h from LSW that had both multiphase CTA and CT perfusion prior to treatment; multiphase CTA had the highest likelihood of discriminating those who would have good clinical outcome after treatment. 144 In the aforementioned Korean study, post hoc analysis using imaging eligibility criteria from the ESCAPE, DAWN and DEFUSE 3 trials did not modify EVT effectiveness; the DEFUSE 3 trial imaging criteria seemed to be slightly superior for patient selection but without reaching statistical significance. 85 In the Safety and Efficacy of Nerinetide (NA-1) in Subjects Undergoing Endovascular Thrombectomy for Stroke (ESCAPE-NA1) trial, patients were treated with EVT 6–12 h from LSW based on ASPECTS and multiphase CTA collateral status. 145 In a post hoc analysis of the trial, treated patients showed similar baseline characteristics to patients included in the DAWN and DEFUSE 3 EVT arms. Despite larger infarct volumes, FFO rates were similar across the three trials (mRS 0–2 in ESCAPE-NA1 45%; DAWN, 49%; DEFUSE 3, 45%). 146
In conclusion, data on standard neuroimaging for EVT patient selection in late time windows are scarce, derived from small retrospective studies in selected stroke study groups. On the other hand, EVT appears to be safe even after 6 h from LSW. A most recent retrospective analysis of the Acute Stroke Registry and Analysis of Lausanne (ASTRAL registry) reported similar rates of procedural complications (either technical or cerebrovascular, including parenchymal hematoma) for early versus late EVT (16.2% versus 16.3%). 147 Carefully designed prospective late time window EVT studies that may integrate multiphase CTA in a CT perfusion protocol of proven efficacy, will allow head-to-head comparison of the two imaging strategies in the future. The ongoing MR CLEAN Late trial will show if a standard imaging protocol based on CTA and collateral assessment may be used for late time windows (https://mrclean-late.nl/). Inclusion and randomization will be restricted to patients with moderate or good collaterals after 100 patients with poor collaterals will have been included in the study.
Expert opinion: data on advanced neuroimaging for patient selection for EVT after 6 h from LSW are of the highest quality. Collateral assessment through CTA is preferred by some EVT experts but supporting data are of low quality. In cases or centers without advanced neuroimaging, multiphase CTA may identify patients with moderate to good collaterals that can possibly be treated safely and effectively with EVT up to 12 h from LSW according to the ESCAPE trial protocol and after 12 h in low-risk patients for hemorrhagic transformation.
Critical comparison of perfusion imaging and collateral assessment by CT/MR angiography
Both imaging modalities depict different facets of reversible (penumbra) and irreversible (ischemic core) brain ischemia. Penumbral imaging is the present of the oligemic brain; collateral status shows the past and predicts the future. If an accurate depiction of the penumbra is a snapshot at the timepoint of imaging, it is through good collaterals that brain parenchyma survived in extended time windows and persisting adequate collateral vessels will salvage the brain until successful recanalization is achieved with EVT. 148 Brain regions with Tmax > 10 s delay are likely to have poor collateral flow. The ratio of the volume of tissue with Tmax > 10 s compared with the Tmax > 6 s volume is referred to as the hypoperfusion intensity ratio (HIR). HIR > 50%, correlates with poor angiographic collaterals, and these patients have larger baseline ischemic core volumes and more rapid infarct growth. 149 A total of 123 DEFUSE 3 patients repeated RAPID automated perfusion imaging at 24 h post-randomization; collateral robustness was assessed with two integrative perfusion metrics: HIR and the CBV index, the relative CBV in the Tmax > 6 s region). 150 Higher HIR and lower CBV index values, reflecting poor collaterals, were associated with larger final infarct volumes than those predicted from baseline imaging. 151 These indices of CTP-derived collateral circulation assessment may be used in clinical practice for patients transferred for emergent EVT after CTP; estimated robust collaterals may obviate the need for repetition of noninvasive brain imaging at arrival at the stroke center.
Baseline multiphase CTA images may also be processed through a simple perfusion reconstruction algorithm (SPIRAL) to produce perfusion maps that have good agreement with a commercially available deconvolution software (CT Perfusion 4D; GE Healthcare). 152 If these preliminary findings are confirmed by subsequent studies, standardized SPIRAL automation may prove equivalent with CTP, saving time, radiation, contrast injection and, eventually, financial resources. One-stop management of AIS patients in the angiosuite is an exciting recent development that integrates advances in penumbral 153 and collateral 154 neuroimaging with the use of flat-panel detector CT and is currently being tested in AIS patients. Such a development will eventually allow for rapid patient selection and prompt treatment with IVT and EVT in eligible patients even in extended time windows. 155
Bridging therapy in WUS and after 4.5 h from LSW
IVT prior to EVT could have several favorable effects: complete and faster recanalization, partial thrombus resolution that prolongs the window of opportunity with a definite treatment through EVT, dissolution of residual peripheral thrombi post-EVT. On the other hand, IVT takes time, might delay treatment, increases the risk of sICH and increases care costs. 156 Two 157,158 out of three157–159 recent RCTs have shown noninferiority of direct EVT compared to bridging therapy within the 4.5-h time window of IVT, provided that the interventional treatment can be performed quickly and reliably. SKIP failed to demonstrate noninferiority. 159 However, in early time windows and when immediate EVT is unavailable, IVT has shown a 18% early recanalization rate in a meta-analysis that included pre-IVT vascular imaging. 22 This substantial effect underlines the importance of pre-EVT thrombolysis when EVT is impossible for technical reasons (e.g. difficult arterial access) and especially in the drip-and-ship paradigm, when transfer delays may render futile EVT recanalization and also explains the strong recommendation of IVT in all eligible patients in early windows.
EVT RCTs in extended windows have recruited a small number of patients that had been unsuccessfully thrombolysed (with regard to recanalization) within 4.5 h from symptom onset. In the DAWN study, 5% of patients (n = 5) were thrombolysed according to guidelines (<4.5 h) and all were transferred cases. Median duration of symptom onset to EVT was 13 h. Although transferred patients had a faster door-to-puncture time, benefits of EVT were similar between direct and transferred patients. In DEFUSE 3 the corresponding patients represented 11% of the study group (n = 10). Among 10 IVT-treated patients, 9 were transferred cases. In the DEFUSE 3 subgroup of transferred cases, the median time interval from symptom onset to EVT was 11 h. No heterogeneity for the EVT treatment effect was observed in direct versus transferred status. In both trials, no IVT was performed in the control group. 85 As a result, a patient that has been unsuccessfully treated with IVT <4.5 h may be treated with EVT in extended time windows if DAWN or DEFUSE 3 criteria are fulfilled, but these patients were a small subgroup of the aforementioned studies and more data are needed.
On the contrary, performing IVT for WUS or AIS with >4.5 h from LSW as part of bridging therapy has not been tested for efficacy or safety in clinical trials and even observational data are scarce. 160 As a result, this is an off-label treatment and, contrary to early time windows, any physician should be hesitant in performing IVT in this subgroup of AIS patients that are destined for EVT. Since most DWI(+)/FLAIR(−) patients have symptom onset <4.5 h it would be logical to propose bridging therapy for WAKE-UP study-eligible patients. Lesion volume was an independent risk factor of hemorrhagic infarction in a post hoc analysis of the WAKE-UP trial; 161 however, it was reassuring that no association was found between ischemic core volume and parenchymal hematoma, which was the only type of hemorrhagic transformation that seemed to affect outcome. Even so, most patients in the WAKE-UP study had ischemic core volume <8 ml (ischemic core volume interquartile range of treated patients 0–7.9 ml) and this could be used as an additional criterion for bridging therapy until data specific for bridging therapy in WUS become available. Moreover, high microbleed burden (>10) has been recognized as an independent risk factor of symptomatic intracranial bleeding. 162 It should be stressed that high microbleed burden does not preclude treatment for WUS according to the WAKE-UP criteria, but it should probably preclude off-label bridging therapy.
The decision for bridging therapy in extended time windows according to the EXTEND trial protocol is harder to make. One can argue that the worse possible outcome for LVO AIS patients is persistent occlusion and therefore support the use of all available recanalization therapies. Since this is a gray area, clinical decisions depend on local availability of resources and individual patient characteristics. The most important factor to consider is, obviously, time. One might consider withholding IVT in a stroke center with a stand-by EVT team, but it would also be appropriate to thrombolyse an LVO patient, who fulfills WAKE-UP or EXTEND criteria and has an overall low risk sICH profile, in a stroke unit and then transfer the patient to a stroke center for emergent digital subtraction angiography (DSA) and, if recanalization has not been achieved, then proceed with EVT. 163 Time metrics are extremely important to reach the right decision and every stroke network should incorporate regularly updated time metrics data in treatment algorithms. By using transcranial Doppler (TCD)monitoring during IVT for LVO strokes (mostly MCA occlusions) in early time windows, we have shown that alteplase leads to sustained complete or partial recanalization in 60% of treated patients at 2 h after bolus. 5 Another study group also reported an increase in recanalization rates during the first 2–3 h after the initiation of alteplase infusion. 164 Hence, EVT recanalization prediction >2 h after IVT initiation could be used as a time limit for deciding whether to proceed with bridging therapy or not. Serendipitously, the same time window of 2 h is currently used in clinical practice in cardiology for ST-segment elevation myocardial infarction: if estimated time to ‘wire crossing’ (reperfusion) of the infarct-related artery through percutaneous coronary intervention (PCI) exceeds 120 min, eligible patients receive the bolus of fibrinolytic and are transferred to a PCI center. 165
For patients presenting directly to a thrombectomy center with ischemic stroke of 4.5–9 h duration (known onset) with CT or MRI perfusion core/penumbra mismatch and who are eligible for EVT, the group members formulating the updated ESO IVT guidelines could not reach a consensus regarding whether IVT should be used before EVT. For patients presenting to a nonthrombectomy center with ischemic stroke of 4.5–9 h duration (known onset) with CT or MRI perfusion core/penumbra mismatch and who are eligible for mechanical thrombectomy, six of nine group members suggested IVT before EVT (expert consensus statement). 24
Individual patient characteristics may also play a role. From the safety perspective, clinicians could opt for direct EVT and skip IVT in LVO patients with large core, especially in the concomitance of risk factors for hemorrhagic transformation as presented in Table 1. From the efficacy perspective, angioarchitecture and clot characteristics on CTA may aid in predicting IVT effectiveness. 166 Smaller clots are easier to dissolve with IVT. 167 Clot migration has been considered a potential adverse event of IVT, precluding thrombus retrieval through EVT, 156 but recent data from the EXTEND trial are reassuring that the risk of thrombus dislodgement from a retrievable to a nonretrievable location does not rise with IVT compared to placebo. 168 Another significant factor is the site of occlusion. Carotid occlusions, either tandem or terminal ICA, are notoriously resistant to complete recanalization with IVT. 55 However, a recent observational study from the German Stroke Registry-Endovascular Treatment in tandem LVO patients undergoing EVT, reports higher odds of successful reperfusion in those pretreated with IVT, presumably through recanalization of the intracranial occlusion. 169 Tandem occlusions are unique in another aspect that increases hemorrhagic risk with IVT: the frequent need to stent an occlusion in the acute phase of stroke necessitates immediate initiation of antiplatelet therapy, ideally double, as well as a heparin IV bolus, drugs that increase hemorrhagic risk in the first 24 h post-IVT. 170 Last but not least, post-IVT blood pressure (BP) management in tandem occlusions is a double-edged sword: overly reducing the BP in a patient with occluded ICA risks extension of the infarct before achieving successful recanalization with EVT, but permissive hypertension post-IVT in order to save brain tissue at risk is associated with an exceedingly high risk of hemorrhagic transformation after IVT. 171
Improved lytic therapies for stroke are an exciting field of research; the Tenecteplase versus Alteplase before Endovascular Therapy for Ischemic Stroke (EXTEND-IA TNK) trial has showed superior efficacy of IV 0.25 mg/kg tenecteplase bolus. 172 Tenecteplase may result in successful recanalization and therefore avert EVT in up to one in five LVO treatments.173,174 However, tenecteplase has not yet been tested in RCTs in late time windows and cannot be recommended.
Expert opinion: bridging therapy has not been tested in RCTs > 4.5 h from LSW or in WUS. Since the DWI(+)/FLAIR(−) pattern is usually seen with symptom onset < 4.5 h, IV alteplase before EVT could be considered in selected FLAIR(−) patients that are considered to carry low overall post-IVT sICH risk, have low (<10) microbleed burden on susceptibility weighted imaging and low infarct volume (<8 ml) on DWI, only when recanalization with EVT is not imminent (predicted time > 120 min). CTP imaging as used in the EXTEND trial may also be applied to select low-risk patients for bridging therapy, if there is small clot burden (indicating higher odds of successful recanalization) and very limited or no ischemic core present.
Discussion
Facing a WUS or an AIS in late time windows, treatment algorithms are dictated by the availability of EVT as well as the imaging studies that a stroke physician may order. A MR perfusion read by an expert neuroradiologist or processed automatically by RAPID software will not be available in most stroke centers and may not be necessary in most late presenters. Recent advances in CT neuroimaging alleviate the need for MRI in most AIS clinical scenarios; IVT for WUS may be an exception, since we now have at our disposal two treatment paradigms, one based on perfusion imaging and another based on MRI, both of which may be used to select AIS patients for systemic thrombolysis. In most centers, clinicians will be obliged to form treatment algorithms according to local infrastructure. In sharp contrast to the exciting advances presented in this narrative review, the current state of AIS treatment in many if not most stroke units around the globe is the linear model of IVT, offered as the only available reperfusion therapy, while patient selection is based solely on noncontrast brain CT (Figure 5).175,176

Absence of advanced neuroimaging and endovascular treatment possibility (on site or at proximity) permits only for this linear recanalization treatment paradigm. A single imaging modality, one recanalization treatment and a single thrombolytic agent, allow only for IVT up to 4.5 h from known symptom onset. This has been the mainstay of AIS treatment for almost two decades but has been rendered obsolete in recent years, precluding life-saving therapy from many AIS patients with salvageable brain tissue (strong recommendation (R), high quality of evidence (Q) according to ESO IVT recommendations). 12
Discrepancies in stroke care have been described in European countries 177 and repeatedly become evident during the annual International Symposium on Collaterals to the Brain, a multidisciplinary scientific conference devoted to key neuroimaging issues in acute stroke therapy across the globe (http://collateralperfusion.org/).We have provided many figures with examples of treatment algorithms from different starting points that could aid in getting the most out of each hospital center’s capabilities, in order to treat as many AIS patients as possible according to the most updated data (Figures 6–12). Standard neuroimaging to select patients for EVT after 6 h from LSW is ardently supported by many opinion leaders in EVT around the globe and data are accumulating for its safety and superior inclusiveness compared to advanced neuroimaging; however, high-quality evidence is still missing. Collateral assessment through multiphase CTA is a promising tool that is readily available in every stroke center and may be used up to 12 h from symptom onset when perfusion imaging is unavailable, according to the ESCAPE clinical trial protocol. Still, the current status of AIS reperfusion treatments in extended time windows is dominated by high-quality data from RCTs that used advanced neuroimaging (CT/MR perfusion or MRI) to select patients.

MRI availability in the acute ischemic stroke phase permits IVT with alteplase in WUS according to the WAKE-UP study protocol (strong recommendation (R), high quality of evidence (Q) according to ESO IVT recommendations; Class I/Level-BR). 12 EVT is unavailable in this scenario.

CT perfusion imaging using an on-site validated perfusion protocol or an automated RAPID protocol, significantly expands IVT applicability. Alteplase may be administered up to 9 h from symptom onset and in WUS according to the EXTEND study criteria (strong recommendation, moderate quality of evidence according to ESO IVT recommendations; Class I/Level-BR): the midpoint of sleep (e.g. 04:00 if the patient fell for sleep at midnight and woke up with symptoms at 08:00) is considered the starting point; IVT can be performed in patients with favorable penumbral imaging up to 9 h after (in this example up to 13:00). The Erlangen study criteria 29 of MQ > 1.4 may be safe in patients with ischemic core <5 ml after 9 h from LSW as off-label IVT treatment (Exp Op). EVT is unavailable in this scenario.
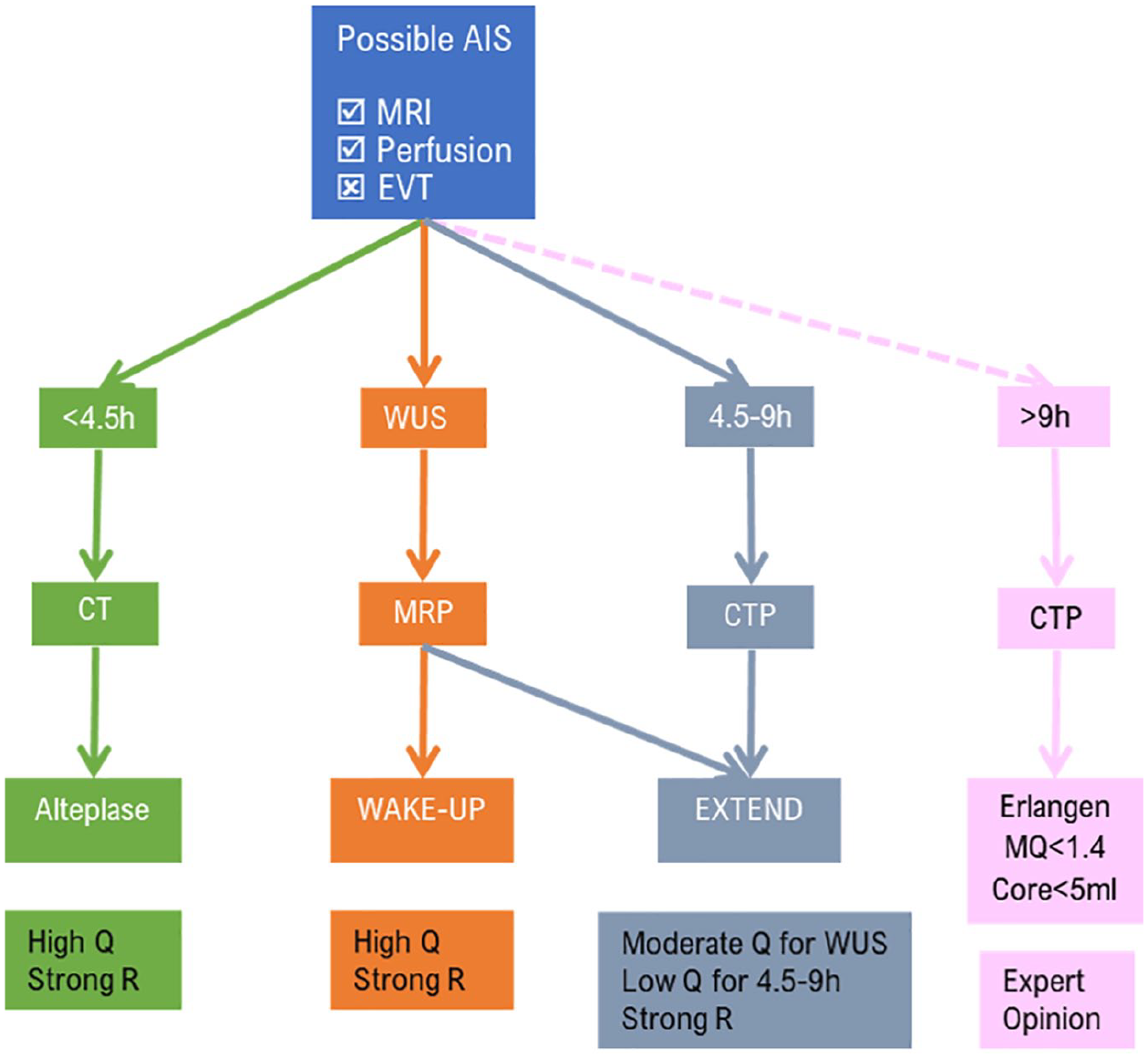
Full neuroimaging package (both MR and perfusion imaging with CT or MRI) does not add much in the applicability of IVT in comparison to CT perfusion. MR perfusion may be preferred over CT perfusion for WUS, as this allows for recanalization treatment in the rare instance of a WUS patient that fulfills WAKE-UP but not EXTEND study criteria (e.g. lacunar stroke <4.5 h from awakening). The Erlangen study criteria 29 of MQ > 1.4 may be safe in patients with ischemic core <5 ml after 9 h from LSW as off-label IVT treatment (Exp Op). EVT is unavailable in this scenario.

EVT availability considerably expands treatment option even in the absence of advanced neuroimaging. Treatment algorithms diverge for LVO and non-LVO AIS patients. Bridging therapy up to 4.5 h has an AHA/ASA Class IA recommendation and high-quality, strong recommendation in the ESO IVT guidelines. 12 Use of TNK 0.25 mg/kg as a bolus IV may be preferred over alteplase (low quality/weak recommendation according to ESO recommendations for IVT; Class IIb/Level B-R). 12 EVT with standard neuroimaging is strongly recommended up to 6 h. In the HERMES meta-analysis, benefit from EVT using only CTA persisted up to 7 h and 18 min from symptom onset to recanalization time 178 ; a time limit also proposed by the ESO/ESMINT guidelines (Exp Op). 73 The latter guidelines suggest (Exp Op) that the ESCAPE trial protocol with the use of multiphase CTA, may be used up to 12 h from LSW for patients with good to moderate collaterals; this may be an option for stroke centers lacking advanced neuroimaging.

MRI availability considerably expands both IVT availability for WUS and EVT in the 6–24-h time window (DAWN protocol). Bridging therapy may be considered for WUS in eligible patients with positive DWI and negative FLAIR, since most of these strokes are <4.5 h, but this therapeutic approach has not been tested in clinical practice (expert opinion according to ESO IVT guidelines). DAWN criteria have been tested in patients up to 6 days from LSW in the Pittsburg study 84 (expert opinion).
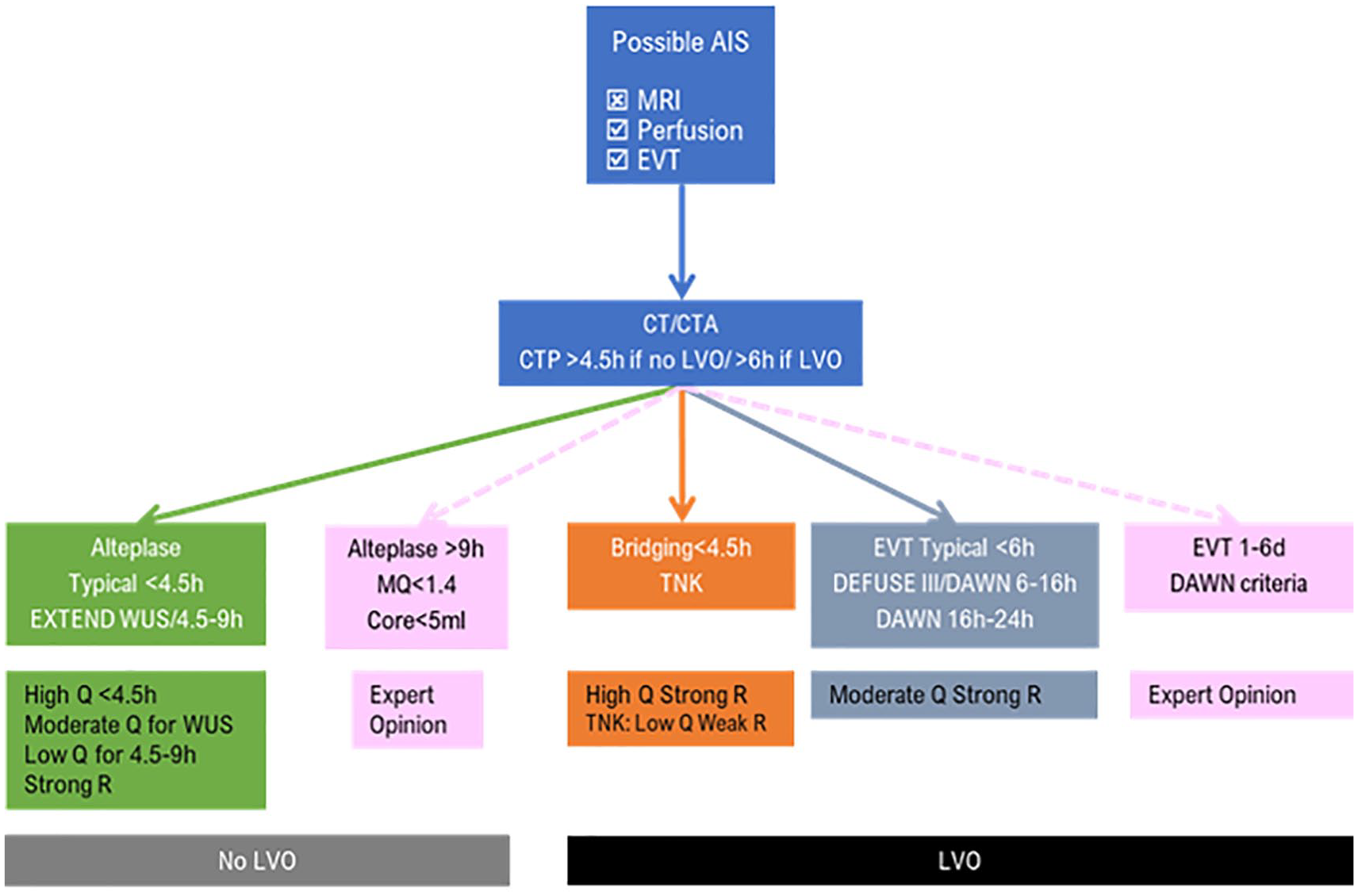
CTP further expands treatment options in EVT-capable stroke centers. Clinicians should bear in mind that CTP means more iodine contrast, radiation and time and should not be part of the imaging protocol for all stroke patients. Some cases with diagnostic difficulties (possible stroke mimics 179 or extensive early ischemic signs on noncontrast CT) may benefit from CTP in early time windows (<4.5 h for IVT and <6 h for EVT). However, most patients eligible for IVT/bridging therapy up to 4.5 h from LSW and EVT eligible patients up to 6 h from LSW do not usually need CTP. CTP may also increase the yield of CTA for distal medium vessel occlusion recognition. CTP may be used for IVT in WUS and 4.5–9 h from LSW according to the EXTEND study criteria. If DEFUSE 3 or DAWN study criteria are fulfilled, EVT is strongly recommended up to 24 h from LSW. The Erlangen study criteria 29 of MQ > 1.4 may allow safe IVT in patients with ischemic core <5 ml after 9 h from LSW as off-label IVT treatment. Bridging therapy using the EXTEND criteria have not been tested during the trial but may be considered only when recanalization is predicted to occur in >2 h for AIS patients with favorable IVT risk–benefit profile, notably extremely limited or no ischemic core (expert opinion). DAWN criteria have been tested in patients up to 6 days from LSW in the Pittsburg study 84 (expert opinion).

State-of-the-art treatment algorithm as of February 2021.The use of CTP means more iodine contrast, radiation and time and should not be part of the imaging protocol for all stroke patients in early time windows (<4.5 h for IVT and <6 h for EVT). Some cases with diagnostic difficulties (possible stroke mimics or extensive early ischemic signs on noncontrast CT) may benefit from CTP in early time windows. However, most patients eligible for IVT/bridging therapy up to 4.5 h from LSW and EVT eligible patients up to 6 h from LSW do not usually need CTP. CTP may also increase the yield of CTA for distal medium vessel occlusion recognition. IVT after 9 h may be offered if MQ exceeds 1.4, according to the Erlangen study. 29 MR perfusion is the optimal imaging modality for WUS, when available; IVT with alteplase in WUS may be performed either by using perfusion criteria as described in the EXTEND trial or the WAKE-UP DWI/FLAIR dissociation criteria (expert opinion). If DEFUSE 3 or DAWN study criteria are fulfilled, EVT is strongly recommended up to 16 h and 24 h, respectively from LSW. Bridging therapy in WUS may be considered, if FLAIR is negative (meaning time of onset is probably <4.5 h). Bridging therapy using the EXTEND criteria has not been tested during the trial but may be considered when EVT recanalization is predicted in >2 h for patients with favorable IVT risk–benefit profile, notably extremely limited or no ischemic core (expert opinion). EVT may be safe using DAWN criteria in patients 1–6 days from LSW as tested in the Pittsburg study 84 (expert opinion).
Reducing inequalities in stroke care involves increasing the percentage of AIS patients that receive acute reperfusion therapies, also in advanced time windows through advanced neuroimaging. A recent report by the Stroke Alliance for Europe (SAFE, https://www.safestroke.eu) estimates that future costs of stroke care in Europe could increase to €86 billion in 2040 if we fail to invest in stroke prevention, acute treatments and rehabilitation. 180 The ESO in cooperation with SAFE has prepared a European Stroke Action Plan for the years 2018–2030, setting a goal for achieving IVT rates for AIS patients above 15% and EVT rates above 5%. 181 Stroke physicians should point out the urgent need to reduce inequalities in acute stroke care to key players such as hospital managers and government officials, to increase access to advanced neuroimaging and expand population coverage with comprehensive stroke centers that offer EVT 24/7. The treatment algorithms presented in this review may help illustrate the increase in the percentage of eligible patients for AIS treatment by investing in neuroimaging and EVT and thus aid healthcare managers to efficiently allocate resources; for example, investing on providing perfusion capabilities in existent CT scanners may be more cost-effective than acute MRI scanner investment. Such cost-effectiveness analyses based on recently published data are currently absent but urgently needed.
Standardization and reducing variations in acute treatment procedures adhering to the latest scientific treatment guidelines may be accelerated through accreditation programs such as the Get with the Guidelines-Stroke or the ESO Stroke Unit and Stroke Center Certification Program.182,183 The final point of this narrative review is that recanalization therapies in late time windows are not investigational nor a luxury for modern acute stroke care; they are irreplaceable life-saving treatment modalities for a major cause of adult disability and death and their widespread use in clinical practice is an urgent necessity.
Conclusions
Translating the recent exciting findings on recanalization therapies in advanced time windows into everyday clinical practice is a major current challenge for stroke medicine. The main barrier to widespread implementation of novel treatment algorithms for AIS in extended time windows is the absence of endovascular and advanced neuroimaging capabilities in stroke centers. Investment in expansion and upgrade of current stroke networks is urgently needed to provide maximal spatial and temporal treatment coverage of AIS patients in the future.
