Abstract
Background:
The literature on endovascular treatment (EVT) for large-vessel occlusion (LVO) acute ischaemic stroke (AIS) has been rapidly increasing after the publication of positive randomized-controlled clinical trials (RCTs) and a plethora of systematic reviews (SRs) showing benefit compared to best medical therapy (BMT) for LVO.
Objectives:
An overview of SRs (umbrella review) and meta-analysis of primary RCTs were performed to summarize the literature and present efficacy and safety of EVT.
Design and methods:
MEDLINE via Pubmed, Embase and Epistemonikos databases were searched from January 2015 until 15 October 2023. All SRs of RCTs comparing EVT to BMT were included. Quality was assessed using Risk of Bias in Systematic Reviews scores and the RoB 2 Cochrane Collaboration tool, as appropriate. GRADE approach was used to evaluate the strength of evidence. Data were presented according to the Preferred Reporting Items for Overviews of Reviews statement. The primary outcome was 3-month good functional outcome [modified Rankin scale (mRS) score 0–2].
Results:
Three eligible SRs and 4 additional RCTs were included in the overview, comprising a total of 24 RCTs, corresponding to 5968 AIS patients with LVO (3044 randomized to EVT versus 2924 patients randomized to BMT). High-quality evidence shows that EVT is associated with an increased likelihood of good functional outcome [risk ratio (RR) 1.78 (95% confidence interval (CI): 1.54–2.06); 166 more per 1000 patients], independent ambulation [mRS-scores 0–3; RR 1.50 (95% CI: 1.37–1.64); 174 more per 1000 patients], excellent functional outcome [mRS-scores 0–1; RR 1.90 (95% CI: 1.62–2.22); 118 more per 1000 patients] at 3 months. EVT was associated with reduced 3-month mortality [RR 0.81 (95% CI: 0.74–0.88); 61 less per 1000 patients] despite an increase in symptomatic intracranial haemorrhage [sICH; RR 1.65 (95% CI: 1.23–2.21); 22 more per 1000 patients].
Conclusion:
In patients with AIS due to LVO in the anterior or posterior circulation, within 24 h from symptom onset, EVT improves functional outcomes and increases the chance of survival despite increased sICH risk.
Registration:
PROSPERO Registration Number CRD42023461138.
Keywords
Introduction
The first positive randomized-controlled clinical trials (RCTs) of endovascular treatment (EVT) in acute ischaemic stroke (AIS) patients treated in the early time window with a large-vessel occlusion (LVO) of the anterior circulation were published in 2015 and provided high-quality evidence for EVT safety and efficacy in AIS due to anterior circulation LVO. 1 Current American Heart Association/American Stroke Association and European Stroke Organization guidelines strongly recommend EVT for AIS patients presenting with anterior circulation LVO and National Institutes of Health Stroke Scale (NIHSS) score ⩾6, Alberta Stroke Program Early CT Score (ASPECTS) ⩽6, up to 6 h from symptom onset based on high-quality data (Class IA) derived from multiple RCTs.2,3 After publication of these guidelines, multiple RCTs and numerous systematic reviews (SRs) have demonstrated EVT efficacy and safety in LVO subgroups. In particular, published SRs on EVT refer to either anterior or posterior circulation, to early or late-time windows, to large or moderate infarct size and to standard or advanced neuroimaging.4–7
However, even a flawless single SR and meta-analysis may offer a shortsighted view of the evidence. 8 Overviews of SRs (‘umbrella reviews’) aim to provide intuitive summaries of the breadth of research to decision makers without demanding from them to assimilate the results of multiple SRs themselves. 9 Overviews, by synthesizing the results of multiple SRs, are broader in scope and may examine the same intervention for different subgroups of patients. 10
These LVO subgroups have different characteristics, but all gain significant benefits from EVT; if risks and benefits remain similar throughout, the idea of continuing using these subgroups may be redundant for any practical or clinical purpose. Overall benefit is also an important piece of information for health policymakers, as both high- and low-income countries strive to develop EVT networks. 11 Cost–benefit analyses have also been published referring mostly to the anterior circulation and specifically to early or late-time windows. 12 It is time to combine all high-quality information available through a rigorous scientific approach rather than extrapolating the results from a subgroup to all patients with AIS and LVO. We have thus performed an overview of SRs and meta-analysis to evaluate the safety and efficacy of EVT in LVO patients. This overview is restricted to primary RCTs published after 2015.
Methods
Standard protocol approvals and registrations
The pre-specified protocol of the present overview of SRs and meta-analysis has been registered in the International Prospective Register of Ongoing Systematic Reviews PROSPERO (Registration Number: CRD42023461138). No amendments were made to the registered protocol. Results are reported according to the Preferred Reporting Items for Overviews of Reviews statement. 13
Data sources, searches and study selection
Following the PICOS format, a systematic literature search was conducted to identify available studies evaluating adult patients with AIS due to LVO (intracranial internal carotid, proximal middle cerebral artery, basilar artery occlusion; P: population) that were treated with EVT (mechanical thrombectomy and/or thromboaspiration) together with best medical treatment [BMT, conservative treatment with or without intravenous thrombolysis (IVT)] (I: intervention) versus BMT alone (C: comparator). Reporting of functional outcome at 3 months, as assessed by modified Rankin scale (mRS; O: outcome) scores, was required for studies to be considered for inclusion. The primary outcome of interest was good functional outcome at 3 months, as defined by mRS-scores 0–2, among patients treated with EVT and BMT versus BMT alone.14,15 Secondary outcomes of interest comprised the following: (i) independent ambulation at 3 months as defined by a mRS-scores 0–3; (ii) excellent functional outcome at 3 months as defined by a mRS-scores 0–1; (iii) symptomatic intracranial haemorrhage (sICH); (iv) all-cause mortality at 3 months and (v) reduced disability as assessed by 1-point reduction across all mRS-scores at 3 months (shift analysis).14,15 Included studies (S: Study design) were SRs of RCTs and primary RCTs. Included SRs provided pre-specified criteria for including and excluding studies and results were reported according to specific guidance for SRs and meta-analyses.
The literature search was performed independently by four reviewers (AS, LP, AHK, KP). We searched MEDLINE, Embase and Epistemonikos databases, using search strings that included the following terms: ‘stroke’, ‘endovascular treatment’, ‘randomized-controlled trial’, ‘LVO’, ‘intracranial occlusion’, ‘trial’, ‘meta-analysis’, ‘review’, and ‘systematic review’. The complete search algorithms used in MEDLINE, Embase and Epistemonikos and the complete inclusion and exclusion criteria are provided in Supplemental Table S1.16–20 We have limited our search in SRs comprising only studies published in 2015 onwards. No language or other restrictions were applied. Our search spanned from 1 January 2015 to 10 October 2023, for each electronic database. 21 We also manually searched reference lists of published articles manually to ensure the bibliography’s comprehensiveness. SRs that included non-controlled studies, case series and case reports were excluded. Commentaries, editorials and narrative reviews were also discarded. Among the studies presenting duplicate data, the ones with the largest dataset were retained, while the others were excluded.
For overlapping reviews, the Jadad algorithm has been adapted and was used independently by two reviewers (OK, KIB); disagreements were settled by consensus after discussion with the corresponding author (GT) (Supplemental Table S2).22,23 Among equivalent SRs, we opted for studies with pooled demographic data that were available to our study group. A supplemental search for primary RCTs published after the publication of each SR has been performed. We applied the respective search string for the same databases used in each selected SR, starting on the date the search was performed in each review and ending on 15 October 2023 (Supplemental Table S3).
Risk of bias assessment and data extraction
Eligible SRs were assessed for bias using the Risk of Bias in Systematic Reviews (ROBIS) tool. 24 For supplemental RCTs, quality assessment has been performed with the Cochrane Collaboration tool (RoB 2). 25 The risk of bias assessment was conducted independently by two reviewers not included in the author lists of the eligible studies (OK, KIB) and disagreements were settled by consensus after discussion with the corresponding author (GT). Data extraction was performed in structured forms, including author names, publication date, study design, country, number of included patients, patient characteristics (age, NIHSS, ASPECTS, sex, stroke onset-to-randomization time, IVT rates, posterior versus anterior circulation) and outcome events independently by two reviewers (AS, LP).
Statistical analysis
Statistical analysis was performed using the Cochrane Collaboration’s Review Manager (RevMan 5.3) Software Package (Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014) and Open Meta Analyst. 26 mRS shift analysis has been performed using an online tool available at: https://www.thembc.com.au/tournament methods. 27 Hundred-person icon arrays (HPIAs) have been previously published to quantify the magnitude of benefit of EVT and IVT versus non-reperfusion in AIS patients with LVO 28 ; we provide updated HPIAs (https://www.iconarray.com) by summation of mRS-scores from each study.
For each dichotomous outcome of interest, the corresponding risk ratio (RR) with 95% confidence interval [95% confidence interval (CI)] was calculated using the random-effects model (DerSimonian and Laird). 16 Subgroup differences were assessed by the Q test for subgroups. 17 For the analysis of the baseline characteristics (age, sex, admission NIHSS, IVT pretreatment, onset-to-recanalization times) of the patients in each arm, we calculated the pooled proportion of each dichotomous variable and the overall mean for each continuous variable. For studies reporting continuous outcomes in median values and corresponding interquartile ranges, we estimated the sample mean and standard deviation using the quantile estimation method. 16 Comparison of the baseline characteristics between the two arms was performed using odds ratio (OR) and mean difference for dichotomous and continuous variables, respectively. Heterogeneity was assessed with the I2 and Cochran Q statistics. For the qualitative interpretation of heterogeneity, I2 values >50% and values >75% were considered to represent substantial and considerable heterogeneity, respectively. The significance level for the Q statistic was set at 0.1. Publication bias across individual studies was assessed using funnel plot inspection and the use of Egger’s test.
Results
Literature search and included studies
The systematic database search yielded a total of 637, 620 and 594 records from the MEDLINE, Embase and Epistemonikos databases, respectively (Figure 1). After initial screening, we retrieved the full text of 114 records that were considered potentially eligible for inclusion. After reading the full-text articles, 111 were further excluded (Supplemental Table S4). Finally, we identified three eligible SRs for inclusion (Table 1),29–31 that included 20 RCTs.32–51 Besides using different statistical approaches (Bayesian mixed-effects model in 1 SR 29 and Random-effects model in 2 SRs30,31), different outcomes and different summary statistics were reported (Supplemental Table S5). To overcome this source of heterogeneity and to present an overall comparison of the two treatment methods, a meta-analysis of RCTs has also been performed.

Flow chart presenting the selection of eligible studies.
Main characteristics of SRs included in the overview (n = 3).

Patients were aware of treatment allocation. Two trials did not blind the outcome assessor to treatment allocation.
Patients were aware of treatment allocation. Minor deviations from intended interventions in three trials. In one trial minor concerns of the randomization process due to inclusion of more patients with diabetes mellitus in the EVT arm compared to control.
Patients were aware of treatment allocation. Some concerns about bias arising in the randomization process in one trial and due to missing outcome data in one study.
AIS, acute ischaemic stroke; Anterior circulation LVO, large-vessel occlusion in the internal carotid artery, middle cerebral artery or anterior cerebral artery; BAO, basilar artery occlusion; mRS, modified Rankin scale; NA, not applicable; NR, not reported; RoB, risk of bias; SD, standard deviation; sICH, symptomatic intracerebral haemorrhage.
Anterior circulation LVO with moderate infarct
The SR by Rajkumar et al. 29 included RCTs on anterior circulation LVO published from 2015 up to July 2020. In total, 12 RCTs were included, reporting a median NIHSS between 16 and 20 (Table 2). Patients were randomized from <4.5 up to 24 h after symptom onset (1 trial <4.5 h; 1 trial <5 h; 4 trials <6 h; 2 trials <8 h; 1 trial <12 h; 1 trial 6–16 h; 1 trial 6–24 h; 1 trial unspecified). We considered only two trials investigating EVT in late-time windows since the trial randomizing patients up to 12 h reported onset-to-randomization times like that of the early time window trials (Supplemental Table S6). IVT rates ranged from 5% to 100% of included patients. All RCTs included patients with internal carotid artery (ICA) and middle cerebral artery segment 1 (M1) occlusions. Seven out of 12 studies included a substantial proportion of patients with segment 2 of the middle cerebral artery (M2) occlusion, whereas other sites of occlusion (anterior cerebral artery, basilar artery) were rare (Table 2). No data on ASPECTS and onset-to-randomization times were provided within the main text and Supplemental Files, we therefore searched primary studies. Mean ASPECTS was found to be 7–9 and mean onset-to-randomization was 169–810 min (Supplemental Table S6).
Table mapping the primary studies contained within included systematic reviews.
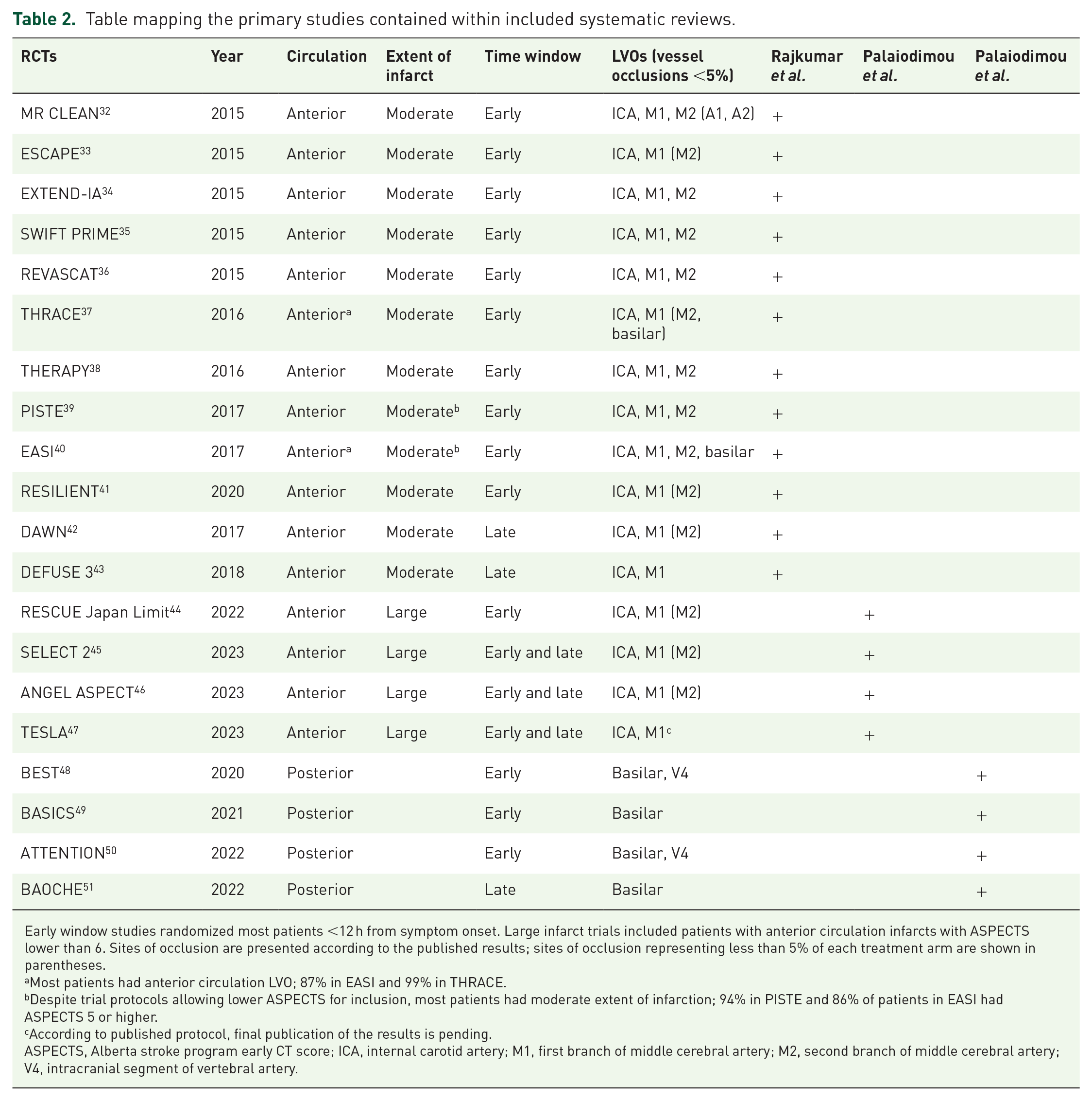
Early window studies randomized most patients <12 h from symptom onset. Large infarct trials included patients with anterior circulation infarcts with ASPECTS lower than 6. Sites of occlusion are presented according to the published results; sites of occlusion representing less than 5% of each treatment arm are shown in parentheses.
Most patients had anterior circulation LVO; 87% in EASI and 99% in THRACE.
Despite trial protocols allowing lower ASPECTS for inclusion, most patients had moderate extent of infarction; 94% in PISTE and 86% of patients in EASI had ASPECTS 5 or higher.
According to published protocol, final publication of the results is pending.
ASPECTS, Alberta stroke program early CT score; ICA, internal carotid artery; M1, first branch of middle cerebral artery; M2, second branch of middle cerebral artery; V4, intracranial segment of vertebral artery.
Anterior circulation LVO with large infarct
The SR by Palaiodimou et al., 30 included RCTs on anterior circulation large-core LVO that reported their results from 2022 to 30 May 2023. In total, four RCTs were included, reporting a median NIHSS of 29 and an IVT pretreatment rate of 23%. Patients were randomized within 24 h of onset in three RCTs and up to 6 h of onset in one RCT, except if there were no FLAIR magnetic resonance imaging early changes, which extended the therapeutic time window up to 24 h. As a result, onset-to-randomization times were low in the latter trial (214–229 min) compared to the rest of the trials (453–587 min). We considered the former trial an early time window trial and the rest both early and late-time window trials (Table 2). All primary RCTs used ASPECTS limits as eligibility criteria (three trials ASPECTS 3–5 and one trial ASPECTS 2–5) and two trials also permitted inclusion based on ischaemic core volume (70–100 ml or at least 50 without upper limit). All included patients had anterior circulation strokes and most had ICA and M1 occlusions. IVT rates ranged from 17% to 29%.
Basilar artery occlusion
The SR by Palaiodimou et al. 31 included RCTs on basilar artery LVO published from 2020 to 29 November 2022. In total, four RCTs were included, reporting a median NIHSS of 24 and an IVT pretreatment rate of 39%. One trial randomized patients <6 h from symptom onset, 1 trial <8 h, 1 trial <12 h and 1 trial 6–24 h. We considered the latter trial as a late-time window trial, whereas the rest as early time window trials since median onset-to-randomization time of the trial randomizing up to 12 h from symptom onset was 4.9 h (Table 2). There was no overlap of primary studies among included SRs (Table 2).
Supplemental search for RCTs that have reported their results since the publication of each SR has been performed (Supplemental Table S3). Only four RCTs (MR CLEAN-LATE, POSITIVE, TENSION, LASTE), not previously included in the SRs, were added to the analysis52–55 (Supplemental Table S7).
We therefore included 3 SRs and 4 additional RCTs, 24 RCTs in total, comprising a total of 5968 AIS patients with LVO (mean age: 67.9 years; 56% men; mean NIHSS-score: 18.4; 50% received IVT; mean onset-to-randomization time: 388 min; Supplemental Figures S1–S5). A total of 3044 patients were randomized to EVT and 2924 patients were randomized to BMT. There were no significant differences between the two arms regarding age, sex and onset-to-randomization times (Supplemental Figures S6–S8). We documented marginally higher NIHSS-scores in the EVT arm at randomization (mean difference 0.39; 95% CI: 0.03–0.74, 20 studies, I2 = 5%, p for Cochran Q = 0.39; Supplemental Figure S9). IVT rates were significantly lower in the EVT arm (OR: 0.86; 95% CI: 0.74–0.99, 18 studies, I2 = 0%, p for Cochran Q = 0.69; Supplemental Figure S10) after removing studies that have not included patients receiving IVT or studies in which all or almost all included patients received IVT.
Quality control of included studies
The risk of bias in the included SRs was assessed by the ROBIS tool 24 and is presented in Supplemental Table S8. Two studies presented a low overall risk of bias30,31; the SR by Rajkumar et al. 29 searched a single database. Supplemental RCTs were examined using the Risk of Bias 2 (RoB2) Cochrane assessment tool. The major concern of the additional RCTs52–55 was the fact that randomized participants and treating physicians were aware of the intervention, also some deviations from intended interventions were noted. Overall, the RCTs were considered of high quality despite the existence of performance bias (Supplemental Figure S11).
Quantitative analyses
Study-level meta-analysis was performed in the 24 included RCTs. An overview of all primary and secondary outcomes, as reported in the included SRs and as estimated in our meta-analysis, is summarized in Table 3. Regarding the primary outcome, EVT was associated with a higher likelihood of achieving good functional outcomes at 3 months compared to BMT (RR: 1.78; 95% CI: 1.54–2.06; 24 studies; I2 = 62%; p for Cochran Q < 0.0001; Figure 2). Regarding secondary outcomes, EVT was associated with independent ambulation at 3 months compared to BMT (RR: 1.50; 95% CI: 1.37–1.64; 24 studies; I2 = 54%; p for Cochran Q = 0.001; Supplemental Figure S12) and with excellent functional outcome at 3 months compared to BMT (RR: 1.90; 95% CI: 1.62–2.22; 24 studies; I2 = 27%; p for Cochran Q = 0.11; Supplemental Figure S13). Regarding safety outcomes, sICH was more common in the patients receiving EVT versus BMT (RR: 1.65; 95% CI: 1.23–2.21; 23 studies; I2 = 0%; p for Cochran Q = 0.52; Supplemental Figure S14). EVT was associated with reduced risk of all-cause mortality at 3 months compared to BMT (RR: 0.81; 95% CI: 0.74–0.88; 24 studies; I2 = 11%; p for Cochran Q = 0.31; Supplemental Figure S15). The odds of reduction of disability (mRS shift) were significantly in favour of EVT (unadjusted generalized OR: 1.64; 95% CI: 1.64–1.81; 24 studies; I2 = 39%; p for Cochran Q = 0.03; Supplemental Figure S16). We have performed leave-one-out analysis in all outcomes. For the primary outcome RRs ranged from 1.72 (95% CI: 1.50–1.98) to 1.83 (95% CI: 1.57–2.12). For mRS 0–3, RRs ranged from 1.47 (95% CI: 1.34–1.61) to 1.52 (95% CI: 1.38–1.67). For mRS 0–1 RRs ranged from 1.75 (95% CI: 1.54–1.99) to 1.86 (95% CI: 1.63–2.12). For sICH RRs ranged from 1.53 (95% CI: 1.13–2.08) to 1.72 (95% CI: 1.27–2.34). For mortality RRs ranged from 0.83 (95% CI: 0.76–0.90) to 0.79 (95% CI: 0.73–0.87). Consequently, the treatment effects for all outcomes did not change direction or lose statistical significance in the leave-one-out analysis, confirming the robustness of all documented associations.
Overview of analyses for primary and secondary outcomes.

AIS, acute ischaemic stroke; BMT, best medical treatment; CI, confidence interval; cOR, common odds ratio; EVT, endovascular treatment; genOR, generalized odds ratio; mRS, modified Rankin scale; OR, odds ratio; RR, risk ratio; RCT, randomized-controlled clinical trials; sICH, symptomatic intracranial haemorrhage.

Forest plot presenting the risk ratio of achieving mRS 0–2 at 3 months among EVT- versus BMT-treated patients.
The pooled proportions of efficacy and safety outcomes, as reported in the included SRs and as estimated in our meta-analysis of the 24 included RCTs, are presented in Table 2. Regarding the primary outcome, 38% of patients after EVT (95% CI: 35–46%; 24 studies; I2 = 93%; p for Cochran Q < 0.001; Supplemental Figure S17) and 22% of patients after BMT (95% CI: 17–27%; 24 studies; I2 = 93%; p for Cochran Q < 0.001; Supplemental Figure S18) achieved good functional outcome (mRS-scores 0–2) at 3 months. Regarding secondary outcomes, 53% of patients after EVT (95% CI: 47–59%; 24 studies; I2 = 91%; p for Cochran Q < 0.001; Supplemental Figure S19) and 35% of patients after BMT (95% CI: 30–41%; 24 studies; I2 = 91%; p for Cochran Q < 0.001; Supplemental Figure S20) retained independent ambulation at 3 months; 23% of patients after EVT (95% CI: 18–27%; 24 studies; I2 = 92%; p for Cochran Q < 0.001; Supplemental Figure S21) and 11% of patients after BMT (95% CI: 8–14%; 24 studies; I2 = 88%; p for Cochran Q < 0.001; Supplemental Figure S22) had excellent functional outcome at 3 months. Regarding safety outcomes, 4.3% of patients after EVT (95% CI: 3.1–5.6%; 24 studies; I2 = 68%; p for Cochran Q < 0.001; Supplemental Figure S23) and 2.1% of patients after BMT (95% CI: 1.4–2.8%; 24 studies; I2 = 32%; p for Cochran Q = 0.07; Supplemental Figure S24) were complicated with sICH. Finally, 23% of patients after EVT (95% CI: 19–27%; 24 studies; I2 = 88%; p for Cochran Q < 0.001; Supplemental Figure S25) and 29% of patients after BMT (95% CI: 24–34%; 23 studies; I2 = 91%; p for Cochran Q < 0.001; Supplemental Figure S26) were dead at 3 months.
Publication bias
Publication bias was evaluated using funnel plots for every outcome of the analysis. Visual inspection of the funnel plots did not reveal evidence of publication bias (Supplemental Figures S27–S32). Egger’s test revealed asymmetry in the outcomes of 3-month mRS 0–1 (p-value = 0.0027), mRS 0–2 (p Value = 0.0014) and mRS 0–3 (p value = 0.01) but not for sICH, 3-month mortality and 3-month disability reduction (p values = 0.99, 0.93 and 0.078, respectively). The rates of IVT pretreatment significantly differed between the two arms and meta-regression analysis was pursued for all examined outcomes. Nevertheless, no significant interaction between IVT pretreatment and achieved mRS-scores 0–2 (omnibus p value = 0.786, Supplemental Figure S33), mRS-scores 0–3 (omnibus p value = 0.671, Supplemental Figure S34) and mRS-scores 0–1 (omnibus p value = 0.214, Supplemental Figure S35) at 3 months was detected. Similarly, there was no significant interaction between IVT pretreatment and sICH (omnibus p value = 0.127, Supplemental Figure S36) or mortality at 3 months (omnibus p-value = 0.985, Supplemental Figure S37). We were unable to perform a sensitivity analysis since many studies have not reported odds of favourable outcomes depending on IVT pretreatment, and those that provided relevant data used different summary statistics (ORs, common ORs, rate ratio, RRs) on different outcomes (mRS shift, mRS 0–3).
To visually present the beneficial effects of EVT for LVO, we calculated the pooled proportions of the scores for mRS 0–2, mRS 0–3 and mortality and we supply HPIA representing the clinical benefit of EVT by encompassing the full spectrum of LVO treatment including extended time windows of treatment, large established infarcts and posterior circulation infarctions (Figure 3).

Hundred-person icon array demonstrating outcomes after EVT and BMT for LVO.
Heterogeneity
Substantial heterogeneity of treatment effect was noted for the primary outcome (good functional outcome; I2 = 62%; p for Cochran Q < 0.0001; Figure 2) and independent ambulation at 3 months (I2 = 54%; p for Cochran Q = 0.001; Supplemental Figure S12). Such discrepancies in treatment effect have already been highlighted in RCTs evaluating EVT efficacy for LVO in the anterior circulation and have been attributed to different study protocols, with more selective studies reporting higher odds of benefit from EVT per patient but excluding more patients that could still derive benefit from treatment. 56 Concerning the posterior circulation, subgroup analysis in the SR by Palaiodimou et al. 31 has suggested that there are statistically significant differences between studies that were conducted in China compared to international studies that were conducted in Europe and North America, while the overall EVT benefit is derived by the RCTs recruiting patients in China.
Subgroup analyses
To further assess for potential reasons of the heterogeneity noted in this analysis, pre-specified subgroup analyses were conducted by stratifying the effect of treatment into three subgroups: anterior circulation versus posterior circulation, early time window versus late-time window, moderate infarct versus large infarct (Supplemental Figures S38–S55). The following subgroup differences were disclosed: sICH risk with EVT compared to BMT was higher in the posterior (RR: 7.48; 95% CI: 2.27–24.62; four studies; I2 = 0%) versus anterior (RR: 1.50; 95% CI: 1.11–2.02; 17 studies; I2 = 0%) circulation (p for subgroup differences 0.01; Supplemental Figure S42), as already shown in the current overview of SRs (Supplemental Figure S5). The RR for good functional outcome with EVT compared to BMT was higher in the late (RR: 2.31; 95% CI: 1.24–4.31; four studies; I2 = 88%) and early and late (RR: 2.41; 95% CI: 1.78–3.26; three studies; I2 = 0%) than in the early (RR: 1.60; 95% CI: 1.40–1.83; 17 studies; I2 = 38%) time window trials (p for subgroup differences 0.03; Supplemental Figure S44); we therefore confirm previous observations known as ‘the late-window paradox’. 57 The RR for mortality was neutral in the early and late treatment group (RR: 1.00; 95% CI: 0.84–1.19; three studies; I2 = 0%) whereas it was lower in the early (RR: 0.77; 95% CI: 0.70–0.85; 17 studies; I2 = 0%) and the late (RR: 0.78; 95% CI: 0.64–0.95; four studies; I2 = 0%) treatment groups (p for subgroup differences 0.04; Supplemental Figure S49); this variation could either be fortuitous or due to the fact that the early and late studies are all more recent, more inclusive studies, excluding patients presenting with better profile and, thus, potentially deriving more benefit from treatment; those are eligible for EVT according to criteria with older, more selective studies. The RR for good functional outcome with EVT compared to BMT was higher with large (RR: 2.49; 95% CI: 1.89–3.29; six studies; I2 = 7%) than moderate (RR: 1.62; 95% CI: 1.39–1.88; 14 studies; I2 = 54%) volume infarcts of the anterior circulation (p for subgroup differences 0.00006; Supplemental Figure S50); the RR for independent ambulation with EVT compared to BMT was higher with large (RR: 1.90; 95% CI: 1.50–2.40; six studies; I2 = 51%) than moderate (RR: 1.39; 95% CI: 1.26–1.53; 14 studies; I2 = 45%) volume infarcts of the anterior circulation (p for subgroup differences 0.02; Supplemental Figure S51).
Discussion
The main findings of our meta-analysis indicate that high-quality data converge into a clear benefit of EVT for a wide variety of indications in AIS patients with LVO: intracranial internal carotid, proximal middle cerebral artery, basilar artery occlusion. Proximal middle cerebral artery consists of the M1 segment in all included studies and, in most included studies of moderate infarcts, M2, too. M2 occlusions were rare in large infarct trials, probably due to the apparition of high ASPECTS scores, mainly in ICA and M1 occlusions. We have limited our overview to include primary studies published from 2015 onward, since older (neutral) RCTs used previous generation materials, intra-arterial thrombolysis as the primary intention recanalization treatment, had low recanalization rates and suboptimal treatment pathways resulting in time delays. 58 The low to moderate heterogeneity found in our meta-analysis supports that, despite our tendency to consider different subgroups of AIS patients, the benefit from EVT is consistent and invariable. Relative risks remained statistically significant for different locations of occlusion, time windows and size of infarcts. It is of note the absence of heterogeneity regarding reduction of disability (mRS shift analysis) and mortality at 3 months in LVO patients treated with EVT. Despite differences in prognosis in the included subgroups, treatment effect seems to be steadily in favour of EVT for all examined subgroups. Distinguishing treatment approaches to early versus late-time window, to moderate versus large infarct size or to anterior versus posterior circulation LVOs used to be necessary since different imaging or clinical criteria have been followed in each respective RCT. However, current knowledge, as presented in this overview, crosses these boundaries, as the treatment effect remains robust across LVO subgroups. The recently published MR CLEAN-LATE trial 52 exemplifies this approach by being a late-time window RCT that randomized patients with both moderate and large infarcts, providing a more pragmatic approach to late-window anterior LVO patient selection for EVT through simple rather than advanced neuroimaging.
During the last decade, we have continuously witnessed reports of improvement in the rates of good functional outcomes after EVT for ever-expanding indications. 59 Novel approaches of post-recanalization intra-arterial thrombolysis have also shown promising results. 60 Hopefully, ongoing trials will succeed in crossing the next frontier, namely the distal artery occlusions. 61 However, from a global health care perspective, focus should be on translating the available data into clinical practice and funnelling funds to provide EVT to as many stroke victims as possible. Despite the improvement in treatment rates in recent years, there are persistent inequalities between countries in access to EVT for acute stroke patients, with most low and middle-income countries lagging. 11 The results of the current overview of SRs may be used to convey the importance of EVT to health policymakers. Epidemiologic data on the prevalence of LVO may be used in conjunction with the RRs reported herein to improve the accuracy of cost-effectiveness and future burden of stroke projection models.62,63
Our umbrella review provides overwhelming evidence that EVT compared to BMT reduces the risk of death at 3 months by 19% (95% CI: 12–26%) with low (11%) heterogeneity across all 24 RCTs. This translates into 61 fewer deaths for every 1000 patients treated with EVT. This finding is in line with a previous meta-analysis from our collaborative group that, after pooling data from 11 RCTs, documented a 17% risk reduction with a number needed to treat of 31 (32 fewer deaths for every 1000 patients treated) with EVT compared to BMT. 60 Given the fact that EVT was associated with reduced disability across all ranks of mRS (ordinal shift analysis), the reduction in mortality with EVT is not associated with increased likelihood of severe disability (mRS-scores of 4–5). This is an important additional benefit of EVT compared to BMT, given the fact that IVT compared to BMT does not reduce 3-month mortality.7,64 In other words, wider implementation of EVT will result in fewer deaths in addition to lower disability rates.
Standard umbrella review methodology extracts statistical results intact and presents them. However, when the literature is as extensive and rapidly evolving as it is in the field of EVT, meta-analyses include different studies and no SR could be viewed as definitive. 65 The fact that there was no overlapping of primary studies among the included SRs permitted us to perform meta-analysis of all primary studies. We also included additional studies published in the last 2 years that would have otherwise been missed. As a result, the strength of our study is the incorporation of 24 RCTs comprising 5968 patients. Our subgroup analyses further solidify the role of EVT as one of the most effective advances in medicine in recent years for all tested LVO subgroups. The selection of subgroups was according to our registered research protocol, and it was based on the stages of clinical research on EVT during the last decade. It may seem counterintuitive that the RR of mortality is 1 (neutral) in the early and late-time window trials but significantly lower in both the early window and the late-window trials in the EVT arm (RR: 0.77 and 0.78, respectively; Supplemental Figure S49) but the main characteristic of the three early and late-window trials is the inclusion of stroke patients with large established infarctions (large-core RCTs). The absence of reduction in mortality risk with EVT in this subgroup confirms the findings of the relevant SR by Palaiodimou et al. 30 However, reduction of disability remains highly significant and primary outcome is significantly higher in the large-core subgroup compared to moderate core of the anterior circulation (Supplemental Figure S50). We therefore conclude that most outcomes of efficacy and safety remain important, irrespective on how we define subgroups: the net benefit of EVT remains robust. Regarding publication bias, no asymmetry was unravelled through funnel plot inspection, but evidence of publication bias was detected for the 3-month functional outcomes using Egger’s test. Furthermore, substantial heterogeneity was present only for the primary outcome but was not significant for all secondary outcomes, despite the differences among treated subgroups of LVO patients. Age, sex and onset-to-randomization times were similar between the two arms in the meta-analysis. The mean NIHSS-scores were slightly higher in the EVT than in the BMT group.
IVT pretreatment rates significantly differed between the two arms, but meta-regression analysis for all examined outcomes failed to identify any interaction between IVT pretreatment rates and the comparative efficacy of EVT versus BMT with regard to primary and secondary outcomes. A very recent SR that included 10 RCTs showed significantly increased odds of functional independence with bridging therapy at the expense of an increase in mortality and sICH as compared to direct EVT. 66 An individual-patient meta-analysis of six RCTs has failed to show non-inferiority of direct EVT to bridging therapy or superiority of bridging therapy in anterior circulation LVO in the context of mothership paradigm (excluding drip-and-ship studies) and in centres with rapid door-to-groin puncture times (median less than 30 min). 67 As far as the posterior circulation is concerned, Palaiodimou et al. performed subgroup analysis after stratification for IVT and found no significant subgroup differences in patients with and without IVT pretreatment. 31 Notably, in the subgroup of patients pretreated with IVT, the effect size of EVT compared to BMT regarding mRS 0–3 was substantially attenuated (data available from two RCTs). The role of IVT before EVT remains established by recent head-to-head RCTs (comparing bridging therapy to direct EVT) and current international recommendations that advocate that all EVT-eligible LVO patients should be pretreated with IVT (if they fulfil the relevant inclusion criteria), especially in the drip-and-ship treatment paradigm.68,69 It remains to be seen if there is reason to omit IVT in patients admitted directly to thrombectomy centres that have proven records of very short groin to recanalization times. 68 Direct EVT versus bridging therapy for BAO will be investigated in the BEST-BAO trial (Direct Endovascular Treatment Versus Bridging Treatment In Basilar Artery Occlusive Stroke; https://www.clinicaltrials.gov; Unique identifier: NCT05631847).
The current overview aims to present the best available evidence to date on EVT benefit for patient-oriented outcomes that were predefined in our study protocol and were used as primary or secondary endpoints in all included RCTs. We have not examined periprocedural complications, recanalization rates, asymptomatic ICH or other surrogate outcomes 70 that have been addressed previously in other SRs. 66 The current review further solidifies the important clinical benefit regarding reduction of dependency and mortality after LVO stroke and our conclusions may be communicated from stroke physicians to policymakers to increase global access to EVT.
A comprehensive approach to improve outcomes for stroke patients will also need to consider that the outcome of a stroke is contingent upon numerous variables, encompassing factors such as age, comorbidities like hypertension, diabetes and asthma, as well as additional elements like smoking and hyperlipidaemia.71–73 Despite advancements in stroke treatment, especially ischaemic stroke, various pre- and postoperative conditions, such as blood pressure control, gastrointestinal motility, malnutrition linked to stroke and pneumonia associated with stroke, persist as reported predictors affecting outcomes. 74 Additionally, determining the optimal blood pressure threshold after intervention for achieving the best outcome remains an ongoing challenge. 75
The main limitation of our study is that it is a study-level meta-analysis, lacking individual patient data. In addition, we documented unadjusted associations that may be prone to residual confounding. Also, the substantial heterogeneity across different studies for the primary outcome of good functional outcomes needs to be taken into account when interpreting our study findings.
Conclusion
The current overview of SRs (umbrella review) of data from randomized-controlled trials strongly supports EVT for AIS patients with LVO. Despite an increase in sICH rates, EVT appears superior to BMT in the anterior circulation even for large infarcts and for posterior circulation LVOs; beneficial effects persist in extended time windows. However, it should be acknowledged that we did not evaluate systematically peri-procedural complications of EVT and that there was substantial heterogeneity across RCTs and SR for the primary outcome (mRS score 0–2) of our umbrella review. The beneficial effects of EVT are related to improved efficacy outcomes in terms of excellent or good functional outcomes, independent ambulation and survival. The consistency of the magnitude of benefit dictates rapid implementation of this lifesaving and disability-sparing treatment modality across the globe.
Supplemental Material
sj-docx-1-tan-10.1177_17562864241246938 – Supplemental material for Overview of systematic reviews comparing endovascular to best medical treatment for large-vessel occlusion acute ischaemic stroke: an umbrella review
Supplemental material, sj-docx-1-tan-10.1177_17562864241246938 for Overview of systematic reviews comparing endovascular to best medical treatment for large-vessel occlusion acute ischaemic stroke: an umbrella review by Apostolos Safouris, Lina Palaiodimou, Aristeidis H. Katsanos, Odysseas Kargiotis, Konstantinos I. Bougioukas, Klearchos Psychogios, Tatiana Sidiropoulou, Stavros Spiliopoulos, Marios-Nikos Psychogios, Georgios Magoufis, Guillaume Turc and Georgios Tsivgoulis in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
None.
Declarations
Protocol registration
The protocol of this systematic review and meta-analysis has been registered to the International Prospective Register of Ongoing Systematic Reviews PROSPERO (Registration number: CRD42023461138).
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
