Abstract
Background:
Characterizing Cladribine tablets prescription pattern in daily clinical practice is crucial for optimizing multiple sclerosis (MS) treatment.
Objectives:
To describe efficacy, safety profile and new disease-modifying therapy (DMT) prescriptions following Cladribine treatment.
Design:
Independent retrospective cohort study in patients followed at six Italian MS centres.
Methods:
Patients diagnosed with relapsing MS (RMS) according to 2017 McDonald criteria, who initiated Cladribine between January 2019 and May 2023, were included. A generalized linear regression model was built for the outcome DMT after Cladribine course. Heatmaps were generated based on weighted pivot tables to visualize the proportion of patients requiring DMT post-Cladribine.
Results:
A total cohort of 352 patients was enrolled, 134 naïve to any DMT, 218 switchers from other DMTs. The last DMT was an injectable first-line DMT for 48 (22%) patients, oral first-line DMT for 141 (64.7%) patients, SP1 inhibitor-Fingolimod for 23 (10.6%) patients, and Natalizumab for 6 (2.7%) patients. Overall, Cladribine was efficacious and well tolerated, 12% of patients required a new DMT prescription after a median time of 24 months. The regression model revealed that patients aged >40 years at Cladribine prescription had a 16% decrease in likelihood of receiving a new DMT. Heatmaps showed patients previously on Fingolimod had a lower rate (72.2%) of being free from therapy after Cladribine.
Conclusion:
In our multicentric real-world Italian study, Cladribine therapy is generally effective during the investigated follow-up period. Understanding key characteristics of patients responding best to Cladribine can help tailor therapeutic strategies for optimal outcomes.
Plain language summary
Independent retrospective cohort study in patients followed at six Italian Multiple Sclerosis centres. The aim was to describe efficacy, safety profile, and new disease-modifying therapy (DMT) prescriptions following Cladribine treatment. Cladribine therapy was generally effective during the investigated follow-up period in this multicentric real-world Italian study. Understanding key characteristics of patients responding best to Cladribine can help tailor therapeutic strategies for optimal outcomes.
Introduction
Multiple sclerosis (MS) is a complex disease characterized by immune-mediated inflammation, demyelination, and neurodegeneration within the central nervous system, leading to a wide spectrum of clinical manifestations and disability. 1 The treatment landscape for MS has undergone significant evolution with the advent of disease-modifying therapies (DMTs) aimed at improving disease outcomes. 2
Cladribine, a synthetic purine nucleoside analog, has emerged as immunotherapy for MS with a mechanism of action involving the selective depletion of lymphocytes, particularly T and B cells, which are central to the autoimmune cascade underlying MS pathogenesis. Clinical trials and real-world studies have demonstrated the efficacy of Cladribine in reducing relapse rates, delaying disability progression, and achieving sustained remission in patients with relapsing forms of MS.3–8
Characterizing Cladribine prescription pattern in daily clinical practice is crucial for optimizing MS treatment. However, significant gaps remain in identifying nonresponders and in establishing a well-defined algorithm for long-term management for enabling personalized patient care.
This study aims to describe new disease DMTs prescription following Cladribine treatment in a real-world multicentre Italian setting to provide neurologists with evidence-based guidance for identifying and optimizing Cladribine prescription pattern.
Methods
Setting and study design
This was an independent retrospective cohort study on prospectively collected data in patients followed at six Italian MS centres.
Study population
We included patients who (a) had a diagnosis of relapsing MS-RMS according to 2017 McDonald criteria 9 and (b) initiated index treatment with Cladribine between January 2019 and May 2023 in the participating centres; (c) starting Cladribine as first DMT or after a failure from previous DMTs for disease activity.
Cladribine was prescribed using the target dose of 3.5 mg/kg and according to Italian prescription rules.
Procedures and covariates definitions
The demographic, clinical, and brain magnetic resonance imaging (MRI) data were recorded retrospectively (up to 12 months) before the start of treatment with Cladribine (the index date) and prospectively until the last available visit of follow-up from the index date.
We excluded data from patients who were lost to follow-up due to continuing treatment in another center.
To further investigate the impact of previous DMT, we then stratified on type of DMT switching from (1) injectable DMTs (Interferons and Glatiramer acetate); (2) oral DMTs (Teriflunomide and Dimethyl fumarate); (3) monoclonal antibodies (Natalizumab); (4) sphingosine-1-phosphate receptor modulators (Fingolimod).
Patient demographics, clinical, laboratory, and MRI variables were extracted from the electronic health record in June 2024.
The data entry portal was iMED© software, and we followed a rigorous quality assurance procedure with a double-entered data system.
Study outcomes
We aimed to explore new DMT prescription after a complete Cladribine course to identify the Cladribine prescription pattern in a real-world setting.
Adverse events (AEs) were collected, according to the European Medicine Agency definitions.
Disease activity was defined as clinical and radiological activity. Clinical activity was defined as the presence of new relapses. Radiological activity was defined as the presence of gadolinium-enhancing (Gd+) lesions on T1-weighted MRI sequences and/or new lesion(s), or increased lesion volume, compared to a reference scan, on T2-weighted MRI sequences
Disability was assessed with the Expanded Disability Status Scale (EDSS) by a Neurostatus-certified MS specialist. Secondary progression was defined according to Lorscheider criteria. 10
Statistical analysis
Data are presented as proportion for categorical variables and mean (standard deviation) or median (interquartile range) for continuous variables.
A logistic regression model was built for the outcome DMT after Cladribine course (as dichotomic 0 = no, 1 = yes): sex (categorical), age at Cladribine prescription (continuous, split into age groups: ⩽40 years and >40 years), Body Mass Index (BMI) (ordinal), disease duration (continuous), number of relapses within 12 months before Cladribine prescription (continous), MRI activity within 12 months before Cladribine prescription (categorical), EDSS at the time of Cladribine prescription (ordinal), number of previous DMTs (continuous); variables with p < 0.05 were inserted into the multivariable model. Multicollinearity was evaluated using the variance inflation factor, where a value >10 was considered an index of collinearity among variables.
The results are presented as odds ratios (ORs) and the corresponding 95% confidence intervals (95% CIs). Significance was settled at 0.05.
To verify the probability of therapy sequencing in our cohort, we built two weighted heatmaps, respectively, for the probability of post-Cladribine treatments based on the number of previous treatments and probability of post-Cladribine treatment based on the last DMT before Cladribine.
Heatmaps were generated based on the weighted pivot tables to visualize the proportion of patients requiring DMT post-Cladribine. Probabilities were calculated using normalized row-wise frequencies. The overall risk of requiring post-Cladribine treatment was computed as the mean of a binary indicator variable for post-Cladribine treatment across all patients.
The sample sizes for each treatment group were calculated to understand the distribution of patients across different treatments and to address the issue of different sample sizes, weights were calculated for each treatment group. The weights were inversely proportional to the sample sizes, ensuring that groups with smaller sample sizes did not disproportionately influence the results. Finally, pivot tables were created to summarize the data. The pivot tables were normalized to show the proportion of patients requiring DMT post-Cladribine.
Incidence rates of subsequent treatment for each prior DMT group were standardized to 100 person-years, allowing for direct comparisons between groups with different follow-up durations.
A forest plot was generated to visually represent the incidence rates (IR) along with their corresponding 95% CI. Heatmaps and IR were calculated on patients with at least 6 months of follow-up after second cycle.
Python Software Foundation (version 3.10), Wilmington, Delaware, USA; https://www.python.org was employed for the analysis.
Results
A total cohort of 352 patients was enrolled (Figure 1). Baseline characteristics are shown in Table 1.

Flow chart of the study.
Whole cohort characteristics.

BMI, body mass index; DMT, disease modifying therapy; EDSS, Expanded disability status scale; IQR, Interquartile range; MRI, magnetic resonance imaging, No., number; SD, Standard Deviation.
Generally, 134 patients were naïve to any DMT, while 218 were switchers from other DMTs. Among switchers, 117 (53.7%) had only one DMT prescription before Cladribine, while other 101 (46.3%) had more than one DMT before.
The last DMT before Cladribine prescription was an injectable first line DMT for 48 (22%) patients, oral first line DMT for 141 (64.7%) patients, SP1 inhibitor-Fingolimod for 23 (10.6%) and Natalizumab for 6 (2.7%) patients.
During the interval between first and second cycle, 7 (1.9%) patients had MRI activity and 15 (4.2%) had a relapse after a median time of 5.1 ± 0.8 months. Among them, two stopped therapy and didn’t complete the second Cladribine cycle for suboptimal response (one proceed to hematopoietic stem cells transplantation and the other one started a monoclonal antibody therapy-Natalizumab) and three for other reasons (two patients had moderate Covid19 infection during pandemic and one woman got pregnant).
During the available follow-up from the second cycle (n = 347 patients), 30 patients (8.6%) experienced a relapse after a median time of 10.5 months (IQR 5.3–21.3, minimum 2, maximum 39). Of these, 26 patients (7.4%) had concomitant MRI activity. Fifteen patients (4.3%) exhibited isolated MRI activity from the last Cladribine cycle to the last available MRI.
Patients with conversion to secondary progressive MS were 11 (3.2%) after a median of 21.5 (IQR 17.5–24.6) months. No serious AEs were collected all over the follow-up. The most frequent AE was Herpes Zoster (six patients; Table 2).
Adverse events.

Heatmaps and Incidence rates
A total cohort of 270 patients had at least 6 months of follow-up and was included for the following analyses.
The median available follow-up after second cycle was 25 months (IQR 14–35 months, minimum 6 months, maximum 58 months). Thirty-three (12.2%) patients started a new DMT after a median time of 24 months (IQR 18–31). All patients with a new DMT prescription had disease activity during the follow-up after second cycle (26 for concomitant clinical and radiological activity, 4 for clinical activity alone and 3 for isolated MRI activity). DMTs prescribed are shown in Figure 2.

New DMTs prescribed.
The generalized linear regression model for the event new DMT prescription, revealed age > 40 years at Cladribine prescription as the strongest predictor. For patients aged > 40 years, the OR was 0.84 (95% CI: 0.71, 0.99), p-value = 0.04, suggesting a 16% decrease in the likelihood of receiving a new DMT prescription (Figure 3). No other variables were retained in the model (Table 3).
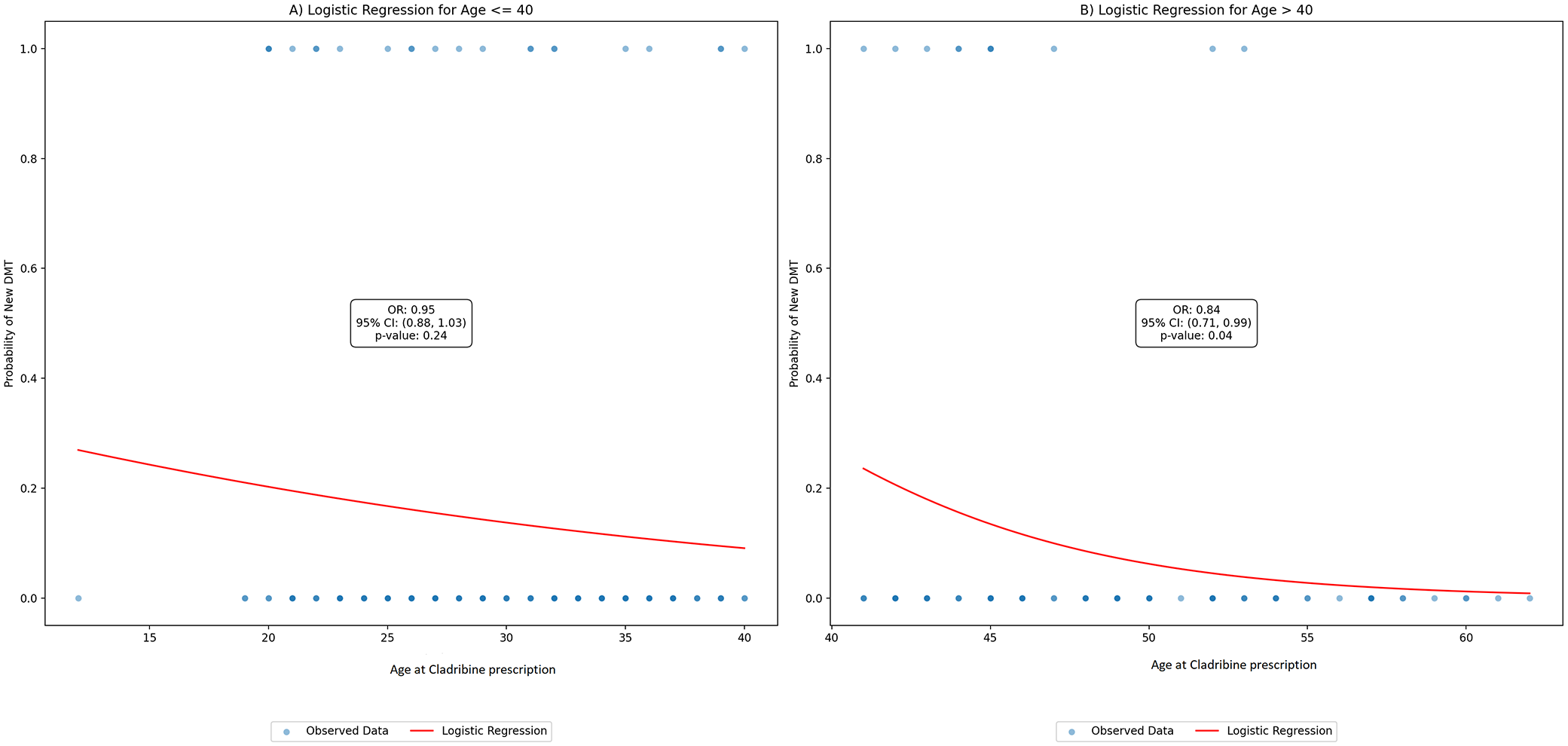
Logistic regression plot for age at Cladribine prescription. (a) Age ⩽40 years and (b) Age > 40 years.
DMT prescription after Cladribine course: Univariable model.

Male sex was used as reference.
BMI, body mass index, DMT, disease modifying therapy; N, number; EDSS, expanded disability status scale; OR, Odds Ratio.
For dichotomic variables the last variable (1 = yes) was employed as reference.
The results of the two different weighted heatmaps for the probability of new DMT prescription are presented in Figures 4 and 5.
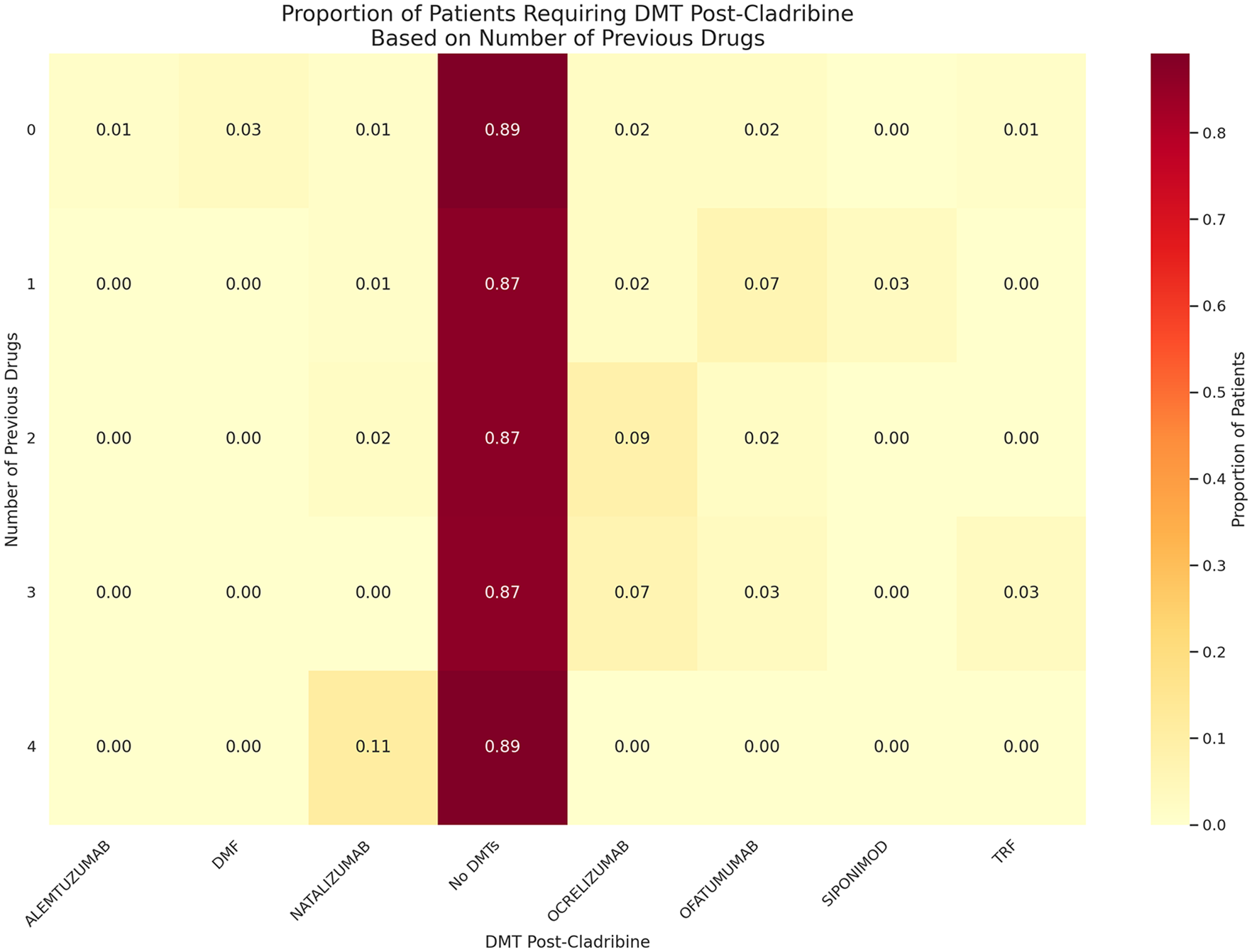
Weighted Heatmap: Proportion of patients requiring new DMT prescription based on number of previous DMTs.

Weighted Heatmap: Proportion of patients requiring new DMT prescription based on previous DMT before Cladribine.
Figure 4 shows that number of previous DMTs not significantly affect the efficacy of Cladribine. The proportion of patients not requiring DMT post-Cladribine remains high regardless of the number of previous drugs (naive patients 89%, four DTMs before 89%).
Conversely, as shown in Figure 5, the type of last DMT before Cladribine influences the efficacy of the treatment: patients previously on Natalizumab, injectable DMTs, oral DMTs showed the highest rates of not requiring post-Cladribine DMT (100%, 94.7%, 86.7%, respectively). Conversely, patients previously on Fingolimod had a lower rate (72.2%).
The overall risk of requiring post-Cladribine treatment across all patients was 12%.
Figure 6 illustrates the IR per 100 person-years, along with their 95% CIs. It highlights the likelihood of patients initiating a new DMT after Cladribine treatment, based on their prior DMT. Fingolimod had the highest IR*persons/year: 11.19, 95% CI: 10.21–12.18), oral DMTs and naïve patients had similar IR*100 persons/year (6.14, 95% CI: 5.82–6.46 and 5.79, 95% CI: 5.46–6.12, respectively), while the injectable DMTs had the lowest IR.*100 persons/year 2.38, 95% CI: 2.05–2.71.

Forest plot-incidence rate of subsequent treatment after Cladribine (100 person-years).
Discussion
In our multicentric real-world Italian study, Cladribine therapy was generally effective during the investigated follow-up period, with 12% of the entire cohort initiating a new therapy after a median follow-up period of 24 months. The emerging data are interesting and worthy of discussion from various perspectives.
First, our aim aligns with recent scientific literature that has explored the positioning of Cladribine, particularly focusing on long-term therapeutic strategies.11–14 Our cohort is very homogeneous, consisting of patients who are either naive or switched due to the inefficacy of another drug.
Age at the time of prescription seems to serve as a protective factor, with individuals over 40 years experiencing a 16% reduction in the likelihood of needing a new DMT prescription.
Current evidence indicates that the effectiveness of Cladribine is not significantly reduced in older individuals with MS.15–17 Additionally, it is generally well tolerated, with no specific safety concerns related to older age, including lymphocytopenia.18,19
Recently an expert panel has recommended the use of Cladribine for individuals aged 45 years or older who were previously on a platform/first-line DMT (Interferons, Glatiramer acetate, Dimethyl fumarate, or Teriflunomide), or for those aged 55 years or older after a high-efficacy DMT (S1P inhibitors, Natalizumab, anti-CD20 agents, Alemtuzumab). 20
Our data suggest that the best profile is observed in patients transitioning from injectable therapies, followed by those on oral therapies and naive patients.
Regarding the data on Natalizumab, although the IR is unreliable due to the small sample size, weighted heatmap analysis shows that patients transitioning from this therapy remained stable with no risk of starting a new DMT. A previous study involving 17 patients who discontinued Natalizumab due to a high John Cunningham virus antibody index (n = 13), disease activity (n = 6), or MRI disease activity (n = 4), and switched to oral Cladribine, demonstrated effective disease suppression over a mean period of 9.7 months with no serious AEs other than the expected lymphopenia.21,22 Although these findings are anecdotal and numerically limited, they underscore the need for further investigation into this type of switch to establish its safety and efficacy in clinical practice.
Another finding confirmed by the literature is the increased risk of subsequent therapy when switching from Fingolimod, necessitating careful case-by-case evaluation, particularly in younger patients. Real-world studies have reported that switching to Cladribine from Fingolimod is associated with a higher rate of disease rebound.23,24 Generally longer time on Fingolimod treatment, younger age at MS diagnosis, and lower lymphocyte levels after discontinuation have been considered as risk factors for rebound.24,25 Another proposed driver was the delay in B-cell suppression, which reaches its nadir 2 months after Cladribine initiation, that may explain the differences in initial disease activity when switching from Fingolimod.24,26,27
The safety profile of Cladribine observed in our study is consistent with findings from previous clinical trials and real-world studies. No serious AEs were reported during the follow-up period, and the most frequent adverse event was Herpes Zoster, which occurred in six patients. This is in line with the safety data from pooled population of patients from early to more advanced relapsing MS. There was no increased risk for infections in general except for a higher incidence of Herpes Zoster. 28
It is essential to incorporate insights from various sources to fully understand the utilization of Cladribine in Italian clinical practice. This requires a thorough understanding of how Cladribine is incorporated into routine care, including treatment protocols, patient selection criteria, and monitoring strategies.
Due to the absence of guidance on long-term management, the main proposal was to classify patients as responders or not based on clinical, MRI activity, and biomarker evaluation with regular and strict disease monitoring based on classification provided from CLARITY study.11,15
The observation that 80% of patients with MRI activity during follow-up did not receive a new therapy prescription, with only 20% initiating treatment, suggests a noteworthy pattern. This emphasizes the value of developing a collaborative decision-making framework to guide treatment strategies effectively.
The limitations of our real world, multicentric study investigating Cladribine include several inherent challenges typical of observational studies. First, the relatively small sample size may limit the statistical power and the ability to detect smaller effect sizes. The absence of a priori power analysis may affect the reliability and generalizability of the results, highlighting the need for careful consideration of these factors in future research. This limitation, coupled with the inherent challenges of long-term studies on immunoreconstituting therapies—such as variability in patient response and potential unforeseen long-term effects—suggests that the findings should be interpreted with caution. Second, the pretreatment drug groups are not very large, which may affect the robustness of subgroup analyses and the generalizability of the results. Although the study spans six centres, the multicentric nature introduces variability that may not be fully accounted for, potentially impacting the consistency of the findings. Additionally, the homogeneity of the cohort, while beneficial for reducing confounding variables, may limit the external validity and applicability of the findings to a different population. These limitations should be considered when interpreting the results and planning future research.
Conclusion
Our real-world data suggest that Cladribine maintains a better profile in terms of treatment persistence and reduced need for subsequent therapies in patients prescribed after 40 years of age and those who are treatment-naive or transitioning from therapies other than Fingolimod. These findings highlight the importance of considering patient age at prescription and treatment history in therapeutic decision-making for MS.29–31
Identifying the optimal patient profile for Cladribine prescription is pivotal as it will enable personalized treatment plans that maximize efficacy and minimize risks. By understanding the key characteristics of patients who respond best to Cladribine, we can tailor therapeutic strategies to achieve optimal outcomes.
