Abstract
Background:
Limited treatment options with a rapid onset of action are available to treat off episodes in Parkinson’s disease (PD) patients. Therefore, the development of rapid onset formulations, for instance with levodopa, is warranted, which was the reason to investigate an inhalable formulation of levodopa.
Objectives:
The primary objective was to determine the duration until maximum effect is reached of inhaled levodopa on the improvement of motor function of PD patients. The secondary objective was to compare the time until maximal effect and the maximal effect of inhaled levodopa versus oral levodopa.
Design:
Open-label randomized two-way one-period crossover trial.
Methods:
Nine PD patients in the ‘off state’ received one dose of inhaled levodopa (90 mg) from Cyclops® and one dose of levodopa orodispersible tablet (100 mg) on two consecutive days in a randomized order. A timed tapping test, Timed Up and Go test (TUG test) and Movement Disorders Society Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) III score were performed pre-dose and on set time points up to 90 min post-dose as measure for motor function. In addition, blood samples were taken for a pharmacokinetic evaluation (Tmax, Cmax and area under the concentration time curve (AUC) 0–3 h).
Results:
The maximal effect of inhaled levodopa was reached at 30 min (tapping test), at 75 min (TUG test) and at 60 min (UPDRS III). The positive effect on the UPDRS was statistically significant within 20 min after inhalation. After oral administration, Cmax and AUC 0–3 h were found to be significant higher (p = 0.028 and p = 0.028, respectively) than after pulmonary administration. Tmax was achieved significantly (p = 0.028) faster after inhalation. The motor function examinations showed a similar maximum clinical improvement after pulmonary and oral administration and although not significant, inhaled levodopa results in a shorter median duration to maximum clinical effect for the TUG and timed finger-tapping test compared with oral administration (TUG: inhalation 55.0 and oral 67.5 min, timed finger-tapping test: inhalation 35.0 and oral 57.5 min). After the levodopa inhalation, there were no adverse events observed and no significant differences found in long-function parameters.
Conclusion:
Inhaled levodopa from Cyclops® shows promising data as a rescue therapy for PD patients with off episodes, not responsive to the current oral therapies.
Trial registration:
The study protocol was approved by the local ethics board ‘Regionale toetsingscommissie patiëntgebonden onderzoek’ (RTPO) in Leeuwarden, The Netherlands (approval number RTPO1019). The study was registered in in the Dutch trial register (LTR) with identification number NL6876. From 5 March 2024 on, the research data on onderzoekmetmensen.nl are known as ‘Overview of Medical Research in the Netherlands’ (OMON). This means the use of the name LTR has thus been dropped. Now, it is registered in the OMON with the same identification number (NL6876, Effectiveness of inhaled levodopa in PD | Research with human participants (onderzoekmetmensen.nl)). All patients provided written informed consent.
Introduction
Parkinson’s disease (PD) is a progressive neurodegenerative disorder characterized by a diminished dopamine production in the central nervous system (CNS) due to loss of dopamine-producing cells in the substantia nigra. 1 The resulting shortage of dopamine causes disruptions of motor circuits in the CNS leading to motor function impairments like tremor, rigidity and bradykinesia. 2 Levodopa, non-ergot dopamine agonists and monoamine oxidase inhibitors are effective in relieving the motor symptoms and signs of the disease. 3 The effectiveness of this treatment subsides over the years and many patients develop fluctuations between so-called on periods, characterized by symptom control or possibly hyperkinesia, and off episodes, in which the motor function is impaired. 4 These off episodes are often caused by a narrowing therapeutic window of levodopa 5 in combination with a delayed onset of effect after orally administered levodopa due to irregular gastrointestinal absorption. 6
There were two options available for the treatment of off episodes in PD patients, namely orodispersible tablets containing levodopa/benserazide (not registered for the treatment of off episodes specifically) and subcutaneous injections of apomorphine. However, these two options are often not satisfactory. Orodispersible tablets containing levodopa appear to work faster than non-orodispersible tablets, but it can still take up to 30–60 min before the desired effect is reached and in some patients, improvement of symptoms does not occur at all. 7 The alternative, apomorphine per injection, has a much faster onset of effect (within 20 min), but its use is often accompanied by nausea, vomiting and pain at the site of injection. 8 Moreover, the injections are considered more burdensome for the patient than other more convenient routes of administration such as oral or pulmonary administration and the patient is frequently dependent on a caregiver for the administration of the injection. Recently, a third option has become available on the market in Europe, a levodopa dry powder inhaler (Inbrija® developed by Acorda Therapeutics, Ardsley, New York, USA).
Inhaled Levodopa seems to be a potent alternative for dispersible levodopa tablets.9,10 After correct inhalation, a major portion of the levodopa is immediately available for absorption. Levodopa inhalation powder facilitates a rapid clinical response because the levodopa is not subjected to the metabolizing enzymes, efflux transporter activity of the gut and first pass metabolism of the liver that occurs after oral administration. 9 Moreover, pulmonary administration bypasses the irregular gastrointestinal absorption of levodopa in off episodes, leading to unpredictable clinical responses after oral administration.
So far, the only registered dry-powder inhalation system for levodopa is Inbrija®, formerly known as CVT-30,11,12 which has been FDA approved since December 2018. This product has a number of drawbacks, related to both the inhaler (capsule-based) and the levodopa formulation (excipients and powder volume), which may limit the suitability of this inhaled levodopa product and leaves ample room for product optimization.13–15 The Cyclops® is a new device developed by Luinstra et al., 16 featuring a novel powder formulation of levodopa suitable for pulmonary administration. Besides levodopa, this powder formulation only contains 2% L-leucine.
L-leucine is an amino acid, a building block for proteins, and thus present ubiquitously in the lungs. 16 The Cyclops® optimizes the drawbacks described for Inbrija® by using fewer excipients and by not using capsules. In a previous study, the tolerability of the pulmonary administered levodopa in PD patients was investigated. The formulation was well tolerated: none of the patients experienced cough or dyspnea and no change in pulmonary function was measured. Also, the pharmacokinetic (PK) results of this study suggest that the tested levodopa formulation may be particularly beneficial for use during an off episode. 13
In this article, we present the data of an open-label crossover trial of pulmonary administered 90-mg levodopa compared with 100-mg oral levodopa in PD patients. The aim of this study was to determine the time until maximum effect is reached on motor function of PD patients during an off episode. Besides the therapeutic effects, we also present a PK evaluation.
Materials and methods
Materials
Levodopa was purchased from Duchefa Farma (Haarlem, The Netherlands) and L-leucine from Sigma-Aldrich (Zwijndrecht, The Netherlands). The levodopa was mixed with 2% w/w L-leucine and micronized. The levodopa with 2% L-leucine powder was weighed manually into the blisters (30.6 mg, corresponding to 30-mg levodopa). The levodopa formulation was inhaled with the Cyclops®; a disposable dry powder inhaler. 17 The comparator medication was an oral levodopa/benserazide orodispersible tablet (Madopar disper) containing 100-mg levodopa and 25-mg benserazide obtained from Roche Pharmaceuticals (Woerden, The Netherlands).
Informed consent and ethics
All patients were treated in accordance with the 1964 Helsinki declaration and its later amendments. The study protocol was approved by the local ethics board ‘Regionale toetsingscommissie patiëntgebonden onderzoek’ (RTPO) in Leeuwarden, The Netherlands (approval number RTPO1019). The study was registered in the Dutch trial register (LTR) with identification number NL6876. From 5 March 2024 on, the research data on onderzoekmetmensen.nl are known as ‘Overview of Medical Research in the Netherlands’ (OMON). This means that the use of the name LTR has thus been dropped. Now it is registered in the OMON with the same identification number (NL6876). All patients provided written informed consent.
Study population
Nine participants with PD were recruited from the ‘Point for Parkinson Centre Groningen’, the Netherlands. The study was performed between May 2019 and January 2022 (which was longer than planned because of the COVID pandemic). Inclusion and exclusion criteria for the study are shown in Table 1. Participants were excluded if they met one or more of the exclusion criteria.
Inclusion and exclusion criteria.

An amendment of the inclusion criteria was made during the study since patients on a morning dose of 100 mg experienced few off episodes, which made inclusion difficult. Because of this, the standard oral morning dose of patients 1–3 and 5–8 (patient 4 was excluded) was limited to 100 mg of levodopa, whereas for patients 9 and 10 this restriction was removed.
UPDRS, Unified Parkinson’s Disease Rating Scale.
The sample size calculation is based on the difference in mean time until maximum effect is reached (Tmax) for inhaled levodopa and oral levodopa (Wilcoxon signed-rank test for matched pairs). Based on an estimated mean of 25 min for levodopa inhalation powder and 40 min for levodopa orodispersible tablet, both with a standard deviation of 10 min and a correlation between groups of 0.5, an effect size of 1.5 is obtained. To show this difference with a power of 90% and an α of 0.05 (two-sided), a sample size of eight patients is required. PD patients in a more advanced stage of their disease sometimes experience a failure of a levodopa dose to induce an on state, a so-called ‘no on’ episode. Anticipating and correcting for this chance of a ‘no on’ episode, a sample size of nine patients is required.
Study design
The study was an open-label crossover trial in which the participants received one dose of inhaled levodopa (90 mg) and one dose (100/25 mg) of levodopa/benserazide orodispersible tablet on two consecutive days in randomized order. The study took 3 days in total for each participant and was performed in the ‘Point for Parkinson Centre Groningen’, the Netherlands. Admission was in the morning of day 1, and the investigational drug administrations on the mornings of days 2 and 3. The study was terminated prematurely if the number of withdrawals is more than four of the included PD patients, for whatever reason. A flowchart of the study design is shown in Figure 1.

Flowchart of the study design.
Randomization
The investigator generated a randomization list for a maximum of 13 participants was produced for the order in which the two intervention treatments (1: 90 mg inhaled levodopa and 2: 100/25 mg orodispersible levodopa/benserazide tablet) were taken. Participants with an uneven enrolment number started with treatment 1 (inhaled levodopa) on day 1 and continue with treatment 2 (orodispersible tablet) on day 2. Participants with an even enrolment number took treatment 2 on day 1 and treatment 1 on day 2.
Dosing
The Cyclops inhaler with a prefilled dose compartment contained 30 mg of levodopa inhalation powder. The 90-mg dose was inhaled by using three 30-mg levodopa inhalers. All levodopa doses administered in this study were at least 7.5 h (five times the half-life time of levodopa) after the last levodopa administration.
Levodopa inhalation
A lung technician trained participants with an empty inhaler until they mastered the inhalation manoeuvre. Flow curves through the inhaler were measured with a differential pressure gauge (PD1 with MC2A measuring converter). LabViews software (National Instruments BV, The Netherlands) conversed the pressure drop across the inhaler into a flow rate. The patient was able to see all flow curves on the computer during the inhalation manoeuvres. This enabled the patients to optimize their inhalation manoeuvre. Additionally, flow curves were measured during the inhalation of the 90 mg levodopa, allowing for the assessment of whether the inhalation was performed correctly.
The levodopa inhaler residue after inhalation was measured by ultraviolet spectrophotometric analyses. Inhaler residue was used for calculation of the delivered dose (blister dose minus inhaler residue).
Spirometry
Lung function was measured as safety check upon admission and again upon discharge from the clinic (end of study). Spirometry was measured using a calibrated pneumotachograph. Forced expiratory volume in 1 s (FEV1), maximum expiratory flow after 50% of the expired forced vital capacity (MEF50) and forced vital capacity (FVC) were measured. If lung function values (FEV1, MEF50, FVC) were >10% lower compared to baseline, the reduction was considered clinically relevant. The measurements were performed by a lung function technician.
Motor function examination
Timed tapping test
In the timed tapping test the participant was instructed to tap the screen of a tablet device with tap recording software successively with the index finger of the most affected hand as rapidly as possible for the duration of 30 s. The software recorded the number of taps.
Timed Up and Go test
The Timed Up and Go (TUG) test measured the time (in seconds) it took an individual to stand up from a chair, walk a distance of 3 m, turn around, walk back to the chair and sit down again. During this exercise no physical assistance was given.
Timed tapping test and TUG test were taken before administration of the levodopa (T = 0) and at T = 10, 20, 30, 40, 50, 60, 75 and 90 min after administration of the levodopa.
Movement Disorders Society Unified Parkinson’s Disease Rating Scale III motor examination
The Movement Disorders Society Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) is a tool to assess severity of PD symptoms. 18 The UPDRS is the primary outcome measure in most clinical trials of PD therapeutics. 19 The UPDRS III motor examination section is a 14-item rating of motor signs. It rates tremor, facial and generalized bradykinesia, and performance on several straightforward tasks. 20
In this study, an accredited MDS-UPDRS scorer rated PD symptoms as defined by the MDS-UPDRS III motor section for each patient, immediately before the administration of the levodopa. Subsequently, the MDS-UPDRS III score was taken at the time points 20 and 60 min after administration of the levodopa.
The MDS-UPDRS motor examinations were recorded on video. A second accredited MDS-UPDRS scorer, who was blind to treatment and the time point at which the score was taken, independently scored the participant videos using the MDS-UPDRS. Rigidity scores were provided for each participant by the first scorer, given that this could not be ascertained from watching the videos. After all videos were scored, marks were compared between raters. If a score deviated by one point (per item), the score of the second scorer was used. In case of larger deviations, the scorers met to come to a consensus.
Blood sampling and analytical methods
A blood sample was drawn immediately before the patient received the study medication. Subsequent blood samples were drawn at time points 5, 10, 15, 20, 30, 45, 60, 90 and 180 min after administration. The blood samples were drawn by a research nurse. Venous blood samples were drawn from an IV line. The samples were collected in an ethylenediaminetetra-acetic acid tube. Within 1 h after sampling, the blood was centrifuged to separate plasma and blood cells. Per 1 ml of plasma, 10 mg reduced glutathione was added and mixed with the plasma. Thereafter, the sample was frozen at −80°C until further analysis. Levodopa was measured by liquid chromatography–tandem mass spectrometry. The blood sample analyses were performed at the University Medical Center Groningen, The Netherlands in the laboratory of Clinical Chemistry.
PK evaluation
Noncompartimental analysis was performed with R using the PKNCA package (noncompartmental analysis calculations for PK data), using sampling times and corresponding serum concentration data. The PK parameters that were calculated were the area under the concentration time curve (AUC) from T = 0 to T = 3 h (AUC 0–3), the maximum levodopa plasma concentration (Cmax) and the time to maximum plasma concentration (Tmax). Levodopa concentration from the pre-dose sample was used as the baseline and subtracted from subsequent samples (to a minimum of zero). For AUCs beyond the last available data point, the concentration data were extrapolated from the last data point and were assumed to be decaying exponentially with the terminal half-life t1/2. Extrapolated AUC values were rejected if the terminal half-life could not be calculated or if the extrapolated part was more than 25% of the interpolated part.
Statistical methods
Statistical analyses were performed with SPSS Statistics version 25.0 (IBM, Chicago, USA). The Shapiro–Wilk test checked for variables normality distribution. A two-tailed paired t test analysed variables following a normal distribution, and the Wilcoxon matched-pairs signed-rank test the variables with skewed distribution. A p-value ⩽0.05 was considered statistically significant.
Study objectives
The primary objective of this study was to determine the time until maximum effect on motor function of PD patients during an off episode after inhalation of levodopa. The secondary objectives were to determine the PK profile (Tmax, Cmax and AUC 0–3 h) of a dose of 90-mg inhaled levodopa in comparison to 100-mg levodopa orodispersible tablet and to determine the maximum clinical improvement and the time to maximum clinical improvement of the motor function of PD patients after inhalation of levodopa in comparison to oral levodopa.
Results
Patients
From May 2019 through January 2022, a total of nine patients (six females) were enrolled. The mean duration of PD was 9 ± 5 years. The patient characteristics are shown in Table 2.
Patient characteristics.

Pharmacodynamic data after inhalation of 90-mg levodopa
From all patients the timed tapping test, TUG test and the MDS-UPDRS III score were taken at all-time points. Patient 4 never started the study. Due to a dose increase in the 4 weeks before admission, the patient did no longer meet all the inclusion criteria. The data of patient 8 were excluded from further analysis because of poor inhalation. The patient exhaled into the inhaler instead of inhaling.
Figure 2 shows a mean change in the pre- to post-dose finger tapping scores. The results show a timed tapping response score of +12.6 after 20 min and +13.6 after 60 min. The maximum effect was measured after 30 min with a score of +16.5.

Timed tapping test: change from before to after dosing for timed tapping response score upon inhalation of levodopa. The figure shows arithmetic means (solid lined) and standard error of the mean (shaded regions), N = 8.
Although not reaching statistical significance, Figure 2 shows a clear trend that points towards a positive effect (more taps) of the levodopa inhalation on the finger tapping test. Not all patients suffered from stiffness or tremor in their hands/arms during an off episode. These patients had a normal score at baseline, so no improvement was expected and seen.
Figure 3 shows the mean changes in the TUG test of −6.3 to −7.1 s after 20–60 min, respectively, with a maximum effect of TUG score of −7.1 s after 50 min. The large standard error of the mean is caused by an exceptionally strong effect observed for one patient.
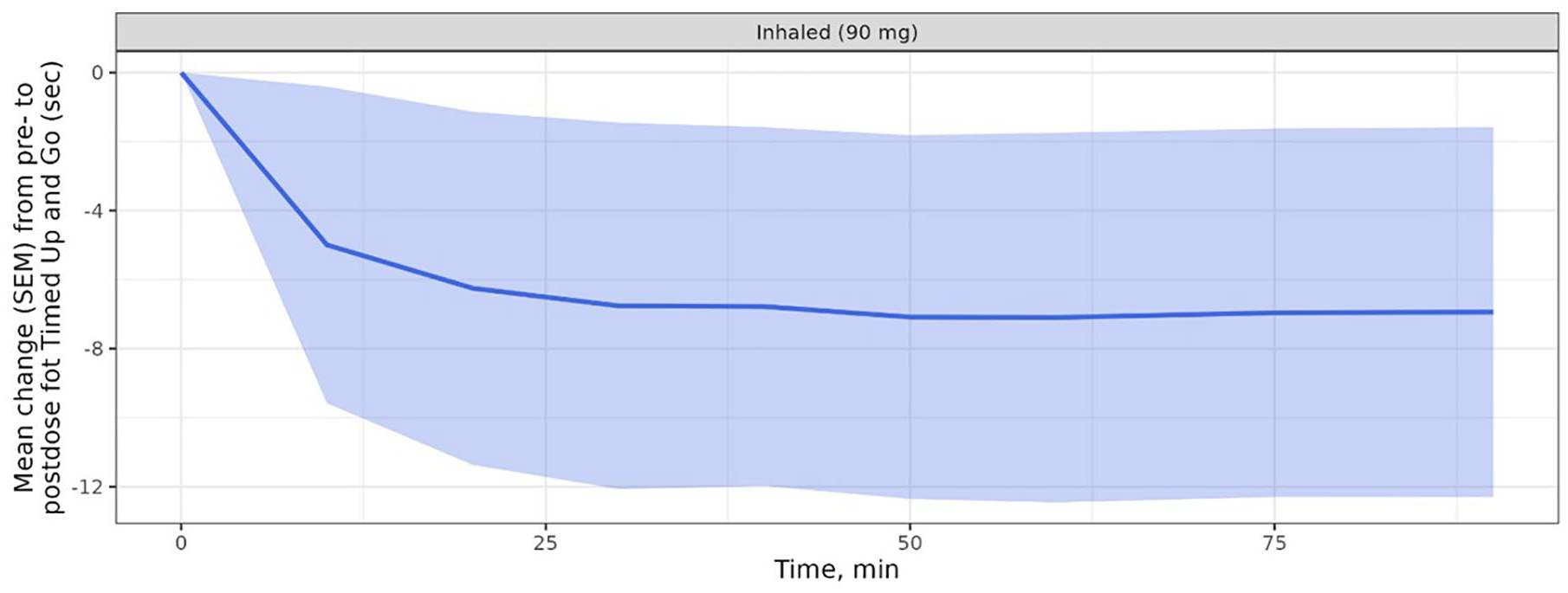
TUG test; pre- to post-dose change for TUG score upon inhalation of levodopa. Figure shows arithmetic means (solid lined) and standard error of the mean (shaded regions), N = 8.
Figure 4 shows a significant pre- to post-dose improvement in mean UPDRS III score of −11.5 points after 20 min and −15.5 points after 60 min. The maximum effect was measured after 60 min with the notion that no assessments were performed at later time points.
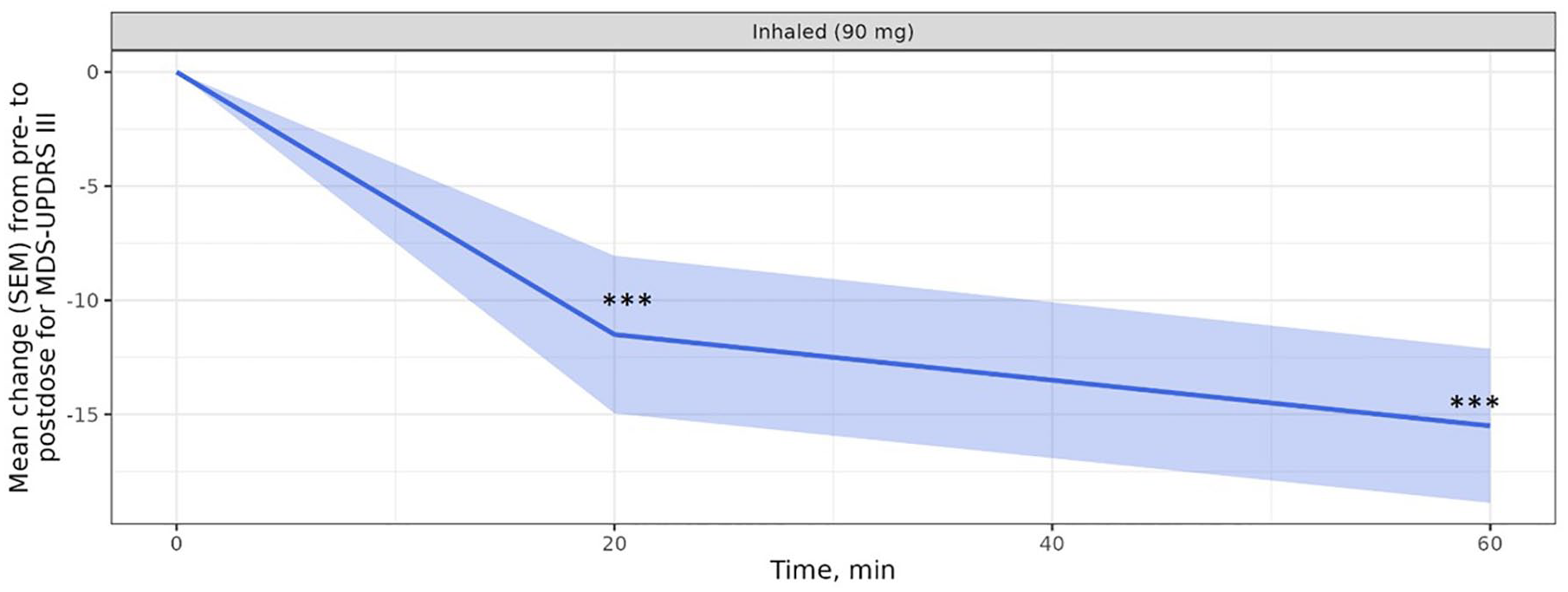
MDS-UPDRS III motor examination; change from before to after dosing for MDS-UPDRS III score upon inhalation of levodopa. Figure shows arithmetic means (solid lined) and standard error of the mean (shaded regions), N = 8 (pt 8 excluded).
Comparison of pharmacodynamic data with oral administration of 100 mg levodopa
Figure 5 shows the clinical improvement on the motor function after oral intake of levodopa. The maximum clinical improvement was seen after 40 min (tapping test), after 75 min (TUG test) and after 60 min (UPDRS III).
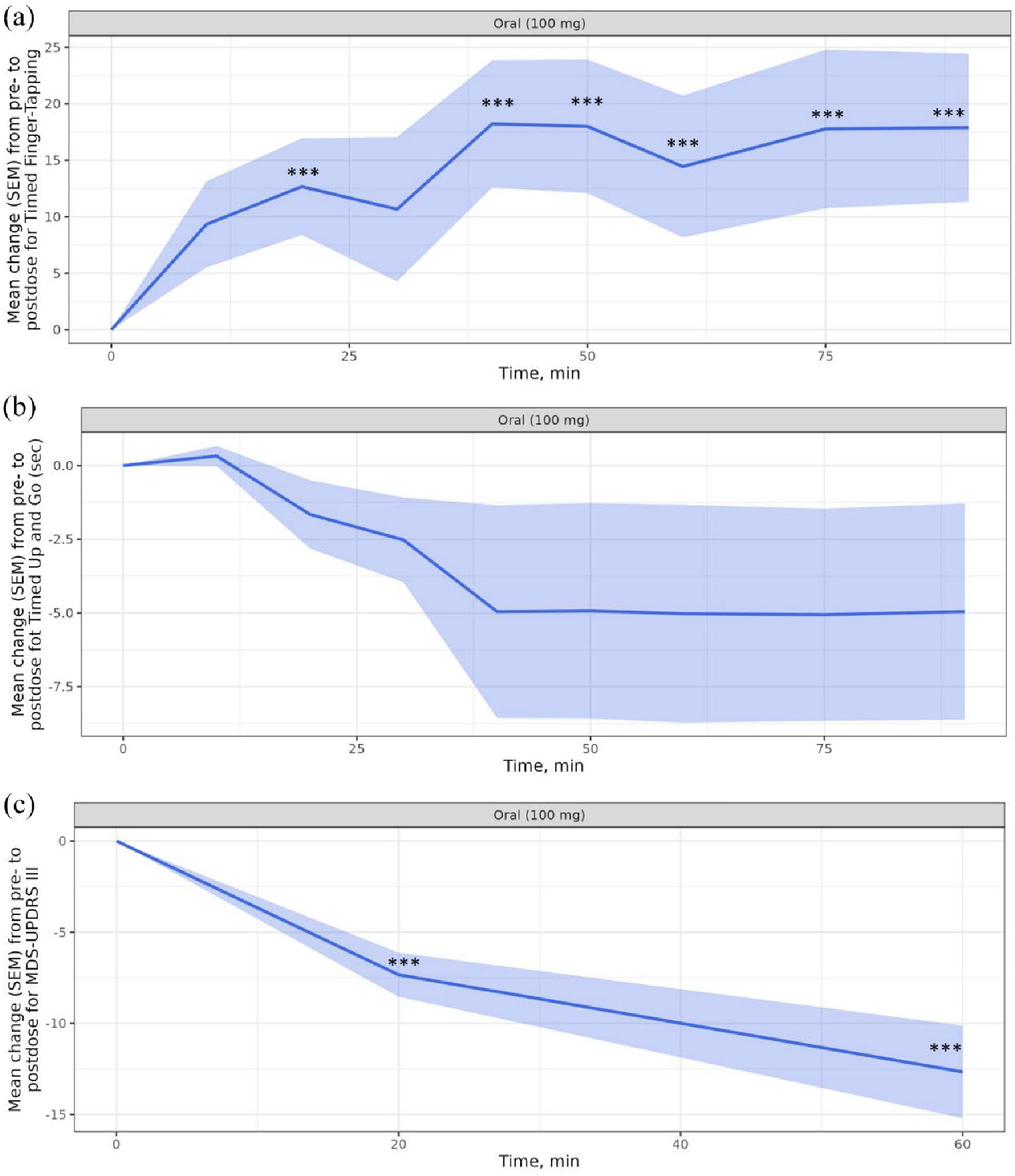
Mean change with standard error of the mean (shaded regions) after oral intake of 100/25 mg levodopa/benserazide. (a) Tapping test. (b) TUG-test. (c) MDS-UPDRS III, N = 9.
The maximum clinical improvements of all patients after oral administration and inhalation were compared (see Table 3). No difference was found between the two forms of administration (tapping test p = 0.484, TUG test p = 0.484 and UDPRS p = 0.309).
Comparison of maximum clinical improvement between inhaled and oral levodopa.

CI, confidence interval; IQR, interquartile range; Mdn, median; TUG, Timed Up and Go.
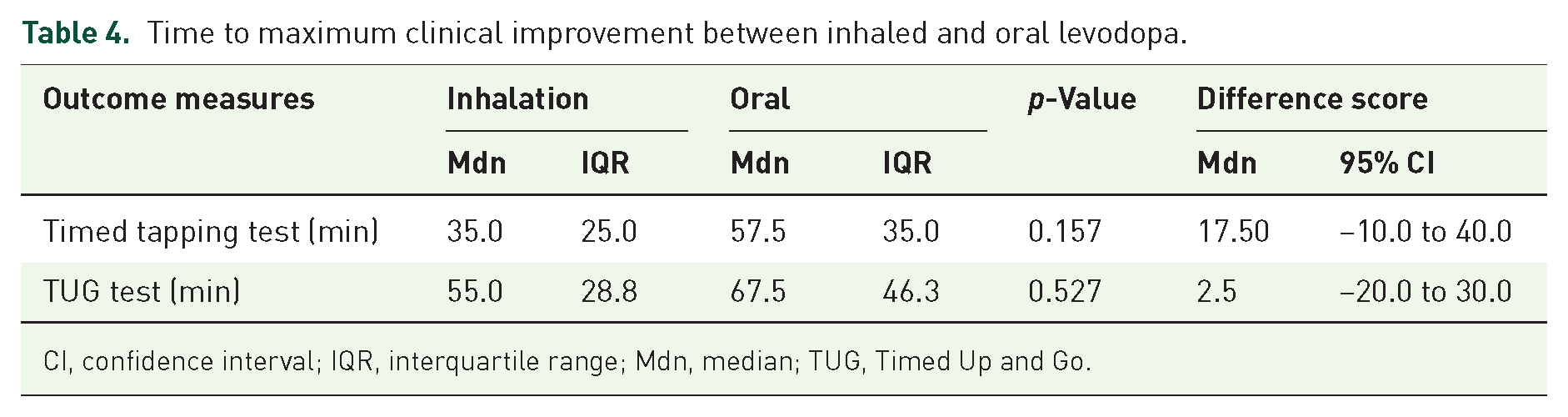
Table 4 shows the time to maximum clinical improvement between inhaled and oral levodopa.
Time to maximum clinical improvement between inhaled and oral levodopa.
CI, confidence interval; IQR, interquartile range; Mdn, median; TUG, Timed Up and Go.
Although not significant, inhaled levodopa resulted in a shorter median duration to maximum clinical effect for the TUG and timed tapping test compared with oral administration. No comparison was made for MDS-UPDRS III score because there were only two assessments (time points 20 and 60 min) after administration of the levodopa.
PK data
A total of 156 blood samples were collected and levodopa plasma concentrations were determined. Twenty-four samples were missed due to problems with the IV line. Sixteen of these missing samples were from participant 9, no blood samples were taken on the inhalation day. The data of patient 8 on the inhalation day were excluded from further analysis because of poor inhalation.
The levodopa plasma concentrations after 90-mg inhaled levodopa and after an oral administration of 100-mg levodopa are shown in Figure 6.

Levodopa plasma concentration. (a) Inhaled dosing. (b) Oral dosing.
Comparison of the PK parameters Cmax, Tmax and AUC 0–3 between the inhaled and oral levodopa are shown in Table 5. After oral administration, the Cmax and AUC 0–3 h (dose-adjusted) were found to be higher than after pulmonary administration. This difference was significant (p = 0.028 and p = 0.028, respectively). Tmax was achieved significantly (p = 0.028) faster after inhalation.
Comparison of pharmacokinetic parameters between the inhaled and oral levodopa.

CI, confidence interval; IQR, interquartile range; Mdn, median.
Spirometry
There were no significant differences in lung-function parameters (FEV1, FVC and MEF50) found after inhalation of the levodopa (end of study) compared to pre-dose. No adverse events were observed, specifically no cough and dyspnoea events occurred.
Discussion
The pulmonary administration of levodopa could be an appealing alternative to oral administration in the treatment of off episodes. It is crucial that patients possess the ability to utilize a dry powder inhaler during an off episode. Luinstra et al. 21 investigated this aspect, and the results suggest that PD patients are capable of using the inhaler during an off episode. Additionally, patients with PD who also have concurrent lung conditions or respiratory disturbances often have prior experience with inhalers, which may facilitate their ability to self-administer inhaled medications. A levodopa inhalation powder has been developed suitable for pulmonary administration with the Cyclops® device. 16 This inhalation powder demonstrates rapid absorption and is well-tolerated by patients. 13 In this study, we assessed the therapeutic effects of an inhaled levodopa dry powder formulation on the recovery from off episodes in PD patients.
The UPDRS III motor score shows a significant improvement from pre-dose to post-dose after pulmonary administration, with a mean of −11.5 points after 20 min and −15.5 points after 60 min. In general an improvement of 5 points is considered as clinically meaningful, 12 a threshold that is already achieved after 20 min in this study (the first assessment time). The fast Tmax (7 min) that has been observed after levodopa inhalation indicates that a clinically meaningful effect may already occur earlier than 20 min. Inbrija® shows a treatment effect (UPDRS III) at 10 min. 12 Based on the (PK) results of our study, a similar time is expected for levodopa administered with the Cyclops® inhaler. Although not significant, inhaled levodopa results in a shorter median duration to maximum clinical effect for the TUG and timed tapping test compared with oral administration (TUG: inhalation 55.0 and oral 67.5 min, timed tapping test: inhalation 35.0 and oral 57.5 min). This could be indicative of a faster onset of effect following inhalation, which would be in line with the shorter Tmax, but it appears that a larger study population would be required to prove statistical significance, given the variation in the data.
The PK evaluation shows that inhaled levodopa is absorbed faster than the oral dose. In all patients, the Tmax of levodopa was reached earlier after inhalation (mean Tmax was 7 min as opposed to 28 min for oral doses). The slow rise of levodopa plasma concentrations after oral administration may be the result of delayed gastric emptying caused by not being in a true fasting state or by the irregular gastrointestinal absorption of levodopa in off episodes. 13 The AUC was half as high after pulmonary administration compared after oral administration. It is remarkable that the motor function examinations show a similar clinical improvement after pulmonary and oral administration. This suggests that a rapidly rising blood level possibly enhances the therapeutic effect.
The overall results of this study show a fast and clinically meaningful positive effect of levodopa inhalation powder inhaled with the Cyclops® on off episodes improvement in PD patients. Since regular oral levodopa treatment was withheld the night and morning prior to levodopa administration, the results in this study were obtained under worst-case conditions (absence of systemic levodopa and decarboxylase inhibitors such as carbidopa and benserazide). Therefore, the effect of levodopa inhalation powder on off episodes in the presence of systemic levodopa and levodopa metabolism inhibitors is expected to be larger when administered as add-on therapy. The time to reach the minimum effective levodopa concentration under such conditions is expected to be shorter due to decreased peripheral conversion of levodopa to dopamine. 22 It may be useful to perform a follow-up trial to investigate the clinical effects of the levodopa inhalation powder if used as add-on to a regular oral levodopa regime. The oral levodopa is in general always combined with a decarboxylase inhibitor.
This study supports earlier findings that our levodopa inhalation powder is safe. 13
None of the patients had a drop in FEV1, MEF50, or FVC, and they did not experience cough or dyspnoea during or after the inhalation manoeuvre. In the study reported by LeWitt et al., 23 using CVT-301, cough was the most common adverse event and the common reason for adverse events related treatment discontinuation. Therefore, the absence of cough is an important advantage of the formulation used in this study.
A satisfactory inhaler technique (preparation and inhalation) is the key to the successful delivery of drug substance by an inhaler. A poor inhaler technique was seen in one patient. This resulted in low levodopa levels, consequently leading to a persistent off episode. In previous studies, PD patients have proven to be able to prepare and operate the Cyclops® inhaler while being off. 24 In a follow-up trail, we will therefore study whether this patient group can successfully operate the inhaler while being unsupervised (in their home situation).
Conclusion
This study shows that inhaled levodopa has a clinically meaningful effect on improvement of off episodes in PD patients within 20 min. The PK evaluation shows that inhaled levodopa is absorbed faster, reaching peak concentration 7 min after dosing, as opposed to 28 min for oral doses. The motor function examinations show a similar maximum clinical improvement after pulmonary and oral administration despite the lower levodopa plasma concentrations after pulmonary administration. This suggests that a rapidly rising blood level possibly enhances the therapeutic effect. Although not significant, inhaled levodopa results in a shorter median duration to maximum clinical effect for the TUG and timed finger-tapping test compared with oral administration. The levodopa powder inhalation powder is well tolerated; there were no adverse events observed and no significant differences found in long-function parameters. The overall results of this study suggest that the tested levodopa inhalation powder may be particularly beneficial as an alternative rescue therapy for PD patients with motor fluctuations.
Footnotes
Acknowledgements
The authors thank the staff of PureIMS for the production of the levodopa Cyclops® product and help in designing the figures. The staff of the Point for Parkinson centre is acknowledged for the help during the admissions, Mr S. Martijn for performing the spirometry measurements and Ms M. de Hoop, Ms J. de Vries and Mr W. Wiersema for drawing the blood samples and Ms J.M. ten Kate, Ms N. Strating, Ms E. Oosterhout, Ms S.M. van Zonneveld, Mr T. Brockötter for assessing the MSD-UPDRS score and Mr R. Nijboer for giving inhalation instructions.
