Abstract
Background:
Dose optimization of sublingual apomorphine (SL-APO), a dopamine agonist for the treatment of OFF episodes in patients with Parkinson’s disease (PD), has been performed under clinical supervision in clinical trials. SL-APO may be a candidate for home dosing optimization which would be less burdensome for patients.
Objectives:
To evaluate the feasibility and safety of home optimization of SL-APO in patients with PD and OFF episodes.
Design:
A multicenter, randomized, crossover study comparing SL-APO with subcutaneous apomorphine was conducted, comprising an open-label dose-optimization phase and a treatment phase. This non-comparative analysis focuses on the outcomes of the dose-optimization phase with SL-APO only.
Methods:
Patients with PD and OFF episodes received SL-APO at an initial dose of 10 mg in the clinic (open-label). Further optimization could continue at home in 5 mg increments during subsequent OFF episodes (maximum dose of 30 mg). Optimization and tolerability were assessed daily by patient-reported feedback via telephone. Patients reporting a FULL ON returned to the clinic for a dose-confirmation visit (DCV). In patients with inadequate response as determined during the DCV, the dose could be further optimized at home.
Results:
Home optimization was continued by 81.4% (83/102) of patients. Of these, 80.7% identified an effective, tolerable dose. Mean time between initial clinic visit and DCV 1 was 6.8 days, and the final optimized dose of SL-APO was 30 mg (mode). In total, 62.7% of patients reported ⩾1 adverse event; the most common included nausea (31.4%), dizziness (9.8%), somnolence (8.8%), dyskinesia (7.8%), and fatigue (5.9%). The safety profile in this study in which most patients performed home dose optimization was consistent with the study utilizing clinic-based optimization.
Conclusion:
After the first clinic dose, home dose optimization of SL-APO appears feasible in patients with PD and OFF episodes, with most patients identifying their optimal SL-APO dose at home.
Trial registration:
This study is registered with EudraCT (2016-003456-7): Clinical Trials register – Search for eudract_number:2016-003456-70.
Introduction
Dopaminergic agonists (DAs) are a common treatment for Parkinson’s disease (PD) and have been associated with class-specific adverse events (AEs), including nausea and orthostatic hypotension (OH).1,2 When initiating DA treatment, including apomorphine, dosage is optimized in a stepwise manner to ensure efficacy and tolerability. Historically, clinical trials for oral/transdermal DAs conducted dose optimization entirely at home in a blinded fashion,3–5 whereas clinical trials of apomorphine formulations conducted open-label dose optimization in clinic.6,7 Apomorphine sublingual film (SL-APO), approved for the acute, intermittent treatment of OFF episodes in patients with PD,1,6 had been optimized using a similar paradigm with dose initiation and identification of the optimal and tolerable dose performed entirely in the clinic.2,6 However, as published data of SL-APO have demonstrated relatively low rates of AEs, home dose optimization following initiation in clinic was later considered possible as it may be less burdensome for the patient.2,6,8
Methods
Study design
A European multicenter, open-label, randomized, crossover study (EudraCT: 2016-003456-7; Clinical Trials register – Search for eudract_number:2016-003456-70), conducted between December 2018 and August 2021, evaluated SL-APO compared with subcutaneous apomorphine (SC-APO) in patients with PD and OFF episodes. Full study design details are provided in a companion article 9 and described briefly herein. The study included an open-label dose-optimization phase and treatment phase; however, the current analysis is non-comparative and describes the outcomes of the dose-optimization phase with SL-APO only.
Patients
Full eligibility criteria are provided in a companion article 9 (Supplemental Table 1 unpublished data). Briefly, adults with PD responsive to and being treated with stable doses of carbidopa/levodopa (CD/LD) and any additional PD medications for ⩾4 weeks (>8 weeks for monoamine oxidase-B inhibitors) were enrolled. Additional key inclusion criteria were stage 1–3 by the modified Hoehn and Yahr scale when ON, ⩾1 OFF episode/day, and ⩾2 h of total daily OFF time. Key exclusion criteria included atypical or secondary parkinsonism, major psychiatric disorder, permanent discontinuation of prior SC-APO administration or prior exposure to SL-APO, and history of symptomatic OH requiring medication.
Procedures
SL-APO dose optimization was initiated in an open-label design in clinic at 10 mg with patients in a practically defined OFF episode 9 (no antiparkinsonian medication after midnight the night prior; Supplemental Figure 1). Assessments included vital signs [orthostatic blood pressure (BP) and pulse rate, before and 60 min after SL-APO administration]. If the patient did not achieve a FULL ON (i.e. period of time the medication provided benefit with regard to mobility, stiffness, and slowness and the patient had adequate motor function to perform their usual daily activities) in ⩽30 min, but tolerated the dose, further optimization could continue at home in 5 mg increments. Dose increases could continue up to a maximum dose of 30 mg during subsequent OFF episodes (⩾2 h and ⩽5 days after the previous dose) without in-person supervision until the patient felt an optimal, tolerable response was achieved. During home dose optimization, clinic staff telephoned patients daily to monitor progress and assess tolerability via patient self-report. Patients did not record BP or pulse at home. Once a tolerable FULL ON was achieved, patients returned to clinic for a dose-confirmation visit (DCV), in which previously described assessments were conducted, including orthostatic BP and pulse readings. If the investigator determined that the ON response was inadequate during the DCV, increased SL-APO doses could be administered during subsequent OFF episodes at home. Additional DCVs could be completed to assess higher doses.
Reports of AEs were collected via daily clinical staff contacts. Initially, use of the antiemetic domperidone was optional if clinically warranted but was not to be used prophylactically. Following a protocol amendment, domperidone use remained optional but could also be used prophylactically or if clinically warranted.
Statistical analysis
The initial sample size calculation for the main study CTH-302 was based on the primary endpoint of the study, the mean change from pre-dose in the Movement Disorder Society-Sponsored Revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) Part III Motor Examination score at 90 min post-dose, evaluated at Visits 3 and 6 after 4 weeks of dosing for each treatment (SL-APO or SC-APO) in the treatment phase (Part B). Assuming that the discontinuation rate is 25% in the dose-optimization phase (Part A), a total of 106 subjects will be randomized into Part A, so that at least 80 subjects are randomized into Part B. Assuming a 30% discontinuation rate in Part B, approximately 55 subjects are expected to complete Part B. With 55 subjects, the study has 90% power to detect a mean treatment difference between SL-APO and SC-APO of 5.5 points for the change in MDS-UPDRS Part III score, assuming a standard deviation of 12 points for the period differences in Part B. The expected mean treatment difference of at least 5.5 points for the change in MDS-UPDRS Part III score was based on the results of the published pivotal study. (Olanow, Factor and Espay, 2020)
The data reported in this subanalysis were analyzed descriptively.
Results
SL-APO treatment was initiated at 10 mg in clinic by 102 patients (Figure 1); FULL ON was achieved by 12 (11.8%) patients with the 10 mg dose. Dose optimization was continued in clinic at subsequent visits by three (2.9%) patients. Four (3.9%) patients discontinued before receiving a second dose: three (2.9%) patients discontinued due to AEs with the 10 mg dose (nausea, n = 1; vomiting, n = 1; nausea and hypotension, n = 1) and one (1.0%) patient withdrew consent. Dose optimization continued at home in 83 (81.4%) patients. Sixteen (19.3%) of these patients discontinued at home [lack of efficacy after reaching the 30 mg dose, n = 13; AE (hyperhidrosis and vomiting (15 mg dose)), n = 1; withdrawal by patient, n = 1; early termination due to sponsor request, n = 1].
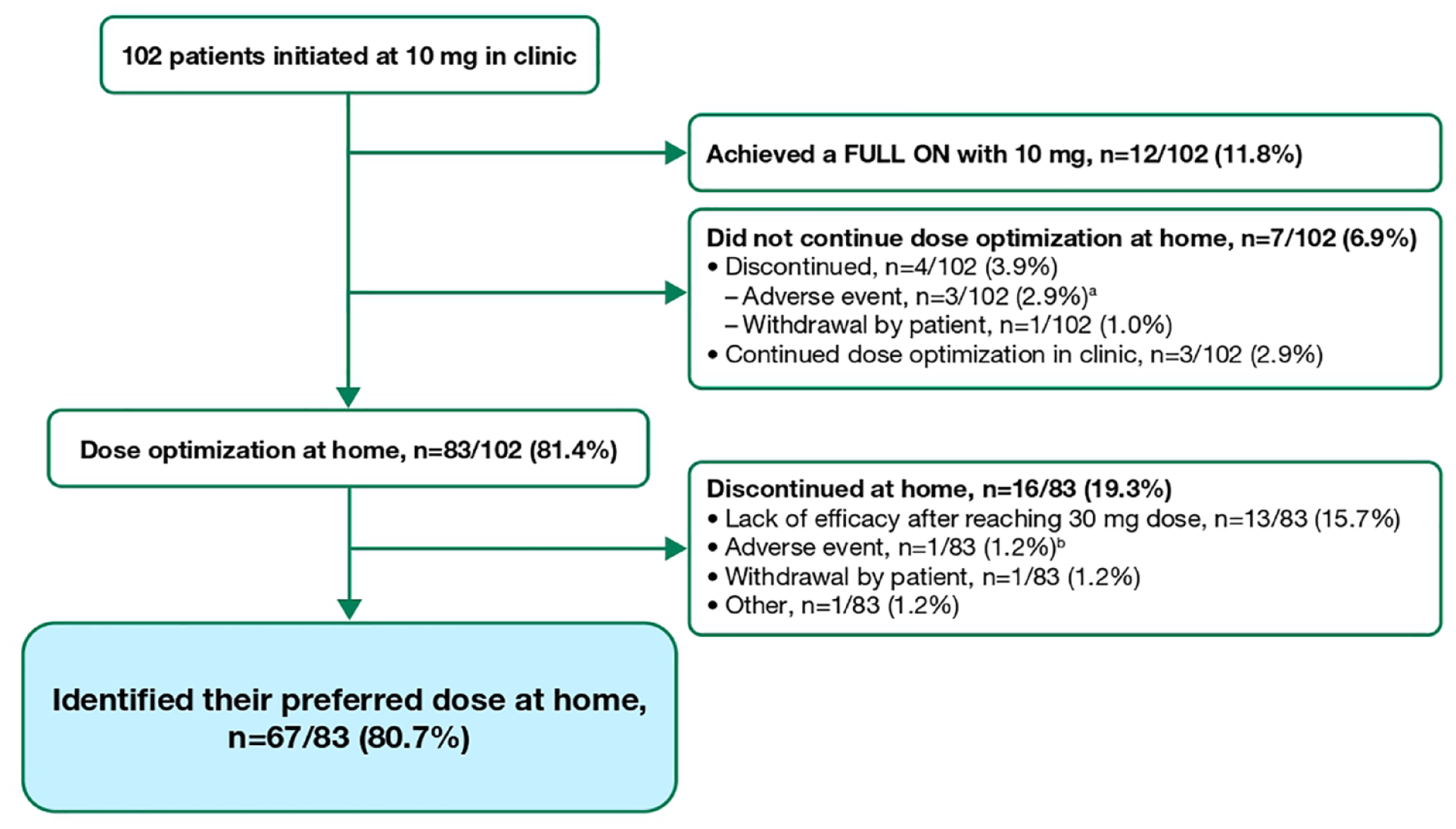
SL-APO home dose-optimization experience (dose-optimization phase safety population).
Overall, 67 (80.7%) patients identified their preferred dose at home, completing home dose optimization. At DCV 1, 54/67 (80.6%) of these patients had identified their dose at home, with 38/67 (56.7%) patients having the same dose confirmed by investigators, 15 (22.4%) had reached the maximum dose of 30 mg and required a dose decrease due to AEs, and one (1.5%) requiring a dose increase (Figure 2). Ten (14.9%) patients had further dose increases at a subsequent DCV, two (3.0%) evaluated all doses and did not reach an effective dose, and one (1.5%) did not attend a DCV. Of the 67 patients who completed home dose optimization, concomitant PD medications included DAs [86.6% (n = 58)], monoamine oxidase-B inhibitors [46.3% (n = 31)], and other agents [43.3% (n = 29)].

SL-APO dose-confirmation experience (dose-optimization phase safety population).
Overall, 54 (80.6%) patients required one DCV, eight (11.9%) patients required two DCVs, three (4.5%) patients required three DCVs, and one (1.5%) patient required four DCVs to complete home dose optimization. The mean (range) time between the first clinic visit (10 mg dose initiation) and identification of a first optimal dose at home before DCV 1 was 3.4 days (1–11). The mean (range) time between the first clinic visit and DCV 1 was 6.8 days (2–17). Among patients who completed dose optimization, the final optimized dose of SL-APO was 30 mg (mode).
Safety findings from the dose-optimization phase of this study are provided in a companion article. 9 During SL-APO dose optimization, ⩾1 AE was reported by 64 (62.7%) patients. The most common (⩾5%) AEs were nausea [31.4% (n = 32)], dizziness [9.8% (n = 10)], somnolence [8.8% (n = 9)], dyskinesia [7.8% (n = 8)], and fatigue [5.9% (n = 6)]. All-cause AEs related to BP (such as dizziness, syncope, presyncope, hypotension, and/or OH) were experienced by 17 patients [16.7% (23 events)]. BP readings were available for 14 of the 23 events (9 of the 23 events occurred on non-clinic days) and 3 of the 14 events were associated with decreased BP that met the definition of OH. Severe AEs that were unrelated to OH-associated AEs were reported by two (2.0%) patients. No deaths occurred during dose optimization.
Discussion
After an initial clinic visit for dose initiation, most patients performed SL-APO dose optimization at home and 80.7% safely completed home dose optimization without in-person supervision based on self-perceived efficacy and tolerability. The safety profile of SL-APO in this study in which most patients performed home dose optimization was similar to that observed for optimization completed entirely within the clinic in the published pivotal study. 6 Nausea was the most common AE in both studies [current study, 31.4% (32/102); pivotal study, 20.6% (29/141)]. 6 Comparable proportions of patients in both studies reported dizziness, somnolence, and fatigue. 6 Reports of dizziness, syncope, presyncope, hypotension, and/or OH were consistent with the pivotal study and other studies investigating SL-APO.2,10 BP readings were not performed at home, and therefore patient-reported AEs of dizziness cannot be correlated with BP.
SL-APO home dose optimization following treatment initiation in clinic offers a valuable option for both patients and healthcare providers. Dose optimization in the home setting is convenient for patients who are unable to attend the clinic while experiencing OFF episodes and provides flexibility for healthcare providers who are unable to accommodate multiple dose-optimization clinic visits. Importantly, it allows for dose identification in a real-world setting, an advantage over the stringent clinic environment. Furthermore, slowly identifying optimal doses at home may allow for gradual development of tolerance to certain possible AEs, such as nausea; however, additional data are needed for confirmation.
This analysis has several limitations to consider. The higher doses to which patients were optimized may reflect investigator and/or patient experience with apomorphine, as some patients had previous experience with SC-APO. Furthermore, investigators may have been inclined to increase the SL-APO dose over the dose identified by patients at home. For the 15 patients who experienced dose reductions at DCV 1, 11 came from two sites (n = 6 and n = 5, respectively). Possibly, these investigators were more inclined to have patients try higher doses at home; three patients did not report AEs, and the rationale for the dose decrease is not reported. Lastly, due to the data collection methods used in this study, rates of AEs experienced by patients in the home dose optimization and in-clinic optimization groups could not be separated. As such, it is not possible to directly compare the safety profiles of these groups.
Conclusion
These results demonstrate that most patients identified their optimal dose of SL-APO at home without in-person supervision and that home dose optimization, which can be regarded as more convenient for patients, appears generally comparable in safety profile with dose optimization completed entirely in clinic.
Supplemental Material
sj-docx-1-tan-10.1177_17562864231209240 – Supplemental material for Feasibility of home dose optimization of apomorphine sublingual film in Parkinson’s disease patients with OFF episodes: results from the dose-optimization phase of an open-label, randomized crossover study
Supplemental material, sj-docx-1-tan-10.1177_17562864231209240 for Feasibility of home dose optimization of apomorphine sublingual film in Parkinson’s disease patients with OFF episodes: results from the dose-optimization phase of an open-label, randomized crossover study by Jan Kassubek, Fabrizio Stocchi, Ernest Balaguer Martinez, Rajesh Pahwa, William Ondo, Yi Zhang, Alyssa Bowling, Eric Pappert, Stuart Isaacson and Stacy Wu in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
The authors would like to thank all CTH-302 study investigators (see Supplemental Table 2) for their contributions to the study. Medical writing and editorial assistance were provided by Maureen Wallace-Nadolski, PhD, and Jessica Deckman, PhD, CMPP, of the Lockwood Group (Stamford, CT, USA) and mXm Medical Communications (Kent, UK).
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
