Abstract
We describe here the first case of cerebral air embolism (CAE) due to a dysfunctional long-term central venous catheter for hemodialysis in a 39-year-old woman with a history of lung transplantation. Air emboli are rare but potentially fatal complications of hemodialysis, in particular, when they involve the brain. Early management with hyperbaric oxygen therapy (HBOT) is critical to prevent deterioration of the patient’s condition. In this case, our patient presented her first symptoms, likely a seizure due to multiple cerebral air emboli, during her hemodialysis session. She was then monitored in the Nephrology Intensive Care Unit in accordance to the medical reference center (with HBOT). Twelve hours later, she experienced secondary deterioration, presenting with acute aphasia, left hemineglect syndrome, and hemiplegia. She was rapidly transferred to the medical reference center for HBOT. The patient fully recovered after receiving three sessions of HBOT. She also presented a seizure during each HBOT session, attributed to hyperoxia. She never experienced another seizure after the episode of CAE. This case highlights the importance of considering patients who have a lung transplant to be at increased risk for air emboli during hemodialysis and the need to rapidly recognize symptoms and start treatment, including HBOT, to optimize recovery.
Keywords
Introduction
Air emboli are rare complications of hemodialysis catheters for which the mechanism is now well understood. They occur in most cases during the insertion or replacement of long-term hemodialysis catheters.1–3 The connection between the vein and the air (by the catheter) and a pressure gradient (i.e., deep inspiration) favor the migration of air into the vein. Air bubbles can then potentially migrate to the pulmonary artery, the arterial circulation, or the cerebral venous system. 4 Although rare, they can be associated with life-threatening organ involvement, especially the brain, in the absence of appropriate treatment.5–7 Thus, knowledge of how to clinically manage such situations is crucial to prevent a fatal outcome.
We present a situation of air embolism that occurred after a hemodialysis session using a long-term jugular catheter in a patient with a lung transplant.
Case description
A 39-year-old woman was admitted to the Nephrology Intensive Care Unit after a hemodialysis session with a suspicion of air embolism due to a sealing dysfunction of a long-term jugular dialysis catheter. The patient’s medical history included cystic fibrosis. She had received two lung transplants and was experiencing chronic graft rejection. End-stage renal disease occurred due to calcineurin inhibitor toxicity, and she received a kidney transplant that failed due to thrombosis of the kidney graft and began chronic hemodialysis. She also experienced a severe cytomegalovirus infection and a nonsymptomatic severe acute respiratory syndrome coronavirus 2 infection 1 month before the air embolism episode.
After a 4-h hemodialysis session, the patient presented with light-headedness and nausea when she was disconnected from the dialysis machine and stood up. She lay down immediately after and clinical examination found blood in the lines of the catheter and on its pad. The obturator appeared to be defective. The lines were cleaned, and no air was observed. Moreover, the patient’s blood pressure was normal, with no dyspnea, hypoglycemia, other complaints, or neurological deficiency. At this point, the medical team decided to keep her in observation in the dialysis center. The first hypothesis was lightheadedness because of hypovolemia due to ultrafiltration. She, thus, received an isosmotic saline infusion on the arterial line of the hemodialysis catheter. Less than 1 h after she had lied down, she experienced acute respiratory distress (88% oxygen saturation with tachypnea and cyanosis of the lips), paresthesia of the extremities, and an inability to speak. An electrocardiogram (ECG) found sinus and regular tachycardia. A blood gas test showed respiratory alkalosis, with no increase in arterial lactate concentration. Pulmonary embolism was quickly excluded by computed tomography (CT), which showed no air trapped in the pulmonary vessels or right heart cavities. She rapidly recovered from her symptoms and was then transferred to the Nephrology Intensive Care Unit to be monitored. At this point, the primary hypothesis was an air embolism, given the chronology of the events. Because of the difficulty of transferring the patient during the night and due to a favorable clinical evolution, it was decided in accordance with the reference center (with a hyperbaric chamber) to monitor her by frequent neurological scoring (each hour) in the first intention.
Twelve hours later, she presented with acute aphasia with left hemineglect syndrome and hemiplegia. The Glasgow Coma Scale score was 9/15 (E4/M4/V1). Blood tests found arterial lactate concentration at 9 mmol/L (normal value <1.6 mmol/L). Cerebral magnetic resonance imaging (MRI) showed multiple distal ischemic lesions compatible with a recent embolic stroke (Figure 1). Thus, the diffusion weight imaging hypersignals associated with the apparent diffusion coefficient restriction were compatible with acute ischemic strokes (Figure 1(a), (b), (d), and (e)). Fluid-attenuated inversion recovery sequences permitted classification of the lesions as strokes that had occurred less (frontal right lesion) or more (temporal left lesion) than 4.5 h before (Figure 1(c) and (f)). The three-dimensional circle of Willis reconstruction (3D-TOF) excluded also thrombosis or stenosis (Figure 1(g)). A body CT scan found no other embolic lesions and no air in the right or left side of the heart. Cardiac echography with a microbubble test showed no intracardiac right-to-left shunt. During the time that the helicopter transfer was being organized and that the tests were ongoing, her clinical symptoms improved, with no intervention, except for increasing the oxygen to 15 L/min. The hyperlactatemia, clinical presentation, and its improvement after oxygen therapy, associated with the MRI findings, support the hypothesis of a seizure due to multiple cerebral air emboli (CAE) but could also be the normal clinical course of air emboli. Her sensory and motor deficiency also improved to only left arm deficiency, but the left hemineglect syndrome was still present.

Cerebral magnetic resonance imaging. (a and d) DWI showing a hypersignal in the frontal right cortex and temporal left cortex (yellow arrow). (b and e) ADC restriction showing acute ischemic stroke (yellow arrow). (c) FLAIR sequence showing no signal and indicating a stroke that occurred less than 4.5 h before (yellow arrow). (f) FLAIR sequence showing a hypersignal compatible with a stroke more than 4.5 h before (yellow arrow). (g) 3D-TOF showing no thrombosis or stenosis.
The patient was rapidly transferred to a medical reference center for hyperbaric oxygen therapy (HBOT). Three sessions were necessary for complete improvement. The HBOT sessions were performed according to current recommendations. 8 The first session was administered at 2.8 bar but the patient experienced a seizure episode at 2.5 bar attributed to hyperoxia. After changing oxygen to air, the patient completely recovered. She also presented with loss of consciousness the night following the first HBOT session, scoring 3/15 (E1/M1/V1) on the Glasgow Coma Scale, without respiratory distress or shock. She then quickly recovered after stimulation (placement of a gastric tube) and regained her previous level of consciousness. The initial cerebral CT scan showed a frontal hyperdensity compatible with hemorrhage which could explain the coma (Figure 2). However, in front of the rapid recovery of consciousness, the patient could also have presented a subclinical epileptic seizure. Then, she had two additional HBOT sessions 24 h after the first one at 2.4 bar because the hemiplegia persisted. She also experienced two new seizures during these sessions but recovered after the oxygen was changed to air and she received anti-epileptic therapy. An electroencephalogram revealed diffuse cerebral distress without epileptic graphoelements, consistent with metabolic or toxic encephalopathy. A second cerebral CT scan was performed (48 h after the first) and showed right frontal, parietal, and occipital subarachnoid hemorrhage, likely due to strokes in different vascular territories (Figure 2). Her chronic antithrombotic treatment (acetylsalicylic acid at 75 mg per day) was, thus, discontinued. After this episode, the patient was once again evaluated by a neurologist, and the cerebral MRI was assessed by a cerebral radiologist. Doppler ultrasound of the carotids was normal and a 24-h ECG showed no atrial fibrillation. Echocardiography found no valve vegetation or thrombi. Based on the initial clinical presentation and all the investigation findings, we concluded that the patient had a seizure with stroke-like symptoms due to CAE following the hemodialysis session and then had seizures due to hyperoxia during the HBOT sessions. She never experienced another epileptic seizure after the episode of CAE.
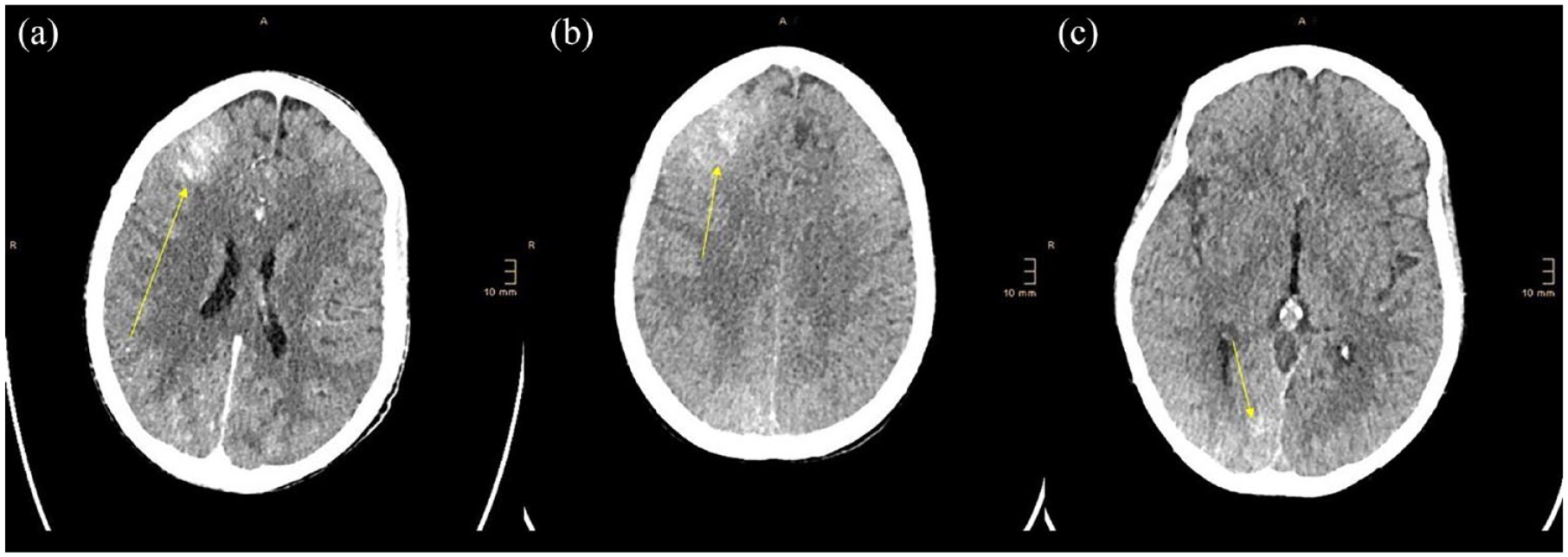
Cerebral CT scans performed after the seizures that occurred during the hyperbaric sessions. (a) First CT scan showing hyperdensity in the right frontal lobe. (b and c) Second CT scan (48 h later) showing hyperdensities in the right frontal (b) and occipital lobes (c) (yellow arrows), suggesting subarachnoid hemorrhage in several vascular territories.
Discussion
Air embolism is a rare but potentially fatal condition.5,6,9 It usually occurs after lung surgery, open cardiac surgery, central venous catheter, and interventional radiology procedures, and, more rarely, after head or neck trauma or surgery. 10 It can also occur following cardiac or pulmonary needle biopsy or diving-related pulmonary barotrauma, during positive pressure ventilation, and sometimes in decompression sickness.7,10 The prognosis of this complication is poor, notably when it is associated with CAE, with 22% mortality, 11 and 50% of patients remaining with long-term neurological sequelae. 12 In this study, we describe the first case of CAE to be reported in a lung transplant patient.
Air embolism is even more uncommon as a complication of hemodialysis in patients treated through a jugular catheter. It was first described in 1969. 13 Usually, this condition is due to leaks or disconnection problems (62% of cases). The mechanism is possibly based on a pressure gradient that facilitates air migration. Such a pressure gradient may be due to inspiration, hypovolemia, or standing up, which changes the hydrostatic pressure in the venous system.14,15 The air usually enters the pulmonary circulation or cerebral venous system by retrograde ascension or arterial circulation, which is known as a paradoxical embolism. 4 Such a paradoxical embolism can be due to a patent foramen ovale, a congenital septal defect, or, in certain cases, pulmonary arteriovenous malformations. 16 The condition of patent foramen ovale is relatively common, and its prevalence has been estimated to be approximately 30% in the healthy population. 17 Pulmonary arteriovenous malformations are rare complications of cardiothoracic surgery that can also be associated with a risk of paradoxical embolism.18–21 A paradoxical embolism can also occur without the presence of an anatomical shunt. In situations of large amounts of air, the air clearance capacity of the pulmonary capillaries is overwhelmed and air passes to the arterial circulation.16,22–24 Hypoxia is also known to increase the risk of the crossover of air to the left cavities of the heart due to pulmonary circulation vasoconstriction.16,25 In our case, it is difficult to differentiate an arterial embolism from a venous embolism through retrograde ascension. It is possible that the patient was in hypovolemia immediately after the dialysis because of ultrafiltration. She stood up, and the deficient obturator allowed the rapid reentry of air when she inhaled. Then, when she lay down, the air that was probably stuck in the right atrium was able to move to the right ventricle and pulmonary circulation. Because the possibility of a persistent foramen ovale was eliminated by cardiac Doppler, two hypotheses can be considered. The first is a paradoxical embolism in a pathological lung, as she had two lung transplants due to cystic fibrosis and chronic graft rejection. The second is venous retrograde air ascension.
The clinical manifestation of air embolism varies depending on the mechanism and location of the air bubbles. The diagnosis of CAE is presumptive and neither cerebral CT nor MRI are recommended, except to exclude other pathologies that could require different therapy. 8 In this case, all the tests were performed during the period we were organizing the transfer of the patient to the HBOT center. Cerebral CT scan is the imaging modality of choice to confirm CAE but is unable to rule out the diagnosis in the absence of intra-cerebrovascular air.26,27 The patient was transferred to the HBOT center 12 h after her initial presentation when she experienced secondary deterioration with neurological symptoms including acute aphasia and hemiplegia. Although the HBOT was delayed, she completely recovered after three sessions. HBOT is the established treatment for CAE and decreases the bubble size and increases tissue oxygenation. 28 Thus, several case studies have shown very good results when HBOT is initiated rapidly, with a direct relationship between the time to HBOT and the probability of a favorable outcome.28,29 A few cases have also shown a response to HBOT more than 24 h after the first symptoms.30–32 In addition, HBOT is always recommended even if the patient shows partial or complete recovery because of the risk of secondary deterioration.
Almost 75% of patients experience a seizure at some point in the first days after sustaining CAE. 33 A small portion is attributable to hyperoxia during HBOT, probably due to the production of reactive oxygen species.34,35 In such cases, seizures occur at a high atmospheric pressure (more than 2.4 bar) and resolve after the discontinuation of hyperoxia and without recurrent seizures after HBOT. In our case, the patient experienced three epileptic seizures, one during each HBOT session. Each of these seizures occurred at an atmospheric pressure of 2.4 bar or higher and resolved after cessation of hyperoxia. The electroencephalogram revealed no epileptic graphoelements, and the patient did not have recurrent seizures after this episode of CAE. The overall presentation is consistent with seizures induced by hyperoxia during HBOT, as described in other case reports.33,35 CAE is also associated with mortality in the acute phase (22% in the review of Pinho et al. 11 ) and with a risk of neurological sequelae, which is reduced by HBOT.27,28
Conclusion
In conclusion, our case report highlights the occurrence of a rare but serious complication of gas embolism in a lung transplant recipient undergoing hemodialysis through a permanent jugular catheter. The patient’s pulmonary status (transplantation and cystic fibrosis) was likely associated with right to left arteriovenous shunting. This case underscores the importance of vigilant monitoring during dialysis procedures, especially for patients with a history of lung transplantation, and the need for prompt recognition and management of gas embolism, specifically HBOT, to prevent potential life-threatening consequences. Further research and awareness among healthcare providers are warranted to better understand the risk factors, pathophysiology, and optimal management strategies for this rare complication. Clinicians should be vigilant in assessing and managing potential risks associated with the dialysis procedure. Early intervention can significantly improve outcomes for these vulnerable patients.
