Abstract
Background:
Due to the rarity of polyneuropathy, organomegaly, endocrinopathy, monoclonal gammopathy, and skin changes (POEMS) syndrome, the best first-line treatment has not been established, although there are several options in guidelines. The preferred treatments vary according to the preference of the physician and anecdote.
Objectives:
First, to analyze the efficacy of a new treatment mode in POEMS syndrome that uses the four-cycle treatment as the induction regimen, followed by sequential transplantation as the consolidation regimen for transplantation-eligible patients, or received another two-cycle treatment for transplantation-ineligible patients. Second, to compare the efficacy and safety of regimens with a proteasome inhibitor (bortezomib–cyclophosphamide–dexamethasone, BCD) or without a proteasome inhibitor (cyclophosphamide–dexamethasone ± thalidomide, CD ± T).
Design:
We conducted a retrospective study using real-world data from Capital Medical University, Xuanwu Hospital.
Methods:
A total of 34 newly diagnosed POEMS syndrome patients met Dispenzieri’s diagnostic criteria, and those who completed at least four cycles of treatment from July 2013 to March 2021 were included.
Results:
The overall vascular endothelial growth factor (VEGF) response rate of this new treatment mode was 100%. The cumulative VEGF complete remission (CRV) rate was 67.9%, and the cumulative complete hematological response (CRH) rate was 55.6%. During the median 49-month follow-up, the 5-year-overall survival (OS) rate was 90.7%, the 3-year-progression-free survival (PFS) rate was 78.4%, and the 5-year-PFS rate was 73.8%. The BCD regimen achieved a 75% CRV rate (median time from diagnosis to CRV = 130 days) and 66.7% CRH rate (median time from diagnosis to CRH = 218 days). In addition, the VEGF response was less than the partial remission (PRV) after four-cycle induction treatment, which, together with a decrease on the Overall Neurological Limitation Scale of less than three points 1 year after consolidation treatment, was an independent poor prognostic factor.
Conclusion:
Bortezomib was well-tolerated by patients with POEMS syndrome. Compared with CD ± T regimen, BCD as the induction regimen achieved better VEGF response and earlier hematological remission. Autologous stem cell transplantation used as consolidation therapy further improved the neurological and hematological remission rates, resulting in better OS and PFS.
Introduction
The polyneuropathy, organomegaly, endocrinopathy, monoclonal gammopathy, and skin changes (POEMS) syndrome is a rare plasma cell proliferative disease that progresses within several months and leads to non-traumatic disability. 1 The incidence of POEMS syndrome is 3 in 1 million, 2 and it predominantly affects those aged 40–50 years.3,4 It is necessary to provide timely and standardized treatment to patients as early as possible. However, because of rarity of POEMS syndrome, potential treatments have mainly been explored in limited clinical trials and case reports.5–10 The best first-line treatment has not been established, although there are several options in guidelines. The preferred treatments vary according to the preference of the physician and anecdote.
The vascular endothelial growth factor (VEGF) level is a good prognostic marker for POEMS syndrome.11,12 Anti-VEGF therapy was suggested in the past for POEMS syndrome but proved inefficient.6,13,14 Currently, POEMS syndrome is treated using plasma-cell-directed therapy.5,15–18 Alkylating agents, immunomodulators, and autologous stem cell transplantation (ASCT) are effective. 1 In particular, ASCT not only achieves VEGF, hematological, and neurological remission, but also improves overall survival (OS) and progression-free survival (PFS).16–18 However, ASCT cannot be performed at the time of diagnosis for most of patients with POEMS syndrome because of the severity of the condition. A low-dose alkylator achieves a similar OS and PFS as ASCT in low-risk patients. 5 ASCT or lenalidomide is recommended for patients with pulmonary hypertension, pleural effusion, and a low estimated glomerular filtration rate. 17 Notably, lenalidomide is effective for patients with POEMS syndrome, but does not provide the same long-term OS or PFS survival benefits as ASCT. 17 Moreover, long-term application of an alkylating agent and lenalidomide may damage hematopoietic stem cells mobilization and affect the subsequent ASCT.19–21
Proteasome inhibitors are included in the first-line combination regimen for multiple myeloma. 22 Peripheral neuropathy is a common side effect of proteasome inhibitors bortezomib. 23 Previously, the concern about exacerbating the peripheral neuropathy induced by the drug, bortezomib has been suggested as second-line therapy for POEMS syndrome. 1 More and more case reports and case series have documented the effectiveness of proteasome inhibitors for treating POEMS syndrome as first-line therapy.24,25 In 2017, a case series reported that reduced-dose bortezomib achieved 100% neurological relief, an 88% VEGF response rate, and a 70% hematological rate. 8 However, that study did not perform a long-term survival analysis due to the short follow-up. And the benefit and risk of proteasome inhibitors bortezomib in POEMS syndrome should be weighed.
This study analyzed the efficacy and safety of a standard dose of the proteasome inhibitor bortezomib, combined with cyclophosphamide and dexamethasone, as the induction regimen. Sequential transplantation was the consolidation regimen for transplantation-eligible patients, while another two-cycle chemotherapy was the consolidation regimen for transplantation-ineligible patients. This study also compared the efficacy and safety of regimens with and without a proteasome inhibitor.
Patient and methods
Patient information
This study retrospectively analyzed patients with POEMS syndrome treated with the bortezomib–cyclophosphamide–dexamethasone (BCD) or cyclophosphamide–dexamethasone ± thalidomide (CD ± T) regimen as first-line therapy at Capital Medical University, Xuanwu Hospital from July 2013 to March 2021. A total of 34 patients met Dispenzieri’s diagnostic criteria, and those who completed at least four cycles of treatment were included. This study was approved by the Institutional Review Board of Capital Medical University, Xuanwu Hospital, and followed the tenets of the Declaration of Helsinki.
Treatment strategy
The BCD regimen from 2013 to 2018 was bortezomib (Velcade®) 1.3 mg/m2, days 1, 4, 8, and 11; cyclophosphamide 0.4 g/m2, days 1/8 or 0.2 g/m2, days 1, 4, 8, and 11; dexamethasone 20 mg, days 1–2, 4–5, 8–9, and 11–12, whereas the BCD regimen since 2019 has been bortezomib 1.3 mg/m2, days 1, 8, 15, and 22; cyclophosphamide 0.4 g/m2, days 1/15 or 0.2 g/m2, days 1, 8, 15, and 22; dexamethasone 40 mg, days 1, 8, 15, and 22. The regimens without a proteasome inhibitor, CD ± T regimen, was cyclophosphamide 0.4 g/m2, days 1/8 or 0.2 g/m2, days 1–4; dexamethasone 20–40 mg, days 1–4 and 9–12; thalidomide 50–100 mg per night. One treatment cycle lasted 28–35 days. Patients who were eligible for transplantation and consented to ASCT achieved at least partial remission after induction treatment followed by ASCT as consolidation treatment. The stem cell mobilization regimen is cyclophosphamide, epirubicin, and granulocyte colony-stimulating factor. Patients who were ineligible for transplantation or refused ASCT after induction treatment underwent another two-cycle therapy as the consolidation regimen. Other therapies included hormone replacement therapy and rehabilitation and supportive therapy. Depending on their willingness, some patients received maintenance therapy after the consolidation treatment.
Response criteria
The response was evaluated every two cycles, 3 months after ASCT, and at the yearly follow-up. If the baseline plasma VEGF level was twice as high as the upper limit of normal, the VEGF response was evaluated. 1 VEGF complete remission (CRV) was defined as a VEGF level back to the normal range. Partial remission (PRV) was defined as a VEGF level that improved by ⩾50% compared to baseline. Stable VEGF (SDV) was defined as a VEGF level that improved or progressed by <50%. VEGF progression (PDV) was defined as a VEGF level that was ⩾50% higher compared with the nadir. Complete hematological response (CRH) was defined as negative serum and urine immunofixation electrophoresis; otherwise, the classification was no hematological response(NRH). 26 Neuropathic remission was defined as a reduction of one point on the Overall Neurological Limitation Scale (ONLS). 27 Neuropathy progression was defined as an increase of one point on the ONLS. 7 OS was defined as the time from the date of diagnosis to the date of death or the end of follow-up. PFS was defined as the time from the date of diagnosis to the date of progression or death event. The last follow-up date was 20 October 2022.
Adverse events
Adverse events (AEs) during chemotherapy were graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events (NCI-CTCAE; version 5.0).
Statistical analysis
SPSS 22.0 software (SPSS Inc., Chicago, IL, USA) was used for the analysis. The two-tailed t test was used to compare two groups. One-way analysis of variance was used to compare more than two groups. The paired t test was used to compare the baseline and post-therapy ONLS scores. Fisher’s exact test was used for analyzing categorical variables. The Kaplan–Meier method was used to generate survival curves and the log-rank test detected differences in survival. For multivariate analysis, Cox proportional hazards model was used to identify independent predictors of PFS. p Values <0.05 were considered significant.
Results
Patient characteristics at diagnosis
A total of 34 patients were included in the study. The baseline characteristics of the patients are displayed in Table 1. The median age at diagnosis was 49.5 (31–75) years, and 24 patients (70.6%) were male. The median time from onset to diagnosis was 11 months. All patients had peripheral neuropathy, as confirmed by electromyography, and 13 patients also confirmed by sural nerve biopsy. The sural nerve biopsy showed axonal loss, demyelination, and epineurial perivascular lymphocytosis. Epineurial and endoneurial edema were also the common pathologic changes. Epineurial neovascularization was relatively more specific. Monoclonal plasma cell-proliferative disorder was detected in all patients and was confirmed by immunofixation electrophoresis in 31 cases, bone marrow flow cytometry in 11 cases which were defined by light chain restricted plasma cells, plasmacytoma of the iliac bone in 1 case who was negative for M protein and monoclonal plasma in bone marrow. All 34 patients had elevated plasma VEGF levels and endocrinopathy. In three patients, Castleman’s disease was confirmed through lymph node biopsy. All four patients with pulmonary hypertension were in the CD ± T group. The pulmonary hypertension was slight in three patients and severe in one. The other clinical features were not significantly different between the BCD and CD ± T groups.
Baseline characteristics of the newly diagnosed POEMS syndrome patients (n = 34).

IVIG, intravenous immunoglobulin; BM, bone marrow; BCD, bortezomib–cyclophosphamide–dexamethasone; CD cyclophosphamide – dexamethasone; CTD, cyclophosphamide – thalidomide – dexamethasone; NS, not statistically significant.
P < 0.05.
Treatment
Thirty-four patients completed at least four cycles of treatment at our institution and were evaluated (Figure 1). In total, 20 patients received BCD, 5 cases received CD, and 9 cases received CTD as the induction regimen. Eleven patients proceeded with sequential transplantation for consolidation, and 22 patients underwent a further two cycles of chemotherapy. One case was not followed up after induction therapy. Two cases in the BCD group received lenalidomide maintenance therapy. In the CTD group, one case received thalidomide, and another received lenalidomide as maintenance therapy.

Flow chart of patient enrolment.
VEGF response
The VEGF response is an important prognostic marker, and an indicator of treatment efficacy, in POEMS syndrome. 12 In total, 16 cases in the BCD group, 3 cases in the CD group and 9 cases in the CTD group were evaluated in terms of the VEGF response. At the end of therapy, the overall cumulative VEGF response rate (CRV + PRV + SDV) was 100% (28/28), and the cumulative CRV rate was 67.9% (19/28). Comparison of the VEGF response was between BCD and CD, as well as BCD and CTD [Figure 2(a)]. After the two-cycle treatment, BCD achieved an 18.8% (3/16) CRV rate and 62.5% (10/16) PRV rate. CD achieved 33.3% (1/3) PRV rate without CR response after the two-cycle treatment. CTD achieved an 22.2% (2/9) CRV rate and 22.2% (2/9) PRV rate. BCD treatment obtained a higher CRV + PRV rate compared with CD treatment (p = 0.1548) or CTD treatment (p = 0.0870), but it is not significant. Comparison of VEGF response was also between regimens with a proteasome inhibitor and that without a proteasome inhibitor (BCD versus CD ± T) [Figure 2(b)]. CD ± T achieved a 16.7% (2/12) CRV rate and 25% (3/12) PRV rate. The CRV + PRV rate with the BCD regimen after the two-cycle treatment was significantly better than that with the CD ± T regimen (p = 0.0497). After the four-cycle treatment, the BCD group, in which only 14 cases were evaluated in terms of the VEGF response, achieved a 85.7% CRV + PRV rate and 71.4% CRV rate. CD achieved 66.7% (2/3) PRV rate without CR response. CTD achieved an 33.3% (3/9) CRV rate and 33.3% (3/9) PRV rate. The BCD treatment obtained a higher CRV rate compared with the CD treatment (p = 0.1023) or the CTD treatment (p = 0.0515), but it is not significant [Figure 2(a)]. The CD ± T group achieved a 66.7% (8/12) CRV + PRV rate and 25% (3/14) CRV rate. The CRV rate with the BCD regimen after the four-cycle treatment was significantly better than that with the CD ± T regimen (p = 0.047). The BCD group achieved a 75% (12/16) CRV rate, CD group achieved a 66.7% (2/3) CRV rate, and CTD achieved a 55.6% (5/9) CRV rate after consolidation therapy. For patients with SDV after four cycles of induction treatment, consolidation therapy did not further improve the VEGF response. The median time to CRV in the BCD group was 130 days, whereas in the CD group it was 188 days and in the CTD group it was 214 days. The majority of patients in the BCD group achieved CRV before ASCT, whereas patients in the CD ± T group often required consolidation therapy to further improve VEGF remission.
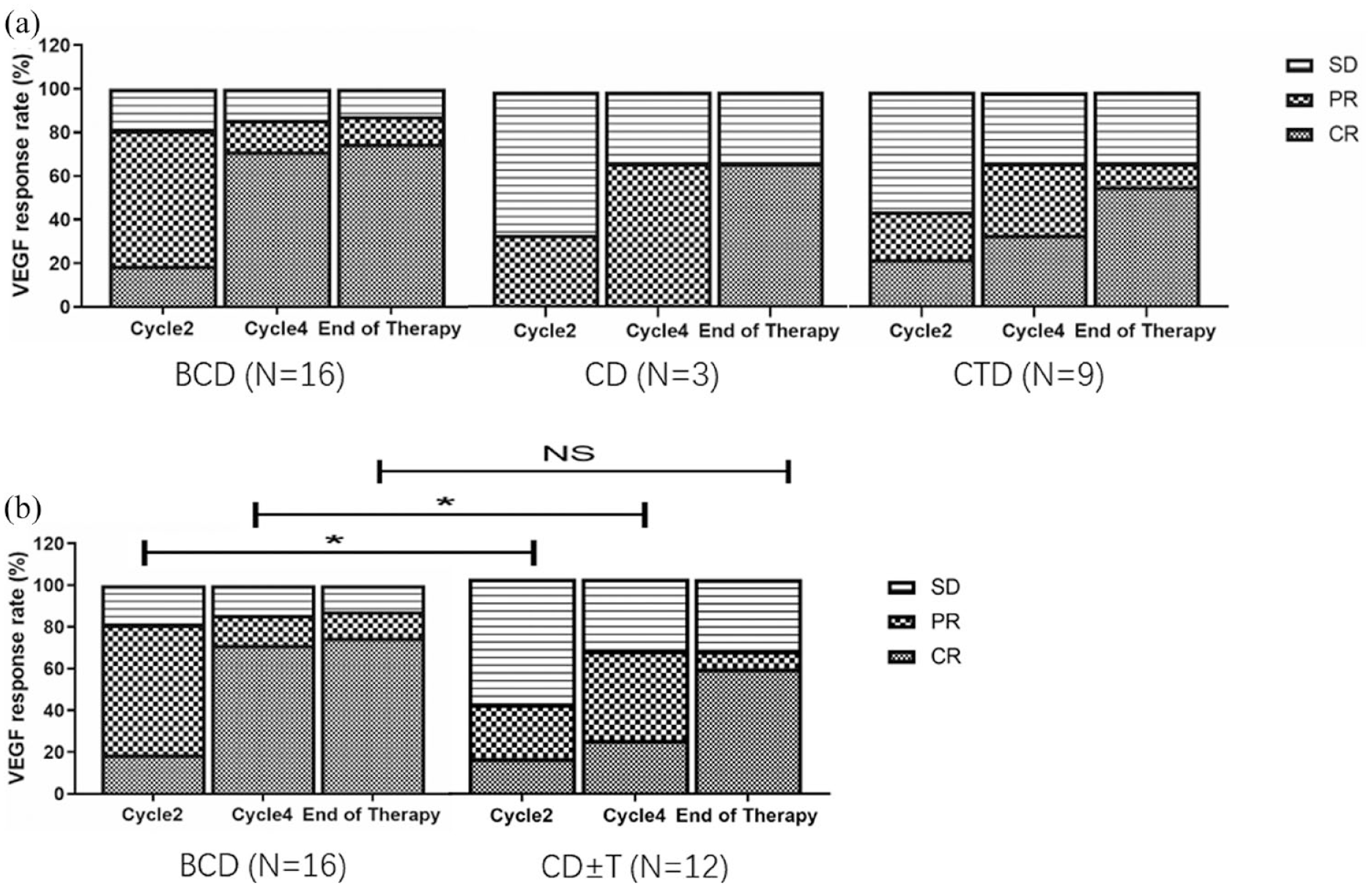
(a) VEGF response of BCD regimen, CD regimen, and CTD regimen. (b) VEGF response of BCD and CD ± T regimens after the second cycle, fourth cycle, and consolidation therapy.
Hematological response
The M protein was evaluated as an indicator of the hematological response. At the end of therapy, the overall cumulative CRH rate was 55.6% (15/27). Comparison of the hematological response was between BCD and CD, as well as BCD and CTD [Figure 3(a)]. The BCD group achieved 13.3% (2/15) CRH rate after two cycles of treatment, 46.2% (6/13) CRH rate after four cycles of treatment, and 66.7% (10/15) CRH rate after consolidation therapy. The median time to CRH in the BCD group was 218 days. The CTD group achieved a 12.5% (1/8) CRH rate after the two-cycle treatment, 12.5% (1/8) CRH rate after the four-cycle treatment, and 50% (4/8) CRH rate after consolidation therapy. Until the end of consolidation therapy, only one case in the CD group achieved CRH. The hematological response in the BCD group is better, but it is not significant [Figure 3(a)]. The CD ± T group achieved a 8.3% (1/12) CRH rate after the two-cycle treatment, 9.1% (1/11) CRH rate after the four-cycle treatment, and 45.7% (5/12) CRH rate after consolidation therapy [Figure 3(b)]. The median time to CRH in the CD ± T group was 455 days. The BCD regimen was associated with earlier hematological remission.

(a) Hematological response of BCD regimen, CD regimen, and CTD regimen. (b) Hematological response of BCD and CD ± T regimen after two-cycle, four-cycle, and consolidation therapies.
Neurological response
The neurological symptoms were evaluated using the ONLS. Thirty-one patients were evaluated by the ONLS and 30 (96.8%) had a neurological response. All the BCD regimen [Figure 4(a)], CD regimen [Figure 4(b)], and CTD regimen [Figure 4(c)] improved the ONLS score (p < 0.05) during therapy. The median neurological response time was 76 days in the BCD group, 64 days in the CD group, and 140 days in the CTD group. Generally, BCD treatment and CD treatment after the two-cycle treatment acquired neurological response, whereas CTD treatment after the four-cycle treatment acquired neurological response. The baseline ONLS score at diagnosis was not significantly different between the BCD group (6.56 ± 2.43) and CD (6.2 ± 2.59), or BCD and CTD group (6.2 ± 2.59). After induction and consolidation therapy, the mean ONLS score in the BCD group (N = 18) decreased from 6.56 ± 2.43 to 2.53 ± 1.91 [Figure 4(d), p < 0.0001]. The mean ONLS score in the CD group (n = 5) decreased from 6.20 ± 2.59 to 2.80 ± 3.03 [Figure 4(d), p < 0.001]. The mean ONLS score in the CTD group (N = 8) decreased from 5.63 ± 2.45 to 2.00 ± 1.31 [Figure 4(d), p < 0.01). No significant difference in neurological response were detected among the BCD regimen, CD regimen, and CTD regimen. The BCD regimen obtained the neurological responses as early as the CD regimen and CTD regimen may delay the neurological responses.

Evaluation of peripheral neuropathy based on the ONLS score. (a) Evaluation of the ONLS score during BCD therapy. (b) Evaluation of the ONLS score during CD therapy. (c) Evaluation of the ONLS score during CTD therapy. (d) Comparison of peripheral neuropathy among the BCD, CD, and CTD group during therapy.
Survival analysis
The 33 patients who completed induction and consolidation therapy were included in the survival analysis. The median follow-up time was 49 (5–114) months. The median OS and PFS have not been reached. The overall 5-year OS was 90.7% [Figure 5(a)], the overall 3-year PFS was 78.4%, and the overall 5-year PFS was 73.8% [Figure 5(b)]. The regimen with a proteasome inhibitor, BCD treatment, obtained a better VEGF response and an earlier hematologic response compared with CD ± T regimen. Therefore, the survival analysis was performed between the BCD group and the CD ± T group. The BCD group achieved a 94.7% 5-year OS and the CD ± T group achieved an 84.6% 5-year OS [Figure 5(c)]. The 3-year PFS and 5-year PFS in the BCD group were both 79.4%, whereas they were 76.9% and 67.3% in the CD ± T group, respectively [Figure 5(d)]. OS and PFS were not significantly different between the BCD and CD ± T groups.

Overall OS (a) and PFS (b) in POEMS syndrome patients. OS (c) and PFS (d) in the BCD induction and CD ± T induction groups. (e) The PFS of the CRV + PRV and SDV patients after four-cycle therapy. (f) PFS of the CRH and NRH patients after consolidation therapy. (g) PFS according to the decrease in ONLS score 1 year after consolidation therapy.
A previous study reported that the VEGF response in patients with POEMS syndrome is independent of the prognostic value. 12 Therefore, the PFS rate was assessed based on the VEGF response after therapy [Figure 5(e)]. The result indicated that the PFS rate improved more in CRV + PRV patients (median PFS not reached) after four-cycle induction therapy than that in SDV patients (median PFS = 10 months, p < 0.01).
The PFS rate was also assessed based on the hematological response after consolidation therapy [Figure 5(f)]. The median PFS has not been reached in CRH patients and the median PFS in NRH patients was 93 months (p = 0.052).
Polyneuropathy is a major feature in patients with POEMS syndrome. We assessed PFS rates based on the neurological response [Figure 5(g)]. The PFS rate improved more in patients with a decrease on the ONLS score ⩾3 at 1 year after consolidation therapy (median PFS not reached) compared with those decrease a score of 0–2 (median PFS = 24 months, p < 0.001).
Effect of ASCT on efficacy and survival
The VEGF, hematological, and neurological responses were compared in the context of consolidation therapy. There were 11 cases in the ASCT group and 22 in the non-ASCT group. Regarding the VEGF response [Figure 6(a)], there were 9 cases in the ASCT group and 19 in the non-ASCT group evaluated. The CRV rate increased in the ASCT group from 88.9% (8/9) before consolidation to 100% (9/9) thereafter. The CRV rate increased in the non-ASCT group from 26.3% (5/19) before consolidation to 52.6% (10/9) thereafter. There were 8 cases in the ASCT group and 19 in the non-ASCT group evaluable for the hematological response [Figure 6(b)]. The CRH rate in the ASCT group increased from 50.0% (4/8) before consolidation to 87.5% (7/8) thereafter. The CRH rate in the non-ASCT group increased from 15.8% (3/19) before consolidation to 42.1% (8/9) thereafter. ASCT consolidation therapy improved the CRH rate [p = 0.043, Figure 6(b)].

(a) Comparison of the VEGF response between the ASCT consolidation and chemotherapy consolidation groups after the four-cycle treatment and consolidation therapy. (b) Comparison of the hematological response between the ASCT consolidation and chemotherapy consolidation groups after four-cycle treatment and consolidation therapy. (c) Comparison of peripheral neuropathy between the ASCT consolidation and chemotherapy consolidation groups during therapy. OS (d) and PFS (e) in the ASCT consolidation and non-ASCT consolidation groups.
The baseline ONLS score was not significantly different between the ASCT group (n = 10, 5.3 ± 1.89) and non-ASCT group (n = 21, 6.65 ± 2.58). After consolidation therapy, the mean ONLS score in the ASCT group decreased from 5.3 ± 1.89 to 1.30 ± 0.95 (p < 0.0001). The mean ONLS score in the non-ASCT group decreased from 6.65 ± 2.58 to 2.00 ± 2.08 (p < 0.0001). The ASCT group achieved a better ONLS score than the non-ASCT group after consolidation therapy (p < 0.05) [Figure 6(c)].
The ASCT group achieved a 100% 5-year OS rate and 88.9% 5-year PFS rate. The non-ASCT group achieved a 86.1% 5-year OS rate and 67.9% 5-year PFS rate. Due to the limited sample size, it cannot be concluded that OS [Figure 6(d), p > 0.05] and PFS were better [Figure 6(e), p > 0.05] in the ASCT than non-ASCT group. Only one case of relapse occurred in the ASCT group during year 4 after first-line therapy, who had not received bortezomib as induction treatment.
Cox proportional hazards model of PFS
The Cox proportional hazards model indicated that a reduction of the ONLS score <3 at 1 year after the consolidation treatment, and a VEGF response below the PRV after the four-cycle induction treatment, were independent predictors of a poor prognosis. The hematological response did not affect PFS (Table 2).
Multivariate (Cox regression) analysis of PFS in POEMS syndrome patients.

Indicate statistical significance. Reduction in ONLS score = best ONLS score − ONLS score at diagnosis.
CI, confidence interval; CRV, vascular endothelial growth factor complete remission; HR, hazard ratio; ONLS, Overall Neurological Limitation Scale; PFS, progression-free survival; PRV, VEGF partial remission; SDV, stable VEGF.
Safety
Table 3 lists the non-neurological toxicities during chemotherapy, including hepatic toxicity (n = 14), hematological toxicity (n = 7), and infection (n = 4) (Table 3). All AEs were grades 1–2. No significant difference in AEs was detected between the BCD and CD ± T groups. Most side effects diminished after chemotherapy. Some side effects required supportive therapy and then returned to normal. None of the patients discontinued therapy because of drug-related side effects.
Adverse events during chemotherapy.

ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BCD, bortezomib–cyclophosphamide–dexamethasone; GGT, gamma-glutamyltransferase.
Discussion
ASCT is effective for VEGF and hematological remission, as well as for improving the OS and PFS for patients with POEMS syndrome.16–18 In most cases, ASCT cannot be performed at the time of diagnosis due to the severity of the condition, where patients may be suffering from severe peripheral neuropathy, thrombosis, pleural effusion, or pulmonary hypertension. In addition, a previous report showed that ASCT for POEMS syndrome was associated with high rates of engraftment syndrome. 28 Therefore, our institution has proposed using the four-cycle treatment as the induction regimen, followed by sequential transplantation as the consolidation regimen, for transplantation-eligible patients, whereas another two-cycle treatment can be used as the consolidation regimen for transplantation-ineligible patients. Some ineligible patients may become transplantation candidates after induction therapy, which would alleviate disease and improve neurological symptoms. Moreover, the use of cyclophosphamide as therapy before ASCT can reduce the likelihood of engraftment syndrome.28,29 The overall VEGF response rate of this regimen was 100%, whereas the cumulative CRV rate was 67.9% and the cumulative CRH rate was 55.6%, consistent with previous reports of the CRV (66.2%) and CRH (49.7%) rates of ASCT for POEMS syndrome. Large retrospective studies on ASCT for POEMS syndrome from Europe 16 and Japan 18 reported 5-year OS, 3-year PFS, and 5-year PFS rates for POEMS syndrome of 89%, 78.3–84%, and 64.8–75%, respectively. The regimen proposed at our institution achieved 90.7% 5-year OS, 78.4% 3-year PFS, and 73.8% 5-year PFS during the median 49-month follow-up. The efficacy and survival benefit of this regimen are not inferior to ASCT. Moreover, no engraftment syndrome has been reported during ASCT for POEMS syndrome at our institution.
A low-dose alkylator with steroids has been the mainstay of treatment for patients with POEMS syndrome unsuitable for transplantation.1,5 Proteasome inhibitors are included in the standard first-line treatment regimen for plasma cell proliferative disorder, 30 which eradicates abnormal plasma cells and inhibits VEGF secretion. 31 As a result, our institution proposed cyclophosphamide, dexamethasone, and a standard dose of the proteasome inhibitor bortezomib as the induction regimen. Compared with previous studies,5,16–18 the CRV rate for the BCD regimen (75%) was not inferior to that of ASCT (66.2%) and was superior to those of melphalan + dexamethasone (MDex, 35.8%) and lenalidomide + dexamethasone (LDex, 47.7%). 17 The cumulative CRH rate for the BCD regimen (66.7%) was superior to those of ASCT (49.7%), MDex (37.7%), and LDex (47.5%). In addition, the VEGF remission rate of the BCD regimen was better than that of the CD ± T regimen. The BCD regimen often achieved CRV after four-cycle induction chemotherapy (after a median of 130 days), whereas the CD ± T regimen achieved CRV following consolidation therapy. Moreover, the median time to achieve CRH was shorter for the BCD than CD ± T regimen. The BCD regimen achieved earlier remission, which may also benefit for a deeper remission. A recent report documented the efficacy of BD for POEMS syndrome. 26 The CRV was 43.9% and the CRH was 46.4%. 26 Therefore, it was concluded that the efficacy of the BCD regimen is not only superior to that of the CD regimen but also to that of the BD regimen. The combination of bortezomib and cyclophosphamide had a synergistic effect.
ASCT as consolidation therapy after induction chemotherapy achieved a 100% CRV rate and 87.5% CRH rate and was thus superior to chemotherapy as consolidation therapy. Transplantation as consolidation therapy achieved a 100% 5-year OS rate and 88.9% 5-year-PFS rate and was thus superior to previously reported POEMS treatments. Only one case of relapse was detected in the ASCT group, during year 4 after first-line therapy. The patient had not received bortezomib as induction treatment. It may be advisable to use BCD as induction therapy and ASCT as consolidation therapy.
Notably, if patients fail to achieve a CRV or PRV after four-cycle induction therapy, efficacy does not improve if another two cycles of consolidation therapy are performed. Our Cox regression analysis revealed that the VEGF response was worse than the PRV after the four-cycle treatment, which was an independent poor prognostic factor of PFS. Therefore, an efficacy evaluation should be conducted after four-cycle therapy. If the VEGF response is worse than the PRV, ASCT or second-line regimen should be suggested. A reduction of the ONLS score of <3 at 1 year after consolidation therapy also predicted a poor PFS. Close follow-up is suggested for patients exhibiting a slow recovery with respect to neurological symptoms.
Peripheral neuropathy is a concern when using proteasome inhibitors. 23 Their use has been recommended for second-line treatment of patients with POEMS syndrome in the past. 1 However, bortezomib was well-tolerated by our patients with POEMS syndrome, even by those who were confined to a wheelchair at diagnosis. After therapy and rehabilitation, their motor function improved gradually, and some could walk without assistance. Both bortezomib and thalidomide have possible neurologic toxicity. The BCD regimen that added bortezomib in the CD regimen obtained neurological response after the two-cycle treatment as early as the CD regimen. However, the CTD regimen that added thalidomide in the CD regimen may delay the neurological response. The neurological symptoms of one case did not improve during therapy. In that case, the high VEGF level and M protein positivity were not resolved during therapy, indicating non-remission of the disease. The use of bortezomib did not aggravate the neurological symptoms in patients with POEMS syndrome. Effective treatment with bortezomib helped to obtain earlier VEGF and hematological remission and improved the neurological symptoms. In addition, ASCT is recommended as consolidation therapy, which improves the neurological response further.
Summary
Bortezomib was well-tolerated by patients with POEMS syndrome. Standard-dose BCD as the induction regimen achieved better VEGF and earlier hematological remission. ASCT used as consolidation therapy further improved the neurological and hematological remission rates, resulting in better OS and PFS. Transplantation after induction chemotherapy reduced the likelihood of engraftment syndrome.
