Abstract
Objective
This study was performed to examine the possible association of iron overload with infectious complications and survival among liver transplant recipients.
Methods
We conducted a systematic review and meta-analysis of studies published in the PubMed, Embase, Web of Science, and Cochrane Library databases up to September 2022. Hazard ratios (HRs) and 95% confidence intervals (CIs) were extracted to estimate the association of iron overload with infectious outcomes and overall survival after liver transplantation.
Results
Eight studies involving 2817 recipients met the inclusion criteria. Iron overload was strongly associated with an increased risk of infection after liver transplantation (HR, 1.66; 95% CI, 1.03–2.68). An increase in the serum ferritin level was associated with an increased risk of infection after liver transplantation (HR, 1.44; 95% CI, 1.09–1.91). Iron overload was a significant predictor of worse overall survival (HR, 1.35; 95% CI, 1.11–1.64). In addition, a high serum ferritin level was significantly associated with an increased risk of death (HR, 1.34; 95% CI, 1.10–1.64).
Conclusion
Iron overload may be associated with a higher risk of infectious complications and a worse prognosis among liver transplant recipients.
Introduction
Liver transplantation (LT) is often a life-saving treatment for the increasing number of patients with end-stage liver disease. Because of improvements in surgical techniques, immunosuppression strategies, and post-LT management of complications, the mortality rate associated with LT has steadily declined. In Western countries, the survival rates after LT are 83% at 1 year and 71% at 5 years. 1 However, the use of immunosuppressants increases the risk of postoperative infection, potentially resulting in severe morbidity. Infection caused by bacteria, fungi, and viruses remains one of the leading causes of morbidity and mortality among transplant recipients worldwide.1–3 Knowledge of modifiable risk factors and their potentially reversible causes is essential to develop targeted preventive strategies.
The Model for End-stage Liver Disease (MELD) was introduced in 2002 and has since become the most widely used method of organ allocation in LT. 4 Additional factors have also been found to be associated with LT-related morbidity and mortality, such as the serum sodium level, 5 renal function, 6 sarcopenia, 7 and the ferritin level. 8 Iron is a critical resource for microorganisms, but it can also increase the virulence of microbes and impair the host immune response.9,10 Ferritin is the major iron storage protein, and it reflects the cumulative iron stores in the bone marrow and tissues. 11 An increase in the serum ferritin level can occur in the context of many hepatic diseases, such as chronic hepatitis C, treatment-naive autoimmune hepatitis, and nonalcoholic steatohepatitis.12,13 However, ferritin is also an acute-phase reactant and is influenced by systemic inflammation. 14 Excess body iron is present in the liver and is very closely correlated with the total body iron stores.15,16 Histologically, iron is not visible in healthy liver tissue. Iron staining of liver biopsy specimens can be used to assess the hepatic iron concentration and has been linked to the severity of liver diseases.17,18 Iron homeostasis plays a critical role in cellular physiology. From an immunological point of view, iron accumulation in macrophages renders them more conducive to supporting the survival and colonization of viruses with other pathogenic microorganisms. 19
Although previous clinical studies have linked iron overload to an increased risk of bacterial infection and mortality after LT,8,20–26 the evidence supporting this hypothesis remains unclear. We therefore performed a meta-analysis of systematic reviews focused on the potential impact of iron metabolism imbalance on patient outcomes.
Materials and methods
We performed this meta-analysis in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. 27
Data sources and search
Two investigators independently screened the abstracts and reviewed the full texts of studies to determine their eligibility. We searched the PubMed, Embase, Cochrane Library, and Web of Science databases from inception to 30 September 2022 using the following terms: “liver transplantation,” “liver grafting,” “hepatic transplantation,” and “iron.” Relevant systematic reviews and cited references were also searched manually. The search was limited to studies published in English. After the initial search, duplicate articles were removed. The titles and abstracts were then screened to progressively exclude nonrelevant studies. The final list of included studies was determined through consensus, and any disagreements were resolved by consulting a third assessor.
Selection criteria
We developed the following inclusion criteria to select studies in accordance with the PICOS framework (population, intervention, comparator, outcomes, and study design).
Studies were included in the systematic review if they met the following criteria: (1) the study investigated the infection and mortality rates after LT; (2) the study evaluated the predictive ability of one or more iron-related markers in blood or hepatic explants (serum ferritin level, stainable hepatic iron, and hepatic iron index) for postoperative infection and mortality in liver transplant recipients; (3) definite infection was defined as laboratory isolation of an organism from a normally sterile site combined with symptoms of infection; and (4) possible infection was defined as the presence of signs and symptoms of infection warranting empirical treatment with antibiotics in the absence of confirmatory evidence.
The exclusion criteria were (1) study abstracts, conference abstracts, editorials, review articles, letters, animal studies, and case studies and (2) studies reporting incomplete or insufficient data.
Data extraction and management
Two reviewers independently extracted the following data into a prepiloted Microsoft Excel-based data extraction form: study characteristics (study design, study period, location, sample size, measures of iron load, and outcome measures) and participant characteristics (number of participants, etiology of liver disease, and pre-LT MELD score). We were unable to quantify differences by race/ethnicity because these data were not available. Next, we extracted the hazard ratio (HR) and 95% confidence interval (CI) associated with the iron load and outcome (infection or death). When critical data were missing or unclear in the published reports, we attempted to contact the authors. Any disagreements in the data extraction process were resolved by discussion.
Quality assessment
Two authors independently assessed the methodological quality of the included studies using the Newcastle–Ottawa Scale (NOS). 28 The NOS is a 9-point scale that consists of three domains: selection, comparability, and outcome assessment. Studies scoring 7 to 9 points were considered to be of high quality, those scoring 4 to 6 points were of moderate quality, and those scoring 0 to 3 points were of low quality.
Statistical analysis
The primary outcome measures were the HRs for infection events and OS. These data were derived from the reported HRs, together with the 95% CIs from the original studies. If the HR and 95% CI were not reported, they were derived indirectly from Kaplan–Meier curves using the methods described by Tierney et al. 29 Pooled HR estimates were generated according to the categories noted above. In the absence of heterogeneity among individual studies, we used a fixed-effects model to pool the data; otherwise, a random-effects model was applied. Heterogeneity among studies was estimated by Cochran’s Q test and the I2 statistic (I2 > 50% indicated substantial heterogeneity). Publication bias was assessed using funnel plots. We also performed sensitivity analyses to examine the robustness of the results. All statistical analyses were performed using RevMan version 5.3 (Cochrane Collaboration).
Results
Identification of studies and study characteristics
The PRISMA flow chart shown in Figure 1 summarizes the literature search and study selection processes. The literature search initially yielded 2819 articles. After removing duplicates, 1018 articles remained. After screening the titles and abstracts, 1005 nonrelevant records were excluded, leaving 14 records for full-text review. Ultimately, eight studies involving 2817 patients met the inclusion criteria for the current systematic review. The overall quality of the eight studies was moderate, and the NOS scores ranged from 7 to 8.

PRISMA diagram showing identification of eligible studies and reasons for exclusion.
All eight studies8,20–26 investigated the relationship of iron overload with infection and survival of patients after LT. A summary of the patients’ baseline information is presented in Table 1. Three studies20,21,23 estimated the correlations between iron overload markers (serum ferritin and stainable iron in the hepatic explant) and susceptibility to infection (cytomegalovirus (CMV) disease, invasive fungal infection, bacteremia) after LT. Mortality outcomes were described in six studies,8,20,22,24–26 which reported the OS HR for serum ferritin and stainable iron in the hepatic explant.
Baseline characteristics of included studies.

RT, retrospective; MELD, Model for End-stage Liver Disease; ALD, alcoholic liver disease; PBC, primary biliary cirrhosis; PSC, primary sclerosing cholangitis; HCC, hepatocellular carcinoma; NASH, non-alcoholic steatohepatitis; AH, autoimmune hepatitis; CLD, cholestatic liver disease; LT, liver transplantation; OLT, orthotopic liver transplantation; CMV, cytomegalovirus; OS, overall survival; NR, not reported
Iron overload and infectious complications in LT
The infectious events after LT are summarized in Table 1. Three studies20,21,23 involving 390 patients examined the relationship between the serum ferritin level, stainable iron in the hepatic explant, and infectious complications after LT. Among these studies, the definition of infectious complications (intra-abdominal infection, bloodstream infection, skin and soft tissue infection, urinary tract infection, CMV disease, and invasive fungal infection) included the time to first infection being at least 1 week after LT. 21
Overall, 103 (26.4%) of these patients had at least one infection event, including bacteremia, CMV disease, and invasive fungal infection. Iron overload was strongly associated with an increased risk of infection after LT (HR, 1.66; 95% CI, 1.03–2.68; p = 0.04) (Figure 2(a)). We analyzed the results using a fixed-effects model because the estimated degree of heterogeneity between studies was 50%. The subgroup analyses showed that an increase in the serum ferritin level was independently associated with an increased risk of infection after LT (HR, 1.44; 95% CI, 1.09–1.91; p = 0.01), with no heterogeneity (I2 = 0%,) (Figure 2(b)).

Forest plot for association between infectious complications after liver transplantation and iron overload. Subgroup analysis was performed according to iron storage indicators. (a) Increased serum ferritin level after liver transplantation and iron stainable by Perls Prussian blue stain in the explanted livers and (b) increased serum ferritin level after liver transplantation.
Iron overload and survival in LT
In total, six articles involving 2536 participants examined the relationship between death and iron markers. Of these, five studies involving 2208 patients estimated the association between the preoperative22,25,26 or postoperative 20 serum ferritin level and OS. One study 24 assessed the association between the hepatic iron index of hepatic explants and OS. We first examined the overall effect of perioperative stored iron markers on survival using a fixed-effects model. The pooled HRs showed that high levels of stored iron markers were significantly correlated with a poor prognosis (HR, 1.35; 95% CI, 1.11–1.64; p = 0.003), and heterogeneity was low (I2 = 34%) (Figure 3(a)).

Forest plots for the association between overall survival and iron overload. Subgroup analysis was performed according to iron storage indicators. (a) Serum ferritin level and iron stainable by Perls Prussian blue stain in the explanted livers. (b) Serum ferritin level and (c) serum ferritin level before liver transplantation.
A previous study showed that the serum ferritin level can indicate the degree of ischemia/reperfusion injury in LT. 30 We performed a subgroup analysis using a fixed-effects model to determine whether the serum ferritin level can be evaluated as a noninvasive prognostic indicator on its own. Three studies assessed the serum ferritin level during the evaluation of patients undergoing LT.22,25,26 In another study, the serum ferritin level was examined within 72 hours after LT. 20
The pooled results revealed that a high preoperative or postoperative serum ferritin level was significantly associated with an increased risk of death (HR, 1.34; 95% CI, 1.10–1.64; p = 0.004), and heterogeneity was low (I2 = 46%) (Figure 3(b)). Pooled analysis using a random-effects model showed that higher preoperative serum ferritin levels were also associated with poor survival (HR, 1.54; 95% CI, 1.06–2.25; p = 0.003), with moderate heterogeneity (I2 = 53%) (Figure 3 (c)).
Publication bias and sensitivity analyses
We tested for publication bias by assessing the symmetry of the funnel plots, and no substantial publication bias was found (Figure 4(a–e)). Sensitivity analyses were conducted to test the robustness of the overall estimated results. The removal of individual studies one at a time did not appreciably change the results.
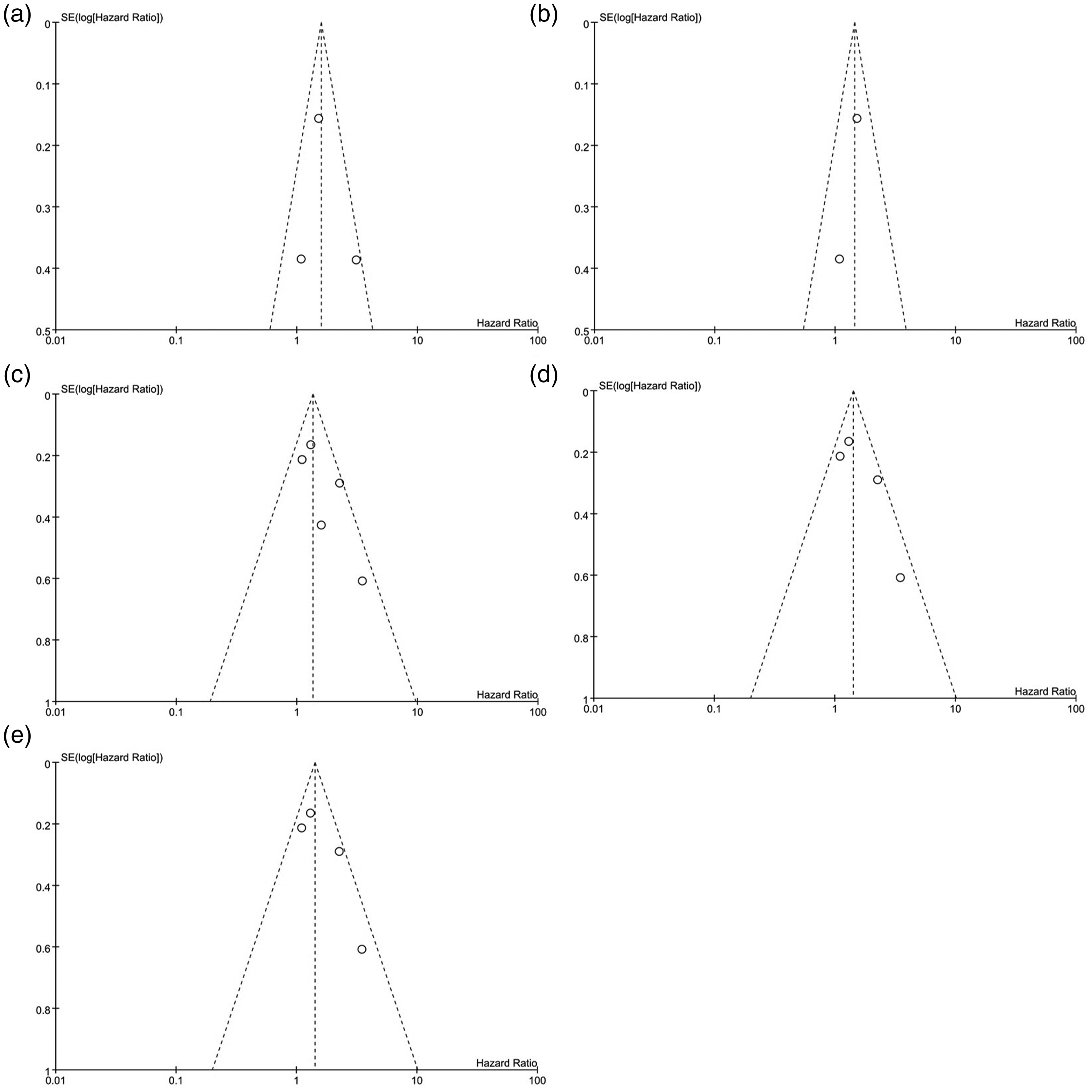
Funnel plots for publication bias. (a, b) Infectious complications and (c–e) Mortality.
Discussion
This systematic review provides several lines of evidence indicating significantly increased risks of infection and death after LT in patients with a high serum ferritin level or hepatic explant iron overload. We included eight studies8,20–26 involving 2817 patients who had undergone LT. Six8,20–22,25,26 of these studies examined the serum ferritin level in LT patients, four8,22,25,26 studies evaluated the serum ferritin level before LT, and two20,21 studies evaluated the serum ferritin level postoperatively. Different pathogen profiles were reported among three studies,20,21,23 including bacteria, fungi, and viruses. Two studies showed that an increased serum ferritin level is an independent risk factor for any infection after LT.20,21 In addition, Alexander et al. 23 found a significant correlation between stainable hepatic iron in explants and invasive fungal infection after LT. Another important finding of the present study is that LT patients with iron overload had a higher risk of death than those without iron overload. The subgroup analysis of serum ferritin as a predictor showed that LT patients with a high serum ferritin level during the perioperative period have poor outcomes.
Liver transplant recipients are on long-term immunosuppressive regimens and are at particular risk of postoperative infection. 3 Infectious complications remain a leading cause of mortality after LT. The latest European surveys from 1968 to 2017 revealed that 4.1% of children who underwent LT died of infection. 31 The most common types infection after LT are bacterial infection (accounting for approximately 70% of all infections), fungal infection, and viral infection. 32 We identified the MELD score as a key consideration for operative urgency in LT. Nevertheless, a previous meta-analysis revealed several risk factors for all-cause infection and mortality that are not directly reflected by the MELD score, such as biliary complications, ascites, atelectasis, and the bilirubin level.33,34 Furthermore, patients with hepatocellular carcinoma undergo LT at a rate that is not restricted by the MELD score. 35 This finding suggests that there may still be unidentified causal risk factors driving these associations.
Iron is the most abundant transition metal ion in biological systems and is an essential nutrient for nearly all organisms and cells. 36 The iron distribution is intimately linked to host defense against pathogens. 37 The main indicators of the iron distribution are the serum ferritin level, hemoglobin level, total iron banding capacity, and transferrin saturation level. 38 The liver iron concentration is also a reliable indicator of total body iron, but its measurement is invasive. 39 Previous meta-analyses showed that that a long operation time, incompatible blood type, operative blood loss, and biliary complications are risk factors for bloodstream infection after LT. 34 Recent evidence has suggested that the serum ferritin level is often increased in patients with liver disease. 13 One study revealed that iron overload triggers the development of nonalcoholic steatohepatitis by interrupting the liver redox balance. 40 Patients with nonalcoholic steatohepatitis are more likely than patients without nonalcoholic steatohepatitis to die of sepsis after LT. 41 Kowdley et al. 42 found that hepatic iron overload is associated with poor clinical survival after LT. A similar finding was reported in patients who underwent hematopoietic stem cell transplantation. 43 In addition, excess iron promotes the rapid growth of bacteria in blood and may enhance susceptibility to sepsis. 44 The invading pathogen must obtain iron from the host, but host nutritional immunity maintains a limited amount of free iron, thereby interrupting iron acquisition by pathogens. 45 However, this relationship remains controversial. Stuart et al. 46 reported that the hepatic iron concentration is not the most important determinant of the outcome after LT.
In the present study, we evaluated the effects of host iron on the clinical outcomes of LT and found that iron overload is a risk factor for infectious complications and poor survival in transplant recipients. However, our study had several limitations. First, all the included studies were retrospective in design, limiting our ability to perform a subgroup analysis to explore potential sources of heterogeneity. Second, the endpoint definitions, study populations, follow-up intervals, and measures of iron storage varied among the studies, thereby increasing heterogeneity. Third, the precise biological significance of the serum ferritin level in LT recipients, including its relationship to iron overload and inflammation, remains unclear. Further research is needed to investigate the role of iron metabolism in the pathogenesis of infection in LT recipients. Finally, the number of included studies was small, and we were unable to evaluate the difference in predictive ability between serum tests and iron staining of grafts through meta-regression.
Conclusion
The current meta-analysis demonstrated the usefulness of serum ferritin measurement and iron staining in the grafted liver for evaluating the probability of infection and predicting mortality in patients undergoing LT. Additional research is necessary to determine whether this new strategy can further inform targeted intervention strategies to reduce the risk of infection and death in liver transplant recipients. Furthermore, future research should examine the risks of adverse events associated with iron overload in LT.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241232920 - Supplemental material for Association of iron overload with infectious complications in liver transplant recipients: a systematic review and meta-analysis
Supplemental material, sj-pdf-1-imr-10.1177_03000605241232920 for Association of iron overload with infectious complications in liver transplant recipients: a systematic review and meta-analysis by Jingpo Zhang, Bingzheng Yan and Xin Shi in Journal of International Medical Research
Footnotes
Author contributions
Jingpo Zhang and Xin Shi designed the study. Jingpo Zhang and Bingzheng Yan performed the literature search, study selection, data extraction, and statistical analyses. Jingpo Zhang and Xin Shi wrote the first draft of the manuscript. All authors contributed to the interpretation of the data and critically reviewed the manuscript.
Data availability statement
All data used in this study are freely available by contacting the corresponding author.
Declaration of conflicting interest
The authors disclose no potential conflicts of interest. All authors have agreed to publish this manuscript.
Ethics statement
This study was approved by The Ethics Committee of the First Hospital of Hebei Medical University.
Funding
The authors received no specific funding for this work.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
