Abstract
Carfilzomib, a selective proteasome inhibitor, is approved for use in relapsed and refractory multiple myeloma. Its link to left ventricular dysfunction is well established but little is known about its effects on the right ventricle. One of its rare complications is pulmonary hypertension, which at its extreme may result in right ventricular dysfunction. Here, we present a case of an elderly male veteran with multiple myeloma status post various failed therapies who developed acute dyspnea after four cycles of carfilzomib and subsequently found to have severe pulmonary hypertension with resultant acute right ventricular failure, which recovered after cessation of carfilzomib. This case highlights the need for careful cardiovascular surveillance while on carfilzomib and the importance of knowing even its rarest complications as these cardiotoxicities are reversible with discontinuation of the drug.
Introduction
Carfilzomib is a selective and irreversible proteasome inhibitor (PI) approved for treatment of relapsed and refractory multiple myeloma. 1 Aggregated safety results from four phase II trials, 2 including a majority of patients with underlying cardiac disease, subsequently found 7.2% of them developed cardiac failure but there were no significant differences in overall mortality between those with and without pre-existing cardiac disorders. 2 Similarly, a small case series from a single center of 67 patients treated with carfilzomib found that most patients who experienced cardiac or vascular-related adverse events subsequent to carfilzomib also had a history of the specific cardiovascular (CV) event they exhibited, reflecting experience in a real-world patient population. 3 The ASPIRE trial compared carfilzomib plus standard therapy (lenalidomide and dexamethasone) to standard therapy alone and showed carfilzomib group to have higher any-grade dyspnea (19.4% vs 14.9%), hypertension (14.3% vs 6.9%), heart failure (6.4% vs 4.1%) and ischemic heart disease (5.9% vs 4.6%). 4 There is little debate regarding the increased CV events associated with carfilzomib, in particular, when it relates to left ventricular function. However, there is scant data on carfilzomib-induced pulmonary hypertension (PH) in the real-world myeloma population and the risks for developing right ventricular (RV) dysfunction. It is imperative for clinicians to be aware and, therefore, be able to identify these complications as these CV sequalae may be reversible with cessation of the medication. Our aim is to report our experience with an interesting real-world case of carfilzomib-induced acute PH with associated RV failure and highlight the need for routine surveillance with echocardiography for early detection of these CV complications.
Case presentation
A 71-year-old veteran male with IgG lambda multiple myeloma, status post failed autologous stem cell transplant with melphalan as well as numerous lines of chemotherapy including bortezomib, presented with acute dyspnea 2 months after he was started on carfilzomib/bendamustine/dexamethasone combination therapy. His other comorbid conditions included systemic hypertension that is treated with carvedilol and amlodipine, remote history of pulmonary embolism now on apixaban, obstructive sleep apnea while compliant with continuous positive airway pressure use and obesity. He had normal pulmonary function test 2 years prior to this index episode. The patient also had a baseline transthoracic echocardiogram (TTE) just before starting carfilzomib revealing normal biventricular and valvular function without evidence of PH, but there was trace circumferential pericardial effusion that’s been chronic for at least 2 years prior. On starting carfilzomib, the patient started noticing worsening dyspnea on exertion until 2 months later when he suddenly developed shortness of breath while ambulating immediately after completing cycle 4 of carfilzomib infusion. He was transferred urgently to the emergency room at which time the vitals were within normal limits except for exertional oxygen saturation of 88% while on room air. The exam was significant for an obese male with mild respiratory distress and mild bilateral lower extremity edema. Cardiac troponins were within normal limits, but brain natriuretic peptide was mildly elevated (148 μg/dL). Electrocardiogram revealed new T-wave inversions in leads V2–V3 (Figure 1). Computed tomography pulmonary angiogram study with contrast demonstrated normal lung parenchyma without evidence of pulmonary embolism or infiltrates. Repeat TTE, however, showed newly dilated RV with significantly reduced RV systolic function (Figure 2) with tricuspid annular plane systolic excursion (TAPSE) of 1.3 cm and an estimated pulmonary arterial systolic pressure (PASP) of 56–61 mmHg (Figure 3).

Electrocardiogram: new nonspecific T-wave changes in V2–V3 when patient became acutely dyspneic after carfilzomib infusion.
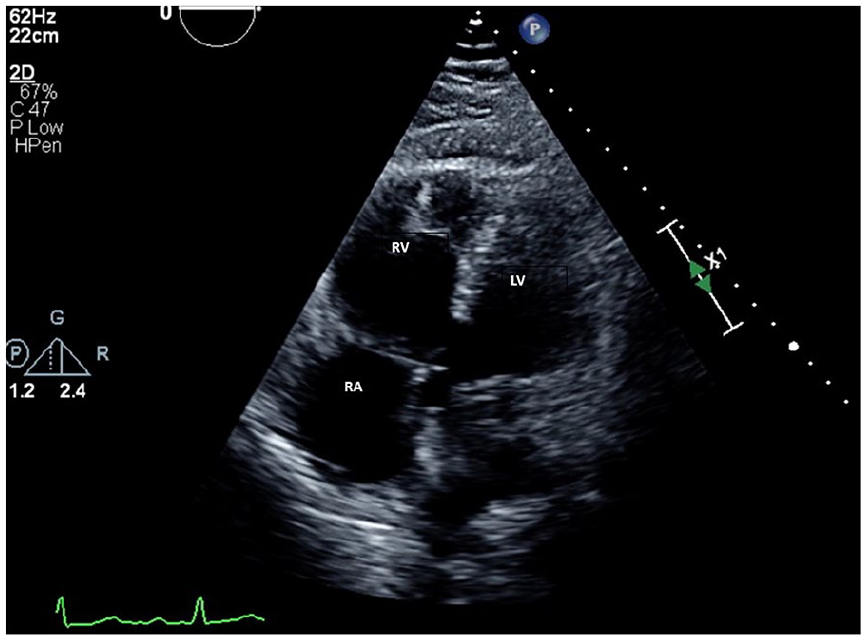
RV focused view during chemotherapy: newly dilated RV with reduced systolic function (TAPSE was 1.3 cm) and lateral wall akinesis. Chronic trace pericardial effusion is also present. LV: left ventricle; RA: right atrium; RV: right ventricle; TAPSE: tricuspid annular plane systolic excursion.

Continuous-wave Doppler during chemotherapy: pulmonary arterial systolic pressure is newly elevated as demonstrated by increased tricuspid regurgitation peak velocity of 3.47 m/s.
Having systematically excluded all major cardiopulmonary disorders (to include pulmonary embolism, acute coronary syndrome, pneumonia, acute anemia) and given the temporal relationship of treatment with carfilzomib to patient’s presentation, decision was made to stop carfilzomib. The patient did not receive any further medical interventions because he was clinically stable once admitted. No diuretics were given. No arterial blood gas was obtained. The patient was discharged after 2 days of monitoring. Right heart catheterization 2 weeks later revealed right atrial pressure of 7 mmHg, pulmonary arterial mean pressure of 28 mmHg, pulmonary capillary wedge pressure of 12 mmHg, normal cardiac output and pulmonary vascular resistance of 2.1 woods unit. Given restoration of near normal pulmonary pressures with discontinuation of the drug, carfilzomib-induced PH was confirmed as the diagnosis. A follow-up TTE one and a half months later (Figure 4) demonstrated normalization of RV size and systolic function (TAPSE was 2.3 cm). The patient’s symptoms and functional capacity also returned to baseline. Unfortunately, given multiple failed treatments for multiple myeloma, patient finally succumbed to complications of the disease 5 months later.

TTE after cessation of carfilzomib: apical four-chamber view demonstrating recovered RV size and systolic function (TASPE was 2.3 cm). LA: left atrium; LV: left ventricle; RA: right atrium; RV: right ventricle; TAPSE: tricuspid annular plane systolic excursion.
Discussion
Carfilzomib-induced PH as documented in the package insert occurs in up to 2% of patients, though ⩾ grade 3 PH adverse event as defined by NCI CTCAE (National Cancer Institute Common Terminology Criteria for Adverse Events) is seen in < 1%. 1 This definition is different from standards set by the 6th World Symposium on Pulmonary Hypertension Task Force, 5 which defines PH as mean pulmonary arterial pressure (PAP) > 20 mmHg. Although our patient did not have direct mean PAP measured, he did have severely elevated PASP during his index episode. With this finding, we presume that his mean PAP was also over 20 mmHg. There is little literature on the association of carfilzomib with acute PH and RV dysfunction. There is one similar case report of carfilzomib-induced PH in a 77-year-old female patient, who developed PH following seven cycles of carfilzomib/pomalidomide/dexamethasone therapy, although she was asymptomatic. A routine screening TTE showed pulmonary systolic pressure of 65–70 mmHg with mild RV enlargement, flattening of interventricular septum but preserved RV systolic function. 6 In contrast, our patient was a male who after only four cycles of therapy developed symptoms with evidence of profound RV systolic dysfunction on TTE. In both cases, PH resolved with discontinuation of carfilzomib and, in our case, RV dysfunction resolved as well.
Carfilzomib’s exact mechanism of action on the CV system remains largely undefined. One theory is PI may lead to intracellular accumulation of misfolded proteins and ultimately abnormal protein homeostasis contributing to apoptosis of cardiomyocytes. 7 Another proposed mechanism is endothelial and oxidative stress leading to vascular dysfunction, increased vascular tone and hypertension. 6 Interestingly, carfilzomib has been shown in the literature to reverse pulmonary arterial hypertension in rat models 8 but not in humans. This discrepancy is not fully understood. Differences between bortezomib and carfilzomib are also not entirely clear but may depend on carfilzomib’s irreversible and highly potent proteasome inhibition activity. 7 Risk factors for cardiotoxicity include prior PI and doxorubicin exposure as well as pre-existing CV conditions such as heart failure and hypertension 9 although these cardiotoxic effects do not appear to be cumulative or dose-related. 10 Combination therapy with carfilzomib and dexamethasone may elevate risks for heart failure. Steroids can induce secondary hypertension and fluid retention, both of which can be exacerbated by routine intravenous fluids administered with this regimen. Bottomline, overall attributable risks and natural history of carfilzomib-related cardiotoxicity are still evolving and incompletely characterized.
There may be several reasons for RV dysfunction found in our case. First, the rise in PAP is likely very rapid in our patient. RV is a low-pressure chamber and when its afterload is acutely elevated, systolic dysfunction may ensue especially when compensatory hypertrophy has not taken place. Second, our veteran is obese. Obesity is an independent risk factor for RV dysfunction. One small study evaluating 35 otherwise healthy obese patients found that isolated obesity had evidence of subclinical abnormalities in RV structure and function by echocardiographic parameters. 11 Finally, our patient had pre-existing CV conditions, such as hypertension and history of pulmonary embolism, which could have contributed to RV abnormalities.
According to a consensus document on management of carfilzomib from European Myeloma Network, a baseline assessment using the SCORE (Systemic Coronary Risk Evaluation) model as a formal and objective way of estimating CV risk is recommended. Echocardiogram is recommended as an initial screening and surveillance tool if concerns for any new CV symptoms or signs arise. 12 Early detection for carfilzomib-induced cardiotoxicity is imperative as these complications are typically reversible with cessation of the medication. This reversal, which was observed in our patient (Figure 4), can be as early as weeks to months. 6
Conclusion
Carfilzomib-induced CV effects are usually attributed to left-sided heart failure, but RV dysfunction can occur especially when acute PH occurs. PH is a known, albeit, rare complication of carfilzomib. It is important to be cognizant of PH as a likely cause for patients with acute dyspnea while on the PI. Routine screening echocardiograms should be considered prior to and during therapy particularly in those with pre-existing CV conditions as these cardiotoxicities are typically reversible with cessation of the drug.
Footnotes
Acknowledgements
The authors thank Bradley McGregor, MD, from Dana Farber Cancer Institute for his expertise and mentorship during editing portion of this paper.
Author contributions
P.-C.M. drafted, conceptualized and revised the manuscript. J.A. and V.B. both made substantial contributions to conception and manuscript revisions. All authors have read and approved the manuscript. All have agreed to be personally accountable for their own contributions.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require Institutional Review Board approval for reporting individual cases or case series.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the research department of the Veterans Affairs Boston Healthcare System.
Informed consent
Written informed consent was obtained from a legally authorized representative for anonymized patient information and images to be published in this article.
