Abstract
By the end of 2022, there had been a reduction in new cases and deaths caused by coronavirus disease 2019 (COVID-19). At the same time, new variants of the severe acute respiratory syndrome coronavirus 2 virus were being discovered. Critically ill patients with COVID-19 have been found to have high serum levels of proinflammatory cytokines, especially interleukin (IL)-6. COVID-19-related mortality has been attributed in most cases to the cytokine storm caused by increased levels of inflammatory cytokines. Dexamethasone in low doses and immunomodulators such as IL-6 inhibitors are recommended to overcome the cytokine storm. This current narrative review highlights the place of other therapeutic choices such as proteasome inhibitors, protease inhibitors and nuclear factor kappa B inhibitors in the treatment of patients with COVID-19.
Introduction
The severe acute respiratory syndrome caused by COVID-19 (severe acute respiratory syndrome coronavirus 2; SARS‑CoV‑2) represents a considerable worldwide threat to global health with the development of a cytokine storm being the primary leading cause of mortality in hospitalized critical cases. 1 Cytokine release syndrome is pathognomonic for severe progressive clinical COVID-19. 2 Interleukin (IL)-6, IL-1, IL-17, tumour necrosis factor-α (TNF-α) and other cytokines are significantly associated with high viral load and severe lung injury. 3 IL-6 is considered to be the pathophysiological hallmark and the main predictor of progression of COVID-19. 4 IL-6-inhibitors, such as siltuximab targeting IL-6 or tocilizumab and sarilumab targeting IL-6 receptors, may provide a life-saving therapeutic tool for critically-ill patients. 5 A previous study observed an improved clinical outcome with a higher survival rate in severe cases following tocilizumab use. 6 Nuclear factor kappa B (NF-κB) plays a central role in the cytokine storm and is linked with severe COVID-19 cases. 7 Transcription factors of NF-κB have an enhancing effect on expression of several cytokines such as IL-1, IL-2, IL-6 and TNF-α. 8 NF-κB also promotes the expression of genes encoding adhesion molecules such as E-selectin and inducible enzymes involved in inflammatory responses such as inducible nitric oxide synthase and cyclooxygenase-2. 8 The NF-κB inhibitor cromolyn was observed to ameliorate cytokine storm-induced inflammation in COVID-19 patients. 9 Interestingly, proteasome inhibitors such as bortezomib and carfilzomib impede viral entry and replication. 10 These agents could be another therapeutic strategy for the treatment of COVID-19 as they might help reduce the cytokine storm associated with SARS-CoV-2-induced inflammation. 11 Clinically proven protease inhibitors such as camostat mesylate could be a prophylactic tool that blocks the cellular entry of SARS-COV-2.9–11
The current narrative review focuses on the potential therapeutic tools that could be used for critically ill patients with COVID-19 and how pharmacogenomic studies are providing promise for targeting different molecules, which could be used in advanced human clinical trials. It is hoped that this research will help in designing an effective protocol for managing severe cases and at the same time could be a potentially prophylactic weapon that increases the efficacy of available COVD-19 vaccines.
The cytokine storm
Clinical and laboratory research has demonstrated a hyperactive dysregulated inflammatory immune response and excessive release of proinflammatory cytokines and chemokines in patients with severe COVID-19.12–14 The excessive and random release of cytokines (known as a cytokine storm) may lead to severe, life threatening and lethal complications. The severity of symptoms depends on the response of patient's immune system to the virus; when there is an aggressive inflammatory response with the release of large amount of cytokines, the symptoms of COVID-19 infection will be severe and may be lethal.15–17 Cytokines, especially IL-6, play a central role in the development of acute respiratory distress syndrome (ARDS), which is considered to be the most severe complication of COVID-19 infections. 18
Targeting and blocking the cytokine storm can help in the discovery of new methodologies to treat COVID-19 infections and reduce its complications.17,19 A previous study showed that the blockage of type 1 interferons in the early stages of COVID-19 infection will determine which cytokines will be involved in the storm and reduce the release of cytokines. 20 Moreover, a number of studies showed that the cytokine storm can be blocked by the use of interleukin inhibitors, especially IL-6 inhibitors, which can be used to treat severe COVID-19 cases.21–26
Targeting NF-κB
COVID-19 is an RNA virus enveloped by outer spike proteins. These spike glycoproteins are recognized by Toll-like receptors (TLRs), especially TLR6, TLR7 and TLR8, which are found on innate immune system cells and this recognition leads to the activation of NF-κB.27–29 Studies have demonstrated that NF-κB is activated in COVID-19 infections,27–29 leading to what is called a cytokine storm because NF-κB is responsible for the secretion and regulation of inflammatory cytokines and different chemokines.7,8,27–29 Knowing the role of NF-κB in severe COVID-19 pathogenesis might lead to the development of new methodologies to treat this infectious disease by the inhibition and blocking of NF-κB, which could suppress the cytokine storm.7,27–33 Known inhibitors of NF-κB include proteasome inhibitors (VL-01, bortezomib, carfilzomib and ixazomib), Bruton tyrosine kinase inhibitor (acalabrutinib), nucleotide analogue (remdesivir), TNF-α monoclonal antibodies (infliximab and adalimumab), N-acetylcysteine and corticosteroids (dexamethasone). 27 Montelukast, a drug that used for asthma patients, has a positive effect in modifying the activity of NF-κB and cytokine storm. 28
Interleukin-6 and COVID-19
Interleukin-6 plays important roles in autoimmunity, 34 inflammatory processes, 34 cytokine storms and cytokine release syndrome (CRS). 35 Moreover, IL-6 induces the secretion of proteins such as C-reactive protein (CRP), ferritin and fibrinogen and inhibits albumin synthesis. 36 In terms of COVID-19, IL-6 can produce a hyper-innate inflammatory response. 37 Serology analysis of COVID-19 patients demonstrated higher serum IL-6 levels in patients in the severe stage of COVID-19 compared with patients in the mild–moderated stage of COVID-19.38,39 In addition, it has been found that IL-6 is linked to COVID-19 stages and radiological findings.40–43 A study conducted in Italy argued that IL-6 can be used as a predictive tool for COVID-19 disease progression. 44
Interleukin-6 blocking agents that have been used for the treatment of COVID-19 include tocilizumab, sarilumab, siltuximab and clazakizumab.45–58 Tocilizumab is used to treat rheumatoid arthritis, systemic juvenile idiopathic arthritis45,46 and the CRS that might develop following the use of some types of immunotherapies. 47 A study of 21 severely ill COVID-19 patients who received tocilizumab treatment demonstrated an improvement in their symptoms and radiological findings. 25 Likewise, another study found that tocilizumab decreased the need for mechanical ventilation and intensive care unit admission in COVID-19 patients. 48 In addition, tocilizumab has been found to treat the neuropsychiatric manifestations of COVID-19.49,50 Findings from a 64-year-old male who received tocilizumab for haemophagocytic lymphohistiocytosis syndrome and COVID-19 showed lower levels of IL-6. 51 Sarilumab is an IL-6 inhibitor that is used for the treatment of rheumatoid arthritis. 52 Results from a study of 28 patients with COVID-19 disease who received a single dose of sarilumab demonstrated an improvement in recovery time. 53 In another study, 53 patients were treated with sarilumab (14 of them were from the intensive care unit) and they exhibited an improvement in their clinical condition. 54 Another study showed similar results on eight patients who were treated with sarilumab. 55 Another monoclonal antibody that binds to IL-6 and neutralizes its effect is siltuximab. 56 In a cohort study, it was reported that siltuximab adjusted the risk of mortality rate. 56 Lastly, clazakizumab is a humanized immunoglobulin G monoclonal antibody, which works against IL-6 and can be used in the treatment of rheumatoid arthritis. 57 In cases of COVID-19 with raised CRP and IL-6 levels, clazakizumab positively affects respiratory function and the level of inflammatory markers, and decreases the need for oxygen therapy. 58
Role of the proteasome system
The cellular proteasome system is the mainstay in the protein degradation process. 59 The ubiquitin-proteasome system is a system that leads to the degradation of protein by affecting proteasome action. 11 Moreover, the ubiquitin-proteasome system plays a role in cell cycle progression, apoptosis, cell transduction and cell transcriptional regulation. 60 Proteasome inhibitors are used for the treatment of multiple myeloma and Mantel cell lymphoma with well-known side-effects including thrombocytopaenia, neutropaenia and peripheral neuropathy.61,62 In terms of COVID-19 disease, proteasome inhibitors affect viral replication, the entry of the virus into the eukaryotic cell, RNA synthesis and the protein structure of the virus.11,63–67 In addition, proteasome inhibitors block NF-κB and inhibit cytokine release,12,68 which makes proteasome inhibitors promising therapeutic options for treating COVID-19 disease.
An example of a proteasome inhibitor is carfilzomib, which provides a higher level of inhibition compared with other proteasome inhibitors. 69 A previous study reported that carfilzomib is the therapeutic choice for treating COVID-19 cases based on the performance of a molecular dynamic simulation followed by a binding free energy calculation, which showed that carfilzomib has a high binding free energy. 70 Another example of a proteasome inhibitor is MG132, which is a synthetic peptide aldehyde. 71 A previous study reported that MG132 suppressed SARS‑CoV‑2 replication by interacting with the early stage of the viral life cycle when compared with other inhibitors such as lactacystin that demonstrated very limited effects on replication. 72 MG132 inhibits m-calpain, which plays a role in SARS‑CoV‑2 replication.73,74 Although MG132 has shown promise in inhibiting proteases, 75 it does not inhibit SARS‑CoV‑2 replication due to proteasome or autophagy impairment. 71 It was reported that MG132 has an antiviral effect on viruses such as herpes simplex virus 1, 76 hepatitis E, 77 human cytomegalovirus, 78 porcine circovirus type 2 79 and coxsackievirus B3. 80 However, MG132 exerts different mechanisms of controlling the cell entry of these viruses by interacting with the ubiquitin-proteasome system rather than inhibiting proteases.76–79
Viral proteases as targets for pharmacogenomics
Viral proteases play an important role in viral entry in the cell, viral replication, maturation of essential viral proteins and the immune response for the virus infection.81–83 Pharmacogenomics plays a role in attenuating the protease genome and designing agents that block their actions. For example, several studies demonstrated that understanding the important role of viral proteases makes them an attractive target for treating severe COVID-19 cases using protease inhibitors.81–83 There are numerous proteases, but this review will only focus on the following: transmembrane serine protease 2 (TMPRSS2), a disintegrin and metalloprotease 17 (ADAM17), main protease (Mpro), 3-chymotrypsin-like protease (3CLpro) and papain-like protease (PLpro).
Studies showed that the angiotensin-converting enzyme 2 (ACE2) receptor acts as a gate for SARS-COV-2 to cross and enter host cells and TMPRSS2 facilitates this process by cleaving the spike protein and enabling it to bind to the ACE2 receptor, which initiates viral entry into the cells.84–94 ACE2 and TMPRSS2 are found in heart, liver, kidneys, brain and other organs, which explains the presence of the extrapulmonary manifestations of COVID-19 infection.84,90 A previous study demonstrated that ARDS treatment outcomes were affected by ACE genotype. 87 The role of ACE2 as a receptor and TMPRSS2 as a primer make these two molecules good targets in the treatment of COVID-19 by suppressing them.89,91–93,95–97 TMPRSS2 inhibitors include bromhexine hydrochloride, camostat mesylate and nafamostat mesylate.89,91,97–101 Bromhexine hydrochloride is used as a prophylactic or as a treatment to decrease the hospital stay, intubation and mortality rates.97,98
The SARS‑CoV‑2 virus can activate ADAM17, 102 which plays a role in the development of a cytokine storm by activating TNF-α and IL-6 receptor.102,103 ADAM17 and TMPRSS2 are essential for viral entry and cell fusion, thus ADAM17 mediates ACE2 shedding and converts it to its active form; and then TMPRSS2 cleaves the virus spike protein to enable its binding with the ACE2 receptor that initiates the viral entry into the cells.84–94,102,104,105 Alpha-1 antitrypsin (A1AT), a protein found in the human body that it is the most common protease inhibitor found in the plasma, works as an antiviral and anti-inflammatory molecule.106–108 Researchers found that A1AT inhibits the activity of TMPRSS2 and ADAM 17, so it interferes with the viral entry into the cell.106–110 Furthermore, A1AT has important anti-inflammatory and immune-regulatory activities by inhibiting NF-κB, IL-8, TNF-α and neutrophil elastase.106,107 These functional characteristics of A1AT make it an attractive target for the treatment of COVID-19 infection.
Several studies reported the importance of discovering that the Mpro inhibitors interfere with viral entry and replication.111–113 Melatonin has a positive effect as an anti-inflammatory molecule, inhibitor of Mpro and inhibitor of ACE2 because it inhibits calmodulin, which is an essential intracellular component for ACE2. 113 Blocking of 3CLpro interferes with viral entry and replication.114–118 Ethacrynic acid, naproxen, allopurinol, butenafine hydrochloride, raloxifene hydrochloride, tranylcypromine hydrochloride and saquinavir mesylate are a group of drugs found to inhibit the activity of 3CLpro. 114 Moreover, phosphate prodrug such as PF-00835231 blocks the proteolytic activity of 3CLpro.115–117 PLpro plays a role in viral replication and innate immune system hyperactivity (i.e. the cytokine storm).83,119–122 As for other proteases, the inhibition of PLpro would be expected to be a treatment for SARS-CoV-2 infection.119–123 Tanshinone II-A sodium sulfonate and chloroxine act as blockers of PLpro. 122
Conclusion
Concepts about COVID-19 and its deleterious impact on human health that are not yet clarified require nuanced research studies in order to overcome severe disease and improve the efficacy of the available vaccines. Targeting different molecules such as proteasome enzymes and NF-kB via pharmacogenetic studies may help in designing novel agents that can be investigated in humans for their efficacy as therapeutic drugs in treating critical cases of COVID-19 or be used prophylactically in adjusted doses after receiving the available vaccines. These studies may be a great breakthrough in combating the cytokine storm that accounts for the serious outcomes observed in severely ill patients. Together with protease inhibitors and IL-6 inhibitors, proteasome inhibitors and NF-kB inhibitors could be a significant potential synergistic combination in therapeutic protocols. Figure 1 presents the molecular targets of potential therapies for critically ill COVID-19 cases. Table 1 summarizes the main published studies that have investigated potential molecular targets for the management of COVID-19.
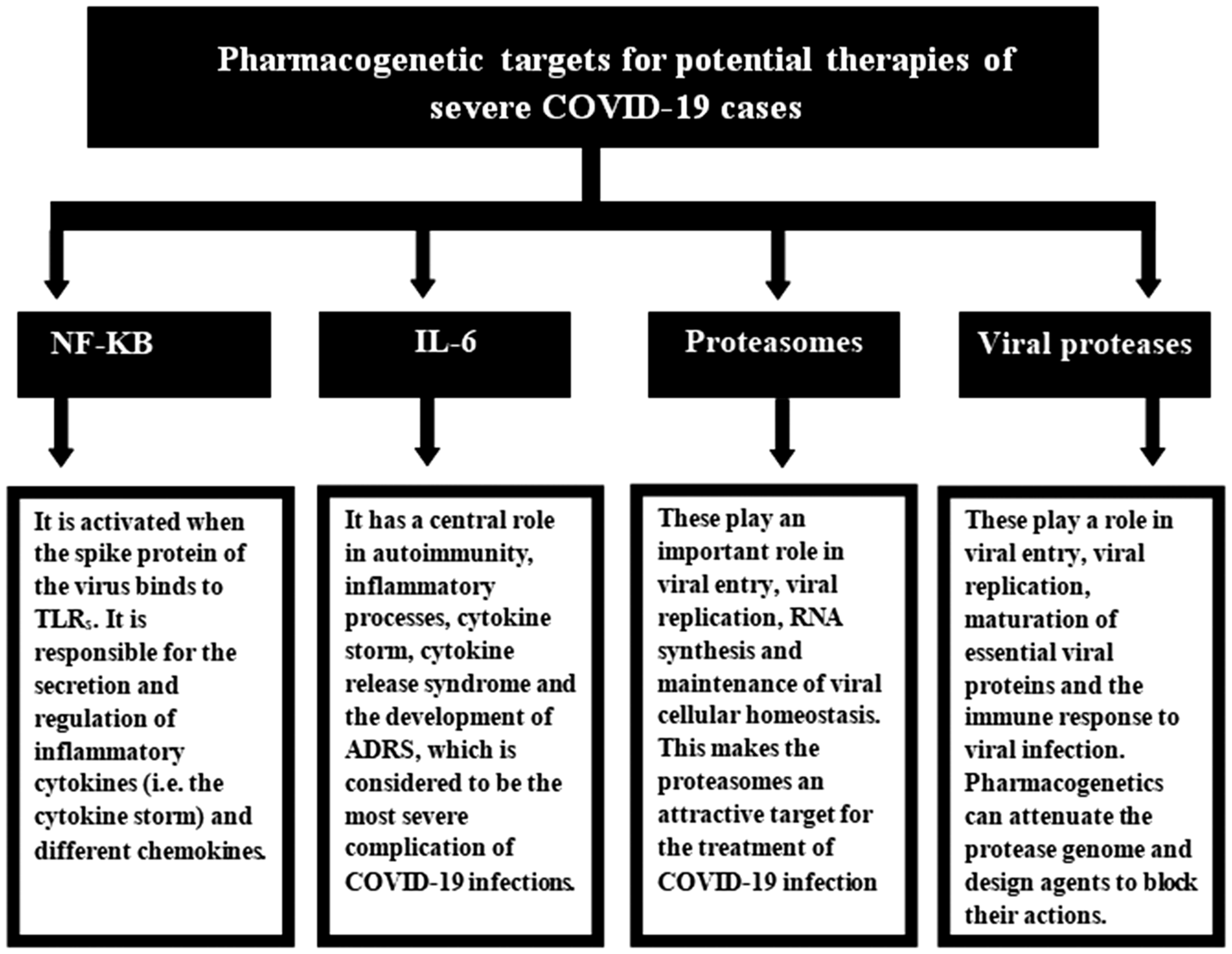
Pharmacogenetic targets for potential therapies for patients with severe coronavirus disease 2019 (COVID-19) caused by the severe acute respiratory syndrome coronavirus 2 virus. NF-κB, nuclear factor kappa B; IL-6, interleukin-6; TLRs, Toll-like receptors; ARDS, acute respiratory distress syndrome.
Summary of the most important recent studies about the management of COVID-19 disease caused by the severe acute respiratory syndrome coronavirus 2 (SARS-COV-2) virus.

IL-6, interleukin-6; NF-κB, nuclear factor kappa B; ACE2, angiotensin-converting enzyme 2; TMPRSS2, transmembrane serine protease 2; ADAM17, metalloprotease 17; A1AT, alpha-1 antitrypsin; Mpro, main protease; 3CLpro, 3-chymotrypsin-like protease.
Footnotes
Author contributions
R.H.T. decided to write the current narrative review and undertook the final editing of the manuscript. N.A.M., O.B.B. and O.M.M. contributed equally to collecting data, editing and revising the manuscript. All authors agree to be accountable for all aspects of the work.
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from funding agency in the public, commercial, or not-for-profit sectors.
