Abstract
Background:
Whether low-dose alteplase is similar to standard-dose bridging alteplase prior to endovascular mechanical thrombectomy in patients with acute ischemic stroke (AIS) remains uncertain.
Aims:
The aim of this study was to compare the efficacy and safety outcomes of low- versus standard-dose bridging alteplase therapy (BT) in patients with acute ischemic stroke (AIS) who are eligible for intravenous thrombolysis (IVT) within 4.5 h after onset.
Methods:
We conducted an indirect comparison of low- versus standard-dose bridging alteplase before mechanical thrombectomy in AIS of current available clinical randomized controlled trials (RCTs) that compared direct mechanical thrombectomy treatment (dMT) to BT. Primary efficacy outcomes were functional independence and excellent recovery defined as a dichotomized modified Rankin Scale (mRS) 0–2 and 0–1 at 90 days. Safety outcomes included symptomatic intracranial hemorrhage (sICH) and any intracranial hemorrhage (ICH).
Results:
We included six RCTs of 2334 AIS patients in this analysis, including one trial using low-dose bridging alteplase (n = 103) and five trials using standard-dose bridging alteplase (n = 1067) against a common comparator (dMT). Indirect comparisons of low- to standard-dose bridging alteplase yielded an odds ratio (OR) of 0.84 (95% CI 0.47–1.50) for 90-day mRS 0–2, 1.18 (95% CI 0.65–2.12) for 90-day mRS 0–1, 1.21 (95% CI 0.44–3.36) for mortality, and 1.11 (95% CI 0.39–3.14) for successful recanalization. There were no significant differences in the odds for sICH (OR 1.05, 95% CI 0.32–3.41) or any ICH (OR 1.71, 95% CI 0.94–3.10) between low- and standard-dose bridging alteplase.
Conclusion:
Indirect evidence shows that the effects of low- and standard-dose bridging alteplase are similar for key efficacy and safety outcomes. Due to the wide confidence intervals, larger randomized trials comparing low- and standard-dose alteplase bridging therapy are required.
Keywords
Introduction
Intravenous thrombolysis (IVT) has been the standard first-line treatment for patients with acute ischemic stroke (AIS) less than 4.5 h after symptom onset.1,2 In the past decade, endovascular treatment using mechanical thrombectomy has been shown to be superior to IVT treatment alone in AIS patients with anterior large-artery occlusion.3–5 Recently, six phase III randomized controlled trials (RCTs) attempted to demonstrate the non-inferiority of direct mechanical thrombectomy (dMT) compared to bridging IVT followed by thrombectomy [bridging alteplase therapy (BT)] in AIS patients, yielding inconsistent results.6–11 Meta-analyses of these six RCTs showed that patients who received dMT were at a similar likelihood of achieving functional independence but were at a lower risk of developing any intracerebral hemorrhage (ICH).12,13 However, the relatively wide 95% confidence intervals (CIs) could not exclude the benefits of IVT before dMT. Previous RCTs and observational data revealed that AIS patients who received IVT using low-dose alteplase had fewer ICH events compared to standard-dose alteplase.14,15 A meta-analysis of several case series detected higher rates of favorable outcome [odds ratio (OR) 1.60, 95% CI 1.07–2.40, p = 0.022] and similar rates of symptomatic intracranial hemorrhage (sICH; OR 0.86, 95% CI 0.41–1.83, p = 0.70) among patients who received standard- versus low-dose alteplase prior to intraarterial therapy. 16 Whether low-dose alteplase is similar to standard-dose bridging alteplase prior to endovascular mechanical thrombectomy remains uncertain and requires investigation. To our knowledge, a head-to-head trial of low- versus standard-dose alteplase in the BT strategy has not been investigated. When direct comparisons are unavailable, indirect comparison based on meta-analysis may be used to evaluate the magnitude of treatment effects across studies. 17 We aimed to compare the efficacy endpoints and safety outcomes of low-dose versus standard-dose bridging alteplase based on current available RCTs data of dMT versus BT in AIS patients who were eligible for IVT, using Bucher’s method of adjusted indirect comparisons. 17
Methods
Search strategy
We searched PubMed, Embase, Cochrane Central Register of Controlled Trials, and the clinicaltrials.gov from inception to July 2022 for all RCTs of IVT-eligible AIS patients receiving BT versus dMT (the only studied common comparator treatment) regarding efficacy endpoints, mortality, and safety (ICH) events. We restricted our analysis to studies reporting event rates or providing sufficient data to calculate event rates.
Data extraction
Two authors independently extracted data for primary efficacy endpoints of 90-day modified Rankin scale (mRS) score (categorical 0–2 versus 3–6, and 0–1 versus 2–6), 90-day mortality, successful recanalization, and safety outcomes including sICH or any ICH. In this analysis, we extracted data of outcomes derived from intention-to-treat analytical strategy when reported. Otherwise, data derived from on-treatment approach were extracted. We also extracted information on inclusion and exclusion criteria, outcome definitions, risk factors for stroke in the dMT and BT group, study design, and statistical analysis strategies to assess the comparability of the trials.
Statistics
We calculated the ORs as our measure of treatment effect in the present analysis because ORs for binary outcomes have less heterogeneity in meta-analysis than risk differences or relative risks. 18 We performed an inter-trial indirect comparison across the RCTs using the Bucher’s method to estimate relative treatment effect size against a common comparator.17,19 We evaluated the level of clinical and methodological similarity among the six RCTs and by comparing outcome event rates among the common comparator (dMT) patients across the different RCTs to assess treatment effectiveness in the common referent (dMT) group. The method to calculate the ORs and 95% CIs of the indirect comparisons was described in previous literature17,20,21 (Table 1). First, the expected effect of ‘standard-dose bridging‘ as a whole versus dMT was estimated as a weighted average using the inverse of the variance of the log (OR) as weights.17,19,20 The second focus in the present analysis was the indirect comparisons of low- versus standard-dose bridging alteplase. We conducted two sensitivity analyses. First, we conducted an indirect comparison between the Direct Mechanical Thrombectomy in Acute LVO Stroke (SKIP) trial (the only RCT using low-dose alteplase in the BT arm) and three RCTs using standard-dose bridging alteplase [Direct Endovascular Thrombectomy versus Combined IVT and Endovascular Thrombectomy for Patients with Acute Large Vessel Occlusion in the Anterior Circulation (DEVT) trial, 7 Direct Intraarterial Thrombectomy in Order to Revascularize Acute Ischemic Stroke Patients with Large Vessel Occlusion Efficiently in Chinese Tertiary Hospitals Multicenter Randomized Clinical Trial (DIRECT-MT) trial, 6 and subgroup data of the DIRECT Endovascular Clot Retrieval versus Standard Bridging Thrombolysis With Endovascular Clot Retrieval (DIRECT-SAFE) trial] 10 representing the East Asian stroke population. We further conducted a separate indirect comparison between SKIP and DEVT for sICH using the National Institute of Health stroke scale (NINDS) criteria, and SKIP versus two trials (DEVT and DIRECT-SAFE) for sICH using the Safe Implementation of Thrombolysis in Stroke-Monitoring Study (SITS-MOST) definitions. We did not assess publication bias because of the small number of eligible studies. Stata 16.0 was used for the statistical analyses and graphical presentation.
Effect size estimate method.

CI, confidence interval; dMT, direct mechanical thrombectomy treatment; OR, odds ratio.
Evaluation of indirect comparisons
We assessed whether the indirect comparisons in the current study meet certain criteria, based on a checklist established by Kiefer et al. 22
Outcomes
The primary efficacy endpoints were 90-day functional independence (mRS 0–2) and excellent recovery (mRS 0–1). The secondary efficacy endpoint was successful recanalization. The safety outcomes included sICH and any ICH.
Results
Baseline characteristics
We included findings from 2334 patients (44.3% women) enrolled in six RCTs, comparing the efficacy and safety profiles of BT to dMT. Among them, the Multicenter Randomized CLinical trial of Endovascular treatment for Acute ischemic stroke in the Netherlands-NO IV (MR CLEAN-NO IV) 9 and the Bridging Thrombolysis versus Direct Mechanical Thrombectomy in Acute Ischemic Stroke (SWIFT-DIRECT) 11 were conducted in European and North America. Primary efficacy endpoints and mortality on the intention-to-treat population were reported across six RCTs. Table 2 summarizes the six RCTs being compared for this analysis. SKIP was the only RCT that compared BT using low dose of alteplase bridging to dMT. 8 Potential clinical and methodological modifiers of relative treatment effects, including patient and intervention characteristics, outcome definitions, clinical moderators, and methodological moderators, such as randomization and blinding, were generally similar across the six included RCTs. Median age and the proportions of female patients were broadly similar across six RCTs, except the SKIP study included a lower percentage (30%) of female patients in the BT arm. Table 3 shows that the inclusion and exclusion criteria were broadly similar across trials. DIRECT-SAFE also included those with basilar artery occlusion (8% in the dMT arm versus 6% in the BT arm), 10 while the other five RCTs only included those with anterior artery occlusion. There was no Alberta Stroke Program Early CT Score (ASPECTS) limit for DEVT, 7 DIRECT-SAFE, 10 and MR CLEAN-NO IV. 9 The SKIP trial only included those with baseline computed tomography (CT)-ASPECTS score ⩾ 6 or diffusion-weighted image (DWI)-ASPECTS score ⩾ 5, which was higher than that of SWIFT-DIRECT (ASPECTS score ⩾ 4). sICH definitions were heterogeneous as follows: NINDS criteria for DEVT and SKIP; SITS-MOST criteria for DEVT, SKIP, and DIRECT-SAFE; the European Cooperative Acute Stroke Study (ECASS) criteria for DEVT and SWIFT-DIRECT, and the Heidelberg criteria for DEVT, DIRECT-MT and MR CLEAN-NO IV. Lost to 90-day follow-up rates were low across the six RCTs (lost to follow-up: DEVT 0/234, DIRECT-MT 2/656, DIRECT-SAFE 0/293, MR CLEAN-NO IV 0/539, SKIP 0/204, and SWIFT-DIRECT 1/408).
Baseline characteristics of included studies.
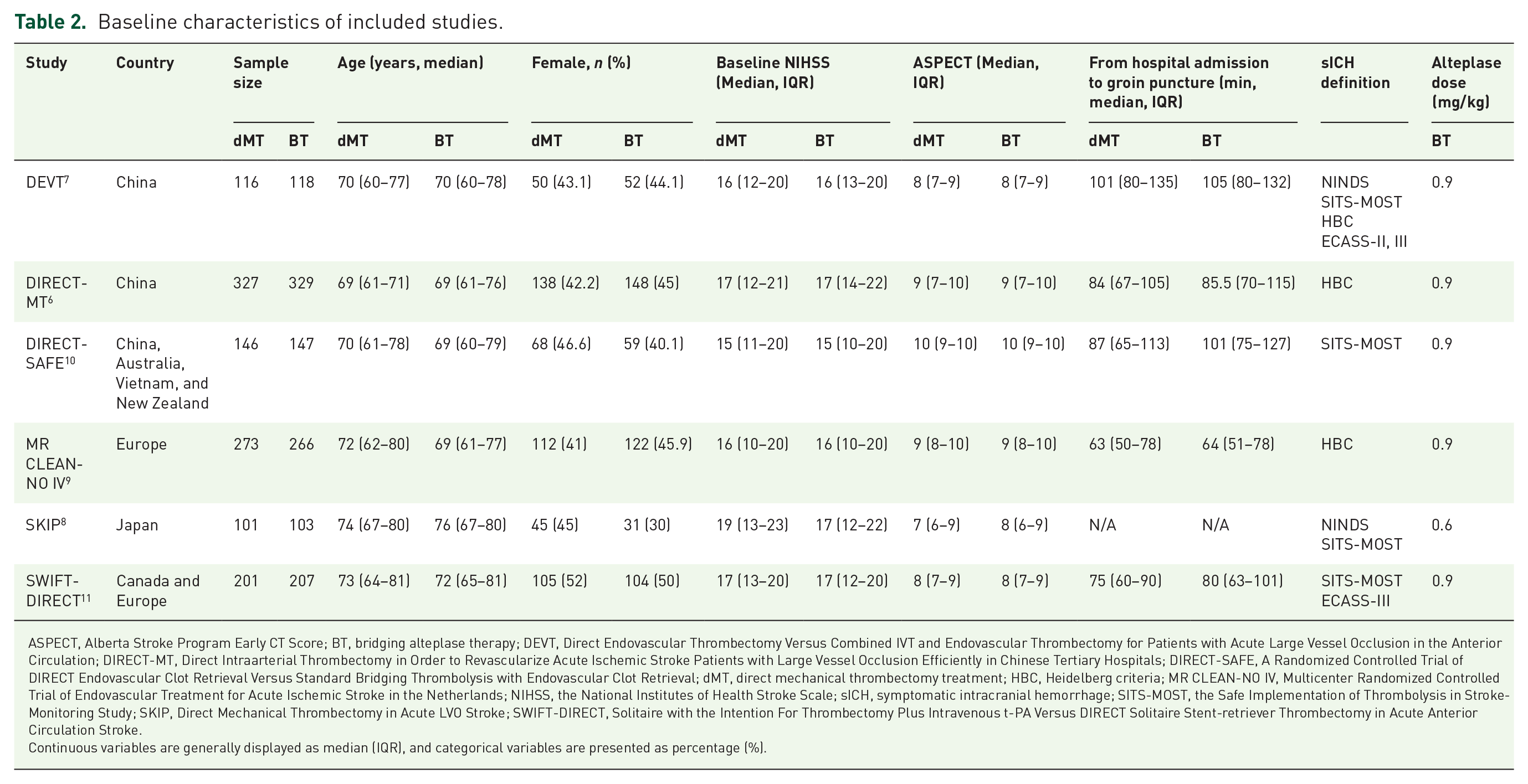
ASPECT, Alberta Stroke Program Early CT Score; BT, bridging alteplase therapy; DEVT, Direct Endovascular Thrombectomy Versus Combined IVT and Endovascular Thrombectomy for Patients with Acute Large Vessel Occlusion in the Anterior Circulation; DIRECT-MT, Direct Intraarterial Thrombectomy in Order to Revascularize Acute Ischemic Stroke Patients with Large Vessel Occlusion Efficiently in Chinese Tertiary Hospitals; DIRECT-SAFE, A Randomized Controlled Trial of DIRECT Endovascular Clot Retrieval Versus Standard Bridging Thrombolysis with Endovascular Clot Retrieval; dMT, direct mechanical thrombectomy treatment; HBC, Heidelberg criteria; MR CLEAN-NO IV, Multicenter Randomized Controlled Trial of Endovascular Treatment for Acute Ischemic Stroke in the Netherlands; NIHSS, the National Institutes of Health Stroke Scale; sICH, symptomatic intracranial hemorrhage; SITS-MOST, the Safe Implementation of Thrombolysis in Stroke-Monitoring Study; SKIP, Direct Mechanical Thrombectomy in Acute LVO Stroke; SWIFT-DIRECT, Solitaire with the Intention For Thrombectomy Plus Intravenous t-PA Versus DIRECT Solitaire Stent-retriever Thrombectomy in Acute Anterior Circulation Stroke.
Continuous variables are generally displayed as median (IQR), and categorical variables are presented as percentage (%).
Inclusion and exclusion criteria each included RCTs.

ASPECTS, Alberta Stroke Program Early CT Score; DEVT, Direct Endovascular Thrombectomy Versus Combined IVT and Endovascular Thrombectomy for Patients with Acute Large Vessel Occlusion in the Anterior Circulation; DIRECT-MT, Direct Intraarterial Thrombectomy in Order to Revascularize Acute Ischemic Stroke Patients with Large Vessel Occlusion Efficiently in Chinese Tertiary Hospitals; DIRECT-SAFE, A Randomized Controlled Trial of DIRECT Endovascular Clot Retrieval Versus Standard Bridging Thrombolysis with Endovascular Clot Retrieval; ICA, internal carotid artery; M1, first segment of the middle cerebral artery; M2, second segment of the middle cerebral artery; MCA, middle cerebral artery; MR CLEAN-NO IV, Multicenter Randomized Controlled Trial of Endovascular Treatment for Acute Ischemic Stroke in the Netherlands; mRS, modified Rankin Scale; NIHSS, the National Institutes of Health Stroke Scale; RCTs, randomized clinical controlled trials; SKIP, Direct Mechanical Thrombectomy in Acute LVO Stroke; SWIFT-DIRECT, Solitaire with the Intention For Thrombectomy Plus Intravenous t-PA Versus DIRECT Solitaire Stent-retriever Thrombectomy in Acute Anterior Circulation Stroke; VT, intravenous thrombolysis.
Table 4 summarizes the baseline characteristics of the common comparator (dMT arm) across the six RCTs. In the dMT arm among all RCTs, SKIP participants had the oldest median age of 74 (67–80) years, the highest median baseline National Institute Health Stroke Scale (NIHSS) score [19 (interquartile range, IQR, 13–23)] and the lowest median ASPECT score [7 (IQR 6–9)]. SKIP participants had the highest proportions of atrial fibrillation (56%) and cardioembolism stroke etiology (66%). Table 5 shows the crude event rates of efficacy endpoints and safety outcomes in the six RCTs. The crude event rates for 90-day functional outcomes in the dMT arms of the six trials were fairly comparable except obvious lower rates for 90-day mRS 0–2 in the DIRECT-MT (36.4%) and mRS 0–1 in the DIRECT-MT (24.5%) and MR CLEAN-NO IV (16.1%). The crude event rates for mortality in the dMT arms of SKIP (7.9%) and SWIFT-DIRECT (10.9%) participants look obviously lower. The crude event rates for successful recanalization in the dMT arms from DIRECT-MT and MR CLEAN-NO IV were lower than 80% (79.4% and 78.7%, respectively). Looking at safety outcomes, the crude event rates for sICH in the dMT arms of DIRECT-SAFE [2/146 (1.4%)] and SWIFT-DIRECT [3/201 (1.5%)] trials look obviously lower. The crude event rates for any ICH in the dMT arms of DEVT [25/115 (21.7%)] and DIRECT-SAFE [31/146 (21.2%)] trials were lower.
Baseline characteristics of the comparator (dMT arm) across the six RCTs.
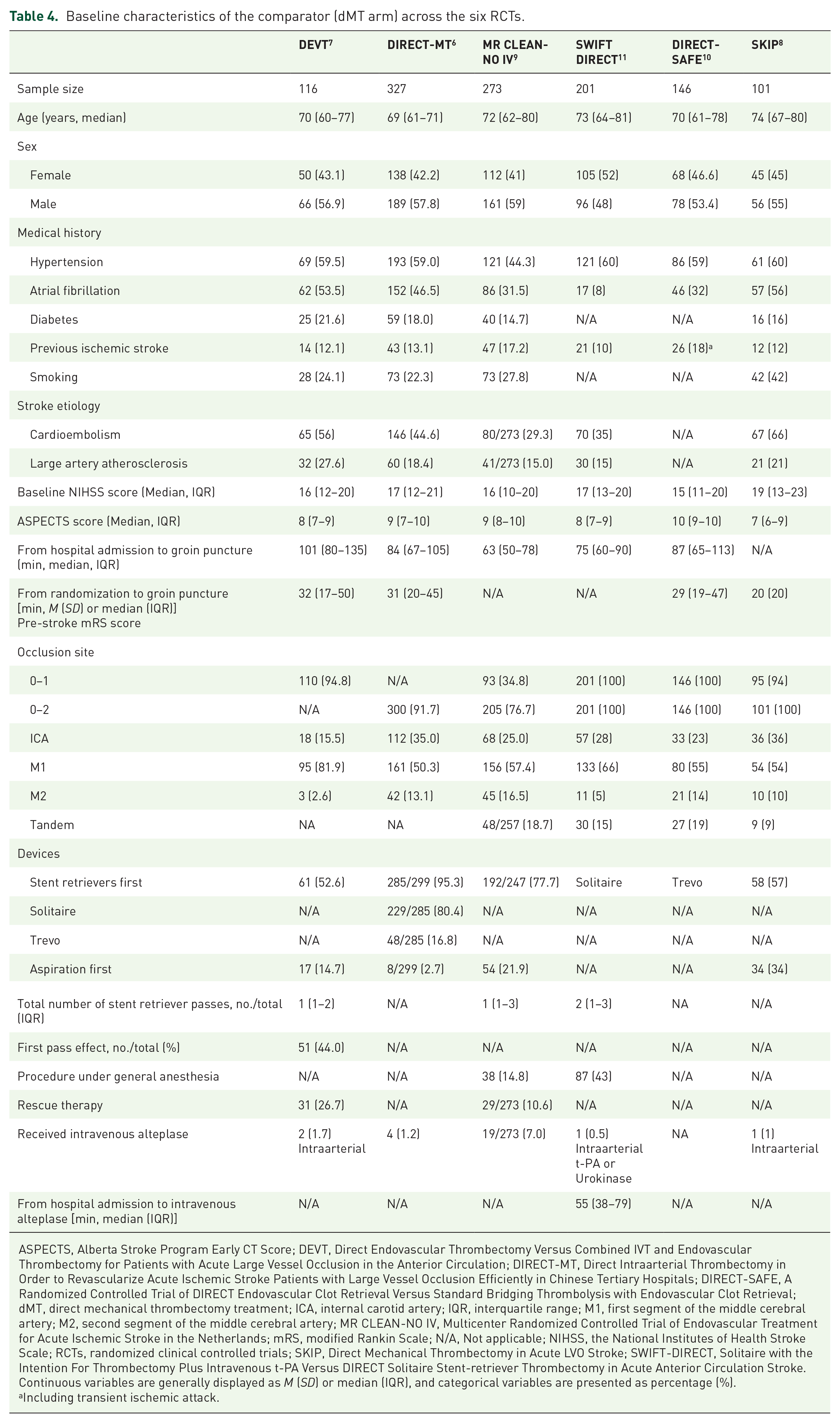
ASPECTS, Alberta Stroke Program Early CT Score; DEVT, Direct Endovascular Thrombectomy Versus Combined IVT and Endovascular Thrombectomy for Patients with Acute Large Vessel Occlusion in the Anterior Circulation; DIRECT-MT, Direct Intraarterial Thrombectomy in Order to Revascularize Acute Ischemic Stroke Patients with Large Vessel Occlusion Efficiently in Chinese Tertiary Hospitals; DIRECT-SAFE, A Randomized Controlled Trial of DIRECT Endovascular Clot Retrieval Versus Standard Bridging Thrombolysis with Endovascular Clot Retrieval; dMT, direct mechanical thrombectomy treatment; ICA, internal carotid artery; IQR, interquartile range; M1, first segment of the middle cerebral artery; M2, second segment of the middle cerebral artery; MR CLEAN-NO IV, Multicenter Randomized Controlled Trial of Endovascular Treatment for Acute Ischemic Stroke in the Netherlands; mRS, modified Rankin Scale; N/A, Not applicable; NIHSS, the National Institutes of Health Stroke Scale; RCTs, randomized clinical controlled trials; SKIP, Direct Mechanical Thrombectomy in Acute LVO Stroke; SWIFT-DIRECT, Solitaire with the Intention For Thrombectomy Plus Intravenous t-PA Versus DIRECT Solitaire Stent-retriever Thrombectomy in Acute Anterior Circulation Stroke.
Continuous variables are generally displayed as M (SD) or median (IQR), and categorical variables are presented as percentage (%).
Including transient ischemic attack.
Crude event rates of efficacy and safety outcomes in the dMT arm of the six RCTs.

DEVT, Direct Endovascular Thrombectomy Versus Combined IVT and Endovascular Thrombectomy for Patients with Acute Large Vessel Occlusion in the Anterior Circulation; DIRECT-MT, Direct Intraarterial Thrombectomy in Order to Revascularize Acute Ischemic Stroke Patients with Large Vessel Occlusion Efficiently in Chinese Tertiary Hospitals; DIRECT-SAFE, A Randomized Controlled Trial of DIRECT Endovascular Clot Retrieval Versus Standard Bridging Thrombolysis with Endovascular Clot Retrieval; dMT, direct mechanical thrombectomy treatment; MR CLEAN-NO IV, Multicenter Randomized Controlled Trial of Endovascular Treatment for Acute Ischemic Stroke in the Netherlands; mRS, modified Rankin Scale; RCTs, randomized clinical controlled trials; sICH, symptomatic intracerebral hemorrhage; SKIP, Direct Mechanical Thrombectomy in Acute LVO Stroke; SWIFT-DIRECT, Solitaire with the Intention For Thrombectomy Plus Intravenous t-PA Versus DIRECT Solitaire Stent-retriever Thrombectomy in Acute Anterior Circulation Stroke.
Data are generally displayed as frequency with percentage (%) of patients.
Table 6 summarizes the baseline characteristics of the BT arm across the six RCTs. In the BT arm, SKIP participants had the oldest median age of 76 (IQR 67–80) years, the lowest proportion of females (30%), and the highest proportion of atrial fibrillation (62%) and cardioembolism stroke etiology (70%). The baseline NIHSS and ASPECT scores in the BT arm of SKIP study were generally similar to other five RCTs. The mean time from randomization to intravenous alteplase in SKIP trial was 14 min, which was longer than those in DEVT and DIRECT-MT (both median 7 min), and DIRECT-SAFE (median 8 min). The trials did not differ in blinding. Table 7 shows that the crude event rates of efficacy outcomes in the BT arm of the six RCTs were generally comparable. SKIP participants in the BT arm experienced the highest proportion of sICH (7.8) and any ICH (50.5%).
Baseline characteristics of the BT arm across the six RCTs.

ASPECTS, Alberta Stroke Program Early CT Score; BT, Bridging Alteplase Therapy; DEVT, Direct Endovascular Thrombectomy Versus Combined IVT and Endovascular Thrombectomy for Patients with Acute Large Vessel Occlusion in the Anterior Circulation; DIRECT-MT, Direct Intraarterial Thrombectomy in Order to Revascularize Acute Ischemic Stroke Patients with Large Vessel Occlusion Efficiently in Chinese Tertiary Hospitals; DIRECT-SAFE, A Randomized Controlled Trial of DIRECT Endovascular Clot Retrieval Versus Standard Bridging Thrombolysis with Endovascular Clot Retrieval; ICA, internal carotid artery; IQR, interquartile range; M1, first segment of the middle cerebral artery; M2, second segment of the middle cerebral artery; MR CLEAN-NO IV, Multicenter Randomized Controlled Trial of Endovascular Treatment for Acute Ischemic Stroke in the Netherlands; mRS, modified Rankin Scale; NIHSS, the National Institutes of Health Stroke Scale; SKIP, Direct Mechanical Thrombectomy in Acute LVO Stroke; SWIFT-DIRECT, Solitaire with the Intention For Thrombectomy Plus Intravenous t-PA Versus DIRECT Solitaire Stent-retriever Thrombectomy in Acute Anterior Circulation Stroke.
Continuous variables are generally displayed as M (SD) or median (IQR), and categorical variables are presented as percentage (%).
Crude event rates of efficacy and safety outcomes in the BT arm of the six RCTs.

BT, Bridging Alteplase Therapy; DEVT, Direct Endovascular Thrombectomy Versus Combined IVT and Endovascular Thrombectomy for Patients With Acute Large Vessel Occlusion in the Anterior Circulation; DIRECT-MT, Direct Intraarterial Thrombectomy in Order to Revascularize Acute Ischemic Stroke Patients with Large Vessel Occlusion Efficiently in Chinese Tertiary Hospitals; DIRECT-SAFE, A Randomized Controlled Trial of DIRECT Endovascular Clot Retrieval Versus Standard Bridging Thrombolysis with Endovascular Clot Retrieval; MR CLEAN-NO IV, Multicenter Randomized Controlled Trial of Endovascular Treatment for Acute Ischemic Stroke in the Netherlands; mRS, modified Rankin Scale; RCTs, randomized clinical controlled trials; sICH, symptomatic intracerebral hemorrhage; SKIP, Direct Mechanical Thrombectomy in Acute LVO Stroke; SWIFT-DIRECT, Solitaire with the Intention For Thrombectomy Plus Intravenous t-PA Versus DIRECT Solitaire Stent-retriever Thrombectomy in Acute Anterior Circulation Stroke.
Data are generally displayed as frequency with percentage (%) of patients.
Relative efficacy of low- versus standard-dose bridging alteplase
Table 8 shows the weighted average ORs and 95% CIs for primary outcomes for all indirect comparisons using the dMT as a common comparator. Indirect comparison of low- with standard-dose bridging alteplase yielded an OR of 0.84 (95% CI 0.47–1.50) for a 90-day mRS 0–2, 1.18 (95% CI 0.65–2.12) for a 90-day mRS 0–1, 1.21 (95% CI 0.44–3.36) for mortality, and 1.11 (95% CI 0.39–3.14) for successful recanalization. There were no significant differences in sICH (OR 1.05, 95% CI 0.32–3.41) and any ICH (OR 1.71, 95% CI 0.94–3.10) between low- and standard-dose alteplase bridging therapy.
Indirect comparison of low-dose versus standard-dose bridging alteplase using dMT as single common comparator.
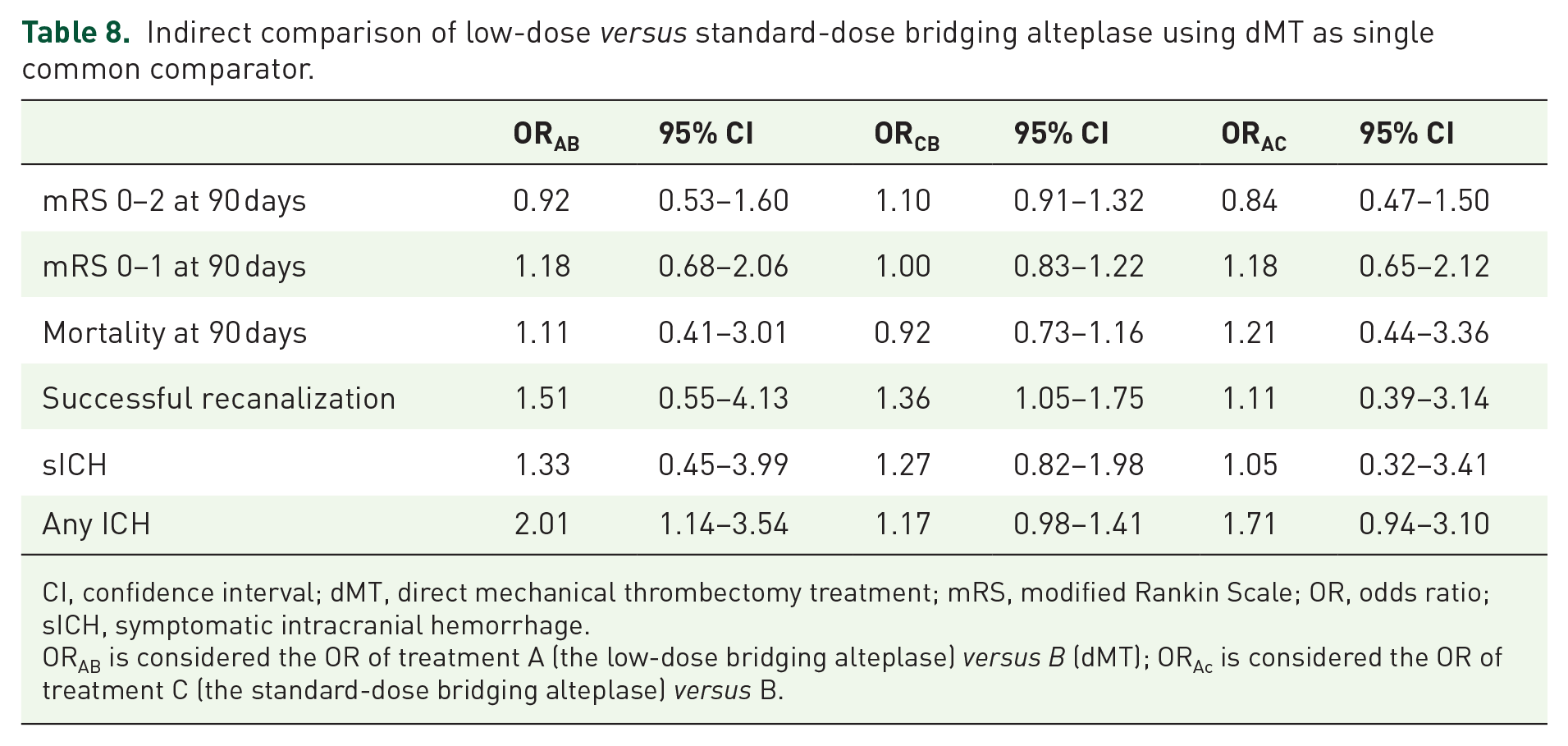
CI, confidence interval; dMT, direct mechanical thrombectomy treatment; mRS, modified Rankin Scale; OR, odds ratio; sICH, symptomatic intracranial hemorrhage.
ORAB is considered the OR of treatment A (the low-dose bridging alteplase) versus B (dMT); ORAc is considered the OR of treatment C (the standard-dose bridging alteplase) versus B.
Sensitivity analysis
Figure 1 shows the direct comparison of the efficacy of BT and dMT in East Asian stroke patients (SKIP, DEVT, DIRECT-MT, and subgroup of DIRECT-SAFE data) stratified by alteplase dose. Indirect comparison in East Asian stroke patients showed that 90-day mRS 0–2 (OR 0.79, 95% CI 0.36–1.74), 90-day mRS 0–1 (OR 1.01, 95% CI 0.43–2.40), mortality (OR 1.10, 95% CI 0.39–3.13), and successful recanalization (OR 1.17, 95% CI 0.41–3.39) were not significantly different between low- and standard-dose bridging therapy. Looking at safety outcomes, low- and standard- dose bridging alteplase did not differ in the odds of sICH (OR 1.03, 95% CI 0.29–3.60) and any ICH (OR 1.57, 95% CI 0.84–2.93, Table 9). A separate analysis of indirect comparison between low- versus standard-dose bridging alteplase detected no significant difference in the odds of sICH (SITS-MOST criteria: OR 1.34, 95% CI 0.27–6.66; NINDS criteria: 1.25, 95% CI 0.35–4.55, Table 10).

Direct comparisons of clinical outcomes between BT versus dMT in East Asian stroke patients, stratified by the alteplase dosage.
Indirect comparison of low- versus standard-dose alteplase bridging using dMT as common comparator in East Asian stroke patients.

CI, confidence interval; DEVT, Direct Endovascular Thrombectomy Versus Combined IVT and Endovascular Thrombectomy for Patients with Acute Large Vessel Occlusion in the Anterior Circulation; DIRECT-MT, Direct Intraarterial Thrombectomy in Order to Revascularize Acute Ischemic Stroke Patients with Large Vessel Occlusion Efficiently in Chinese Tertiary Hospitals; DIRECT-SAFE, A Randomized Controlled Trial of DIRECT Endovascular Clot Retrieval Versus Standard Bridging Thrombolysis with Endovascular Clot Retrieval; dMT, direct mechanical thrombectomy treatment; mRS, modified Rankin Scale; OR, odds ratio; sICH, symptomatic intracranial hemorrhage; SKIP, Direct Mechanical Thrombectomy in Acute LVO Stroke.
ORAB is considered the OR of treatment A (low-dose bridging alteplase) versus B (dMT); ORAc is considered the OR of treatment C (standard-dose bridging alteplase) versus B. The indirect comparison results were derived from the SKIP versus DEVT, DIRECT-MT, and subgroup of DIRECT-SAFE as a whole.
Indirect comparison between the low- versus standard dose of alteplase bridging regarding sICH.

CI, confidence interval; dMT, direct mechanical thrombectomy treatment; mRS, modified Rankin Scale; OR, odds ratio; sICH, symptomatic intracerebral hemorrhage.
SITS-MOST criteria.
NINDS criteria.
ORAB is considered the OR of treatment A (the low-dose bridging alteplase) versus B (dMT); ORAc is considered the OR of treatment C (the standard-dose bridging alteplase) versus B.
Evaluation of indirect comparisons
Table 11 shows that the current study generally meets certain criteria of the indirect comparisons, except the study question was not to be addressed established in advance.
Checklist for evaluation of indirect comparisons.

Discussion
Our indirect comparison analysis showed that patients who received low- versus standard-dose bridging alteplase had a similar likelihood of achieving functional independence or excellent recovery at 90 days. Moreover, low-dose bridging alteplase did not translate into a lower risk of sICH or any ICH. Notwithstanding the inherent limitations of indirect comparisons, Bucher’s approach remains a useful method to estimate the comparative clinical efficacy and safety profiles when head-to-head trials are unavailable.17,19 We additionally compared the SKIP population to those of DEVT, DIRECT-MT, and subgroup participants of the DIRECT-SAFE, attempting to obtain relevant results in East Asian stroke population.
Previous studies have directly compared the efficacy endpoints and safety outcomes among AIS patients who received low- and standard-dose intravenous alteplase. A prospective, multicenter, observational study showed that AIS patients who received standard- versus low-dose alteplase had higher proportions of sICH (ECASS definition, 8.0% versus 2.6%) and mortality within 3 months (12.8% versus 6.9%). 23 The Enhanced Control of Hypertension and Thrombolysis Stroke Study (ENCHANTED) trial failed to show non-inferiority of low- to standard-dose intravenous alteplase in terms of 90-day functional outcomes and mortality, though fewer ICH events were observed for ENCHANTED participants who received low-dose alteplase thrombolysis. 14 Notably, endovascular thrombectomy treatment was similar in the low- and standard-dose arms (64/1654 versus 60/1643). 14
Although the potential impact of alteplase dosage on the outcomes of AIS patients who received BT is of clinical interest, direct comparisons between low- and standard-dose bridging alteplase in AIS patients are currently unavailable and are likely to remain so for the near future. A small prospective observational study of AIS patients with middle cerebral artery occlusion showed that patients who received standard-dose bridging alteplase had higher sICH rates [4/14 (28.6%) versus 0/18 (0)] and any ICH rates [3/14 (21.4%) versus 1/18 (5.6%)]. 24 In contrast, two retrospective observational studies detected no significant differences in 90-day functional independence, mortality, and ICH complications between low- versus standard-dose alteplase bridging therapy.25,26 However, these above-mentioned observational studies were limited by high selection bias and the small sample sizes. Intention-to-treat data of the Korean ENCHANTED Study showed no significant difference in 90-day mRS 0–2 between standard- and low-dose alteplase among 67 patients who underwent cerebral angiography (39% versus 21%; OR 2.39, 95% CI 0.73–7.78). 27 However, direct comparisons between low- and standard-dose bridging alteplase in terms of functional and safety outcomes were not provided. 27 Our study adds to these data by performing an indirect comparison derived from the most up-to-date high-quality RCT data. Unexpectedly, indirect comparison of low- versus standard-dose bridging alteplase yielded a higher likelihood of any ICH (OR 1.71, 95% CI 0.94–3.10), although it did not reach statistical significance. One possible source of bias might be due to SKIP participants having the highest proportions of atrial fibrillation and cardioembolic stroke etiology, which have a higher ICH risk.28,29 Moreover, more than half experienced any ICH at 36 h from onset in the BT arm of the SKIP trial, which appears obviously higher than the other five RCTs (ranging 21.8–42.2%). Nevertheless, our findings need to be validated by future large sample-size RCTs or individual patient data meta-analyses.
There are several strategies available to conduct indirect comparisons, among which Bucher’s method employing a common comparator is considered the gold standard method when head-to-head RCT data are unavailable.17,19,21 To our knowledge, RCTs of bridging alteplase versus placebo in IVT-eligible patients are unavailable, so dMT was the only common comparator for indirect comparisons. We used intension-to-treat data for primary efficacy endpoints and mortality, which is considered the preferred approach for analyzing data from an RCT to maintain random treatment allocation. Adjusted indirect comparisons using Bucher’s method assumes that the trials are broadly similar to one another in terms of potential modifiers of treatment effects.17,19 Moreover, the relative efficacy of a given intervention compared with a common comparator would be homogeneous across each of the study participants.17,19 Indirect comparisons in current study generally meet the assumption of homogeneity. Our analysis suggest that a very large number of study participants would be required to detect non-inferiority in a head-to-head RCT addressing alteplase dosage prior to mechanical thrombectomy. Recently, an artificial intelligence-based prediction model of subarachnoid hemorrhage outcomes has been shown not to be inferior to those of previous statistically calculated prediction models. 30 Whether artificial intelligence-based prediction models may have a role in determining the optimal dose of alteplase needs to be investigated in future studies.
Strengths and limitations
Strengths include the indirect comparisons based on the most recent high-quality RCT data. Moreover, our analysis included East Asian stroke populations with considerable participant characteristics homogeneity. We acknowledge limitations. Because this is an indirect (inter-trial) comparison that addresses the efficacy endpoints and safety outcomes reported in the RCTs, cautions are needed when interpreting our findings due to the fundamental challenges as follows. First, this indirect comparison was without adjustment for clinical variables. Moreover, almost all the results were inconclusive. Although median age and the proportions of female patients were broadly similar across the six RCTs, some critical differences in baseline characteristics (i.e. proportion of atrial fibrillation, and time interval from hospital admission to intravenous alteplase) were observed in the SKIP when compared to other RCTs. Previous studies showed that the odds of ICH and unfavorable outcomes were profoundly increased in AIS patients who had atrial fibrillation.28,29 The optimal dosage of alteplase bridging in AIS patients with atrial fibrillation remains unclear and needs to be investigated. Second, the definition of sICH across the six RCTs was heterogenous. However, the widely used ECASS, SITS-MOST, and Heidelberg criteria share one thing in common (hemorrhage with a neurological decline NIHSS ⩾ 4), minimizing the heterogeneity. 31 Moreover, even if both comparator and treatment groups differ in their baseline characteristics, indirect comparison using the Bucher’s method could obtain an unbiased estimate of treatment effect. 17 Third, deviations from similarity are potentially more confounding in this approach than in a traditional meta-analysis. However, efficacy endpoints were consistent across the included RCTs (mRS 0–2 and mRS 0–1). Fourth, there was only one relatively small low-dose bridging trial (SKIP), which might contribute toward fairly wide CIs for the indirect comparison. Moreover, this approach does not allow adjustment for participant demographics and modifiers of relative treatment effects in different trials. For example, there was a significant interaction between patients ⩾70 years of age and those <70 years of age regarding the primary outcome (90-day mRS 0–2) in the SWIFT-DIRECT trial. 11 The ongoing Improving Reperfusion strategies in Ischemic Stroke (IRIS) collaboration pooled analysis of individual patient data from the six RCTs might contribute to more tailored decision-making concerning prior alteplase use in AIS patients who require EVT. 32
Summary
Despite the limitations of the indirect comparison analysis, our study detected no significant differences in efficacy endpoints and safety outcomes between the low- and standard-dose alteplase in bridging alteplase for AIS. Future head-to-head direct comparison RCTs would better address this issue in patients with AIS who received bridging thrombolysis.
