Abstract
Background:
Epilepsy is one of the important long-term sequelae of neonatal hypoxic-ischemic encephalopathy (HIE) and is typically characterized by drug resistance and poor surgical outcomes. Vagus nerve stimulation (VNS) is a promising neuromodulation therapy for refractory epilepsy.
Objectives:
The present study aimed to first evaluate the effectiveness of VNS in patients with refractory HIE-induced epilepsy and scrutinize potential clinical predictors.
Methods:
We retrospectively collected the outcomes of VNS in all patients with refractory HIE-induced epilepsy and at least 2 years of follow-up. Subgroups were classified as responders and nonresponders according to the effectiveness of VNS (⩾50% or <50% reduction in seizure frequency). Preoperative data were analyzed to screen for potential predictors of VNS effectiveness.
Results:
A total of 55 patients with refractory HIE-induced epilepsy who underwent VNS therapy were enrolled. Responders represented 56.4% of patients, and 12.7% of patients achieved seizure freedom at the last follow-up. In addition, the responder rate increased over time with rates of 23.6%, 38.2%, 50.9%, and 56.4% at the 3-, 6-, 12- and 24-month follow-ups, respectively. After multivariate analysis, neonatal seizure was identified as a negative predictor (OR: 4.640, 95% CI: 1.129–19.066), and a predominant seizure type of generalized onset was identified as a positive predictor (OR: 0.261, 95% CI: 0.078–0.873) of VNS effectiveness.
Conclusion:
VNS therapy was effective in patients with refractory HIE-induced epilepsy and was well tolerated over a 2-year follow-up period. VNS therapy demonstrated better effectiveness in patients without neonatal seizures or with a predominant seizure type of generalized onset.
Keywords
Introduction
Hypoxic-ischemic encephalopathy (HIE) is a significant and frequent cause of acute mortality and chronic neurological disability in neonates 1 and is characterized by clinical manifestations, including neonatal seizures, breathing difficulty, and reduced tone and reflexes. 2 Epilepsy is one of the important long-term sequelae of HIE and is typically combined with other sequelae of neurodevelopmental disorders, such as cerebral palsy, mental retardation, learning disabilities, and behavioral problems. 3 The reported rate of epilepsy following HIE ranges from 9% to 33%.4–6 Children with a history of HIE have a fivefold increased risk of developing epilepsy compared with those without. 7 In addition, neonates exhibiting severe encephalopathy, neonatal seizures, or extensive brain injury on magnetic resonance imaging (MRI) were more likely to develop epilepsy after neonatal HIE.8,9 Based on severe brain injury and neurodevelopmental impairment, HIE-induced epilepsy is characterized by a high risk of drug resistance and disappointing surgical outcomes. 10 Therefore, it is of utmost importance to explore new therapeutic methods for HIE-induced epilepsy.
Vagus nerve stimulation (VNS) is a neuromodulation therapy used as an adjunctive therapy for patients with medically refractory epilepsy who are not eligible for epilepsy resection surgery, or in whom surgery has failed. Since its first human implantation in 1988 and more than 100,000 subsequent implantations, VNS has generated growing interest in the management of refractory epilepsy.11–13 The effectiveness of VNS in treating focal epilepsies and various types of generalized epilepsies, including genetic generalized epilepsy (GGE) and Lennox–Gastaut syndrome (LGS), has been demonstrated in numerous studies.14,15 Surgical outcomes in terms of seizure control account for an average 50–60% improvement and are characterized by an increasing benefit over time. 16 In addition, VNS has been reported to be effective in specific epilepsy conditions, such as tuberous sclerosis, drop attacks (tonic/atonic seizures), and refractory postencephalitic epilepsy.17,18 VNS demonstrated better effectiveness and a lower incidence of side effects in these conditions. 19 Thus, VNS may shed some light on therapeutic advances in refractory HIE-induced epilepsy.
Currently, there are no relevant studies on HIE-induced epilepsy treated by VNS. Due to the impaired structural brain connectivity following neonatal brain injury, these patients might respond differently to VNS than patients with other types of epilepsy. 20 In this study, we aimed to first define the effectiveness of VNS in patients with refractory HIE-induced epilepsy and evaluate the potential predictors of VNS effectiveness.
Materials and methods
Definition of HIE-induced epilepsy and refractory epilepsy
Neonatal HIE was diagnosed according to the following criteria:21,22 (1) ⩾36 weeks gestational age; (2) a definite history of perinatal asphyxia (umbilical cord compression, placental abruption, prolonged labor, etc.); (3) an Apgar score ⩽3 at 1 min and ⩽5 at 5 min of age as well as cord blood gas pH < 7.0 or base deficit ⩽16 mmol/L; (4) different degrees of neonatal encephalopathy according to Sarnat criteria at 1–6 h of age 23 characterized by clinical manifestations, such as abnormal consciousness, decreased spontaneous activity, seizures; and (5) without intrauterine or perinatal infection, major anomalies of the brain or other major organ systems, or evidence of congenital metabolic disease. Epilepsy is one of the most frequent sequelae of neonatal HIE. Currently, a clear diagnostic criterion for HIE-induced epilepsy is not available. All of the patients in this study reported a definite neonatal HIE history, definite MRI lesions of HIE, and epilepsy diagnosis. All recruited patients harbored ischemia-hypoxia lesions in MR images based on definite fluid-attenuated inversion recovery (FLAIR)/T2 abnormal signals, and some lesions were also accompanied by different degrees of encephalomalacia or encephalatrophy. Representative MR images of the two patients are shown in Figure 2. In addition, patients denied other definite etiologic factors for epilepsy (brain malformation, brain tumors, traumatic brain injuries, encephalitis, chromosome disease, metabolic, or mitochondrial diseases) and history of conditions involving ischemia and hypoxia beyond the perinatal period (hypoxic hypoxia, ischemic hypoxia, circulatory dysfunction-induced hypoxia, as well as poisoning-induced hypoxia). Thus, the form of epilepsy occurring in these patients was classified as HIE-induced epilepsy as defined in the present study.
Refractory epilepsy in this study referred to drug-resistant epilepsy, which was defined as patients with “failure of adequate trials of two tolerated and appropriately used antiepileptic drug schedules (used as monotherapies or in combination) to achieve sustained seizure freedom.” 24
Patient selection
This study was performed in accordance with the principles of the Helsinki Declaration and approved by the ethics committee of Sanbo Brain Hospital, Capital Medical University (SBNK-2017-15-01). All participants were informed of the purpose of the study and provided written informed consent. For the children included in this study, we obtained written informed consent from the guardian.
We retrospectively studied the effectiveness of VNS in patients with refractory HIE-induced epilepsy from Sanbo Brain Hospital, Capital Medical University between September 2008 and February 2020. In our comprehensive epilepsy center, each patient was evaluated through MRI and video electroencephalography (VEEG) before the operation. Some patients might also undergo positron emission tomography-computed tomography (PET-CT), neuropsychological assessment, and magnetoencephalography (MEG). The treatment strategies for all patients were discussed and determined at a multidisciplinary team (MDT). VNS was recommended in the following conditions based on our previous strategy: 25 (1) patients whose epileptogenic focus could not be precisely localized; (2) patients with an epileptogenic focus involved in the eloquent areas; and (3) patients who did not accept surgical resection or had early surgical failure. SEEG and the Wad test were conducted to determine whether the epileptic foci were involved in eloquent areas. All patients recruited in this study were followed up for at least 2 years.
Clinical data collection
The medical history of patients, including sex, age of epilepsy onset, history of neonatal seizure, age of VNS implantation, epilepsy duration, predominant type and frequency of seizures, number of preoperative AEDs, history of mental retardation, status epilepticus (SE) and febrile seizures, brain surgery before VNS, spatial distribution of EEG, and brain MRI, was collected.
Neonatal seizures referred to seizures occurring within the first 28 days of life for full-term infants, 26 which is defined by clinical seizures and definite EEG correlates according to the ILAE classification of neonatal seizures published in 2020. 27 In our study, neonatal seizures further referred to the onset of epilepsy and excluded acute symptomatic and febrile seizures. After the occurrence of the first seizure during the neonatal period, the patients began to experience recurrent spontaneous seizures, which met the diagnosis of epilepsy. For this research, seizure type was defined as the most frequent seizure type of each patient based on the medical documents provided by the physician. According to the 2017 ILAE classification of epilepsy, 28 seizure types were classified as “focal onset” and “generalized onset.” As noted in our previous study, 29 the monthly frequency of seizures was categorized into “<80 times” and “⩾80 times.”
Preoperative evaluation
The standard 10-20 system of electrode placement was used for 64-channel long-term video EEG monitoring in all patients for at least 24 h. The interictal epileptic discharges (IEDs) were classified as focal, multifocal, and generalized according to our previous study: 25 (1) focal: the IEDs only involved one lobe or contiguous lobes; (2) multifocal: the IEDs involved ⩾3 multiple lobes; (3) generalized: the IEDs were bilateral synchronous and generalized in both hemispheres. Similarly, for patients whose seizures were recorded, the ictal onset rhythms were also classified as focal, multifocal, and generalized. Brain 1.5-T MRI scans with T1, T2, and FLAIR sequences were performed in all patients. To further verify whether patients were eligible for VNS surgery, PET and MEG were conducted in some patients to help locate the epileptic area. All results of the preoperative evaluation were analyzed in detail by experienced neurologists, neurosurgeons, neuroradiologists, and electrophysiologist. VNS implantations were performed by two neurosurgeons according to standard procedures. 30
Programming strategy of VNS
VNS parameter setting was performed in accordance with a previous programming strategy in our epilepsy center. 31 In the 55 patients recruited in our study, two models of vagus nerve stimulators were implanted: Model 103 (Demipulse, LivaNova) implanted in 76.4% (42/55) of patients and Model G111 (Beijing PINS Medical Co., Ltd.) implanted in 23.6% (13/55) of patients. The stimulation was initiated at 7 days after implantation of the stimulator. Initial parameters were set at 0.5 mA for the out current, 30 s for the signal on time, and 5 min for the signal off time. The signal frequency (30 Hz) and the pulse width (250 ms) were kept constant, and the magnet current was set as 0.25 mA higher than the output current. Within 1 month of discharge, the current intensity of effective treatment was increased to 1.25–1.5 mA at the outpatient clinic. Thereafter, the parameters were modified to 0.25 mA every 3–6 months according to the improvement in seizure control and patient tolerance.
Seizure outcome and follow-up
All patients included received follow-up for at least 2 years after VNS therapy. The outcomes of VNS therapy were determined by a questionnaire survey performed when patients were readmitted to the hospital for adjustment of stimulus parameters or remote follow-up via online methods. The effectiveness of VNS was evaluated according to our previous study. 29 Response was defined as a ⩾50% reduction in baseline seizure frequency of the predominant seizure type. Seizure freedom in the present study was defined as complete freedom of all types of seizures at the last follow-up. VNS outcomes were collected at 3, 6, 12, and 24 months and at the last follow-up after VNS treatment. The last follow-up results were used to define the overall effectiveness and potential predictors of VNS.
Statistical analysis
All analyses were performed using SPSS software version 23.0, and a p value of <0.05 was considered statistically significant. The median (interquartile range, IQR) was used to describe the continuous variables, and frequency was reported for categorical variables. In univariate analysis, the Mann–Whitney U test was employed to compare continuous variables, and categorical variables were assessed using Pearson’s chi-square or Fisher’s exact test. Variables showing a p value of <0.05 in the univariate analysis were then entered into a multivariate logistic regression model based on the backward stepwise method.
Results
Demographic characteristics
The overall flow of patient recruitment is shown in Figure 1. A total of 63 refractory HIE-induced epilepsy patients with VNS implantation were collected, and 8 patients were excluded based on a follow-up time <2 years. The present study was based on the remaining 55 patients with refractory HIE-induced epilepsy (40 men and 15 women) managed during the period of 2008–2020. No serious adverse effects were reported in the enrolled patients.

Flow chart for recruiting patients who satisfied the inclusion and exclusion criteria.
Among the recruited refractory HIE-induced epilepsy patients, the median age of VNS implantation, age at seizure onset, and duration of epilepsy were 13.0 (IQR 8.4–20.0) years, 2.0 (IQR 0.2–5.0) years, and 10.0 (IQR 5.6–16.0) years, respectively. Neonatal seizures were found in 13 (23.6%) patients. Thirty-six (65.5%) patients had mental retardation, 13 (23.6%) patients experienced febrile seizures before VNS, and 5 (9.1%) patients reported a history of cerebral palsy. Other patient characteristics are shown in Table 1.
Patients’ demographic and clinical features and their relationship with VNS effectiveness.

AEDs, antiepileptic drugs; EEG, electroencephalogram; IEDs, interictal epileptiform discharges; MEG, magnetoencephalography; PET, positron emission computed tomography; SE, status epilepticus; VNS, vagus nerve stimulation.
p < 0.05.
Results of preoperative evaluation
Brain MRI results were reviewed in all patients. All MR images of the included patients revealed ischemia-hypoxia lesions based on definite FLAIR/T2 abnormal signals, some of which were also accompanied by different degrees of encephalomalacia or encephalatrophy. Representative MR images of the two patients are shown in Figure 2. During scalp EEG monitoring, IEDs were observed in all patients: 18 (32.7%) were focal, 30 (54.5%) were multifocal, and 7 (12.8%) were generalized. Seizures were recorded in 52 (94.5%) patients, including 5 (9.1%) with focal epileptic discharges, 15 (27.2%) with multifocal epileptic discharges, and 32 (58.2%) with generalized epileptic discharges. In addition, to help locate the epileptogenic focus, PET was performed in 4 (7.3%) patients, and MEG was performed in 9 (16.4%) patients.
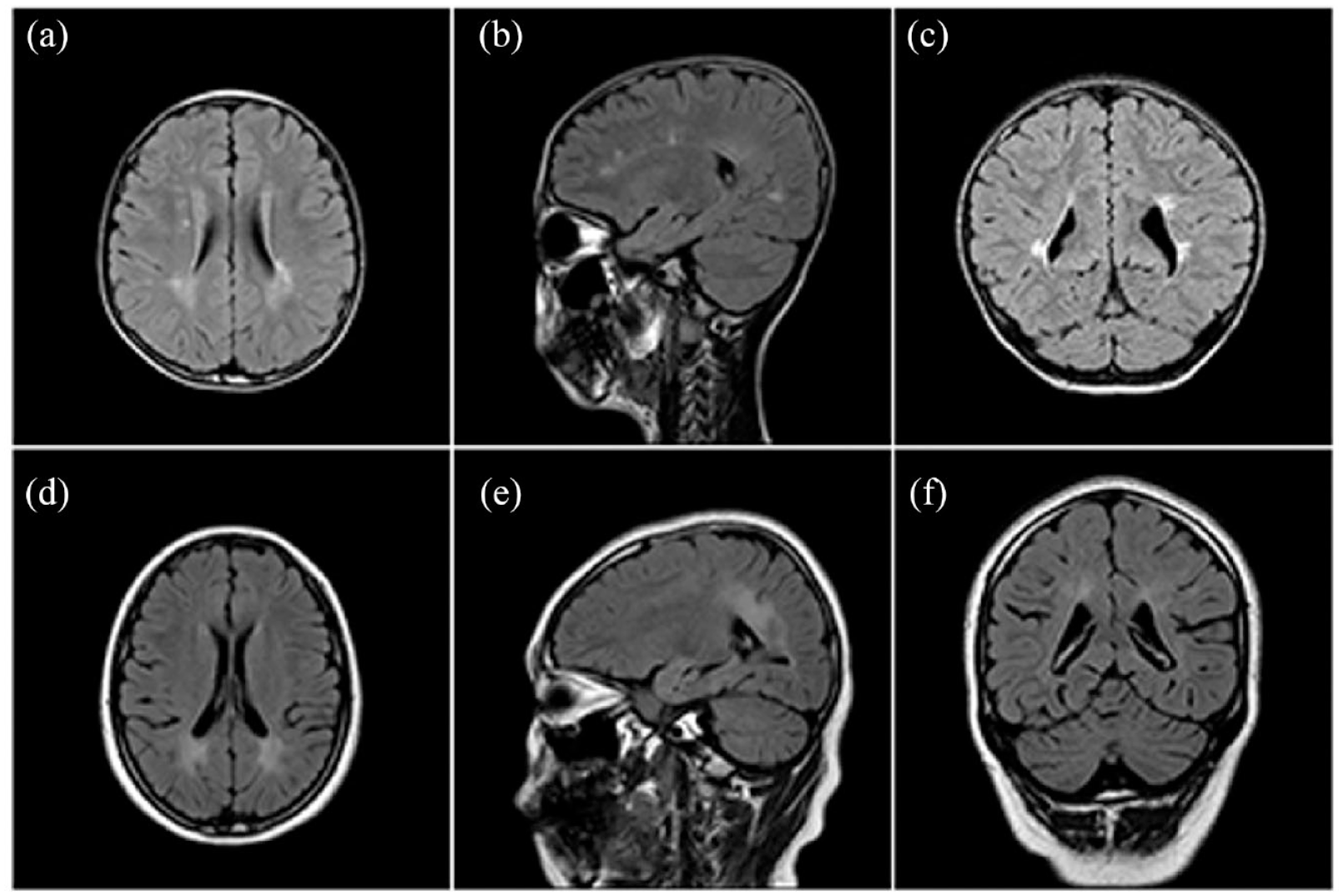
Representative FLAIR MR images of two patients with refractory HIE-induced epilepsy. There were representative FLAIR MR images of two patients with refractory HIE-induced epilepsy in the axial (a, d), sagittal (b, e), and coronal (c, f) planes. (a–c) An 8-year-old boy with HIE due to postnatal asphyxia, with a seizure onset at 6 years old. The abnormal FLAIR hyperintensity was observed in white matter of bilateral periventricular areas, and bilateral parietal and occipital lobes. The patient got seizure freedom after 2 years following the VNS therapy. (d–f) A 10-year-old girl with HIE due to hypoxia during delivery, with a seizure onset at 20 days after birth. The abnormal FLAIR hyperintensity was observed in white matter of bilateral parietal and occipital lobes. The patient got no significant reduction of seizure frequency after 2.5 years following the VNS therapy.
Outcomes of VNS
The median time of the last follow-up was 3.5 (IQR 2.4–5.0) years for all participants, ranging from 2.0 to 12.0 years. The last follow-up outcomes of VNS therapy showed that seizures were reduced in 36 (65.5%) refractory HIE-induced epilepsy patients and reduced by ⩾50% in 31 (56.4%) patients. In addition, seizure freedom occurred in 7 (12.7%) patients. McHugh and modified Engel seizure outcome classifications were used to evaluate the last follow-up outcomes (Table 2). The modified Engel scale revealed that 7 (12.7%) of 55 patients with refractory HIE-reduced epilepsy were classified as class I, 11 (20.0%) patients were class II, 13 (23.6%) were class III, and 24 (43.7%) patients were class IV. The McHugh scale showed that 21 (38.2%) of 55 patients were classified as class I, 10 (18.2%) patients were class II, 5 (9.1%) patients were class III, and 19 (34.5%) patients were class IV–V.
Seizure outcomes evaluated by modified Engel and McHugh seizure outcome classifications.
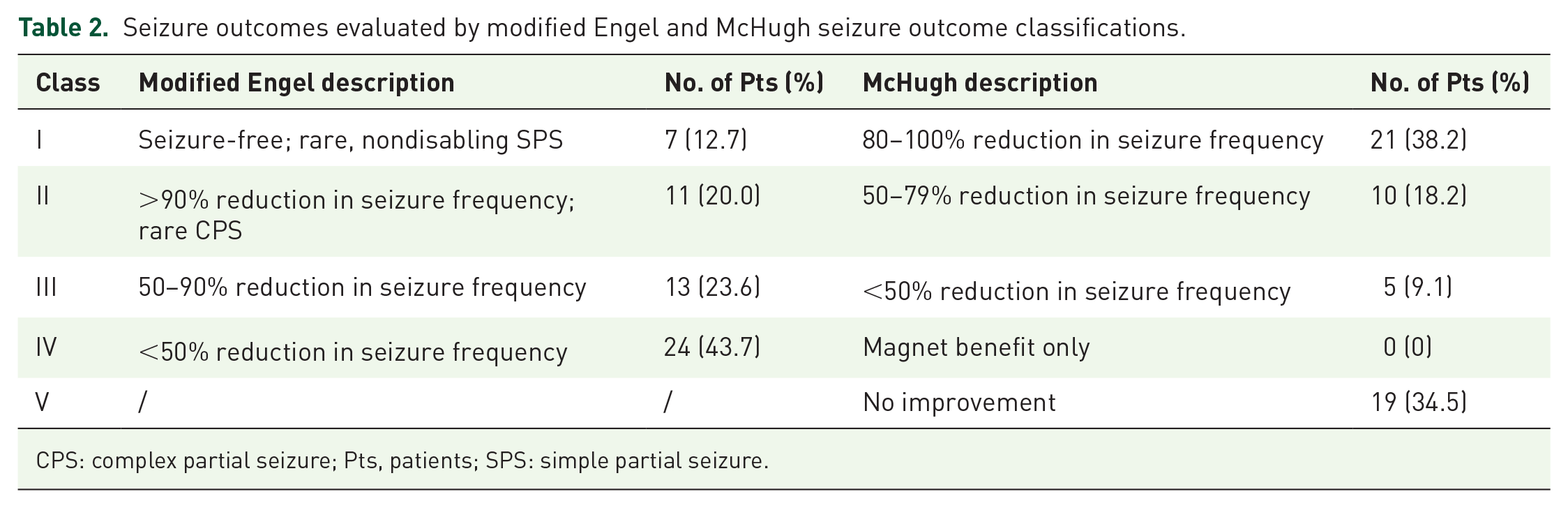
CPS: complex partial seizure; Pts, patients; SPS: simple partial seizure.
After VNS therapy, the effectiveness outcomes of 55 recruited patients were evaluated at the 3-, 6-, 12- and 24-month follow-ups [Figure 3(a) and (b)]. The detailed assessments of VNS outcomes based on the McHugh description at different follow-up time points are shown in Figure 3(a). Both the responder and seizure freedom rates gradually increased over time [Figure 3(b)]. At 3, 6, 12, and 24 months of follow-up, the number of responder patients was 13 (23.6%), 21 (38.2%), 28 (50.9%), and 31 (56.4%), respectively, and the number of patients with seizure freedom was 1 (1.8%), 2 (3.6%), 2 (3.6%), and 6 (10.9%), respectively. Among the 13 responders at 3 months, 2 (15.4%) patients adversely became nonresponders at 6 months, and the situation lasted to the last follow-up. Out of the 42 nonresponders at 3 months, 10 (23.8%) patients turned as responders at 6 months, and 1 of the 10 patients reversely returned to nonresponders at the last follow-up (5 years after VNS therapy); 7 (16.7%) patients got response at 12 months, and 1 of the 7 patients adversely returned to nonresponders at 24 months; 4 (9.5%) patients got response at 24 months and 1 patient got response over 2 years (4.4 years) after VNS therapy. Seven patients (12.7%) of the study population achieved seizure freedom at the last follow-up, among them six patients got seizure freedom within the 2 years after VNS therapy and one patient got that over 2 years.

Seizure outcomes of patients with refractory HIE-induced epilepsy after VNS. (a) Seizure outcomes described by McHugh outcome classification at 3-, 6-, 12-, and 24-month follow-up after VNS therapy. Arrows indicated changes in VNS effectiveness between follow-ups. (b) Both the rates of responder and seizure freedom gradually increased over time.
During the follow-up period, physicians could adjust the medication treatment according to the seizure outcome of patients. Among the 55 recruited patients in this study, the number of AEDs at baseline and the last follow-up was 2.0 (IQR 1.0–3.0) and 3.0 (IQR 2.0–3.0), respectively. No significant changes in the number of AEDs (p = 0.320) were noted. Thus, the AED treatment remained stable from baseline to the last follow-up. In the present cohort, at least one concomitant AED was changed in 27 (49.1%) patients, and no changes in baseline concomitant AEDs were observed in 28 (50.9%) patients. Of the 27 patients with AED changes, 5 (9.1%) patients had a reduction in the number of AEDs from their baseline medication. Furthermore, we analyzed the effectiveness of VNS in 33 (60%) patients with no changes or with a reduction in AED numbers. The responder and seizure freedom rates at the last follow-up were 57.6% (19/33) and 9.1% (3/33), respectively.
Analysis of prognostic factors for VNS effectiveness
In the univariate analysis (Table 1), the following factors were associated with seizure outcomes: age at seizure onset, neonatal seizure, seizure type, and cerebral palsy. Among the five patients with cerebral palsy, no patients responded at the last follow-up. Therefore, the sample size in the responder group was zero, and the odds ratio (OR) value could not be calculated in the multivariate logistic analysis. 32 Thus, the variable of cerebral palsy was not further included in the multivariate logistic regression model. The remaining three statistically significant factors in the univariate analysis were then entered into the multivariate logistic regression model using the backward stepwise method. After multivariate analysis, a predominant seizure type of generalized onset (OR: 0.261, 95% CI: 0.078–0.873) was identified as a positive predictive factor for VNS effectiveness in refractory HIE-induced epilepsy, whereas neonatal seizure (OR: 4.640, 95% CI: 1.129–19.066) was identified as a negative predictive factor for VNS effectiveness (Table 3).
Predictors of VNS effectiveness for refractory HIE-induced epilepsy on multivariate analysis.

CI, confidence interval; OR, odds ratio; VNS, vagus nerve stimulation.
p < 0.05.
Neonatal seizures further referred to the onset of epilepsy and excluded acute symptomatic and febrile seizures in the present study. Thirteen (23.6%) patients experienced neonatal seizures in the study population. After univariate and multivariate analyses, neonatal seizures were significantly related to nonresponders. Patients with neonatal seizures had a lower rate of response than those without [Figure 4(a)]. At the last follow-up (⩾24 months), 4 (30.8%) of 13 patients with neonatal seizures achieved a response, and 1 (7.7%) achieved seizure freedom. In total, 27 (64.3%) of those without neonatal seizures achieved a response, and 6 (14.3%) achieved seizure freedom.

Responder rate in patients classified by predictors of VNS effectiveness. (a) Patients with neonatal seizure had lower rate of VNS responders than those without. (b) Patients with predominate seizure type of generalized onset were more likely to get response after VNS therapy than those with focal onset. (a, b) The responder rate gradually increased over time in all subgroups of patients with refractory HIE-induced epilepsy.
The seizure type in this study was classified as “focal onset” and “generalized onset” according to the 2017 ILAE classification of the epilepsies. 28 After univariate and multivariate analyses, the predominant seizure type of generalized onset was significantly associated with responders. The responder rate of patients with generalized onset was greater than that noted for those with focal onset [Figure 4(b)]. At the last follow-up (⩾24 months), the responder rates of patients with predominant seizure types of generalized onset and focal onset were 67.6% and 38.1%, respectively. Their seizure freedom rates were 17.6% and 4.8%, respectively.
Of note, five (9.1%) patients with cerebral palsy in this study were all found to be nonresponders at the last follow-up after VNS therapy (Table 1). Among them, 40.0% (2/5) showed a reduction in seizure frequency of less than 50%, and 60.0% (3/5) showed no improvement in seizure control. Therefore, cerebral palsy may also represent a negative predictor for VNS effectiveness in refractory HIE-induced epilepsy. Further studies with a larger sample are needed to confirm this finding.
Univariate analysis of other factors, such as sex, age at VNS implantation duration of epilepsy, monthly seizure frequency, the type of stimulator, and EEG features, did not reveal significant differences between the two groups of responders and nonresponders.
Discussion
In this study, we first reviewed the clinical data of VNS effectiveness for refractory HIE-induced epilepsy with a minimum follow-up of 24 months. The results demonstrated that VNS was an effective approach to reduce seizure frequency in those patients. Out of 55 patients with refractory HIE-induced epilepsy, 31 (56.4%) patients experienced a greater than 50% reduction in seizures at the last follow-up, including 7 (12.7%) patients who achieved seizure freedom. This result was consistent with previous studies on the effectiveness of VNS reported in the last 30 years with a 50% or greater reduction in seizure frequency achieved in 45–65% of patients and infrequent complete seizure freedom. 33 No significant side effects were reported in the recruited patients of this study. In addition, baseline concomitant AEDs remained unchanged or were reduced in 60% (33/55) of the patients at the last follow-up in the present study. Analysis of this subset of patients allowed a more accurate assessment of the positive effects of VNS, and a similar response rate was noted in this group (57.6%) compared with the entire population of this study (56.4%). Thus, the safety and effectiveness of VNS therapy in patients with refractory HIE-induced epilepsy are reported.
In the current study, the responder rates were 23.6%, 38.2%, 50.9% and 56.4% at 3, 6, 12, and 24 months after device implantation, respectively, and the seizure freedom rates were 1.8%, 3.6%, 3.6%, and 10.9%, respectively. The responder and seizure freedom rates gradually increased over time, which was consistent with the progressive increase in overall response to VNS treatment that has been previously reported.34–36 In a meta-analysis identifying 74 clinical studies with 3321 patients who received VNS implantation for intractable epilepsy, 36 seizure frequency was reduced by an average of 45% with a 36% reduction in seizures at 3–12 months after surgery and a 51% reduction after over 1 year of therapy. At the last follow-up, approximately 50% of the patients had a 50% or greater reduction in seizure frequency, and complete seizure freedom was rarely (<5%) attained. These results confirmed a delayed benefit to sustained VNS therapy, although complete seizure freedom is achieved in only a minority of patients.
In further analysis of predictors for VNS effectiveness in refractory HIE-induced epilepsy, neonatal seizures were associated with nonresponders (OR: 4.597, 95% CI: 1.039–20.328). Neonatal seizure, a clinical emergency occurring in 3–5 cases out every 1000 live births, is one of the most common complications of HIE with seizures aggravating acute brain injury and chronic developmental disorders.37,38 Newborns with seizures have a mortality rate of up to 20% and an increased risk of long-term consequences, including epilepsy, cerebral palsy, and psychomotor disorders.39,40 Children with clinical neonatal seizures have higher rates of postneonatal epilepsy (20–30%),41–43 and patients with EEG-confirmed neonatal seizures have postneonatal epilepsy rates of approximately 50%. 44 Furthermore, a high risk of severe neurodevelopmental impairment (81%) and drug-resistant seizures (32%) are noted among those who developed epilepsy. 44 Similar to previous findings, the percentages of cerebral palsy (15.4% versus 9.1%) and mental retardation (76.9% versus 65.5%) were higher in the 13 patients with clinical neonatal seizures in the present study compared with the overall patient population (n = 55). Thus, due to severe brain injury and subsequent neurodevelopmental disorders, neonatal seizures may predict adverse VNS therapy outcomes in refractory HIE-induced epilepsy.
In addition, we also found that the age of seizure onset in the responder group was significantly older than that in the nonresponder group. Prior studies have also provided similar results. In a systematic review of 101 studies, a later age at seizure onset was associated with better seizure outcomes following VNS implantation. 45 A retrospective analysis of 158 patients with medically refractory epilepsy also reported that patients with age at seizure onset ⩾15 years old were ideal candidates for VNS. 46 Thus, a later age of seizure onset may also indicate a better VNS effectiveness in refractory HIE-induced epilepsy.
Our results indicated better VNS therapeutic efficacy in patients with a predominant seizure type of generalized onset compared with those with focal onset. Similarly, in a systematic review of VNS therapy including 2869 patients across 78 studies, 47 a predominantly generalized seizure type was found to be a predictor for seizure freedom (OR: 1.360; 95% CI: 1.010–1.820).
In this study, we also analyzed some other sequelae of HIE combined with epilepsy, such as cerebral palsy and mental retardation. Among 55 patients with HIE-induced epilepsy, 5 (9.1%) patients had cerebral palsy, all of whom were nonresponders at the last follow-up. The finding indicated that patients with cerebral palsy may rarely benefit from VNS therapy, and this notion needs to be further confirmed in studies with a larger sample in the future. Of 36 (65.5%) patients with mental retardation in this study, 17 (47.2%) patients responded to VNS therapy. The responder rate in this group of patients was also lower than that of the overall population, although the difference was not statistically significant (47.2% versus 56.4%). Thus, for those patients with other severe or complicated sequelae after HIE, VNS effectiveness on seizure frequency reduction may be limited to some extent.
In addition to decreasing seizure frequency, VNS has also been reported to improve behavior and cognition in epilepsy patients.48,49 In a European long-term study in 347 children with VNS therapy for drug-resistant epilepsy with up to 24 months of follow-up, 50 researchers found improved assessments of concentration, energy, mood, verbal communication, and progress with schoolwork over time. In addition, clinical assessment of the patient’s overall condition revealed that one-third of the patients were considered ‘much improved’ or ‘very much improved’ following 12 months of adjunctive VNS therapy, and the condition continued to improve for up to 24 months. These indicators were not analyzed in this study. However, it is important to note that a moderate effect on seizure frequency may not signify low utility of VNS. For those with complicated HIE-related sequelae, VNS treatment may offer other benefits, such as improvements in mental retardation, motor disorders, or emotional problems. Irrespective of the unsatisfactory effect on seizure frequency, the possible reduction in overall improvement in well-being may make this treatment worth considering. Therefore, more research on VNS therapy for HIE-induced epilepsy is expected to further explore this issue in the future.
Several limitations of the present study should be recognized. First, the inherent biases of the retrospective study and the relatively small sample size could not be excluded in this study, and more prospective studies with large samples are expected to provide clearer conclusions in the future. Second, clinical assessments of emotion, cognition, and overall life quality were not included in this study, all of which are important for the curative effect of patients with refractory HIE-induced epilepsy. Despite these limitations, this study suggested the effectiveness of VNS in reducing seizure frequency in patients with refractory HIE-induced epilepsy. In addition, neonatal seizure and a predominant seizure type of generalized seizure onset may represent independent predictors for VNS effectiveness in refractory HIE-induced epilepsy.
Conclusion
Our data demonstrated that VNS therapy was effective in patients with refractory HIE-induced epilepsy and was well tolerated over a 2-year follow-up period. VNS therapy exhibited increased effectiveness in patients without neonatal seizures or with a predominant seizure type of generalized onset.
